Abstract
Practical relevance:
Mammary tumours are among the most common neoplasms in both cats and dogs, but the prevalence of malignant histological types is far higher in cats (ratio of malignant:benign is at least 4:1).
Clinical challenges:
The more aggressive nature of mammary neoplasia in cats poses challenges for management. Prognosis is affected by tumour size and, therefore, early recognition and treatment of mammary tumours is paramount. Although the primary tumour can be excised surgically, no studies have shown that chemotherapy significantly extends survival time; hence, metastatic spread remains an important clinical problem.
Patient group:
Mammary tumours usually affect older female cats, mainly entire females. Siamese and Oriental breeds may be predisposed. Male cats can develop mammary neoplasia, but this is rare.
Evidence base:
This review summarises the current literature relating to aetiology, pathology, presentation, diagnosis, staging, treatment and prognosis of feline mammary tumours.
Epidemiology
Mammary neoplasia is the third most common tumour type affecting female cats after lymphoma and skin tumours, accounting for 17% of tumours. 1 Reported incidence is 25.4 per 100,000 female cats per year. 1 Although precise statistics are not available, the incidence of mammary tumours may vary globally depending on cultural acceptance of neutering policies. Neutering is less common in Scandinavia and some other parts of Europe than in the UK, for example.

Aetiology
As in humans and dogs, hormonal fluctuations associated with repeated oestrous cycles may influence the development of mammary tumours in cats. This is supported by a case control study reporting that cats spayed before 1 year of age had a decreased risk of developing mammary cancer, 9 and that intact cats were seven times overrepresented in a population of cats with mammary tumours compared with a control group. 10 However, cats spayed at less than a year old do still develop mammary tumours so the effect of early neutering does not eliminate the risk of mammary tumours, 8 and early studies reporting more tumours in intact cats may possibly have reflected neutering practices at the time rather than a true increased risk. 11
Other evidence to support a hormonal aetiology is that oestrogen and progesterone receptors are found in normal mammary tissues and benign tumours but are often lost in malignant tumours and metastases.12–17 In addition, exogenous progesterone administration to prevent pregnancy, or for behavioural aggression, causes tumour development (benign and malignant) in both male 18 and female cats. 19 A possible dose-related effect may occur, with increased risk of mam-mary carcinoma if given regularly rather than intermittently. 10
There is no definitive evidence for a viral aetiology for mammary tumours in cats, although this was proposed in early literature, and obesity has not been implicated, unlike in dogs.
Presentation
Cats have four pairs of mammary glands (two thoracic and two abdominal) and, although any gland may be affected, some studies have reported a predisposition for mammary tumours in the caudal glands.11,20 Mammary tumours present as a single subcutaneous nodule or mass within the mam-mary glands (Figures 1 and 2), which may be discrete and mobile, or attached to underlying tissues and possibly ulcerated in appearance (Figure 3). A few may appear cystic. It is difficult to distinguish benign from malignant nodules in cats, so all should be treated as potentially malignant. Multiple mammary masses within several glands are common (usually ipsilateral but occasionally bilateral) (Figure 4) and were reported in 60% of cats in one study. 8 Sometimes, the true extent of the disease cannot be appreciated without clipping the fur. Drainage lymph nodes (inguinal or axillary) may also be visibly or palpably enlarged.

Mammary mass in the thoracic glands of an 11-year-old female entire (FE) domestic shorthair (DSH) cat

Mammary mass in the thoracic glands and axillary lymph node of an 8-year-old FE DSH cat

Ulcerated mammary mass in the second abdominal gland of a 21-year-old male neutered DSH cat

Bilateral mammary masses before (a) and after (b) clipping
In particularly aggressive inflammatory carcinomas with extensive lymphatic involvement, the glands may be swollen, hot and painful. 4 This presentation may be difficult to differentiate from fibroadenomatous hyper-plasia (fibroepithelial hypertrophy, feline mam-mary hypertrophy), although the latter more commonly affects young cats. 21

Diagnosis
If a mammary mass is palpated, confirmation that it is neoplastic requires biopsy of tissue or a fine needle aspirate (FNA) for cytology. Since most feline mammary tumours are malignant, FNAs are more reliable than in dogs and are useful to confirm the diagnosis (Figure 5).
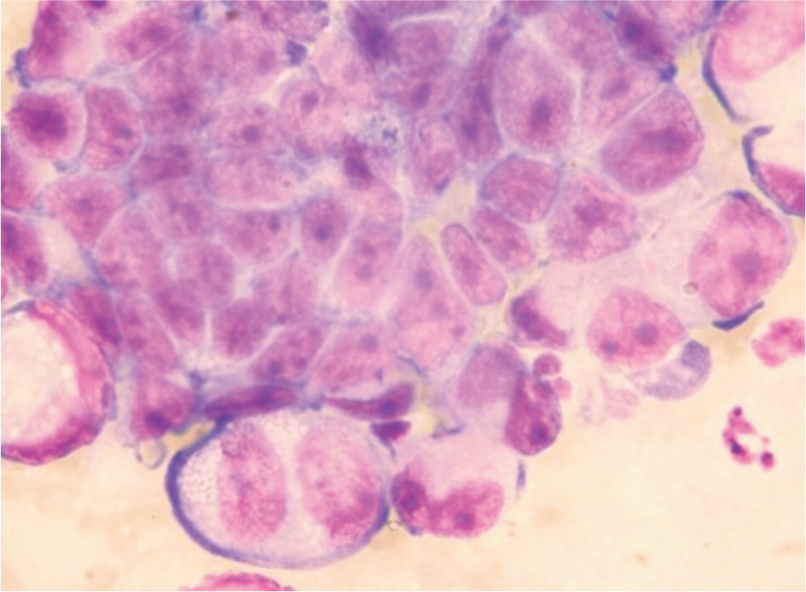
Cytology of feline mammary carcinoma, showing a raft of polygonal neoplastic epithelial cells with anisocytosis and anisokaryosis, some multinucleate cells and prominent nucleoli, often several per nucleus. Courtesy of Elizabeth Villiers
Most feline mammary tumours derive from glandular epithelium and all strictly are adenomas or adenocarcinomas, although the latter are often loosely referred to as carcinomas. Benign tumours are uncommon but, of these, fibroadenoma is most frequently reported, with simple adenoma or duct papilloma rarely seen. The main histological type of mammary tumour in the cat is simple adenocarcinoma derived from the luminal epithelium of the mammary ducts and alveoli (Figure 6). Complex or mixed tumours involving both luminal and myoepithelial cells are extremely infrequent in cats compared with dogs, although they may be associated with a better prognosis.22,23 In cats, carcinomas may be tubulopapillary, solid, cribriform or mucinous, although squamous cell carcinoma and mixed carcinosarcoma are also reported. 24

Histology of feline mammary simple adenocarcinoma. Low power views showing primary tumour invading muscle (a), and pulmonary metastasis (b) with tumour cells in blood vessels and lung tissue. Courtesy of Tim Scase
Inflammatory mammary carcinoma, which has a particularly poor prognosis owing to an additional inflammatory component blocking the lymphatics and affecting lymph drainage and causing swollen, painful glands, has been reported in three cats with underlying highly malignant, papillary mammary carcinomas. 25
Staging
If a mammary tumour is suspected or confirmed, investigations should be carried out to determine the local extent and degree of spread throughout the body prior to surgical excision. As so few mammary masses are benign, and gross appearance alone is an unreliable basis on which to distinguish benign from malignant tumours, complete staging should be routine for all mammary masses. The WHO staging system is usually applied (Table 1); 26 however, with many tumours being noticed earlier and in less advanced stages, fewer larger tumours over 5 cm diameter are being reported. Measurement of the primary tumour with calipers is important since the size of the tumour influences the prognosis: those <3 cm diameter being associated with better survival rates than those >3 cm (see later).
TNM and clinical staging system for feline mammary tumours

Adapted from Owen 26
Staging of confirmed tumours should include palpation and aspiration of local drainage lymph nodes, since more than a quarter of cats have regional metastasis at the time of diagnosis. 27 Involvement of multiple axillary lymph nodes is commonly detected by lymph angiography (58–75% cases) but a single inguinal lymph node predominates (84–94% cats). 28 Although the axillary and inguinal lymph nodes are those mainly reported as being affected in feline mammary neoplasia (80% cats), the sternal lymph node may also be involved (30% cats). 27
For more distant spread, three-view chest radiography (preferably performed under anaesthesia with inflated lungs) and abdominal ultrasonography should be carried out, since the most common sites of metastasis are the lungs, medial iliac lymph nodes and abdominal organs (Figure 7). Pulmonary metastases usually appear as a miliary pattern on thoracic radiographs, but pleural surfaces can also be affected and in some cases metastatic lung disease can cause pleural effusion (Figure 8). More uncommonly, metastasis to bone can be detected.

Enlarged left medial iliac lymph node chain with surrounding hyperechoic fat detected by abdominal ultrasound at routine staging of an 11-year-old female neutered (FN) Abyssinian with mammary carcinomas of the left caudal abdominal and right cranial thoracic glands

Left lateral (a) and dorsoventral (b) radiographs of the thorax of the cat imaged in Figure 7. These views show general increased opacity within the thoracic cavity, with severe retraction of the lung fields from the dorsal wall and effacement of the cardiac silhouette and diaphragm, consistent with pleural effusion
Advanced imaging (ie, computed tomography [CT]) of the lungs provides more accurate assessment of metastases (see ‘case notes’), and should be used where there is any concern about the radiographic appearance of the lungs (Figure 9).

Left lateral radiograph (a) of a 12-year-old FN DSH cat with a mammary carcinoma of the second abdominal gland. A couple of faint, ill-defined soft tissue opacities superimposed on the cardiac silhouette, and suspicious for metastasis, were noted on this view (arrows) but not the right lateral view. CT of the chest confirmed the presence of a 2–3 mm hyperattenuating nodule in the middle right lung lobe and (b) a 4.8 mm hyperattenuating nodule in the caudal part of the left lung lobe (arrow)
Since most affected cats are elderly, haematology, biochemistry and urinalysis should also be performed to assess for concurrent disease. Paraneoplastic conditions are rarely reported with mammary tumours, and feline leukaemia virus and feline immunodeficiency virus are not impli cated in the aetiology. However, if further treatment, including chemotherapy is being considered, it is important to evaluate viral status since viral immunosuppression may influence whether treatment goes ahead.
Surgical treatment
The mainstay of treatment for mammary tumours is still surgical resection. The extent of surgery is influenced by the lymphatic drainage in the feline mammary gland (see box), as tumour cells spread readily beyond the primary site and complete excision should encompass all known drainage pathways.
Recommended approach
The recommendation based on drainage studies is to perform unilateral or bilateral mamma-ry strips because of possible contact between individual glands and between left and right sides. While radiological imaging studies would suggest that this may not be necessary in every case, additional prognostic analyses do support the use of unilateral or bilateral mam-mary strips since the extent of surgery appears to make a significant difference to local recurrence/disease-free interval (DFI) 33 and survival time. 34 For bilateral strips a 2-week interval is recommended between surgeries although simultaneous bilateral mastectomy can also be performed (Figure 10). Tumour fixation to skin or abdominal fascia necessitates removal of these structures en bloc. 35

Bilateral mammary strip in a cat with mammary carcinoma. Courtesy of Kathryn Pratschke

Chemotherapy
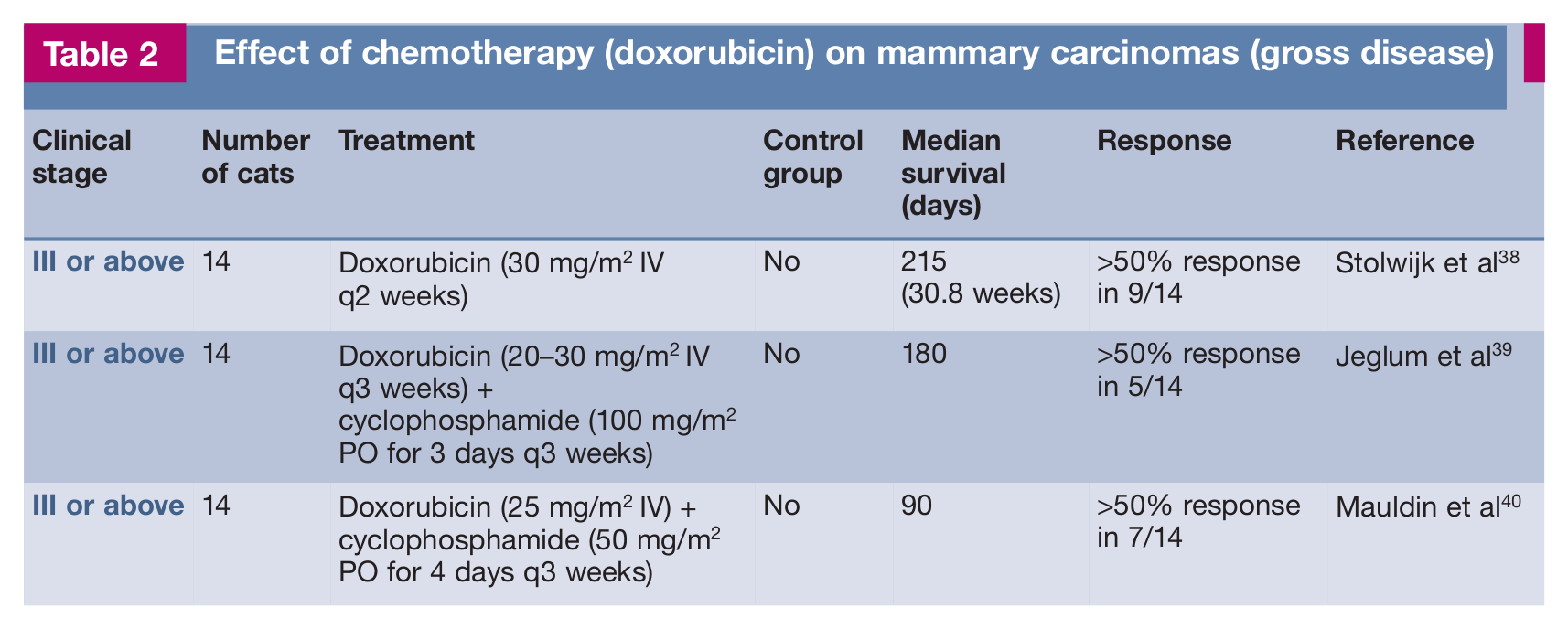
There is some data to show that chemo therapy may be effective on mammary cell lines in vitro,36–38 and that treatment of non-resectable disease in vivo with doxorubicin and cyclophosphamide may shrink tumour size in 50% cases and possibly increase survival (Table 2).38–40 The benefit of using chemotherapy as an adjunct to surgical excision of mammary tumours in cats, however, is still not clear (Table 3).
Effect of chemotherapy (doxorubicin) on mammary carcinomas (gross disease)
Effect of chemotherapy (doxorubicin) as an adjunct to surgical removal of mammary tumours

DFI = disease-free interval
Doxorubicin 1 mg/kg IV q3 weeks
Doxorubicin dose not given (four cases doxorubicin alone)
Cyclophosphamide dose not given
Doxorubicin 1 mg/kg IV q3 weeks (one case had vincristine 0.7 mg/m2 IV and 13 cases had cyclophosphamide 250 mg/m2 IV, 1 week after doxorubicin)
Meloxicam 0.2 mg/kg PO on day of surgery, then 0.1 mg/kg q24h for 5 days, then 0.025 mg/kg
A large, multicentre study of 67 cats receiving adjunctive doxorubicin reported a median survival time of 448 days. 41 Although there was no control group in the study, this survival time was deemed longer than historical controls and was similar to that in another study of 23 cats without a control group (460 days) combining adjunctive doxorubicin with the COX-2 inhibitor meloxicam. 42 A further study of 73 cats, which included a control group of 36 cats undergoing surgical excision only, reported increased survival time and DFI for the cats receiving postoperative doxorubicin and cyclophosphamide (1406 versus 848 days [survival time] and 676 versus 372 days [DFI]); 34 however, the difference was not statistically significant.
It is possible that with greater numbers and more statistical power, a true benefit of aggressive chemotherapy may become apparent. Alternatively, a different approach with anti angiogenic metronomic (low dose) chemotherapy may prove effective, although low dose chemotherapy using vincristine, cyclophosphamide and methotrexate did not prevent recurrence or metastasis in one report. 8
Other therapies
Although immunomodulators such as bacillus calmette-Guerin (BCG), 43 Corynebacterium parvum, 44 liposome-encapsulated muramyl tripeptide phosphatidylethanolamine (L-MTP-PE) 45 and oral levamisole 46 have been tried as intratumoural injections (BCG) or adjuncts to surgical excision of feline mammary tumours, none have proved successful in extending survival time or altering recurrence rate. There are no reports of using antioestrogens such as tamoxifen in cats, probably since most malignant feline mam-mary tumours lack oestrogen receptors and expected benefits would, therefore, appear to be minimal.
The small molecule inhibitors that target receptor tyrosine kinases (receptor tyrosine kinase inhibitors or RTKIs) are effective in the management of some types of veterinary cancers, particularly those with altered TK activity. 47 Imatinib and masitinib are well tolerated in cats;48–51 however, there is no information about their efficacy against feline mammary tumours. (Further discussion on the use of targeted therapies in cats, with a particular focus on the idiosyncracies of feline patients, is provided in an accompanying article in this special issue.)
Prognosis
Prognosis is guarded for most cats with mammary tumours, with deaths mainly attributable to local recurrence or metastasis. The average time between detection and death is reported to be 10–12 months;20,35 however, as already mentioned, several factors affect prognosis of feline mammary tumours (Table 4).
Prognostic factors for feline mammary tumours

Clinical parameters
Prognosis is very much related to the size of the tumour at initial presentation, with tumours of large volume (>27 cm3) or diameter (>3 cm) being associated with shorter survival times (4–12 months).8,52,53 The degree of spread at initial presentation (eg, regional lymph node metastasis) also dramatically affects prognosis,20,33,53 as does the extent of surgery carried out. Radical mastectomy produces a significantly longer DFI,33,53 and histological completeness of resection correlates with survival. 27 The cat’s age may also influence prognosis,27,33 although more recent studies dispute this. 53
Histological parameters
A wealth of literature exists on histological markers detected by pathologists, 54 some of which influence prognosis. However, few are routinely used in diagnostic laboratories to offer helpful information to clinicians.
The most useful assessment by pathologists is histological grading, which is significantly related to both overall survival and DFI in univariate and multivariate analyses.55–57 Mitotic index also correlates with survival time, 58 but although other proliferation markers such as Ki67,57,59 AgNOR 60 and PCNA 61 may help determine high histological grade, they have not been shown to be independent prognostic markers. Similarly, although expression of hormone receptors (ER and PR) has been examined in feline carcinomas, receptor status does not correlate with overall survival and is not routinely assessed.14,15 The epidermal growth factor receptor 2 (Her2/neu) is variably over-expressed in feline carcinomas and, while one study has associated this with survival, 62 more recent data has not. 63 High COX-2 expression in mammary carcinomas has also been linked with poor prognosis, 64 but this association is complicated by the fact that it also correlates with expression of the angiogenic factor vascular endothelial growth factor (VEGF), which itself is significantly correlated with overall survival. 65 More recently, expression of the cytoplasmic protein kinase, AKT, which is activated by numerous receptor tyrosine kinases such as EGF or by loss of the tumour suppressor gene PTEN (phosphatase and tensin homologue), has been significantly associated with shorter DFI. 66
Footnotes
Key Points
Case notes
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The author does not have any potential conflicts of interest to declare.
