Abstract
Practical relevance:
To safely and effectively treat cats with cancer it is important to understand the drugs being used and some species-specific concerns in relation to chemotherapy.
Clinical challenges:
While many of the same principles in treating cats with chemotherapy and targeted agents hold true as for other species, including dogs, cats display altered metabolism of drugs and species-specific toxicities that can present particular challenges for veterinarians.
Audience:
This article is aimed at practitioners who treat feline cancer or who help manage cats undergoing cancer therapy.
Evidence base:
The article reviews the known literature regarding species differences between dogs and cats relating to the use of chemotherapy and targeted therapies. For many of the drugs mentioned there are limited studies and caution must be exercised when using drugs that have a low therapeutic index.
Idiosyncracies of the cat
While cancer is in many respects similar across species lines, distinct differences do occur. This is particularly true for cats with regard to the spectrum and locations of cancers seen, the biologic behavior of tumors and the way in which cats respond to treatment. Chemotherapy raises specific concerns, including dosing differences and toxicity profiles, that make understanding that cats are not small dogs vital in planning and carrying out the safest and most effective treatment.
Dosing of chemotherapeutic agents
A cat’s size and weight can make dosing of chemotherapy drugs difficult. As there are no chemotherapy or targeted agents approved for use in cats, pill sizes are not produced in dosages that are appropriate for their weight. This is even more of an issue than for small dogs, which often weigh more than the average cat.
Many chemotherapeutic drugs are dosed based on body surface area (BSA) in m2, as opposed to body weight. BSA is used in humans and in veterinary patients for drugs that have a narrow therapeutic range as multiple physiologic processes, including renal function and energy metabolism, are proportional to surface area. 1

Feline body weight to body surface area conversion chart

This table is derived from the formula: BSA = 10.0 × (Body weight [g])0.67/10,000
Note there is controversy over the use of this formula to calculate drug dosages in cats (see text)
The BSA formula is most accurate for subjects that differ in size but not in shape. For cats, the value most commonly used for the constant K is 10. 2 This differs from the value of 10.1 that is commonly used for dogs. 2 Complicating the use of 10 as a K value in cats is the fact that the study that derived this value involved kitten cadavers from which the intestinal contents had been removed. The weights obtained in this experimental setting might, therefore, not correlate well with weights derived for adult cats that are not fasted. Also, the methods used to determine the measured BSA in the study are subject to an error rate of up to 10%.
There are several additional limitations to using BSA to calculate drug dosages. Individual cats may have variations in drug metabolism, distribution and excretion. Furthermore, there may be differences in protein binding, absorption, metabolism and excretion among individual cats, leading to variation in drug levels attained. The exponent for body weight of 2/3 (0.67) that is used in the BSA calculation for dogs and cats has also been questioned. The value for adult cats has been suggested to be as low as 0.4. Obesity can also affect the exponent, with obese cats having a lower value compared with those of normal body condition. 2
Taken together, this may explain why several of the chemotherapy drugs that are dosed on the basis of BSA in dogs, are dosed on the more traditional mg/kg basis in cats.

General toxicity concerns
Consideration of chemotherapy side effects is of the utmost importance when treating a cat with cancer. The most common side effect and the dose-limiting toxicity for most drugs is neutropenia (Figure 1). The nadir, or lowest neutrophil count, generally occurs about 1 week after chemotherapy but does vary with the agent used. In order to avoid sepsis or other severe infections it is important always to monitor white blood cell counts before giving a subsequent dose of chemotherapy.

Neutropenia in a blood smear prepared from a cat 1 week after receiving chemotherapy. Courtesy of Dr Andrew Graham Burton
Gastrointestinal signs are another common side effect. Cats also are more susceptible to weight loss and inappetence than dogs. Antiemetics should be used in any cat that becomes anorexic after being treated with chemotherapy. Combinations of antiemetics that work through different mechanisms of action can be more effective than a single agent on its own. Effective combinations include metoclopramide along with a 5-HT3 antagonist such as ondansetron or dolasetron or a substance P inhibitor such as maropitant. Often appetite stimulants, such as mirtazapine or cyproheptadine, can prove useful as well.
Of concern to owners, but less likely to affect the patient’s quality of life, is that cats will often break or lose their whiskers. These will generally grow back after therapy is complete (Figure 2). While generalized alopecia is very rare it is common for cats to lose their guard hairs, giving them a softer coat overall.

Whiskers regrowing following chemotherapy. Courtesy of Antony Moore

Chemotherapy agents with specific concerns
A number of commonly used chemotherapy agents in veterinary medicine have special considerations for use in cats. Dosing information for these drugs is provided in Table 2.
Dosages of chemotherapy drugs commonly used for treating cancer in cats
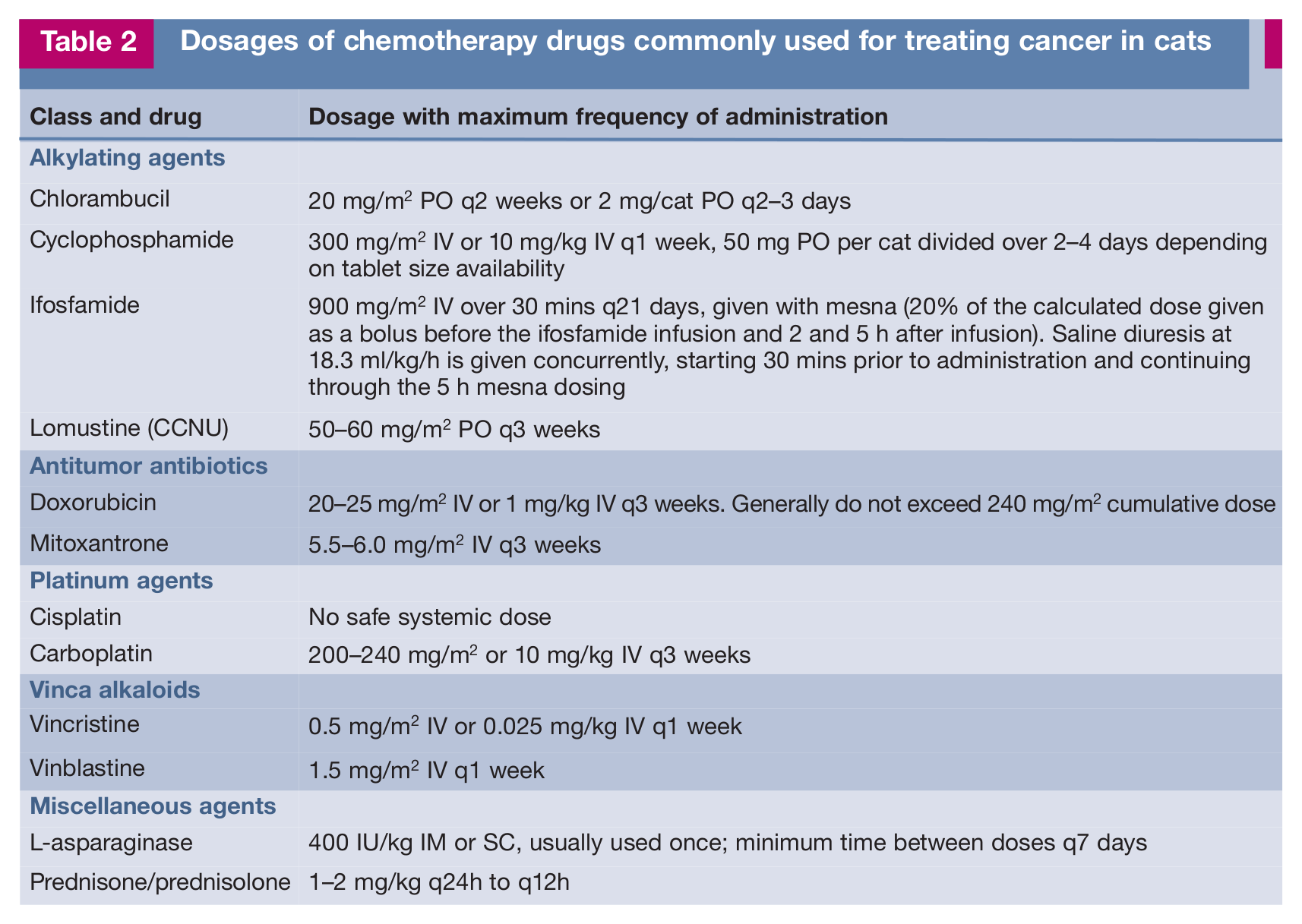
Alkylating agents
Chlorambucil
Chlorambucil is an oral alkylating agent that has been used to treat lymphoid malignancies including chronic lymphocytic leukemia and small cell lymphomas in the cat. It is considered to be mildly myelosuppressive and usually does not cause much in the way of gastrointestinal side effects. 5
Cyclophosphamide
Cyclophosphamide has been used in cats, usually in combination with other chemotherapeutic agents, for the treatment of a variety of tumors including lymphoma, mammary masses and injection site-associated sarcomas. While the dose-limiting toxicity for this drug remains bone marrow suppression, a major concern in dogs is the development of hemorrhagic cystitis. This has not been reported in the cat so remains less of a concern; however, if clinical signs of stranguria, pollakiuria, dysuria or hematuria develop, this should be considered a differential and, unless ruled out, cyclophosphamide should be discontinued and another drug used in its place. It is important to note that, if using the oral formulation, pills should not be cut.
Ifosfamide
Ifosfamide has mostly been studied in cats for the treatment of injection site-associated sarcomas. The safe tolerable dose in this species has been determined to be 900 mg/m2, which is considerably higher that what can be used in the dog.6,7 The dose-limiting toxicity is neutropenia, which can be severe and may occur between 5 and 28 days after administration. This drug can also be renally toxic and, being related to cyclophosphamide, can induce severe hemorrhagic cystitis. To counteract the effects on the bladder, it is given in conjunction with high-rate saline diuresis and mesna. Cats with heart disease or other conditions that do not allow a high rate of saline diuresis should not receive this drug. An anaphylactic type reaction has been documented with administration, so careful monitoring of cats receiving this drug is important. Gastrointestinal signs including drooling during administration and anorexia have also been reported, so antiemetic therapy may be needed.
Lomustine
Lomustine (CCNU) is an alkylating agent that has been used for the treatment of mast cell tumors and lymphoma, either alone or in combination protocols. The most common side effects of this drug in the cat are hematologic, with both neutropenia and thrombocytopenia reported. 8 While a relatively high incidence of hepatotoxicity has been reported in dogs associated with the use of this drug, this appears to be much less common in the cat. 9 Gastrointestinal side effects seen in dogs also appear to be less common in cats. There are rare reports of pulmonary fibrosis developing in cats after receiving lomustine chronically to a high cumulative dose. 10
Antitumor antibiotics
Doxorubicin
Doxorubicin is commonly used in the cat for the treatment of a variety of lymphoid tumors, carcinomas and sarcomas. The dose-limiting toxicity of this chemotherapy agent is neutropenia. Anorexia can be common. Somewhat unique to the cat and important to note is that renal damage and failure is possible with high cumulative dosing. 11 To date, the acute cardiac toxicity seen as arrhythmias and the chronic cumulative dose-dependent toxicity of dilated cardiomyopathy have not been reported in the cat. Due to the risk of renal damage, however, most clinicians will limit the cumulative dose in cats to 180–240 mg/m2.
Mitoxantrone
Mitoxantrone is an antitumor antibiotic that has been used in protocols for feline lymphoma, mammary tumors and squamous cell carcinoma. While this drug is more myelosuppressive than doxorubicin, there appears to be less risk to cats of renal damage than when doxorubicin is used. Mitoxantrone can additionally cause gastrointestinal side effects including vomiting, diarrhea and anorexia. 12
Platinum agents
Cisplatin
While the safe intralesional use of cisplatin has been reported in cats, its systemic use is contraindicated and, at most doses, lethal. In dogs the organ system of concern is the renal system; in cats it is the respiratory system. At doses as low 40 mg/m2 cats have been reported to develop pleural effusion, pulmonary edema and dyspnea, leading to death. On necropsy examination this was associated with an inflammatory reaction in the lungs. 13
Carboplatin
Although in the same class of drugs as cisplatin, carboplatin can be given safely to cats, and has been used for the treatment of a variety of carcinomas and sarcomas. The reported dose that is safe to give is less than that for dogs. Neutropenia is the dose-limiting side effect of this drug and can occur 7–21 days after administration. 14 Some cats are more affected than others and it may be possible to escalate the dose to as high as 240 mg/m2 IV in some cats. 15 Nephrotoxicity has been reported and so carboplatin should be used with care in cats with renal insufficiency. Renal function should always be monitored in cats receiving this drug.

Vinca alkaloids
Vincristine
Vincristine has mostly been used in cats as part of combination protocols to treat lymphoma (Figure 3). While relatively sparing of the bone marrow, it can cause gastroenteritis and anorexia in the cat. Peripheral neuropathy is also a reported toxicity in cats. 16

Cat receiving a dose of vincristine chemotherapy for its high grade intestinal lymphoma
Vinblastine
Like vincristine, vinblastine is mostly used for treating lymphoma, but it has also been studied as a treatment for mast cell tumors. While more myelosuppressive than vincristine, the risk of gastrointestinal side effects is lower.
Miscellaneous agents
L-asparaginase
L-asparaginase, which is an enzyme derived from Escherichia coli, catalyses the amino acid L-asparagine to aspartic acid and ammonia. It has been used in cats for the treatment of lymphoid tumors as it depletes whole body L-asparagine and tumor cells have a decreased or absent ability to produce this amino acid. Since normal cells are able to produce this amino acid and lymphoid neoplasms have a relatively high requirement for this amino acid, this agent is able to selectively kill tumor cells.
A study evaluating the effects of L-asparaginase treatment of feline lymphoma showed reduced asparagine levels in cats by 2 days post-administration. 17 These effects were lost within 7 days of administration, which is a much shorter duration of effect than reported for the dog, which can last weeks. This may be due to cats, being a true carnivore, consuming high protein diets coupled with a high rate of amino acid synthesis. The authors of the study documented an overall response rate (complete and partial responses) of 30% to single agent L-asparaginase. 17 While the reported response rate is lower than that in dogs, there is demonstrated clinical benefit in the use of L-asparaginase in the treatment of feline lymphoma.
Side effects reported with the use of L-asparaginase in cats are minimal; however, since it is an exogenous protein an anaphylactic reaction may potentially develop with repeated administration. Resistance to this drug is common and due to upregulation of asparagine synthetase in the surviving tumor cells. While not investigated, this is a likely mechanism of tumor resistance in this species.
Corticosteroids
Prednisone and prednisolone, the most commonly used corticosteroids in cats, are included as a component of cancer therapy in multiple lymphoma and mast cell tumor protocols. While most veterinarians consider prednisone and prednisolone to be equipotent in the dog, this may not be so in the cat. Prednisone must be converted by the liver to its active form prednisolone. In one study involving six healthy cats there were decreased blood levels of prednisolone when prednisone was administered compared with the same dosage of oral prednisolone. 18 The authors concluded that either oral prednisone was not as bioavailable as prednisolone or that hepatic metabolism of prednisone to prednisolone was not as efficient in cats. Given the small study size and clinical evidence in multiple studies that cats do respond to prednisone, more investigation is needed before discontinuing the use of this drug in cats.
Long-term corticosteroid use in the cat is associated with the risk of diabetes mellitus with insulin resistance, although this is more of a problem with long-acting injectable formulations.
Targeted therapies
Over the past several years, targeted therapies have been developed for the treatment of various canine cancers and approved for use in melanoma and mast cell tumors. Their utility in the treatment of other cancers is promising and currently under investigation. While presently there are no targeted agents approved for use in cats, there are several studies and reports in the literature of their use (Table 3). It is important to realize that targeted therapies can have toxicities associated with them and that cats receiving these agents need to be monitored as carefully as those being treated with traditional chemotherapy drugs.
Drug dosages for targeted therapies in cats

Toceranib phosphate
Toceranib phosphate targets the Kit, VEGFR, PDGFR and FLT-3 tyrosine kinase receptors. 19 This drug, while not licensed for use in the cat, is approved for use in the dog. Currently, there is very limited data on its use in feline medicine. When given at high doses (6.5 mg/kg q24h), vomiting, diarrhea, anorexia and weight loss were reported. 20 While there are several ongoing clinical trials assessing the use of this drug in cats, there are no published studies in the cat and it should be used with extreme caution and only if no other therapies are available.
Imatinib mesylate
Imatinib mesylate is the prototypic small molecule inhibitor that inhibits the tyrosine kinase receptors BCR-Abl, PDGFR and C-Kit. There have been a few studies investigating its use for the treatment of mast cell tumors and injection site-associated sarcomas in the cat.21,22 However, as this drug has had limited evaluation in the cat, the full range of possible toxicities is not known. In the published literature there are reports of elevated liver and renal enzymes, leukocytosis and a case report of a cat developing proteinuria while being treated with imatinib. 23 The idiosyncratic hepatotoxicity seen in dogs has not been reported as a problem in cats.
Masitinib mesylate
Masitinib mesylate targets the tyrosine kinase receptors c-Kit, PDGFR and Lyn. This drug is currently approved for use in dogs in the United States and Europe for the treatment of mast cell tumors. It has been studied to some degree in the cat, and a pharmacokinetic and a dose escalation study have been published. The pharmacokinetic study reported a bioavailability of approximately 60% and suggested that an oral dose of 10–15 mg/kg was needed to achieve adequate plasma concentrations. 24 In a safety study performed in healthy cats given 50 mg either daily or every other day, some gastrointestinal signs and other toxicity effects were seen, including proteinuria, neutropenia and elevated creatinine levels. 25 As the dosing and efficacy have not been established in the cat this drug should be used with extreme caution.

Footnotes
Key Points
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The author does not have any potential conflicts of interest to declare.
