Abstract
Feline panleukopenia
This oldest feline viral disease known to the veterinary profession is caused by a typical parvovirus (feline panleukopenia virus, FPV) that infects a wide variety of carnivores including all felids, mustelids, procyonids and most canids. The dog and the coyote are the only carnivores that will not be infected, or will not develop disease, as the receptor on their cells will not bind the virus. 1

All parvoviruses are very stable and may stay infectious in the environment for months. They are highly contagious, which means that only a few virus particles are required to induce an infection. As an affected animal sheds enormous amounts of virus, soon the shelter or household is heavily contaminated, which causes a threat to non-vaccinated – or, rather, non-immune – animals, like cats and other carnivores.

Virus replication is restricted to mitotically active tissues, such as the bone marrow, lymphatic cells, the gut epithelium and the developing fetus. The hallmark of infection is a viraemia, which carries the virus from the initially infected cells of the tonsils and other regional cells to the target tissues. Neutralising antibodies can greatly influence the viraemia and are even able to prevent virus spread through the organism. Animals that have survived the disease are protected from reinfection, most likely for their entire life.
Vaccination is highly efficacious, and cats that have actively responded to vaccination and show antibody titres are protected for several years, and probably also life-long. However, the success of vaccination is greatly influenced by neutralising antibodies. In the case of passively acquired antibodies this has severe consequences. Maternal antibodies interfere with vaccination, and the time-point of the first vaccination is therefore important. To minimise the risk of this interference, kittens are vaccinated twice or even three times during their first weeks of life.
Primary vaccination course
A minimum of two doses – one at 8–9 weeks of age and a second 3–4 weeks later (at a minimum of 12 weeks of age) – should be administered to cats living in low-risk situations.
In higher-risk situations, a third vaccination, at 16 weeks, is recommended. Maternal antibodies may persist beyond week 12 in some cats, as field data suggest,
2
such that vaccination at 12 weeks may fail to induce protection [

Feline herpesvirus infection
Feline herpesvirus (FHV) is the agent of feline viral rhinotracheitis. Together with feline calicivirus and other pathogens, it is involved in the feline upper respiratory tract syndrome. Typical acute FHV disease results in rhinitis, conjunctivitis, and superficial and deep corneal ulcers, in particular dendritic ulcers. Latent chronic infection is the typical outcome of an acute infection, and reactivation gives rise to intermittent viral shedding in oronasal and conjunctival secretions.
Conjunctivitis may be associated with corneal ulcers, which may develop into chronic sequestra. FHV is the most important cause of corneal ulceration. 3 Stromal keratitis is a secondary, immune-mediated reaction due to the presence of virus in the epithelium or stroma. Damage to the nasal turbinates during acute disease is a predisposing factor for chronic rhinitis.
Molecular diagnosis is now in regular use, especially to identify FHV DNA in corneal samples. The practitioner should avoid the use of fluorescein and topical anaesthetics in the eye before sampling because these compounds can affect the sensitivity of some polymerase chain reaction (PCR) methods, 4 unless permitted by the diagnostic laboratory.
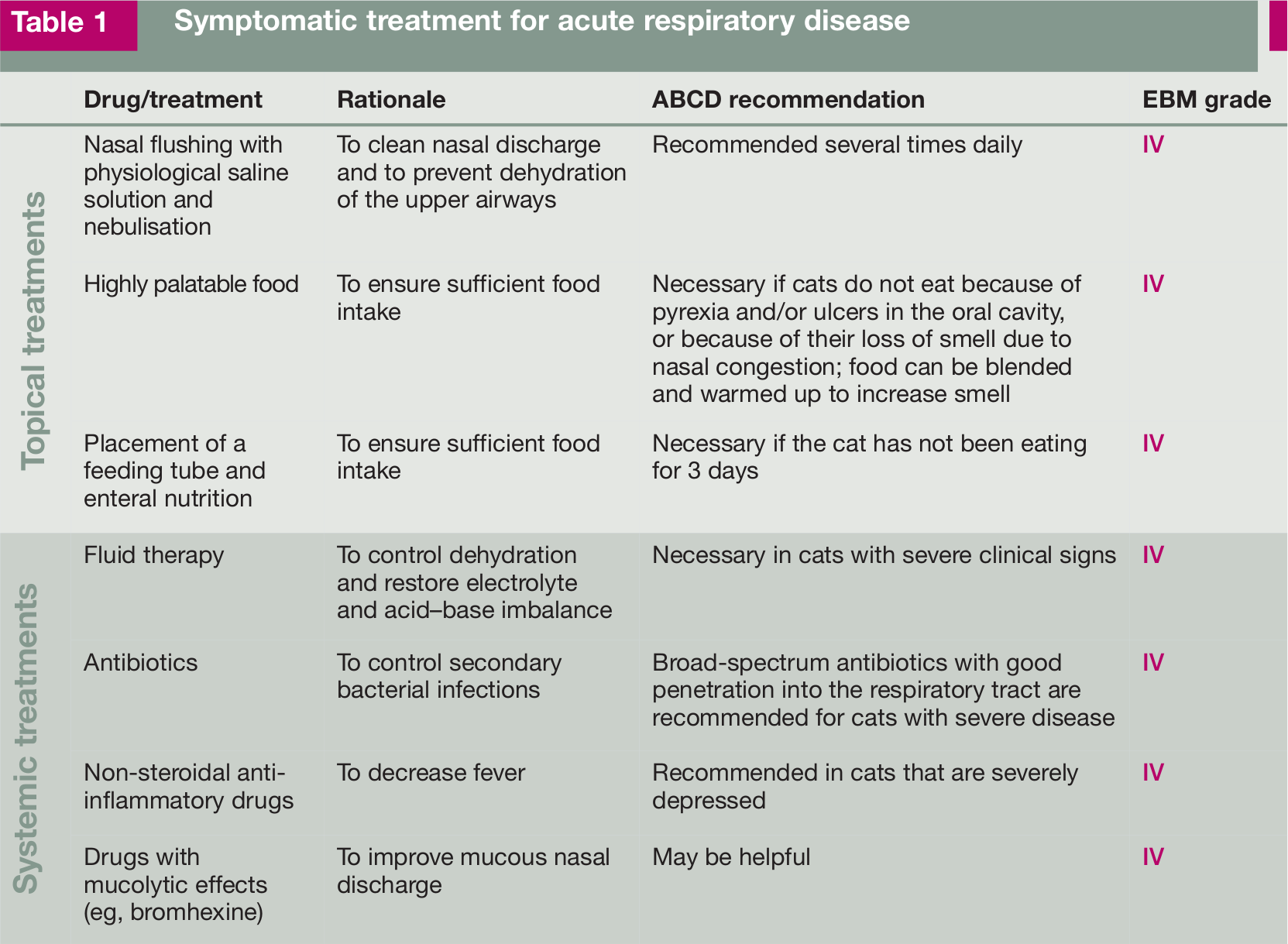
The symptomatic treatment for acute respiratory disease where FHV infection may be involved is summarised in Table 1. The symptomatic treatment for acute ocular disease is outlined in Table 2.
Symptomatic treatment for acute respiratory disease
Symptomatic treatment for acute ocular disease (conjunctivitis and keratitis)

Anti-herpesviral drugs have come to the market, which now allow recommendations to be made for the topical and systemic treatment of acute FHV ocular disease (Table 3). The drugs listed may not be readily available or licensed for cats. Other drugs have been proposed, like bromovinyldeoxyuridine, HPMA, ribavirin, valaciclovir, vidarabine, foscarnet and lactoferrin, but their efficacy has not been proven.
Antiviral drugs recommended for topical and systemic treatment of acute FHV ocular disease. The drugs are listed in decreasing order of preference

ND = not determined, SPF = specific pathogen-free
Maternally derived (passive) immunity can interfere with the response to vaccination until about 8 weeks of age [
FHV is common in multicat households, and infections can pose a problem in shelters. Management to limit the spread of infection is as important as vaccination. In shelters where incoming cats are mixed with resident cats, high infection rates are frequent. Newcomers should therefore be quarantined for the first 3 weeks, and kept individually – unless known to come from the same household.
Feline calicivirus infection
Feline calicivirus (FCV) remains an important cause of disease and a frequently diagnosed infection in both ill and healthy carrier animals. The virus evolves quickly and is very variable, particularly at antigenic sites on its surface exposed to the feline immune response. As such, many strains co-circulate, even at the level of the population of cats seen by a typical veterinary surgery. 23 As well as causing consistent oral ulceration and upper respiratory disease, the virus has also been associated with superficial ocular disease. 24 Nearly all cats with chronic lymphoplasmacytic gingivitis/stomatitis complex test positive for FCV. In addition, outbreaks of highly virulent and often lethal FCV infection in domestic cats have been described in the United States and in Europe; recently, a single outbreak has also been described in exotic captive felids in the USA.

Transmission is usually through contact with infected cats but secretions can remain infectious in the environment for many days. Fleas that have lived on infected cats have also recently been shown to shed infectious faeces, although how significant this is in the field is unknown. Diagnosis is based on clinical signs, supplemented by virus isolation or PCR. Because of the frequent asymptomatic carrier phase, care should be taken when interpreting any FCV-positive result.

Feline interferon-ω (licensed for the treatment of canine parvovirus and feline leukaemia virus infections in some European countries) has been shown to inhibit FCV replication in vitro. However, controlled field studies are lacking to support its use in cases of uncomplicated oral/respiratory disease. There is also some suggestion that strains of FCV may vary in their sensitivity to interferon. In cases of chronic stomatitis, topical oral interferon has been shown to lead to statistical improvement in clinical scores, but this improvement was generally not different from cats receiving steroid alone [
FCV vaccines provide protection mainly by inducing virus neutralising antibodies. As the virus can mutate quickly, field strains could evolve resistance to any vaccine-induced immune response, particularly if a vaccine is used for a prolonged period of time in the population. Some laboratory studies lend support to this hypothesis [
The most commonly used vaccine strains of FCV are F9, which is the oldest, isolated in the 1950s, FCV 255, and two new strains G1 and 431.
Maternally derived antibody can interfere with vaccine-induced immunity. The higher the level, the greater the interference. Where a more rapid response to vaccination is required, a modified-live vaccine may be preferred [
Dogs and cats in shelter or kennel environments should be housed separately, and flea control should be implemented to minimise the risk of transmission of FCV and other diseases. Point of care assays for identifying cats lacking protective immunity on admission to shelters have been described, 30 but their practicality is not known. They should not be used as a replacement for rigorous biosecurity.
Feline leukaemia
Feline leukaemia virus (FeLV) is a gamma-retrovirus of worldwide occurrence that is important for all small felids. Infected cats may become persistently viraemic and shed large amounts of virus through saliva and faeces, whereby they can infect other cats in close contact. Several variants of FeLV are known that differ in virulence and host cell spectrum. As is the case for all retroviruses, a DNA copy of replicating FeLV RNA is integrated into the DNA of host cells (as so-called provirus), where it remains for the lifetime of the infected cell.

Infection starts in the oropharynx from where the virus will spread via leukocytes to lymph nodes and the bone marrow. Once the bone marrow is infected, viraemia will develop and, as a consequence, FeLV replication will take place in many organs. In most cases, viraemia will be persistent and lead to clinical signs such as aplastic anaemia, immunosuppression and a variety of tumours. These cats are called progressor cats. In a fraction of infected cats, viraemia will either not develop or will be overcome after several weeks or months (regressor cats). Antibodies to FeLV and/or T lymphocytes are observed in regressor cats, suggesting that a functioning immune system is essential. Regressor cats remain latently infected for some time, and viraemia can be reactivated by stress and/or high doses of corticosteroids.

The diagnosis of FeLV infection is mostly done by in-house tests that detect the viral core protein p27. Presence of p27 is a marker for infection and correlates well with viraemia. In addition, proviral DNA can be detected through PCR techniques in blood, and viral RNA, eg, in saliva. Information that can be drawn from test results is discussed in the box above.
Treatment of viraemic cats can be tried using raltegravir, an antiretroviral drug used in AIDS treatment, at up to 40 mg/kg body weight per day. Under experimental conditions, a 15-week treatment regimen led to a strong decrease in viral loads; however, viraemia was not overcome completely [
Good (recombinant) FeLV vaccines are available, which protect from disease but not from infection. The ABCD suggests that cats at risk (eg, with access to outdoors) are primarily vaccinated. Cats living in an apartment with no contact with other cats may not need to be vaccinated. Vaccination should take place at 8–9 weeks, again at 12–13 weeks and 1 year later. Thereafter, vaccination frequency can be lowered to an interval of 2–3 years.
Feline immunodeficiency virus infection
Feline immunodeficiency virus (FIV) infection may induce clinical signs of immunodeficiency that lead to opportunistic infections or lymphomas, although in some cats the clinical signs are mild. A few studies have shown that the survival time of infected cats was not decreased; such variable outcomes are likely a result of the heterogeneity of viral isolates as well as management practices. FIV-infected cats kept in single cat households with regular veterinary care are more likely to remain healthy than those in multicat households lacking regular health checks. Prompt supportive treatment dramatically increases the survival time for FIV-infected cats.

The infection is usually diagnosed using in-house serological assays to detect antibodies against major viral proteins, and it is important that such results are properly interpreted. Situations where a confirmatory test should be used are described in the box below.
Several laboratories offer PCR tests for the diagnosis of FIV but these tests are not necessarily more reliable than serological tests. PCR tests are highly prone to contamination and must be conducted under rigorous conditions with appropriate controls to minimise false-positive results. False-negative results may arise in situations where the primers are not homologous to the infecting field strain and, therefore, infection is not detected.

Few antiviral drugs are effective against FIV and do not induce adverse side effects. Treatment with nucleoside analogues that inhibit the reverse transcriptase, such as AZT or PMEA, may reduce clinical signs but these drugs, as well as derivatives such as PMPDAP, induce anaemia. Bicyclams bind to the FIV co-receptor molecule CXCR4, thereby inhibiting viral replication. Treatment with the bicyclam AMD3100 leads to a significant decrease in proviral load in cats naturally infected with FIV and may be a useful therapy, but this treatment should not be combined with PMEA [
A whole inactivated virus vaccine has been available commercially to veterinarians in the USA since 2002, and in Australia and New Zealand since 2004. However, its use is not recommended in Europe by the ABCD since the antibodies induced interfere with the serological diagnosis of infection. Also, the vaccine has not proven effective against primary field strains of FIV circulating in Europe.
Feline rabies
Rabies is still endemic in some middle and east European countries, and in 2011 the total number of animal cases in Europe was 5801, mainly involving foxes (source WHO). However, on very rare occasions isolated rabies cases in companion animals appear also in western European countries that are considered as rabies free. Often the source of these is illegal importation of a pet from endemic areas.
Due to testing of pets for neutralising antibodies, which has been required by some countries before entering their territory, interesting data have been published about duration of the antibody response after vaccination. Though differences in efficacy between some commercial rabies vaccines used in Europe have been demonstrated,37–39 following a single vaccination these products have been shown to induce in most animals a titre above 0.5 IU/ml, the internationally accepted threshold antibody level [
There is increasing evidence that the persistence of antibody after the first rabies vaccination may be much shorter than generally believed, especially in dogs. It has been demonstrated that antibody titres fall below 0.5 IU/ml in almost 21% of dogs within 4–6 months after a single vaccination [

A non-adjuvanted recombinant rabies vaccine for cats was recently marketed in some European and other countries. It has been shown to induce immunity lasting 3 years [
Feline infectious peritonitis
The pathogenesis and epidemiology of feline infectious peritonitis (FIP), a bone of contention until recently, have been further elucidated. The hypothesis of mutants arising in individual cats upon bursts of replication (eg, under immune-suppressive stress) has been experimentally corroborated and explains the sporadic, non-epidemic occurrence of FIP. 42 Functional expression of one of the non-structural proteins (3c) is crucial for feline coronavirus (FCoV) replication in the gut, but dispensable for systemic replication of the feline infectious peritonitis virus (FIPV) mutant. While intact in all FCoVs, the 3c gene was found mutated in >70% of FIPV strains – but not in all, implying that mutation in 3c is not the (single) cause of FIP.

Most cats with FIP had no detectable intestinal FCoV and had seemingly cleared the primary infection. In those with detectable intestinal FCoV, the virus always had an intact 3c and seemed to have been acquired by FCoV superinfection. 43 Apparently, 3c-inactivated viruses replicate not at all – or only poorly – in the gut, explaining the rare incidence of FIP outbreaks. After experimental infection with FIPV strains with an intact 3c gene, the virus was shed in the faeces. Also, faecal virus from these cats was not infectious for other cats. 44
Sequencing of many different FIPV and FCoV strains revealed two alternative codons in the S gene that correlated with the FIP phenotype in >95% of cases. This again supports the internal mutation theory and might also be a basis for identifying the virulent FIPV phenotype in cats suspected of having FIP. 45
Chlamydophila felis infection
Chlamydophila felis is primarily an ocular pathogen, causing both acute and chronic conjunctivitis. Serological surveys have shown that infection is widespread in cats but most clinical cases occur in young cats under 9 months of age, particularly pedigree kittens from multicat households. Conjunctivitis is a common reason for presentation of cats to practitioners and Chlamydophila is the single most common infectious cause and possibly the most common cause overall. The conjunctivitis is usually bilateral but may be unilateral early in infection or in chronic cases. Chlamydophila will not usually cause corneal lesions.

PCR testing of ocular swabs is now the preferred method of diagnosis. Care is necessary in collecting swabs as the organism is essentially intracellular and some conjunctival cellular material is required to optimise the chance of detecting the organism.
While other antibiotics, such as fluoroquinolones, have some activity against Chlamydophila, systemic doxycycline (10 mg/kg q24h) is regarded as the treatment of choice [
Both inactivated and modified-live vaccines are available to assist in control. These are only available as part of multivalent vaccines. Both types of vaccines will prevent or reduce the severity of clinical signs but do not prevent infection. These are not always included in routine vaccination programmes for pet cats but may be particularly indicated in catteries with endemic infection or in high-risk (most frequently multicat) situations such as breeding catteries and rescue shelters. Chlamydial polymorphic membrane proteins have been identified as immunodominant and considered as potential serodiagnostic antigens and novel vaccine candidates. 46
Bordetella bronchiseptica infection in cats
Bordetella bronchiseptica is primarily a respiratory pathogen. The bacterium may particularly play a role in multicat environments, where other factors like overcrowding, stress and poor hygienic conditions exist. In general, clinical signs are mild, although pneumonia has been reported, especially in young kittens less than 10 weeks old.
B bronchiseptica is shed with oral and nasal secretions of infected cats. After experimental infection, B bronchiseptica could be isolated for 19 weeks after infection. Transmission is through direct contact, although indirect transmission cannot be excluded. However, B bronchiseptica is susceptible to common disinfectants. Epidemiological studies suggest that dog-to-cat transmission occurs. Also, a few reports describe cats as a possible source of infection of immunocompromised human patients. It is therefore justified to consider B bronchiseptica as a rare but potential zoonotic infection.47,48
For a diagnosis of B bronchiseptica infection, both bacterial culture and PCR are available. B bronchiseptica can be isolated from oropharyngeal or nasal secretions; alternatively, transtracheal wash/bronchoalveolar lavage are used. Nasal swabs were shown to be more often positive then oropharyngeal swabs. 49 Both methods lack sensitivity.
Antibacterial therapy is indicated, including in cases with mild clinical signs, to prevent more severe disease due to colonisation of the lower respiratory tract. The choice of antibiotic should be based on antibiotic sensitivity testing. If not available, tetracyclines, and in particular doxycycline, are the antibiotics of choice. Resistance has been detected against clavulanate-potentiated amoxicillin, and to a higher extent against ampicillin and trimethoprim [

The control of B bronchiseptica infections requires similar measures as used for other respiratory pathogens (ie, FCV and FHV). These are aimed at preventing spread of the infectious agents and reducing their concentration in the environment. An intranasal modified-live vaccine is available but not considered a core vaccine. Vaccination should be limited to cats living in high-risk environments such as shelters and boarding catteries, and where B bronchiseptica has been identified as the problem. The modified-live vaccine is licensed for use as a single vaccination with annual boosters. Interestingly, vaccination with an intranasal live FCV and FHV vaccine also reduced the signs of an experimental B bronchiseptica infection [
Influenza A virus infection in cats
Felids can be naturally and experimentally infected with influenza A viruses; outcomes range from subclinical infection to fatal disease. The virulence of H5N1 highly pathogenic avian virus strains for the domestic cat was established at the very beginning of the avian flu epidemics in Asia. During the same period, infection of household cats and outbreaks of fatal disease in tigers and leopards were reported from Thailand. Then, in Europe, three cats were found dead on the island of Rügen, Germany. The susceptibility of the cat to influenza A viruses was confirmed during the H1N1 pandemic in 2009/2010; indeed, several cases of respiratory disease in domestic cats were linked to the pandemic H1N1 virus. 51 Luckily neither H5N1 nor H1N1 virus infections spread among domestic cats in Europe.52,53

Cats can be infected via the intratracheal and oral routes and, for H5N1 virus, by feeding them infected chickens. Infection with the H5N1 virus may occur through contact with infected birds, and with H1N1 likely with infected owners. Therefore, a cat living in a household with human cases of influenza is at risk. To date, no cat-to-person transmission has been reported, but the risk for persons can presently not be predicted.
If influenza in cats is suspected, their case histories need to be recorded. Differential diagnosis should exclude other infections leading to similar systemic and respiratory signs, including infections with FHV, FCV and B bronchiseptica. Diagnosis should always be confirmed by laboratory testing. In the absence of specific preventive measures, the risk of a cat acquiring an influenza A virus infection must be minimised through management measures. Especially for H1N1, but also for other human influenza A virus infections, patients must be advised to avoid physical contact with cats.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article. The ABCD is supported by Merial, but is a scientifically independent body.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
