Abstract
Practical relevance:
Feline injection site-associated sarcomas (FISSs) have been the cause of much controversy and concern since they were first reported in the early 1990s. While not solely associated with vaccination, there are implications for vaccination sites and schedules and, while guidance has been published, this appears to be permeating only slowly through to general practice.
Clinical challenges:
Up to one-quarter of cats with this difficult condition have metastatic lung involvement. The mainstay of treatment is aggressive surgery, but even in cases where full excision with clean margins is achieved, tumour recurrence is anticipated in about one-third of cases. The role of radiotherapy and chemotherapy as adjuvant treatments has yet to be clearly defined.
Patient group:
FISSs are often seen in younger cats, with a peak presentation at 6–7 years and a second peak at 10–11 years.
Evidence base:
This review summarises the diagnosis and management of FISS with reference to the latest published treatment results. It focuses on surgical excision but also covers adjuvant radiotherapy and chemotherapy, and gives median survival times for the different treatment approaches.
FISS – what’s the history?
Feline injection site-associated sarcoma (FISS) was originally called vaccine site-associated sarcoma due to the tendency for these aggressive soft tissue sarcomas to occur in vaccination sites. They were first reported in 1991 1 and linked to rabies vaccination and the use of adjuvants, particularly those containing aluminium, in vaccines. 2 However, various studies investigating the association of vaccination with injection site sarcomas have not conclusively implicated either one particular vaccine or the presence of aluminium in adjuvanted vaccines.
Rabies vaccination has been documented to cause local post-vaccine reactions that were twice the size of those following feline leukaemia virus (FeLV) vaccination. 3 Adjuvanted-killed vaccines caused local inflammatory reactions that were greater than non-adjuvanted vaccines of similar antigenic composition, and aluminium-containing vaccines resulted in greater local inflammation post-vaccination than non-aluminium-containing vaccines. 4 Reactions are more common with adjuvants containing aluminium; less common with non-aluminium adjuvant vaccines and not seen with non-adjuvant live vaccines. 3 However, the size of reaction is not related to the presence of aluminium. 3 Two large epidemiological studies failed to produce any evidence that the risk of sarcomas is any greater with aluminium-containing vaccines.5,6 The reported incidence of FISS varies from 1.3/1000 vaccinations 7 to 1/10,000 vaccinations.8,9
The concern about adjuvanted vaccines (particularly aluminium adjuvants) led to the development of non-adjuvanted recombinant vaccines. A recent study comparing the association of different vaccinations and other injectable drugs with sarcoma formation found that cats suffering from FISS in the hind limbs were significantly more likely to have received inactivated rabies vaccines than recombinant rabies vaccines compared with control cats. 10 For cats with FISS in the interscapular region, the frequency of administration of long-acting corticosteroid injections was also higher compared with control cats. 10 Vaccine site-associated sarcomas were renamed as injection site-associated sarcomas as an acknowledgement that they can occur after injection of a variety of medications, including antibiotics, long-acting corticosteroids and insulin. 11 Similar reactions are seen with intramuscular and subcutaneous vaccination, although injection site reactions are less common with intramuscular injections.
Cats have an increased tendency to respond to inflammation by fibroblast proliferation and ultimately tumour genesis compared with dogs, although injection-associated sarcomas have been reported in other species including dogs and rabbits. Microscopic observation of transition zones that pass from inflammation to sarcoma in cats supports this theory. 2 The sarcomas have also occurred at the site of other sources of inflammation such as suture material, microchips and retained surgical swabs, and at previous surgical or implant sites. Various risk factors have been identified (see box above right). 11


Characteristics of injection site-associated sarcomas
Injection site-associated sarcomas are often seen in younger cats, with a peak presentation at 6–7 years of age and a second peak at 10–11 years. 6 They usually occur in the subcutis, whereas non-injection site-associated fibrosarcomas are dermal in origin (Figures 1 and 2). FISSs are also often larger at presentation than non-injection site-associated sarcomas, which may be related to the difficulty in palpating subcutaneous masses in the interscapular region or the more aggressive biological behaviour of FISS.

Feline injection site-associated sarcoma (FISS) in an 8-year-old female neutered (FN) domestic shorthair (DSH) cat
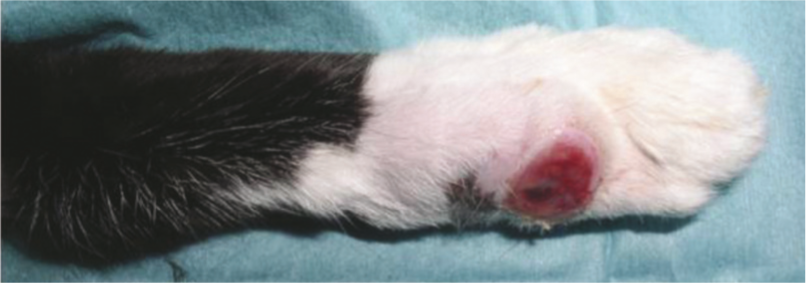
Fibrosarcoma on the hind limb of a 13-year-old FN DSH cat. Courtesy of Prue Neath
Diagnosis
Full haematology and biochemistry profiles, along with FeLV and feline immunodeficiency virus (FIV) status and urinalysis, are recommended to pick up any concurrent disease or immunocompromise which may affect response to treatment. A biopsy is required to definitively diagnose FISS; cytology is not reliable. The biopsy is best performed after imaging to prevent any biopsy-associated inflammation confusing the imaging findings. If an owner wishes to have a biopsy result prior to imaging, an incisional wedge or punch biopsy should be taken. The biopsy tract should be close to any further planned surgical treatment area and should not extend through the sarcoma into clean tissue planes. Hence, care must be taken if using a Tru-cut needle biopsy unless the mass is large. The biopsy site should be closed with clean instruments and the dead space opposed carefully to avoid wound problems or seroma formation.
Inflammatory reactions can occur after injections but should resolve in 6–8 weeks. If a granuloma is picked up post-injection on biopsy, marginal excision can theoretically be performed as at present there is no evidence that granulomas predispose to sarcoma formation. Excision is prudent if the granuloma persists longer than 3 months or is over 2 cm in size.
Staging
In cases of FISS, thoracic computed tomography (CT) or three-view inflated thoracic radiography should be used to screen for metastatic lung involvement, which occurs in 10–25% of cases.13,14,18 The most common site for metastases is the lungs, but metastasis can also occur to the subcutaneous tissue (Figure 3), liver and local lymph nodes. The draining lymph nodes should be assessed by palpation and cytology.

Dorsal (left) and transverse (right) magnetic resonance (MR) images of a FISS in a 12-year-old FN DSH cat. The FISS is on the lateral thoracic wall but the MR images also reveal a subcutaneous metastasis in the dorsal midline, which was not palpable on physical examination
If an owner is considering treatment, magnetic resonance imaging (MRI) or CT is advised to determine the extent of the tumour and to assess whether 3–5 cm margins are achievable (Figures 3 and 4). Due to the inflammatory nature of FISS, MRI or CT may overestimate tumour volume, athough this should improve the chance of full excision with subsequent surgery. Typically, the volume of the tumour determined by CT is much greater than that determined by palpation.9,19

CT reconstruction of an interscapular FISS in a 13-year-old Maine Coon, demonstrating the relationship of the tumour with the cervical dorsal spinous processes. Courtesy of Cambridge Radiology Referrals
At present, there is insufficient evidence in the literature to determine whether preoperative advanced imaging improves survival times for FISS but it should make surgical planning easier. FISSs generally show strong contrast uptake on CT. If CT is used for FISS in the interscapular region, an additional post- contrast scan with the fore limbs positioned caudally along the body, in addition to the standard cranial position of the fore limbs, allows better evaluation of the relationship between the tumour and surrounding muscles. 20
If advanced imaging is not possible for financial or logistical reasons, careful palpation of the tumour will determine whether surgical resection is feasible, although physical palpation tends to underestimate the tumour volume. Even if located in the subcutis and mobile, these tumours often have palpable stalks or tendrils which extend to the underlying muscles.
Surgical treatment
Radical surgery with 3–5 cm lateral margins and two muscle layers or bone as a deep margin is the recommended surgical treatment advice. Even in cases of FISS that are fully excised, about a third will experience recurrence of the tumour; thus surgery is often palliative. Radiotherapy can be used as an adjuvant treatment, either postoperatively, preoperatively or as a combination pre- and postsurgery. The first surgery is the most effective as far as curative surgery is concerned; thus, masses in predilection sites should not be treated with excisional biopsy unless the owner has no further wish to treat any tumour that is diagnosed.

Such radical surgery is easier with the guidance of advanced imaging in the form of CT or MRI. Prior to starting surgery, the tumour extent and proposed incision site is drawn out on the skin in order that the required margins are achieved (Figure 5). Often there is sufficient skin in the interscapular area to enable primary closure of the wound, particularly after resection of the epaxial muscles and cervical spinous processes (Figures 6 and 7). The skin should be assessed preoperatively and, if there is doubt as to whether primary closure will be possible, the application of skin stretchers consisting of Velcro bands will recruit additional skin in 2–3 days (Figure 8). Alternatively, an advancement flap can be used from the dorsal abdominal skin.

Surgical approach to a FISS. The planned incision site with 5 cm margins is drawn onto the skin with a sterile marker prior to incision

En bloc excision of the skin, subcutis and muscles

Appearance after resection of the dorsal spinous processes of the cervical vertebrae
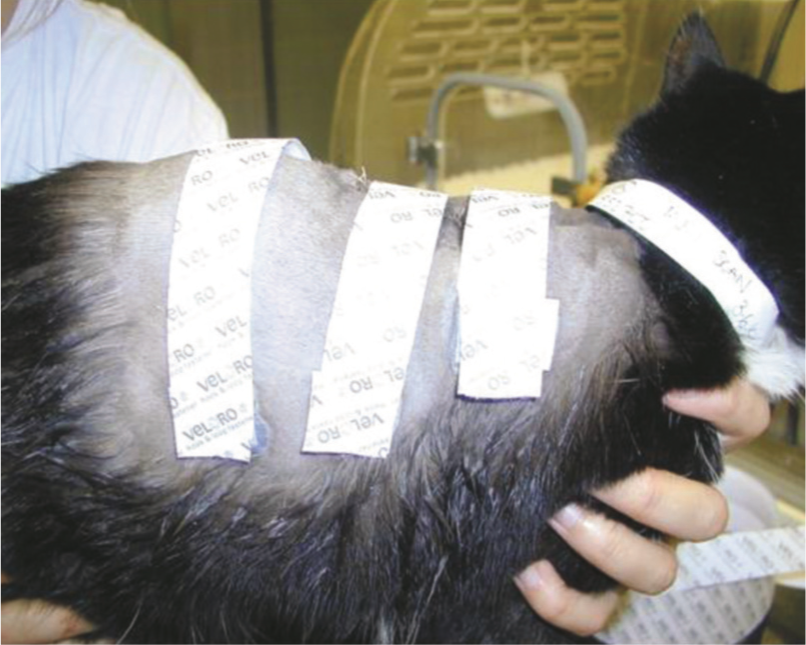
Skin stretching with Velcro bands. Courtesy of Prue Neath
Any structures that fall within the prescribed margins should be resected, including epaxial muscles (eg, splenius and semispinalis, which support the head in the cat), thoracic wall, dorsal spinous processes or dorsal scapula. Surgical excision of injection site-associated sarcomas in the interscapular region usually involves resection of the neck muscles and dorsal spinous processes of the underlying cervical vertebrae. Resection of the spinalis, semispinalis and rectus capitis dorsalis minor results in weakness of the neck and a flexed head carriage. This improves over 6–8 weeks, with full head movement returning (Figure 9).

Head carriage after resection of the cervical dorsal spinous processes and associated muscles 8 weeks previously
Sometimes the dorsal aspects of the scapulae are in the surgical field. Partial resection of more than 50% of the scapulae, if bilateral, may result in a poor gait for the first 6–8 weeks, with the fore limbs being excessively mobile. If the sarcoma is situated over one scapula, a total scapulectomy or forequarter amputation is required. With rear limb sarcomas, amputation by coxofemoral disarticulation should be performed unless the tumour is subcutaneous and two muscle layers can be achieved as a deep margin without amputation. Hemipelvectomy is occasionally required if the tumour is positioned on the proximal limb. For injection site-associated sarcomas that occur over the flank, full thickness excision of the abdominal wall is required. These deficits can usually be closed without the use of surgical mesh, though mesh (usually polypropylene) may be required for wounds over the lateral thoracic wall.
After excision of the tumour, the surgical margins are painted with Indian ink (Figure 10) prior to submission for histopathological analysis. Small fragments (2–3 mm) are taken from the wound bed in the areas where the surgical margins are considered to be the narrowest to assess the tissue left in the body. The pathology submission report should contain full details about the history of the patient and site of the lesion, and request that surgical margins are evaluated.

Tumour inking after removal. Courtesy of Prue Neath
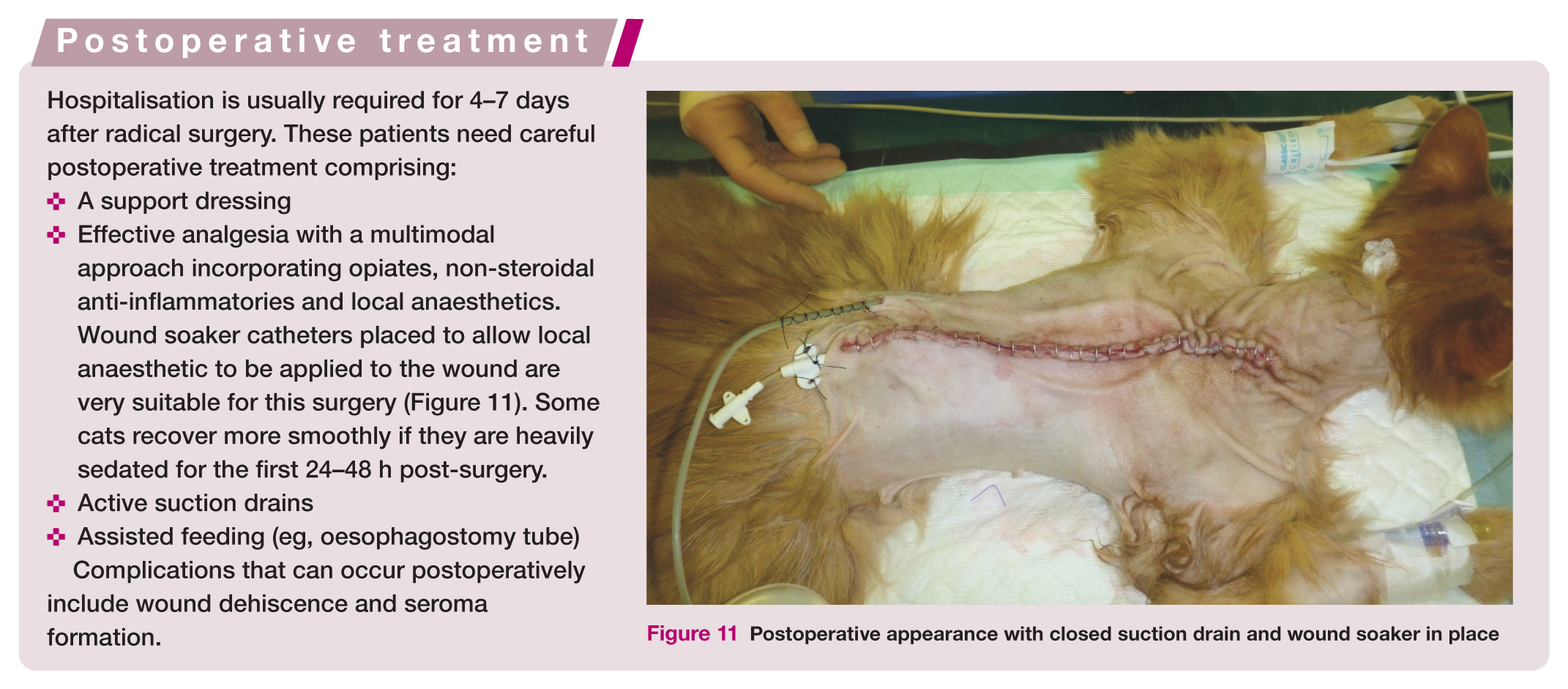
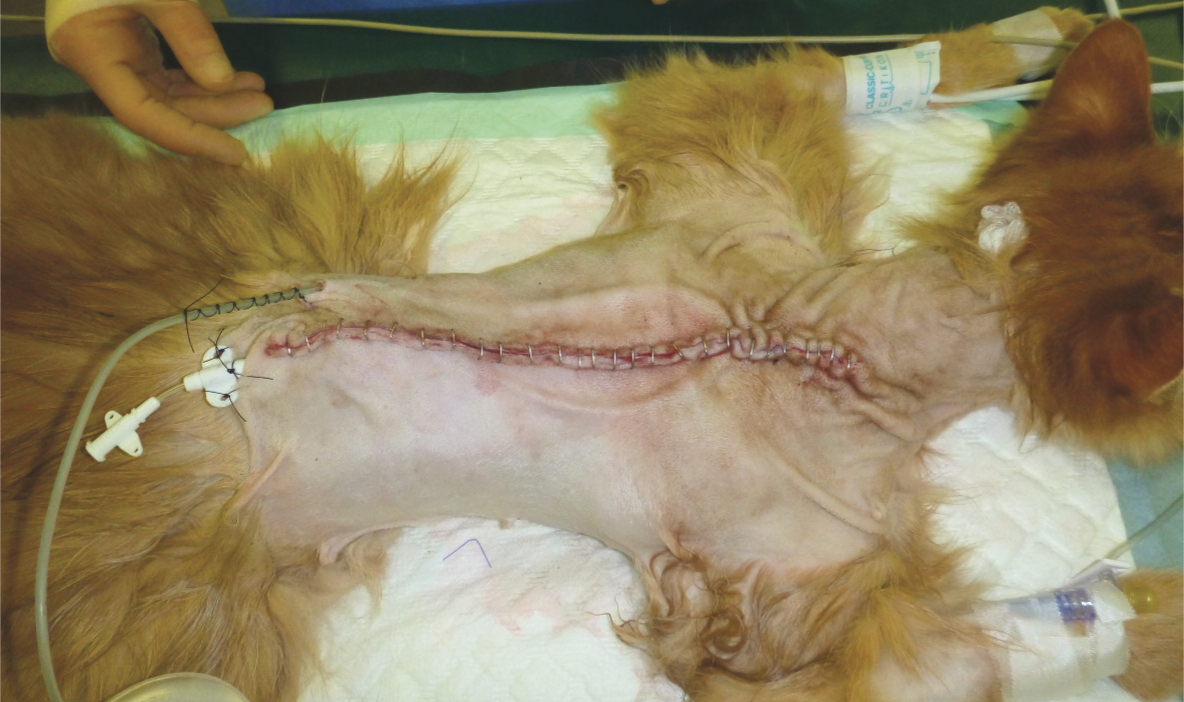
Postoperative appearance with closed suction drain and wound soaker in place
Results following surgery
The outcome following surgery is dependent on many factors including the experience of the surgeon, the number of previous surgeries the cat has undergone and the extent of the surgery. Radical excision with 5 cm margins appears to give the best results following surgery as a sole treatment, 12 and results are better if this is the first excision (Table 1).21,22 The success rate of surgery is also influenced by the position of the FISS, with amputation for FISS affecting the hind legs having the highest chance of cure. 23
Results following surgical excision

Results following radiotherapy


Following surgery, cats with complete excision of the tumour have a longer tumour-free interval and survival time compared with those with incomplete margins. In one study, 21 the difference was >16 months versus 4 months for tumour-free interval and >16 months versus 9 months for survival time. The recurrence rate in cases with dirty margins after surgery is 10 times higher than in cases with clean margins. 23 Even for cats that have clean margins after surgical excision, a good prognosis is not guaranteed as about 20% will develop metastatic lesions, while local recurrence rates in cats with histopathological clean margins range from 14–50%.8,12,14,23
The fact that tumour recurrence occurs with such a high prevalence in cases with clean surgical margins has led to debate. Does the invasive nature of this tumour make accurate evaluation of clean margins in veterinary histopathology extremely difficult? Or in cats that develop FISS does ongoing inflammation, potentially resulting from the surgical excision, lead to further tumourigenesis? This latter theory is supported by the fact that recurrence is commonly seen a year or more after excision in cases with clean margins but usually more rapidly in cases with dirty surgical margins. The difficulty in assessing the margins in FISS due to the infiltrative nature of the tumour has led to a three-dimensional histological technique being recommended, whereby a strip 3–4 mm in thickness is taken from the whole surgical margin. 24 However, even using this technique, there was a 19% recurrence rate in tumours excised with clean margins. 24
If, following a first surgery that failed to obtain clean margins, further surgery is contemplated, the survival time is improved by referring to a specialist surgeon.
Radiotherapy
As the recurrence rate following surgical excision is relatively high, even in the face of clean surgical margins, radiotherapy has been used as a neoadjuvant and adjuvant therapy. Radiotherapy as a sole treatment is generally unrewarding, but it has been used both preoperatively 23 and postoperatively in the treatment of FISS.22,25,26 Pre- and postoperative radiotherapy can also be combined.
The rationale behind preoperative radiotherapy is that the tumour is likely to shrink and, perhaps more importantly, the tendrils of microscopic tumour that invade the subcutaneous tissues will be reduced and viable tumour seeding will be decreased at surgery. Postoperative radiotherapy is used to mop up residual microscopic disease; however, the surgery may damage the blood supply to the cells, especially at the periphery of the tumour, 27 making the tumour cells more radioresistant. This is supported by the fact that in one study evaluating postoperative radiotherapy on soft tissue sarcoma recurrence rate in dogs, delaying the radiotherapy until 6 weeks postoperatively was associated with a decreased recurrence rate compared with the traditional timing of 3–4 weeks. 27
However, the benefit of radiotherapy has not been conclusively proven in the literature. Although Eckstein et al reported median survival times of 1300 days with surgery and postoperative radiotherapy, a number of the cases had adjuvant chemotherapy. 26 In one study, the use of postoperative radiotherapy in cats was associated with a decreased median survival time compared with treatment by surgery alone. 14 Interestingly, in a study by Cohen et al, cats that had conservative excision followed by radiotherapy had a longer median survival time (502 days) than cats that had wide excision followed by radiotherapy (476 days). 22 This may be related to the fact that the cats that underwent conservative excision had radiotherapy 2 days after the surgery, while with wide excision there was a delay.
Chemotherapy
The efficacy of chemotherapy on FISS is unproven, with varying results in studies, although the high metastatic rate encourages the use of chemotherapy. Effects on FISS cell culture by chemotherapeutic drugs have been demonstrated, with doxorubicin and mitoxantrone being the most effective. 28 Many of the clinical studies are retrospective and use different combination therapy; therefore, clearly determining the additional benefit of chemotherapy is difficult. In the Cohen et al study, combining doxorubicin and cyclophosphamide with surgery and postoperative electron beam therapy did not significantly alter the rate of metastasis or median survival time. 22 A second study of 69 cats also found no difference in recurrence rate or metastatic rate when doxorubicin was combined with radical surgical excision. 29 Doxorubicin, when added to surgery and radiation in another study, actually decreased the time to first recurrence and survival time compared with surgery and radiation (median survival time of 674 days versus 842 days). 30 However, this was a non-randomised study of only 25 cats. Poirier et al reported a 5.6% metastatic rate in cats treated with surgery and chemotherapy in the form of liposome-encapsulated doxorubicin, 31 which is notably lower than most other studies, and a prolonged disease-free interval compared with cases treated by surgery alone (388 versus 93 days).
Due to the high metastatic rate associated with FISS, particularly grade 3 tumours, chemotherapy is still an appealing treatment option. It may be that some of the newer agents that are becoming available, such as the tyrosine kinase inhibitor, imatinib, will have a more convincing effect on FISS, although currently there are limited studies available. Imatinib inhibited the growth of FISS in a murine xenograft model, 32 and would seem tailored to FISS due to the presence of PDGFR (platelet derived growth factor receptor), which is a receptor tyrosine kinase. In one phase 1 clinical trial of imatinib, 4/9 cases trialled had FISS. In these four cases a response to treatment was noted and consisted predominantly of short-term tumour stabilisation. 33

Immunotherapy
The most successful immunomodulation therapy reported so far has involved xenogeneic cells secreting human interleukin-2 (IL-2), which was trialled in 16 cats with FISS, two of which had local recurrence and three had metastatic disease. The median survival time was 16 months versus 8 months in cats that did not receive IL-2. 34 A second study used human and feline IL-2 and demonstrated a lower tumour recurrence rate compared with control cats not receiving immunotherapy following surgery and iridium-based radiotherapy (39% and 28% versus 61% for the controls). 35 Recombinant feline interferon-omega has been assessed for safety but, to date, reports on its efficacy are lacking. 36 These are, as yet, not widely available treatments.
Prevention
In response to the appearance of FISS, the Vaccine-Associated Feline Sarcoma Task Force (VAFSTF) was formed in 1996 in North America. This group devised recommendations about the sites at which vaccines should be given and the frequency of vaccinations. Other bodies, including the European Advisory Board on Cat Diseases (ABCD), the American Association of Feline Practitioners (AAFP) and the World Small Animal Veterinary Association (WSAVA), have also issued guidelines on cat vaccination.37–39
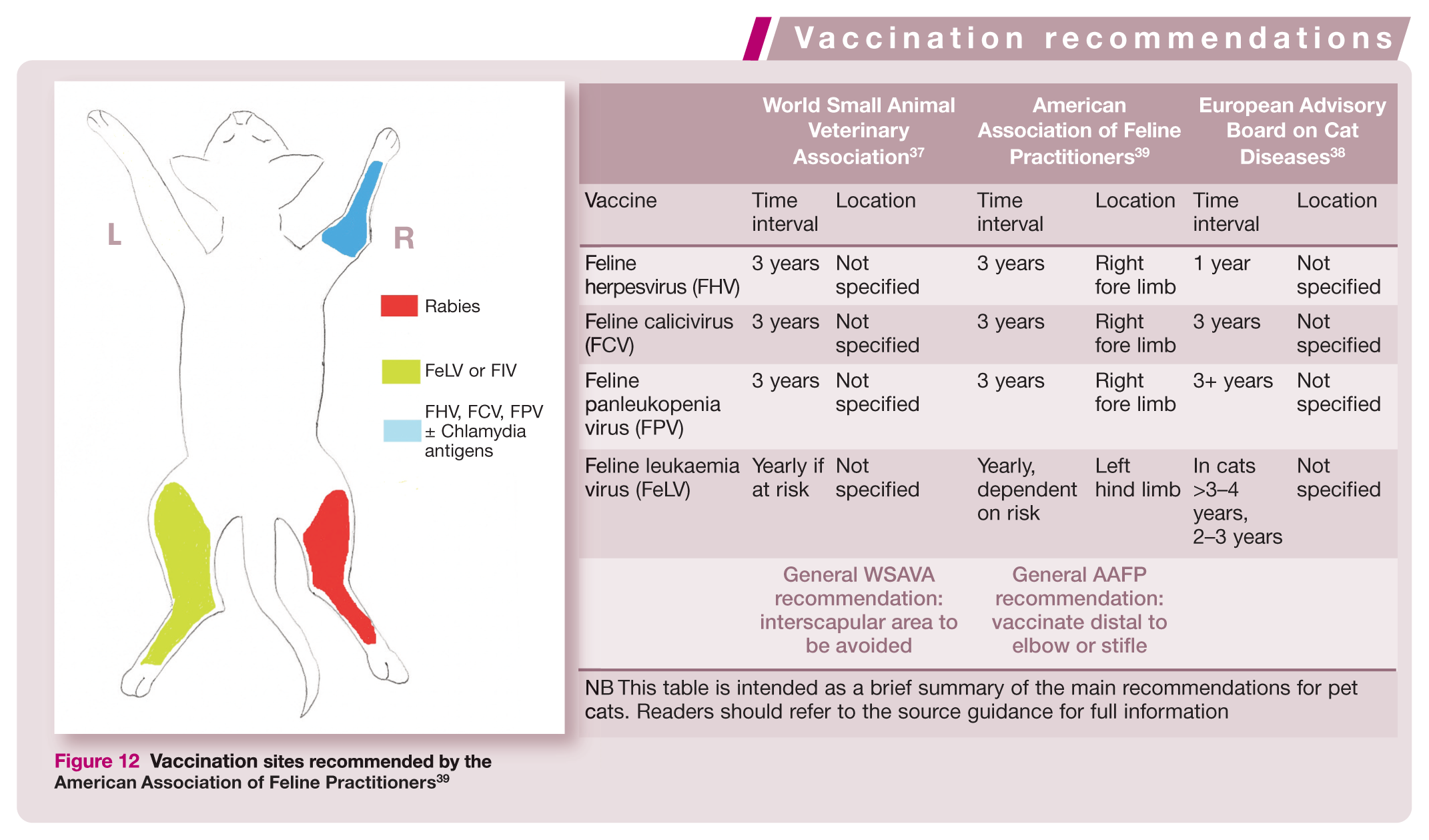

NB This table is intended as a brief summary of the main recommendations for pet cats. Readers should refer to the source guidance for full information
A recent study on feline vaccination practices in the UK, 40 revealed that the most common site of vaccine administration was still the interscapular region, despite both the AAFP and WSAVA specifying that this region should not be used. Likewise, in a large American epidemiological study published in 2003, 11 84% of 662 cats with FISS had tumours located in the interscapular region despite the AAFP guidelines having been published in 2000. In a group of 392 cats that were recruited from 1990–2006, the incidence of interscapular FISS reduced from 53% to 40% as the incidence of FISS in the limbs increased, although the interscapular region was once again the most common location by far. 41 The AAFP recommends using the distal limbs (see box, Figure 12), while the WSAVA advises using the lateral abdomen or thorax and changing the site of administration each time a vaccine is given. The majority of practitioners in the UK study were also giving the FHV, FCV and FPV vaccine annually, although both the WSAVA and AAFP guidelines are for 3-yearly vaccinations. FeLV vaccination is still recommended on a yearly basis, dependent on risk, although in older cats the ABCD advises administration every 2–3 years.

Vaccination sites recommended by the American Association of Feline Practitioners 39

The lack of adherence to published guidelines may result from poor awareness of the guidelines or may reflect the opinion that the guidelines are unfeasible in practice. Even if the guidelines are deemed difficult to follow, one would hope the current practice of interscapular injections will change, perhaps using the hind limbs rather than distal hind limbs as vaccination at the level of the stifle is tolerated by many cats and still allows good surgical margins with amputation if an injection site-associated sarcoma develops. As a number of FISSs have also occurred following the administration of long-acting corticosteroids 10 and long-acting penicillins, it would be good practice to avoid injecting these drugs into the scruff of the feline neck. Once this message pervades into general practice, we will hopefully improve the prognosis associated with these tumours.
Footnotes
Key Points
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The author does not have any potential conflicts of interest to declare.
