Abstract
Practical relevance:
Feline chronic kidney disease (CKD) is frequently encountered by veterinarians. Timely diagnosis and staging may facilitate the initiation of adequate therapy and improve the prognosis for patients.
Clinical challenges:
Feline CKD is diagnosed based on the presence of compatible clinical signs and renal azotaemia, which implies that urinalysis (particularly urine specific gravity) is mandatory to confirm the diagnosis. Although the diagnosis of advanced feline CKD and associated complications is usually straightforward, based on complete blood and urine examination, all routine blood and urine tests have their limitations in detecting early CKD. Therefore, diagnosing early or non-azotaemic CKD is much more challenging. Although determination of glomerular filtration rate (GFR) would be ideal to identify early kidney dysfunction, practical limitations hamper its routine use in clinical practice.
Patient group:
CKD is typically a disease of aged cats, but may affect cats of all ages. Conclusive breed and sex predispositions for feline CKD are not reported.
Audience:
This review is directed at practising veterinarians and provides an overview of the required diagnostic tests, the classification system established by the International Renal Interest Society, and the importance of and possible techniques for early detection of CKD.
Evidence base:
Staging of cats with CKD is essential as it directs management and provides a prognostic guide. Given that diagnosis at early disease stages is associated with more prolonged survival times, simple, inexpensive and accurate methods for early CKD diagnosis are needed. Techniques currently under investigation include limited sampling strategies to estimate GFR, clearance marker cut-off concentrations to identify cats with low GFR, new indirect GFR markers and urinary biomarkers.
Minimum database for diagnosis
Feline chronic kidney disease (CKD) is diagnosed based on the presence of combined renal azotaemia and poorly concentrated urine (urine specific gravity [USG] ?1.035), with compatible historical and/or physical examination findings.1,2 If the urinary bladder is palpable, urine can be easily collected by cystocentesis (Figure 1).
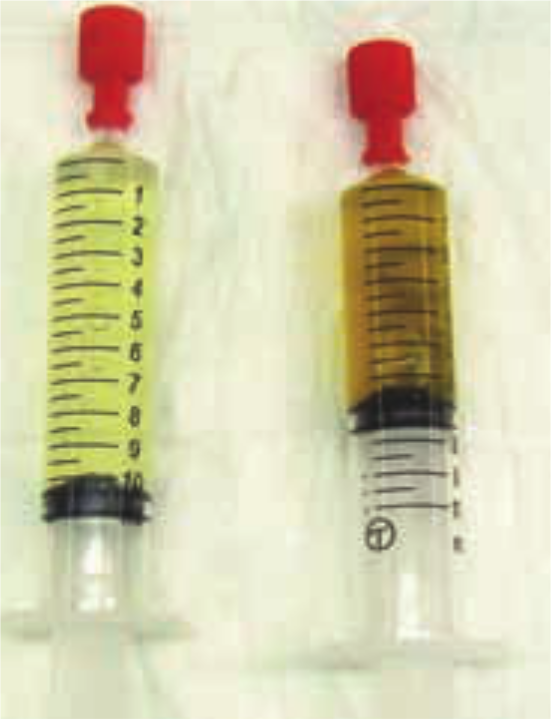
Urine samples taken by cystocentesis from (left) a cat with IRIS stage 3 CKD and (right) a healthy geriatric cat. The cat with CKD had poorly concentrated urine (USG 1.014); the healthy cat concentrated urine (USG 1.045). In the absence of bilirubinuria, the difference in urinary concentration is macroscopically visible

Signalment
The signalment may be helpful for diagnosing feline CKD. Veterinarians should have an increased index of suspicion for CKD in senior and geriatric cats. Although CKD typically is a disease of older cats and some breeds have been overrepresented in some studies, CKD may affect cats of all ages and conclusive breed and sex predispositions for feline CKD are not reported.3–6
History
Cats with CKD are presented at various stages of illness. Some are incidentally diagnosed during health screening; some demonstrate mild clinical signs; while others suffer from end-stage CKD with severe signs such as emaciation or dehydration.3,4 The illness duration prior to admission is highly variable. Although feline CKD is typically chronic, an acute history of illness is not uncommon. 3
Clinical signs
The most common clinical signs are non-specific and include inappetence, polyuria, polydipsia, weight loss, lethargy, halitosis and vomiting.3–5,7 Signs associated with nephrotic syndrome or hypertension are uncommon in cats with CKD.3,4,7 Physical examination reveals thin body condition, dehydration, periodontal disease, unkempt hair coat, abnormal findings on kidney palpation (small, irregular or enlarged kidneys) and pale mucous membranes (Figure 2).3,4

Cat presenting with severe clinical signs due to acute-on-chronic kidney disease. Thin body condition (body condition score 3/9) and unkempt hair coat with scaling are visible (a,b). The cat also had pale mucous membranes and was salivating because of uraemic stomatitis and nausea (c). After stabilisation with infusion, supportive therapy (nasoesophageal tube feeding, antiemetics, antacids, analgesics) and antibiotics, the cat was discharged. Follow-up 2 weeks later revealed IRIS stage 3 CKD
Serum creatinine
Renal azotaemia is defined as increased serum creatinine and urea concentrations due to intrinsic renal pathology (see box on the right). 8 Creatinine is more reliable than urea as an indirect marker for glomerular filtration rate (GFR) because creatinine is less influenced by extrarenal factors (eg, intestinal protein content, liver function) and undergoes glomerular filtration without tubular reabsorption. The daily production rate of creatinine depends on muscle mass, which may be of clinical importance in geriatric cats with age-related muscle wasting or when muscle mass gradually declines during CKD progression.9–11
The (modified) Jaffé and enzymatic assays are frequently used commercial creatinine assays. Both methods correlate well with a reference method. However, the Jaffé assay may overestimate low concentrations and underestimate high concentrations of feline serum creatinine. The enzymatic assay only slightly overestimates feline creatinine and appears to be the preferred method. 12 Although the (modified) Jaffé assay is being replaced gradually by enzymatic assays, many diagnostic laboratories still use this assay to measure serum creatinine concentrations. Reference intervals (RIs) for serum creatinine can differ widely between laboratories, which may lead to misclassification of samples as normal or abnormal.13,14 Given that assays and RIs often differ between laboratories, clinicians are encouraged to consistently determine serum creatinine in a single laboratory with good quality control. Development of age-dependent RIs for laboratory parameters, especially serum creatinine, would improve health screening in aged cats.9,15
Urine specific gravity
In veterinary medicine, USG is routinely measured by refractometry. Human hand-held optical refractometers can overestimate feline USG. 16 However, these errors are not clinically relevant and are unlikely to change clinical decision making. 17 Veterinary refractometers with a separate feline USG scale avoid these errors. 16 Alternatively, a conversion calculation is available (feline USG = [0.846 x SG of human refractometer] + 0.154), a formula that dates from 1956.16,17
Most cats with CKD have isosthenuric urine (USG 1.007–1.015),3,4 but this finding is less consistent compared with dogs. 18 Some cats, with spontaneous as well as with experimentally induced CKD, can retain their urine concentrating ability despite being azotaemic, particularly in the early stages of disease.3,4,19 With disease progression, USG usually gradually declines.4,20
Proteinuria – is it renal?
Although severe proteinuria is uncommon, low level proteinuria (urinary protein:creatinine ratio [UPC] <1) commonly affects feline CKD patients and is an important prognostic factor and therapeutic target.7,21–23 Therefore, quantification and longitudinal monitoring of proteinuria is very important in all cats with CKD.

Proteinuria may be prerenal, renal or postrenal in origin and persistent renal proteinuria indicates CKD. A step-wise diagnostic approach (see box on page 17) must be followed to eliminate prerenal (eg, haemoglobinuria, myoglobinuria, Bence-Jones proteinuria), postrenal urinary (eg, urolithiasis, cystitis, ureteritis, bladder or urethral neoplasia) and postrenal extraurinary proteinuria (eg, genital tract inflammation). 24
Routine tests for proteinuria
Several methods exist to evaluate whether cats with CKD are proteinuric. However, the UPC, which provides an index of total urinary protein loss and correlates closely with the gold standard of 24 h urinary protein excretion, is the only reliable method to determine the clinical implications.24,25 Because the UPC can vary depending on the methodology, monitoring of UPC requires that the same laboratory assay is consistently used. 26
In practice, dipstick tests are often used as a measure of urinary protein. Urine dipstick tests primarily measure albumin; are easy, rapid, in-house tests; and provide semiquantitative assessment of proteinuria. Unfortunately, urine dipsticks only reliably detect severe feline proteinuria. In cats with low level proteinuria, false-positive and false-negative results are common.27,28 A positive dipstick test can be confirmed by the more sensitive and specific semiquantitative sulfosalicylic acid (SSA) turbidity test, which has a lower detection limit (5 mg/dl) compared with dipstick tests (30 mg/dl). Dipstick and SSA results must be interpreted in the light of USG, because positive results in concentrated urine reflect less severe protein loss than in dilute urine.9,29 If a false-negative dipstick result is suspected, the SSA test or a species-specific microalbuminuria test (see below) can be employed.29,30
Measurement of UPC is recommended in all animals with positive semiquantitative proteinuria tests.9,24,30
(Micro)albuminuria
Microalbuminuria is defined as the presence of a small amount (1–30 mg/dl) of albumin in the urine, below the limit of detection of urinary dipstick tests.30,31 Microalbuminuria may also remain undetected by UPC determination. 28 Higher urinary albumin concentrations (>30 mg/dl) are termed (overt) albuminuria and are usually detected using urine dipstick tests or UPC.30,31 Persistent renal (micro)albuminuria may be indicative of renal disease;24,31 however, (micro)albuminuria has been observed in healthy cats and in cats with a wide variety of non-renal diseases (eg, infectious, inflammatory, endocrine, neoplastic and urinary tract disease).31–35 It is currently unknown whether microalbuminuria is a negative prognostic factor in cats, as it is in humans.31,34
(Micro)albuminuria can be measured with the urinary albumin:creatinine ratio (UAC) by quantifying urinary albumin with an enzyme-linked immunosorbent assay (ELISA) using a feline-specific albumin antibody. 31 Until now, an apparent benefit of UAC measurement over UPC has not been found.21,36,37 Alternatively, feline microalbuminuria can be detected with a commercial in-house semi-quantitative ELISA-based dipstick test (ERD-Health Screen, Heska Corporation, Fort Collins, Colorado, USA).31,32,38 Although most cats with a negative microalbuminuria dipstick test have a UPC <0.4,32,38,39 a negative microalbuminuria dipstick result does not preclude elevated UPC.32,35 A positive microalbuminuria test is very likely in feline CKD patients with a ?2+ urine dipstick result, positive SSA test or ?trace dipstick result combined with a positive SSA test. In any of these scenarios, quantification of UPC or UAC is warranted. Conversely, microalbuminuria is unlikely in CKD patients with UPC <0.2. 39
Routine evaluation for the presence of (micro)albuminuria in cats is not warranted for a number of reasons: principally, because (micro)albuminuria occurs with various diseases, UAC measurement is not widely commercially available, UAC lacks benefit over UPC, semiquantitative test interpretation might be difficult and negative microalbuminuria tests do not rule out proteinuria.9,27 However, there are some indications for (micro)albuminuria assessment, particularly in cats at risk of renal disease without overt proteinuria (see box below).9,30 It is important to remember that (micro)albuminuria is not necessarily associated with CKD and diagnostic tests to define the underlying cause are recommended.31,33

Additional diagnostic tests
Blood and urine examination
Additional blood and urine parameters need to be monitored carefully in cats with CKD, mainly to facilitate early recognition and treatment of complications.
Approximately 15% of cats with mild CKD (IRIS stage 2) and up to 100% of cats with end-stage CKD have hyperphosphataemia.3,5,7,42 Hyperphosphataemia might be overlooked if the phosphate RI is based on healthy young cats, again indicating the need for age-dependent RIs. 15 Total and ionised calcium concentrations in cats with CKD vary from increased, to normal to decreased.20,42,46 Ionised calcium concentrations tend to decline with increasing severity of CKD.20,42 Because total calcium poorly predicts ionised calcium concentration, particularly in CKD patients, measurement of total calcium is of little value and ionised calcium determination is required to assess calcium status in CKD cats.42,46
Blood pressure measurement
Hypertension frequently complicates CKD (20–65% of cases)53–55 and renal dysfunction is the most common underlying cause of feline hypertension (31.9–87% of cases).56–59 In humans, hypertension is considered to be both a cause and a consequence of CKD and a contributor to progressive CKD. 60 Similarly, and regardless of the underlying cause of hypertension, azotaemia is observed in many hypertensive cats.56,58,59,61 Idiopathic hypertension is diagnosed in 13–20% of hypertensive cats; however, it is uncertain whether these non-azotaemic and non-hyperthyroid cats have primary hypertension or hypertension secondary to early, subclinical, non-azotaemic CKD.57–59,62
Although an association between hypertension and progressive kidney disease is generally presumed, it remains uncertain whether feline systemic hypertension might cause CKD and what role it plays in CKD progression. 62 Nevertheless, blood pressure should be measured in all cats with kidney disease and renal function should be assessed in all hypertensive cats.58,61,63

Techniques for blood pressure measurement are reviewed in detail elsewhere.63,64 Because it is inexpensive, easy and accurate, the Doppler ultrasonic technique (Figure 3) is the most suitable method for indirect systolic blood pressure (SBP) measurement in practice. Oscillometric techniques are less accurate in cats, but may be advantageous in patients that prefer minimal restraint.63,65 Hypertension is considered and further diagnostic tests are advised if SBP, measured with a Doppler device, exceeds 160 mmHg on repeated occasions or on a single occasion with clinical manifestations of hypertension.63,64,66

Systolic blood pressure measurement in cats using the Doppler ultrasonic technique; in a sitting position (a), in sternal recumbency (b) and in lateral recumbency (c). Points of note are to restrain the cat gently in a comfortable position and to hold the cuff at the level of the heart base. In cats, the authors always use headphones to avoid stress hypertension due to the sounds of the Doppler machine and to improve audibility of the Doppler sounds

Dorsal ultrasound view of the right kidney of a 13-year-old Persian cat with PKD. The kidney is severely enlarged (length >6 cm) and contains multiple, well-defined cysts of different sizes. The presence of multiple cysts in the cortex and medulla results in complete distortion of the normal kidney structureMedical imaging

Sagittal (a) and transverse (b) ultrasound views of the left kidney of a 1-year-old domestic shorthair cat with ureteral obstruction due to ligation. Note the moderate pyelectasia (3.6 mm) and dilation of the proximal ureter (1.8 mm). The kidney was mildly enlarged (4.6 cm)

Dorsal (a) and transverse (b) ultrasound views of the right kidney of a 9-year-old domestic shorthair cat with renal lymphoma. The hyperechoic cortex is surrounded by a thick hypoechoic subcapsular halo. The kidney was severely enlarged (6.5 cm)

Left–right lateral (a) and ventrodorsal (b) projections of the abdomen of a 4-year-old domestic shorthair cat with acute-on-chronic renal failure and nephrolithiasis. Both kidneys are small and markedly irregular in outline. An asymmetry in kidney size is visible. The kidneys both contain radiopaque, well-defined, mineralised elements (nephroliths) in the pelvic area
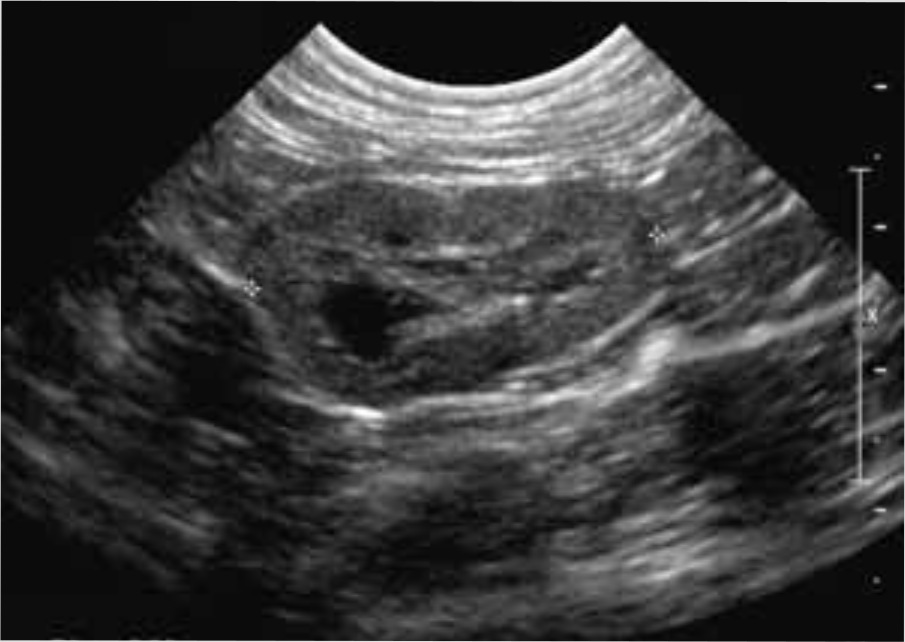
Dorsal ultrasound view of the left kidney of a 7-year-old British Shorthair cat with IRIS stage 3 CKD. The left kidney is decreased in size (2.9 cm) and irregular in outline. The cortex is hyperechoic and the corticomedullary demarcation is mildly reduced
Uncontrolled hypertension leads to end-organ damage at the level of the kidneys, heart, brain and eyes. The majority of hypertensive cats have ocular or cardiac abnormalities on physical examination.54,56–58,61,64

Glomerular filtration rate
Determination of GFR is considered to be the gold standard method for evaluation of kidney function. 72 For research purposes, plasma clearance of an intravenously administered marker, commonly iohexol or creatinine, is frequently used in cats to estimate GFR. Unfortunately, iohexol assays and injectable creatinine are not commercially available. Inulin and radioisotopes have also been used as clearance markers, but inulin assays are technically challenging and not widely available and radioisotopes require specialised equipment and carry the risk of radiation exposure.11,73 Such multi-sample techniques for GFR estimation are additionally labour-intensive, time-consuming and may be stressful or painful for the patient, which limits their practical use in cats.
However, GFR determination might be valuable for cats with doubtful renal function (eg, unexplained weight loss or polyuria/ polydipsia; IRIS stage 1 CKD patients; idiopathic hypertension; or azotaemia, poorly concentrated urine or pathological renal proteinuria as a single laboratory abnormality). GFR determination is also recommended to guide dosage adjustment for potentially toxic drugs that primarily undergo renal excretion.9,10
Several limited sampling strategies have been described to estimate feline GFR based on a reduced number of blood samples. Unfortunately, in most of the studies to date no or only few renally impaired cats were evaluated and none of the methods have been sufficiently validated in cats with CKD to be used in practice.74–82 The authors’ group is currently working on limited sampling strategies in cats with a wide range of GFR. 83
Renal biopsies
Kidney histology in cats with CKD often cannot reveal the underlying cause. However, some primary causes such as renal lymphoma, amyloidosis and FIP can usually be identified on renal biopsies, regardless of the disease stage. 3 Renal biopsy should be considered when knowledge of morphological alterations in renal structure will substantially influence patient management; for example, in cats with renal lymphoma, amyloidosis or glomerulonephritis. However, this is not true for the majority of cats that suffer from chronic generalised tubulointerstitial nephritis, glomerulosclerosis, tubular necrosis or PKD, or for cats with significant azotaemia or end-stage CKD regardless of the underlying cause.5,10
Maximum information will be obtained by evaluating kidney biopsies with light, electron and immunofluorescent microscopy, which is particularly recommended in patients with persistent severe proteinuria (UPC ?2) without severe azotaemia (IRIS stages 1 to early 3). Potential underlying diseases leading to proteinuria should be ruled out before taking kidney biopsies.29,84

Screening for early CKD
Routine health screening of aged cats is important for early detection of chronic conditions, such as CKD.15,87,88 Survival rates for cats with CKD are significantly associated with azotaemia and proteinuria, and cats diagnosed early in the disease course live longer than cats diagnosed with more severe azotaemia.6,21 So, an even better prognosis might be expected for cats diagnosed in the non-azotaemic disease stage (IRIS stage 1) because timely therapeutic intervention might prevent or delay disease progression and complications.9,89 Similarly, in humans, many adverse events of CKD such as progressive deterioration of kidney function, complications of decreased kidney function, and cardiovascular disease can be prevented or delayed by early detection and treatment.90–94
Unfortunately, diagnosis of early feline CKD is challenging. Over two-thirds of functional renal mass must be lost before kidneys lose their urine concentrating ability and over three-quarters must be lost before azotaemia develops. Thus, serum creatinine and urea concentrations and USG are often within RIs in cats with early CKD, particularly because some cats may maintain their urine concentrating ability.8,11,72
Practical, inexpensive and accurate methods to detect early feline CKD are urgently needed. Until these are available, veterinarians should improve owner awareness of early signs of CKD. Poor body condition, weight loss and polyuria/polydipsia are not always recognised by cat owners.15,95,96 Regular nutritional assessments (diet history, body weight, body and muscle condition score) may improve early detection of chronic diseases.97–99

Minimum laboratory database
The minimum laboratory database for CKD screening consists of serum creatinine, USG and proteinuria or (micro)albuminuria measurements.15,87,88 If a blood pressure device is not available, fundoscopy may be used to investigate for the presence of hypertension. 58
Physical and laboratory parameters should be compared with values obtained at previous health screenings to detect clinically relevant changes. Increasing serum creatinine concentrations, even within the RI, may indicate early kidney dysfunction, particularly in cats with weight loss or muscle wasting or USG consistently below 1.035.9,89,95 However, many factors influence USG and daily USG fluctuations can be seen in healthy animals. Thus, low USG without other indicators of CKD does not necessarily suggest kidney dysfunction.8,9,71
In a study of healthy non-azotaemic geriatric cats, plasma creatinine concentration combined with UPC was predictive of the development of azotaemia, indicating that high–normal creatinine concentrations and/or UPC values consistent with borderline or overt proteinuria might indicate early kidney dysfunction. 37
More advanced tests
More advanced tests to evaluate kidney function might be considered in cats returning doubtful routine blood and urine test results. As discussed earlier, GFR estimation would be ideal but has important practical limitations. Some of these limitations might be avoided by limited sampling strategies, but research is ongoing. Because, in daily practice, detection of early renal dysfunction is more important than knowing the exact GFR value, the authors’ group recently developed cut-off concentrations for creatinine, exo-iohexol and endo-iohexol at three time points after intravenous injection of iohexol and creatinine. These cut-off points identified cats with low GFR with high sensitivity and specificity. 103
Studies evaluating the value of serum cystatin C (sCysC) as an indirect marker of GFR in cats are also ongoing. Cystatin C, a low molecular weight protein produced at a constant rate by all nucleated cells, meets the criteria required for endogenous GFR markers. 104 Serum CysC is superior to serum creatinine for the detection of renal dysfunction in humans 104 and also has some advantages over serum creatinine in dogs. 105 Cats with CKD have higher sCysC concentrations compared with healthy cats,106,107 but evidence showing advantages of sCysC over serum creatinine in the detection of early feline CKD is currently not available.
Another pathway for identifying kidney disease involves the use of urinary biomarkers for tubular or glomerular damage.108,109 Retinol binding protein (RBP), N-acetyl-β-glucosaminidase (NAG) activity, urinary cystatin C (uCysC), transforming growth factor-β1 (TGF-β1), interleukin-8 (IL-8) and (micro)albuminuria (see above) are promising candidate urinary biomarkers for cats.31,32,36,107,109–114 Low molecular weight proteins (NAG, uCysC, RBP) and tubular enzymes (NAG) are not present in the urine of healthy animals, whereas patients with CKD might have detectable urinary concentrations secondary to tubulointerstitial damage or inflammation. Tubulointerstitial inflammation or fibrosis might also result in overexpression and increased urinary concentrations of inflammatory cytokines (TGF-β1, IL-8).108,109
In humans, careful selection of biomarkers allows detection of site-specific changes (glomerular versus tubular). 108 Whether the same is true in cats, and whether these urinary biomarkers have benefit over routine parameters in the detection of early feline CKD, is currently unknown.
Footnotes
Key Points
Funding
The preparation of this article was supported by an educational grant from Boehringer Ingelheim.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
