Abstract
Practical relevance:
Chronic kidney disease (CKD) is one of the most frequently encountered disorders in cats, having increased in prevalence in recent decades. Although the underlying cause is rarely identified, the common final outcome of feline CKD is tubulointerstitial fibrosis. Knowledge of CKD pathophysiology is necessary for optimal individualised patient management, especially with regard to diagnosis and treatment of extrarenal complications.
Patient group:
CKD is most common in senior and geriatric cats, but should be considered in any feline patient with ureterolithiasis, hyperthyroidism, retrovirus infection, systemic hypertension, cardiovascular disease or urinary tract infection.
Evidence base:
Most of our knowledge of the pathogenesis of CKD is extrapolated from human nephrology and experimental animal studies. There is, therefore, a need for further studies in cats. The prevalence of clinical signs in feline CKD is well documented. Several concurrent diseases associated with CKD have also been reported in cats, especially in the geriatric population, but there is no or only limited published evidence demonstrating a cause-and-effect relationship between most of these conditions and CKD. Studies performed over the past 15 years have nevertheless allowed identification of major risk factors (proteinuria, plasma phosphate and plasma creatinine) influencing the progression of feline CKD.
Clinical challenges:
Clinical signs occur in the late stages of renal disease, so populations at higher risk of CKD should be screened routinely. CKD-associated complications (systemic hypertension, secondary renal hyperparathyroidism, hypokalaemia, anaemia, metabolic acidosis) must not be overlooked as they may affect the progression of disease. Disease progression is itself unpredictable and renal function may remain stable for extended periods. Most cats with early CKD do not progress to end-stage CKD before they die.
Audience:
General practitioners play a major role in screening feline patients at risk of development or progression of CKD.
Introduction
Chronic kidney disease (CKD) is the most common renal disease in cats and is defined as structural and/or functional impairment of one or both kidneys that has been present for more than 3 months. 1 CKD may result from any condition that causes progressive and irreversible damage to the kidneys and indeed is intimately associated with an entire spectrum of diseases that occur following initiation of kidney damage. The severity of CKD is denoted by the International Renal Interest Society (IRIS) staging system according to the degree of azotaemia, with stage 1 representing non-azotaemic disease, through to stage 4 representing severe end-stage renal azotaemia.
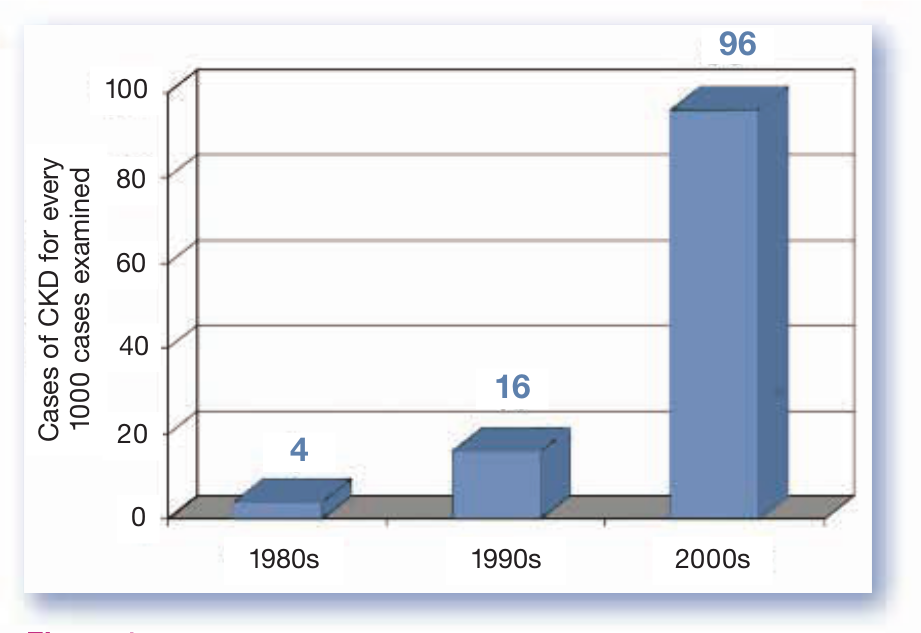
The prevalence of feline CKD is high and seems to be continuing to rise rapidly (Figure 1).
Aetiology of feline CKD
CKD is a non-specific term that does not indicate the cause of kidney damage and/or impaired kidney function. The causes are heterogeneous and most often not identified. They can be divided into two groups – congenital and acquired.
Congenital diseases
Congenital diseases are present at birth and include inherited disorders. The most common genetic disease in cats is autosomal dominant polycystic kidney disease (Figure 2), which in the past has affected up to 38% of Persian and Persian-cross cats (recent data suggest the prevalence is lower as a result of selective breeding efforts). 4 Other congenital feline kidney diseases are renal amyloidosis in Abyssinian, Siamese and Oriental cats, 4 glomerular disease in Abyssinian cats 5 and juvenile renal dysplasia. 6

Ultrasonographic appearance of a polycystic kidney in a cat
Acquired diseases
Acquired diseases have been identified, or are suspected, in many cases of CKD in cats.
Hyperthyroidism can mask and even exacerbate co-existing CKD, with renal disease becoming apparent in 15.3–39% of cats after treatment of hyperthyroidism.12,14,15 Proteinuria is an indicator of all-cause mortality in hyperthyroid cats after treatment but not a mediator of progression of CKD in these patients. 15
Exclusive feeding of an inappropriate

’Big kidney–little kidney’ syndrome in an azotaemic cat. Abdominal radiographic findings such as these are indicative of ureteral obstruction
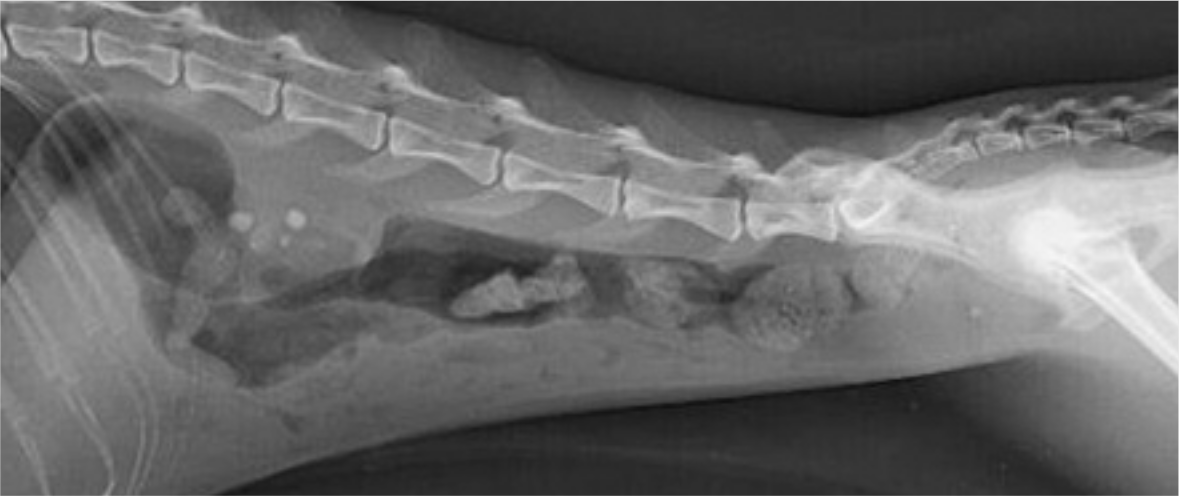
Abdominal radiograph showing multiple nephroliths in a cat with advanced CKD

Cytology of a fine-needle aspirate from the kidney of a cat with renal lymphoma. Large blastic lymphoid cells typically have a high nucleocytoplasmic ratio, microvacuolated basophilic cytoplasm and a round nucleus with reticulated chromatin and prominent basophilic nucleoli
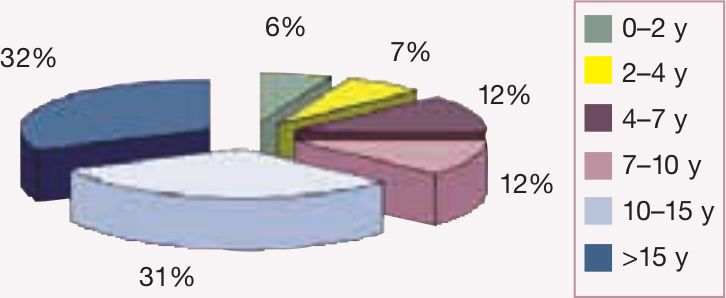
Distribution of ‘renal failure’ in cats of different ages based on 2228 case records from patients diagnosed at 23 US veterinary colleges 3

Pathogenesis of feline CKD
Tubulointerstitial fibrosis appears to be the most common final outcome of feline CKD (Figures 7 and 8).2,3,10,48 Primary glomerular diseases are rare – glomerular disease develops secondarily to systemic disease in most cases, specifically neoplastic (eg, leukaemia, lymphoma), infectious and non-infectious inflammatory disorders (eg, pancreatitis, immune-mediated diseases). 49 The presence of interstitial fibrosis is the strongest histomorphometric predictor of plasma creatinine concentration. 38 Tubular damage occurs early in the course of disease, before elevations in serum urea and creatinine concentrations. 50 Although cats are able to maintain urine concentrating ability after subtotal nephrectomy, 51 urine specific gravity decreases as plasma creatinine increases in cats with CKD,52,53 suggesting a close relationship between glomerular and tubular function during the progression of feline CKD.

Distribution of renal lesions in 64 cats with CKD 10

Renal histology in a cat with end-stage CKD. Note the interstitial inflammation, fibrosis and tubule dilation. H&E, original magnification x200. Courtesy of Dr A Poujade and Dr C Dally, Laboratoire d’Anatomie Pathologique Vétérinaire du Sud-Ouest, Toulouse, France
Although most of the factors that initiate kidney damage remain unknown in cats, it is hypothesised that the process is similar to that described in other species (see box below). 54
Sustained and chronic kidney injury induces irreversible inflammation with infiltration of inflammatory cells, which in turn serves as a primer that initiates renal fibrogenesis. Inflammatory cells produce profibrotic cytokines (eg, transforming growth factor [TGF-β1]) which promote fibrogenesis by activation of matrix-producing cells. Urine excretion of TGF-β1 is increased in feline CKD. 55
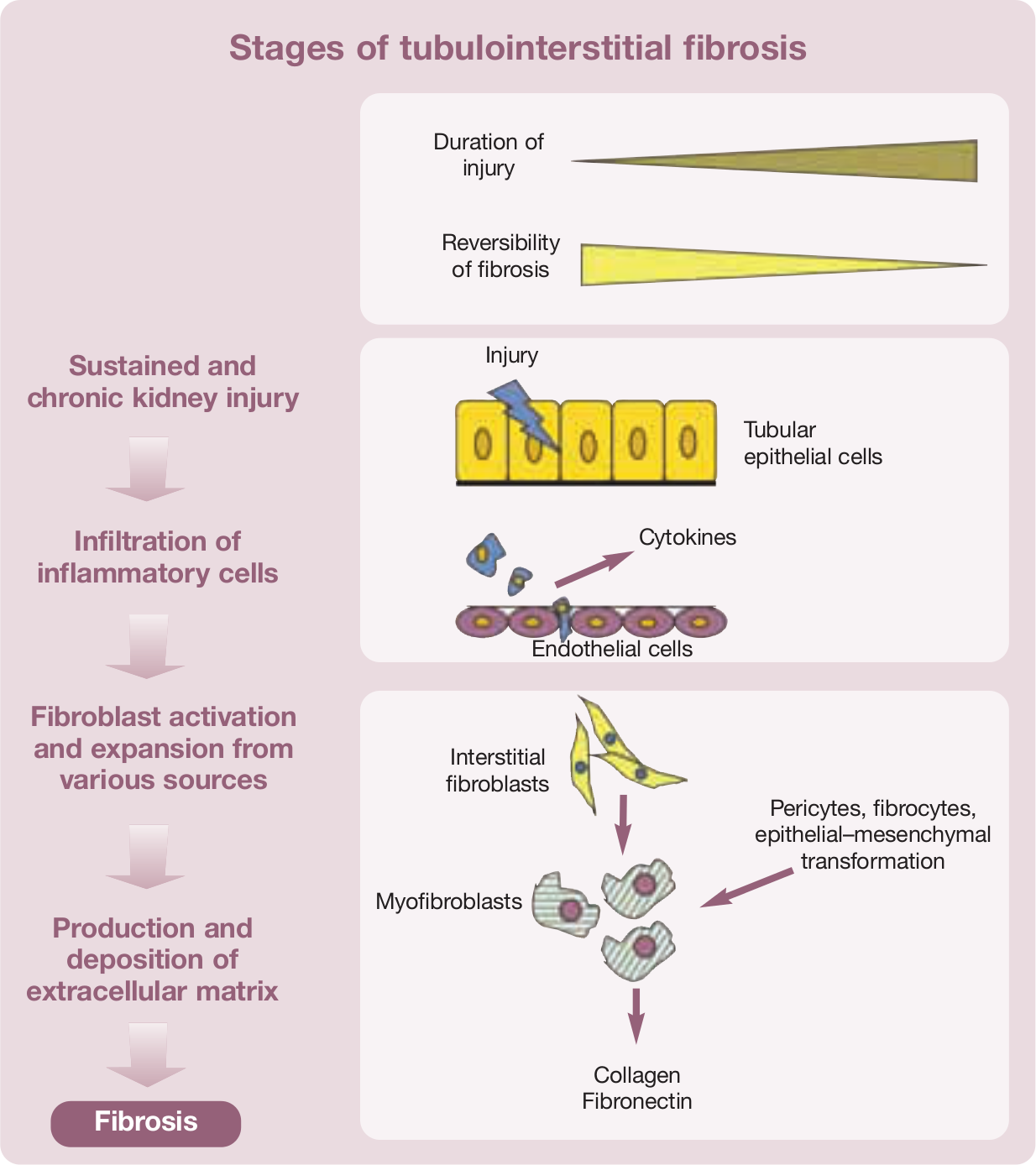
Activated fibroblasts, also referred to as myofibroblasts, produce large amounts of extracellular matrix components (collagen, fibronectin). Other sources of myofibroblasts are pericytes, fibrocytes and tubular epithelial cells that undergo epithelial–mesenchymal transformation. Myofibroblasts express α-smooth muscle actin and vimentin. In cats with CKD, interstitial expression of these markers correlates with the extent of tubulointerstitial lesions, interstitial fibrosis, interstitial fibronectin deposition and renal dysfunction.56,57 Chronic hypoxia is often also present and is associated with increased oxidative stress, 54 which is likely to be important in the pathogenesis of feline CKD.58,59
The renin–angiotensin–aldosterone system (RAAS) is additionally activated in cats with naturally occurring or experimental CKD.39,60–63 Angiotensin II induces vasoconstriction of the efferent arteriole (and consequently glomerular hypertension) and accumulation of extracellular matrix mediated by TGF-β1. 64 In cats with CKD, angiotensin-converting enzyme (ACE) inhibitors decrease glomerular capillary pressure, the ratio of efferent to afferent arteriolar vascular resistance and proteinuria, but have not been shown to improve histopathological changes in the kidneys or survival times.46,65 Insufficient statistical power might, however, explain the lack of effect on survival. Aldosterone could also contribute to the development of CKD via several pathogenic mechanisms (see box on the right). 66
Progression of feline CKD
The progression of CKD is postulated to result from a self-perpetuating vicious cycle. It is attributed to maladaptive functional and structural changes in surviving (or remnant) nephrons after renal injury; in cats with experimental CKD, these are principally increases in single nephron glomerular filtration rate (GFR), glomerular pressure and glomerular volume. 67
Because of the large reserve capacity of the kidneys, at least 75% of renal function needs to be impaired before azotaemia is detected. 68 In cats, GFR remains stable for up to 1 year after induction of experimental CKD.69–71 Cats with spontaneous CKD show an unpredictable time course of the disease, with extended periods of stable renal function before decompensation (Figures 9 and 10).27,40,72,73 In one study, for example, 81% and 37% of cats with stages 2 and 3 CKD, respectively, did not progress to stage 4 before they died. 73 The median survival time for advanced stage 2 (serum creatinine 205–250 µmol/l, 2.3–2.8 mg/dl), 3 (serum creatinine 251–440 µmol/l, 2.9–5.0 mg/dl) and 4 (serum creatinine >440 µmol/l, >5.0 mg/dl) was estimated at 1151, 778 and 103 days, respectively, but large individual variation was observed. 33


Percentage of cats with progression of CKD (ie, ≥25% increase in plasma creatinine concentration relative to the concentration at diagnosis) within 1 year of diagnosis, according to IRIS stage of renal disease 73

Decline in glomerular filtration rate (dotted line) during the progression of CKD from IRIS stage 1 to stage 4. The rate of progression varies according to the nature of the kidney damage and among individual cats. Cats in stages 2 and 3 can have stable renal function for sustained periods until decompensation
Pathophysiology of secondary changes occurring in feline CKD
The clinical signs and complications of CKD progress with declining GFR due to decreased elimination of, and hence overexposure to, endogenous substances (so-called uraemic toxins). These complications are also considered in themselves to be major contributors to the progression of renal disease, and increase the risk for morbidity and mortality in patients with CKD.
Systemic hypertension
Systemic hypertension is commonly associated with CKD. The pathogenesis of hypertension in cats with CKD is multifactorial and includes impaired excretion of sodium, RAAS activation, stimulation of the sympathetic nervous system, arterial structural changes, endothelial dysfunction and oxidative stress. 35 Systemic hypertension may adversely affect renal function by inducing glomerular hypertension and proteinuria; it also increases the risk of other end-organ damage (ie, ocular, neurological and cardiac) (Figure 11). 75
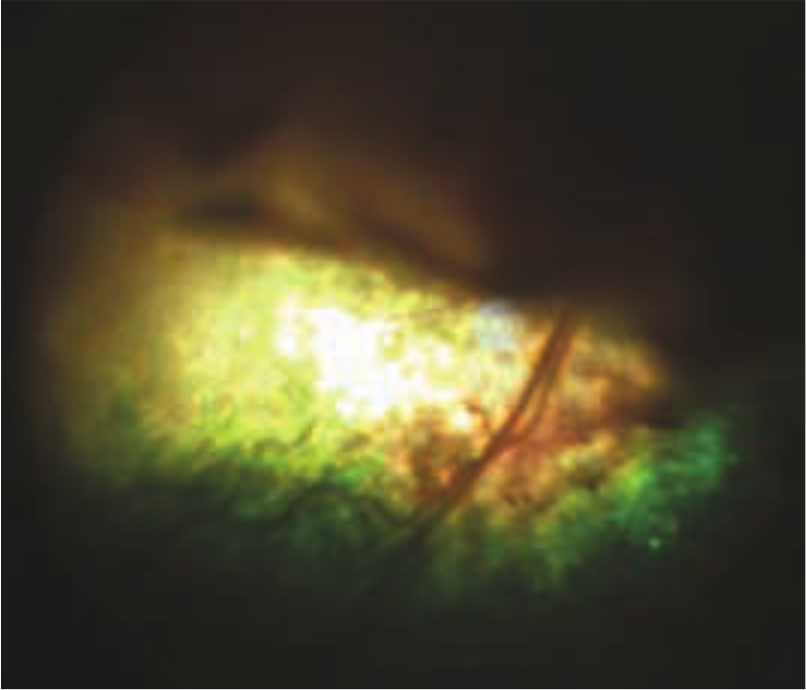
Dorsal retinal detachment in a cat with systemic arterial hypertension
Renal secondary hyperparathyroidism
Renal secondary hyperparathyroidism (RHPTH) is an adaptive and ultimately maladaptive process that develops in response to declining kidney function, phosphate retention and failure to bioactivate vitamin D (see box above). 76 It is well recognised that phosphate retention is a major contributor to the progression of CKD. Hyperphosphataemia and increased plasma parathyroid hormone (PTH) concentrations are observed in 13–100% and 47–100%, respectively, of cats with CKD.3,53,77 RHPTH is particularly common in stages 3 and 4 CKD, as the severity of RHPTH increases with the degree of azotaemia.3,53,77 Plasma fibroblast growth factor 23 (FGF-23) is a phosphaturic hormone involved in the pathogenesis of RHPTH in humans. In cats with CKD, plasma FGF-23 is strongly correlated with plasma creatinine concentration and PTH, whereas there is an inverse relationship between FGF-23 and GFR.78,79

Clinical signs induced by RHPTH may result from calcium phosphate precipitation into the tissues (Figure 12) but are relatively uncommon in cats with CKD. Metastatic paw calcification 80 and osteoporosis 10 have been reported.

Soft tissue mineralisation in the tongue of a cat with end-stage CKD. H&E, original magnification x200
Hypokalaemia
Hypokalaemia is a common finding in cats with CKD (prevalence 18–30%), especially those in IRIS stages 2 and 3.3,10,26,27,52,81 CKD is also the most common cause of hypokalaemia in cats. Cats with severe hypokalaemia are 3.5 times more likely to have CKD. 82 Hypokalaemia in feline CKD may result from multiple causes (see box below). It may also develop in renal-impaired cats fed a high protein diet. 83 Potassium depletion may induce metabolic acidosis, kidney dysfunction (hypokalaemic nephropathy), muscle weakness and cardiac arrhythmia.81,84 Therefore, CKD could lead to potassium depletion, which would in turn contribute to progression of the renal disease and worsening of the clinical condition.
By contrast, hyperkalaemia is uncommon in cats with CKD (prevalence 5.8%),46,53 except in end-stage oliguric CKD. 81
Anaemia of renal disease
A normochromic, normocytic, hypoproliferative anaemia is frequently diagnosed in cats with CKD, especially in end stages of the disease;85,86 about 30–65% of cats with CKD develop anaemia during progression of their renal disease. 86 The PCV decreases as the severity of CKD increases.52,53 CKD-induced anaemia results mainly from inadequate production of erythropoietin by the peritubular fibroblast type-1 interstitial cells. Other contributing factors to anaemia include malnutrition (eg, iron deficiency), shortened red blood cell life span, myelofibrosis, metabolic alterations affecting erythropoiesis, uraemic toxins, hyperparathyroidism and gastrointestinal blood loss. 86 Anaemia adversely affects the quality of life of cats with CKD. 86

Metabolic acidosis
Metabolic acidosis is also frequent, with a prevalence of 22–88% dependent on the severity of CKD.3,10,52 Metabolic acidosis generally occurs late in the time course of CKD. 72 An increase in anion gap due to low plasma bicarbonate and chloride concentrations is observed, 52 with development of hypochloraemia preceding acidosis. 72 Metabolic acidosis may result from potassium depletion, 84 but hypokalaemic cats are generally able to maintain normal venous blood pH and bicarbonate concentration. 52 Renal ammoniagenesis, the main renal process for urinary hydrogen ion excretion, could also be impaired in CKD. 52 Hypochloraemia may result from the loss of chloride through the gastrointestinal tract due to gastric hyperacidity or vomiting. 72
Metabolic acidosis promotes many adverse clinical effects in the renal patient, including anorexia, vomiting, lethargy, weakness and malnutrition. 27
Role of proteinuria in disease progression
Proteinuria is a sign of kidney damage, but also a strong indicator for progression of CKD.87,88 Small proteins, such as albumin, are filtered by the glomeruli and reabsorbed by the proximal tubule cells by receptor-mediated endocytosis. Dysfunction of these two processes results in proteinuria. Proteinuria accelerates progression of CKD by induction of tubular chemokine expression and complement activation; this, in turn, leads to inflammation and fibrogenesis. Proteinuria is associated with interstitial fibrosis and glomerular hypertrophy, 38 and might, therefore, be a marker of tubular dysfunction associated with tubulointerstitial fibrosis.
Proteinuria in cats with naturally occurring CKD is generally mild compared with their human counterparts,31,34,46 with 90% and 49% of cats with CKD having a UPC of <1.0 and <0.25, respectively. 31 The severity of proteinuria, however, has prognostic significance in terms of survival time (Figure 13).31,46 Despite proteinuria being generally mild, the ACVIM consensus statement on the treatment of proteinuria recommends therapeutic intervention when UPC ≥0.4 in cats with CKD causing azotaemia. 87 Plasma creatinine concentration and SBP are positively correlated with increasing UPC. 31

Effect of proteinuria on survival in cats with CKD. 31 The hazard ratio for death or euthanasia was 2.9 and 4.0 for a UPC of 0.2–0.4 and >0.4, respectively, compared with the baseline group (UPC <0.2)
Whether proteinuria is contributing to CKD progression or whether proteinuric CKD is intrinsically more rapidly progressive remains unknown.
Major clinical signs of feline CKD
The prevalence of clinical signs in cats with CKD is presented in Tables 2 and 3.
Survival time in relation to various clinical criteria

PCV = packed cell volume
Adapted from Boyd et al 33
Prevalence of clinical signs detected by owners in cats with CKD
Maximum percentage of cases showing the clinical sign.
Prevalence of physical examination findings in cats with CKD

Maximum percentage of cases showing the clinical sign.
Clinical signs may be non-specific and manifest late in the disease process, once major renal tissue damage has occurred. Many cats remain almost symptom-free until they reach advanced stages of CKD. The frequency of clinical signs is similar in IRIS stages 2 and 3, but stage 4 is associated with increased frequency and severity of signs. 46
Polyuria and polydipsia represent the earliest and most common clinical manifestations of CKD. 27 Cats with CKD are more likely to have had polydipsia and polyuria in the year before they are diagnosed. 89 Polydipsia, which is a compensatory response to polyuria, is more easily recognised by owners. In advanced stages, polyuria results in dehydration as fluid loss from the body exceeds fluid intake.
Gastrointestinal signs are the most prominent indicators of uraemia and probably represent the main reason for the owner to consult a veterinarian. Hypergastrinaemia, reported in cats with CKD, 90 may induce gastric hyperacidity, uraemic gastritis, gastrointestinal bleeding, anorexia and vomiting. While diarrhoea is uncommon, constipation is more frequent in cats with CKD than in dogs. It is attributable to dehydration but also to the use of phosphate binding agents. 91
Other reported uraemic clinical complications are lethargy, weakness, depression, tremor, myoclonus, seizures, myopathies, uraemic pericarditis and pneumonitis, hypothermia, anaemia and renal osteodystrophy. These disorders are all multifactorial and their pathogenesis has not generally been investigated in cats.
Footnotes
Key Points
Funding
The preparation of this article was supported by an educational grant from Boehringer Ingelheim.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
