Abstract
Overview:
Toxoplasma gondii infection is common in cats, but the clinical disease is rare. Up to 50% of cats, especially free-roaming ones, have antibodies indicating infection and the presence of cystic stages.
Disease signs:
Clinical signs only appear in few cats when they become immunosuppressed – in these situations cystic stages can be reactivated. Commonly affected are the central nervous system (CNS), muscles, lungs and eyes.
Human infection:
Cats can pose a risk for humans when they shed oocysts. However, this happens only once in their lifetime, usually only for 3–10 days after ingestion of tissue cysts. Thus, cats that have antibodies to T gondii no longer shed oocysts, and do not pose a risk to humans.
Agent properties
Toxoplasma gondii is an obligate intracellular coccidian parasite that can infect virtually all species of warm-blooded animals, including people. Domestic cats and other felids are the natural hosts – non-feline species serve only as intermediates.1,2
Three infectious structures can be distinguished: sporozoites in oocysts, tachyzoites (the actively multiplying stage), and bradyzoites (the slowly multiplying stage) enclosed in tissue cysts. Oocysts are excreted in faeces, whereas tachyzoites and bradyzoites are found in tissues and milk.1,2
Pathogenesis
Enteroepithelial life cycle
This cycle is found only in the feline host. Most cats are infected by ingesting intermediate hosts – typically rodents – infected with tissue cysts. Bradyzoites are released in the stomach and intestine from the tissue cysts when digestive enzymes dissolve the cyst wall. They enter epithelial cells of the small intestine and give rise to schizonts, initiate five types of predetermined asexual stages, and merozoites released from the schizonts eventually form male and female gamonts. After fertilisation, a wall is formed around the fertilised macrogamont to form an oocyst. Oocysts are round to oval, 10 x 12 μm in size, and are still unsporulated (not infectious) when passed in faeces. After exposure to air and moisture for 1–5 days they sporulate to contain two sporocysts, each with four sporozoites.1,2

The cycle is usually completed within 3–10 days of ingestion of tissue cysts, which is the route of infection in up to 97% of naive cats. In the rare event that cats ingest oocysts or tachyzoites, formation of new oocysts is delayed and shedding can occur for up to 18 days (occasionally longer). However, only 20% of cats fed oocysts will shed.1,2
Extraintestinal life cycle
The extraintestinal development of T gondii is the same for all hosts, including cats, dogs, and people, irrespective of whether tissue cysts or oocysts have been ingested. After the ingestion of oocysts, sporozoites hatch in the lumen of the small intestine and enter intestinal cells, including those in the lamina propria. Sporozoites divide into two by an asexual process known as endodyogeny, thereby becoming tachyzoites. These are lunate (falciform) in shape, approximately 6 x 2 μm, and multiply in almost any cell of the body. When the cell ruptures, releasing the tachyzoites, these infect new cells. Otherwise, tachyzoites multiply intracellularly for an undetermined period, and eventually encyst. Tissue cysts vary in size from 15–60 μm and usually conform to the shape of the parasitised cell. Tissue cysts are formed mainly in the CNS, muscles and visceral organs, and probably persist for the life of the host. They can be reactivated after immunosuppression, which may then lead to clinical signs.1,2
Parasitaemia during pregnancy of the host can cause placentitis and spread of tachyzoites to the fetus. Many kittens born to queens infected with T gondii during gestation become infected transplacentally or when suckling. Clinical signs are common in these kittens, varying with the stage of gestation at the time of infection; some of these newborn kittens shed oocysts.1,2
Epidemiology
Antibody prevalence to T gondii varies geographically: in Portugal, 24% of cats had antibodies in one study; 3 in the USA, 16–40% were antibody-positive, depending on the state. 2 However, only 3/326 faecal samples from cats in California and 1/252 in Switzerland contained T gondii oocysts. 4 The annual burden in the environment is about 90–5000 oocysts per square metre. 5 The age of the cat does not play a role in the frequency of T gondii shedding, but the season does: shedding is more common between July and December in the northern hemisphere. 6
The three major modes of transmission of T gondii in all host species are congenital infection, ingestion of infected tissue, and ingestion of oocyst-contaminated food or water. 2 Less important are blood transfusions and organ transplantations.1,2 Lactogenic transmission is suspected because the organism has been detected in queen’s milk. 7
T gondii blocks the innate aversion of rats for cat urine, instead making them attracted by the feline pheromone, which can increase the likelihood of a cat capturing an infected rat. This reflects adaptive ‘behavioural manipulation’ by T gondii in optimising the chances of completing the parasite’s life cycle: it reproduces only in the feline intestine. The behavioural manipulation hypothesis postulates that a parasite will specifically manipulate host conduct essential for its transmission. However, the neural circuits for innate fear, anxiety and acquired fright all overlap, raising the possibility that T gondii can disrupt all of these non-specifically. 8 Experimental infections have shown that T gondii can change chemical messages in the CNS that affect rodent behaviour because the infection can lead to cyst formation in the CNS with production of tyrosine hydroxylase, resulting in a lack of dopamine.9 –14
Meat contaminated with T gondii cysts has been the primary source of infection in people, and antibody prevalence in humans is relatively high. Exposure from oocyst-contaminated soil or water is common. Indeed, water-borne outbreaks of toxoplasmosis have been reported worldwide and support the theory that exposure to environmental oocysts poses a significant health risk. 15
Clinical signs
Clinical signs develop very rarely in infected cats and are caused by inflammation and tissue necrosis resulting from intracellular growth of tachyzoites. 2 Congenital infection tends to be more serious than infection of the adult cat. 2
Clinical toxoplasmosis develops during dissemination and intracellular replication of tachyzoites. It usually originates from reactivation of a latent infection rather than after a newly acquired infection. If a carrier cat is immunosuppressed, bradyzoites in tissue cysts replicate rapidly and disseminate again as tachyzoites. Clinical toxoplasmosis has been documented in some cats infected with feline immunodeficiency virus (FIV) or feline leukaemia virus (FeLV) [
The most commonly affected tissues are the CNS, muscles (Figure 1), lungs (Figure 2) and eyes. Hepatic and pancreatic involvement is less likely. Cats with toxoplasmosis show neurological signs (eg, seizures, ataxia), muscle hyperaesthesia, dyspnoea, uveitis, icterus, diarrhoea, fever, depression, anorexia and weight loss.
2
Transplacentally or lactogenically infected kittens develop more severe signs and frequently die of pulmonary or hepatic disease [

Myositis in a cat caused by T gondii cysts. The cat presented in lateral recumbency, was unable to get up, and showed severe muscle hyperaesthesia. Courtesy of Katrin Hartmann, Ludwig Maximilians University, Munich, Germany

Thoracic radiograph (laterolateral view) of a cat with pulmonary toxoplasmosis. Courtesy of Katrin Hartmann, Ludwig Maximilians University, Munich, Germany
Immunity
Immunity to T gondii in the cat is poorly understood. In the mouse and in humans, it is highly dependent on cell-mediated effector responses. 23
All infected cats develop IgG and IgA antibodies; about 80% also have IgM antibodies. IgG can take 4–6 weeks to appear, and maximal antibody titres are achieved within 2–3 weeks of first appearance. 2

Diagnosis
Oocyst shedding is diagnosed by microscopy of faecal samples. Diagnosis of the disease is only confirmed when the organism is found in body fluids or tissue. If suitable samples cannot be taken, a tentative diagnosis is sometimes based on rising IgM titres, exclusion of other causes for the clinical signs, and a favourable clinical response to anti-T gondii drugs [
Detection of oocysts in faeces
T gondii oocysts are 10 μm in size and best demonstrated by centrifugation using Sheather’s sugar solution (saccharose solution with a specific gravity of 1.27 g/ml) during the shedding period. T gondii oocysts are morphologically indistinguishable from those of Hammondia hammondi, Besnoitia oryctofelisi and Besnoitia darlingi. 2 A cesium chloride method for easy purification of T gondii oocysts from faeces of infected cats has been described. 15
Detection of tachyzoites
Ante-mortem diagnosis of clinical toxoplasmosis ideally is based on the detection of the organism by cytology or polymerase chain reaction (PCR) [
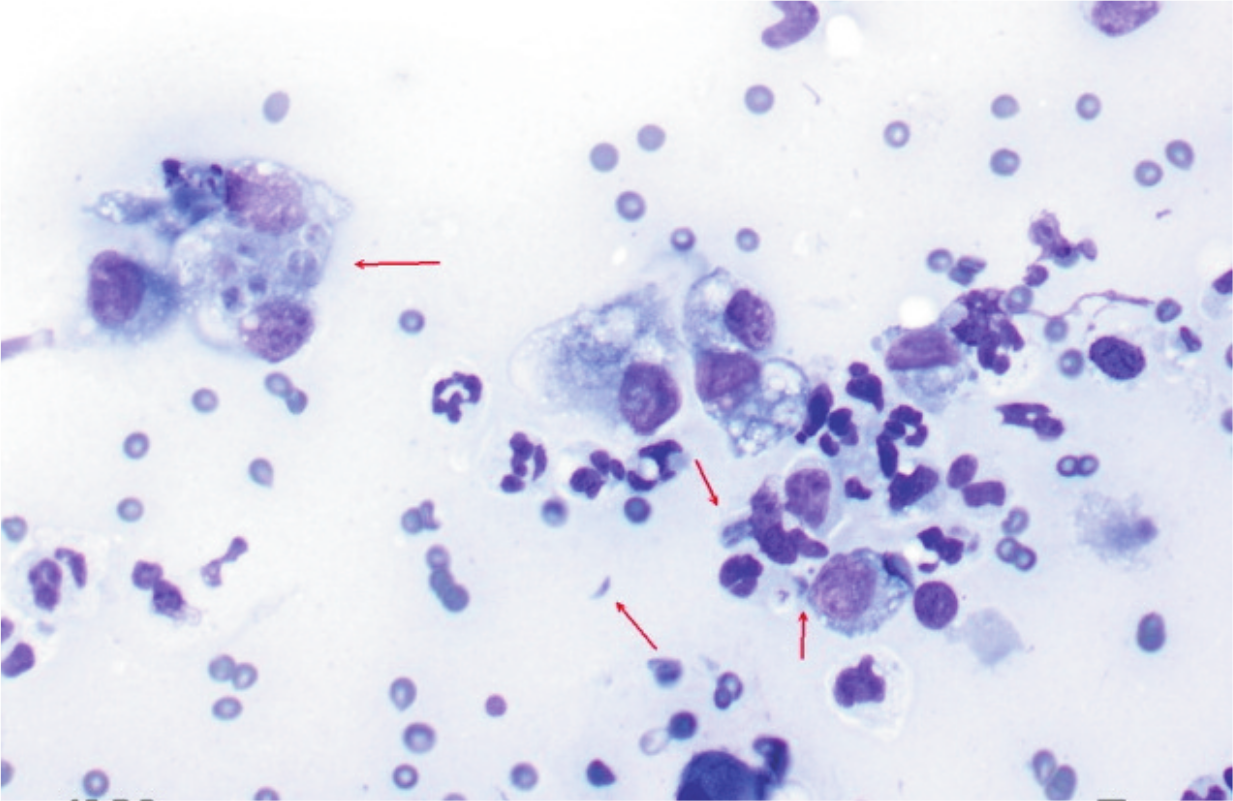
Cytology of a fine-needle aspirate from a cat with pulmonary toxoplasmosis and lung consolidation, showing numerous intracellular and extracellular T gondii tachyzoites and cysts (arrows). Courtesy of George Reppas, Vetnostics, Australia
Detection of antibodies
Using the immunofluorescence assay (IFA), antibodies of the IgM, IgG and IgA isotypes can be detected. For assessing human health risks, antibody test results from healthy cats are useful. An antibody-negative cat can be shedding oocysts (early during infection, before antibodies have had time to develop) and will likely shed oocysts if exposed for the first time. An antibody-positive cat does not shed oocysts: antibodies need 2–3 weeks to develop, by which time cats usually no longer shed; and a cat sheds only once in its lifetime. It is also unlikely to shed oocysts if re-exposed or immunosuppressed [
Antibodies are commonly found in both healthy and sick cats. Thus, their presence does not prove clinical toxoplasmosis. Antibodies of the IgM class are also commonly detected in healthy cats and do not correlate with clinical signs.
Treatment
Clindamycin is the treatment of choice
24
and should be administered at 10–12 mg/kg orally q12h for 4 weeks (Table 1) [
Treatment of toxoplasmosis

Clinical signs not involving the eyes or the CNS usually begin to resolve within the first 2–3 days of clindamycin administration. CNS and ocular toxoplasmosis tend to respond more slowly. In cases of pulmonary toxoplasmosis, radiographic abnormalities might not resolve for several weeks. The prognosis is usually poor in pulmonary or hepatic disease, particularly in immunocompromised animals. 26
Prevention of infection
Preventing toxoplasmosis in cats involves measures intended to reduce the incidence of infections and the shedding of oocysts into the environment. Cats should preferably be fed commercially available, processed food. Prevalence of feline T gondii infection is higher in countries where raw meat is fed. Freezing or irradiation can kill tissue cysts without affecting meat quality. Pets should be prevented from hunting and eating intermediate hosts (rodents) or mechanical vectors, such as cockroaches and earthworms. If meat is fed, it should be thoroughly cooked, even if frozen. Cats should be prevented from entering buildings where food-producing animals are housed or where feed storage areas are located. 1

Footnotes
Key Points
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article. The ABCD is supported by Merial, but is a scientifically independent body.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
