Abstract
A 4-year-old domestic shorthair cat presented with tetany. Laboratory testing confirmed severe hypocalcemia and primary hypoparathyroidism. The cat subsequently developed congestive heart failure secondary to myocardial failure and was treated with pimobendan, enalapril, furosemide, calcitriol and calcium salts. All clinical signs resolved and cardiac function returned to normal within 1 month, and remained normal after cessation of all medications except calcitriol. Hypocalcemia-associated cardiomyopathy is a rare, reversible condition that has not previously been reported in a veterinary patient, and it should be considered as a differential diagnosis in patients with myocardial failure.
Case Report
A 4-year-old, neutered male domestic shorthair cat presented to the hospital with a 4-day history of tetany, lethargy and decreased appetite progressing to anorexia. On physical examination, the cat had altered mentation (periodically obtunded, disoriented and, at times, hypersensitive to stimuli), mydriasis oculus urteque and generalized tetany. The patient was an indoor cat with no known exposure to topical flea and tick preventatives, or other toxins. The client reported that the patient had been episodically hypersensitive to stimuli for the past month, and that these episodes were becoming more pronounced prior to the onset of tetany. Review of the past medical history revealed that the patient had presented with a complaint of episodes of sneezing 2 months prior. At that time, a complete blood cell count (CBC) revealed mild neutrophilic leukocytosis (Table 1). Serum biochemical abnormalities included mildly increased alanine aminotransferase (ALT) activity, mild hypocalcemia (7.8 mg/dl) and mildly increased creatinine kinase (CK) activity (504 IU/l) (Table 2). Serum ionized calcium level was not evaluated. A total thyroxine (T4) was normal (0.8 μg/dl; 0.8–4.0 μg /dl). Urinalysis, obtained by cystocentesis, revealed well-concentrated urine [urine specific gravity (USG) 1.047] and a benign sediment. At that time, the patient was treated with marbofloxacin (Zeniquin; Pfizer), with treatment completed several days prior to the current presentation.
Hematology results for the patient during the course of treatment (all testing by Antech Diagnostics, a veterinary reference laboratory)

Indicates values above the reference interval, †Indicates values below the reference interval, RBC = red blood cells; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; WBC = white blood cells
Selected serum biochemistry values for this patient during the course of treatment (all testing by Antech Diagnostics, a veterinary reference laboratory)
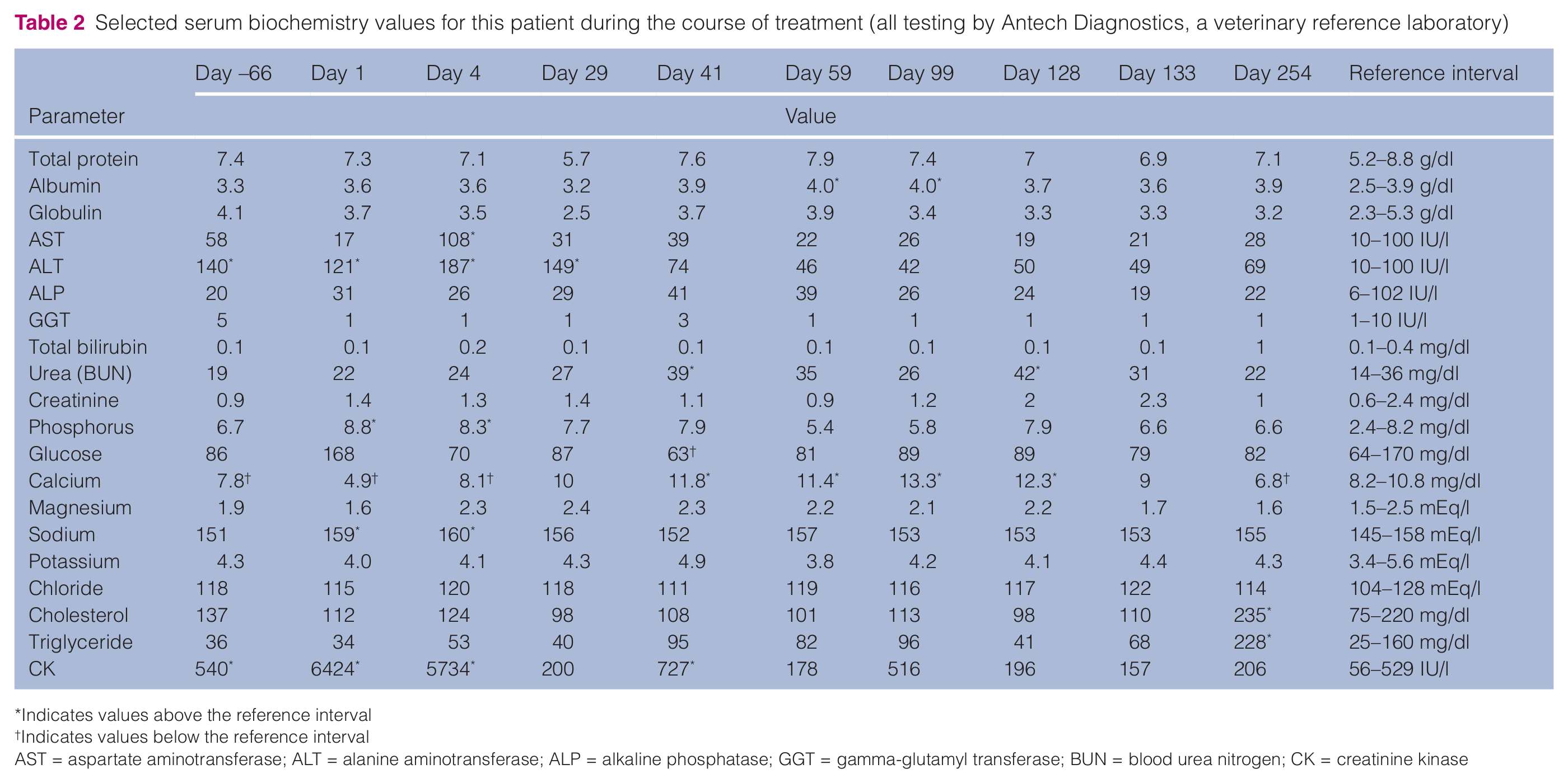
Indicates values above the reference interval
Indicates values below the reference interval
AST = aspartate aminotransferase; ALT = alanine aminotransferase; ALP = alkaline phosphatase; GGT = gamma-glutamyl transferase; BUN = blood urea nitrogen; CK = creatinine kinase
Baseline vital parameters on admission included the following: body weight = 4.9 kg; body condition score (BCS) = 4/9; rectal temperature = 39°C; respiratory rate (RR) = 40 breaths per minute with normal effort; heart rate = 190 beats per minute with regular rhythm and strong femoral pulses; blood pressure = 130/75 mmHg; mean arterial pressure 95 mmHg (PetMAP; Ramsey Medical). Ophthalmologic examination was unremarkable apart from sluggish pupillary light reflexes (direct and consensual). The patient was hospitalized and given intravenous (IV) sodium chloride 0.9% qs 20 mEq/l KCl (Hospira) at a rate of 4 ml/kg/h. A CBC revealed a mild neutrophilic, eosinophilic leukocytosis (Table 1). Serum biochemical abnormalities included mildly increased ALT activity, mild hyperphosphotemia, marked hypocalcemia (4.9 mg/dl), normal serum albumin, mild hypernatremia, and markedly increased CK activity (6424 IU/l) (Table 2).
Initial treatment for hypocalcemic tetany consisted of calcium gluconate (APP Pharmaceuticals) 250 mg (1.16 mEq) IV q6h. Differential diagnoses that were considered for the marked hypocalcemia included primary hypoparathyroidism, pancreatitis, nutritional secondary hyperparathyroidism and spurious (laboratory error). Given the history of previous mild hypocalcemia, unusual behavior and lack of gastrointestinal signs, diagnostic testing for pancreatitis was not pursued. Nutritional secondary hyperparathyroidism was less likely given the normal dietary history of a veterinary therapeutic diet (Iams Veterinary Low-Residue dry and canned diets) owing to another cat with gastrointestinal disease in the household. Primary hypoparathyroidism was diagnosed based on an inappropriately low parathyroid hormone (PTH) concentration (14.3 pg/ml; 0–36 pg/ml; Antech Diagnostics) given a marked ionized hypocalcemia (0.68 mmol/l; 1.16–1.34 mmol/l; Antech Diagnostics). Although the patient’s PTH fell within the published reference interval, an appropriate physiologic response to this level of hypocalcemia would require an elevated PTH concentration. 1 Also, the patient had hyperphosphatemia (8.8 mg/dl; 2.4–8.2 mg/dl; Antech Diagnostics) and elevated ALT activity (140 IU/l; 10–100 IU/l) at the time of initial diagnosis, and these change have been reported in 8/9 cases and a third of cases, respectively, of feline primary hypoparathyroidism. 1 Continued treatment included IV crystalloid fluids (4 ml/kg/h, 1.5× maintenance) and calcium gluconate [250 mg (1.16 mEq) IV q6h].
The following morning the patient was slightly agitated with improved tetany, body weight of 5.2 kg, a BCS of 4/9, a rectal temperature of 39.9°C, a respiratory rate of 50 breaths per minute with mildly increased respiratory effort, and a heart rate of 200 beats per minute. The IV fluid rate was decreased to 3 ml/kg/h and thoracic radiographs revealed a moderate-to-marked, diffuse interstitial pulmonary pattern in the cranioventral and left and right caudal lung lobes, mild pleural effusion as evidenced by rounding of the lung lobe margins and moderate generalized cardiomegaly (Figure 1a, b). The main differential diagnoses for the interstitial pulmonary infiltrates included cardiogenic pulmonary edema (precipitated by IV fluid resuscitation in a patient with occult cardiac disease), non-cardiogenic pulomonary edema (neurogenic, acute lung injury, vasculitis, impaired lymphatic drainage), pneumonia (aspiration versus infectious) or pneumonitis. The IV fluid administration rate was further decreased (2 ml/kg/h), and within 10 h the patient’s respiratory rate and effort had normalized. Diuretic therapy was not initiated. As the patient’s respiratory rate and effort were improving, no antibiotics were administered and pneumonia was considered less likely. Owing to persistent hypocalcemia, the dose of calcium gluconate was increased to 2.32 mEq IV q6h. The patient’s tetany resolved gradually over the first 48 h of treatment and, after 4 days of hospitalization, the patient was discharged for home care on oral calcitriol ((vitamin D3, Rocaltrol; Roche) 250 μg PO q24h and calcium carbonate 500 mg PO q24h.

Right lateral (a) and ventrodorsal (b) thoracic radiographs (day 2). There is a moderate diffuse interstitial pulmonary pattern that is more severe in the right and left caudal lung lobes (ventrodorsal view), and mild pleural effusion evidenced by rounded lung lobe margins on the lateral view. There is moderate cardiomegaly (vertebral heart size = 9.5). These findings are consistent with cardiogenic pulmonary edema due to congestive heart failure
The patient re-presented on day 29 with a 1-day history of acute dyspnea, and the client reported that the patient’s respiratory rate and effort had gradually increased over a 2-week period. On presentation, the cat was tachypneic (RR = 50) and mildly dyspneic with visible abdominal effort, but was clinically stable with a heart rate of 180 beats per minute, a body weight of 4.74 kg and a BCS of 4/9. A CBC was unremarkable, serum biochemistry was unremarkable except for a mildly increased ALT 149 (10–100 IU/l) with normal total calcium and creatinine kinase activity, and urinalysis was unremarkable, including a USG of 1.043 (1.015–1.060) and a benign sediment. Thoracic radiographs revealed moderate pleural effusion, a marked generalized interstitial pulmonary pattern and marked cardiomegaly (Figure 2a, b). An echocardiogram (GE Logiq 5) was performed and revealed moderate-to-severe left atrial (LA) dilation [LA diameter = 19.3 mm (12.1 ± 1.8 mm), LA diameter to aortic (Ao) diameter ratio (LA:Ao) = 1.79 (1.29 ± 0.23)], severe left ventricular eccentric hypertrophy [increased left ventricular end diastolic diameter (LVd) = 23.0 mm (15.1 ± 2.1 mm)] and severe myocardial failure [increased left ventricular end systolic diameter (LVs) = 18.0 mm (6.9 ± 1.1 mm), decreased left ventricular fractional shortening (FS) = 21.7% (55.0 ± 10.2%)]. 2 These abnormalities were consistent with a diagnosis of severe myocardial failure and congestive heart failure, with differentials including idiopathic dilated cardiomyopathy, taurine deficiency-induced myocardial failure, myocarditis and hypocalcemia-induced myocardial failure. Although there was a diet history of Iams Veterinary Formula Low-Residue canned and dry, a whole blood taurine level was submitted to evaluate for taurine deficiency (testing performed at the Wisconsin Veterinary Diagnostic Laboratory). Treatment was initiated with furosemide 2 mg/kg IM, and the patient’s respiratory rate and effort improved dramatically. As the patient responded well to this initial therapy, it was deemed unnecessary to perform therapeutic thoracocentesis. The cat was discharged on furosemide 10 mg PO q12h (2.1 mg/kg), enalapril 2.5 mg PO q12h, pimobendan (Vetmedin; Boehringer Ingelheim) 0.625 mg PO q12h and taurine 250 mg PO q24h while awaiting the taurine blood levels. The current doses of calcitriol and calcium carbonate were continued.

Right lateral (a) and ventrodorsal (b) thoracic radiographs (day 29). There is marked diffuse pulmonary interstitial pulmonary pattern, and moderate pleural effusion that partially obscures the cardiac silhouette. There is progressive severe cardiomegaly (vertebral heart size = 10.0) These findings are consistent with worsening of the congestive heart failure
On day 41, the patient was presented for follow-up evaluation. The client reported that the patient had not had any further dyspneic episodes, despite a lower furosemide dose of 1 mg/kg PO q12h. The whole blood taurine level was normal [460 nmol/ml (300–600 nmol/ml)] and the taurine supplement was discontinued. Recheck CBC revealed a mild eosinophilia (Table 1). Serum biochemistry profile revealed mild azotemia and hypercalcemia (Table 2). A T4 level was repeated, and it confirmed that the patient remained euthyroid (1.3 μg/dl). The USG was 1.040 (1.015–1.060) and the remainder of the urinalysis was unremarkable. The patient was eupneic (RR = 24) with quiet bronchovesicular lungs sounds and normal respiratory effort. The patient’s doses of calcium carbonate and furosemide were both decreased.
One month after congestive heart failure was diagnosed and 59 days after treatment of hypocalcemia was instituted, the patient was presented for recheck evaluation and cardiac imaging, including echocardiography. The patient was asymptomatic and thriving. Thoracic radiographs were normal, with resolution of pulmonary infiltrates, pleural effusion and cardiomegaly (Figure 3a, b). Compared to only 4 weeks prior, the echocardiogram revealed a normalized left atrial diameter (LAd) [LAd= 11.9 mm (previous 19 mm), LA:Ao = 1.54 (previous 1.79)], normal-to-near-normal left ventricular size [LVd = 17.2 mm (previous 23 mm)], markedly improved and borderline–to-mild myocardial failure [LVs = 10.2 mm (previous 18 mm), FS 41% (previous 21%)]. The CBC and serum biochemistry profiles were unremarkable (Tables 1 and 2). Given the dramatic improvement in the patient’s cardiac size and function, the calcium carbonate and remaining cardiac medications were tapered incrementally over the next 4 weeks.

Right lateral (a) and ventrodorsal (b) thoracic radiographs (day 59). There is complete resolution of the pulmonary infiltrates and pleural effusion. The heart has returned to normal size (vertebral heart size = 7.5). This is a normal thoracic study
Approximately 6 weeks later (day 99), the patient was presented for a recheck evaluation and echocardiography. Calcitriol was the only medication being given; all cardiac medications had been stopped for 2 weeks. The echocardiogram revealed normalized left atrial size (LAd = 12 mm, LA:Ao = 1.44), smaller and normalized left ventricular size (LVd = 13.1 mm) and normalized systolic function (LVs = 5.5 mm, FS = 58%). The CBC was unremarkable (Table 1). The serum biochemical profile revealed persistent hypercalcemia (Table 2). A urinalysis revealed a USG of 1.035 and benign sediment. The PTH was 6.3 pg/ml (0–36pg/ml) with ionized calcium 2.03 mmol/l (1.16–1.34 mmol/l). In response to persistent hypercalcemia, the calcitriol dosing frequency was adjusted from q12h to q24h.
On day 128 the patient was presented for evaluation of polyuria and polydipsia. A CBC was unremarkable (Table 1). Serum biochemical profile revealed azotemia and hypercalcemia (Table 2). Urinalysis revealed a reduced USG of 1.025 compared with the patient’s previous values of 1.035–1.040. Calcitriol therapy was discontinued temporarily with a plan to re-initiate therapy in 5–7 days after assessment of calcium levels. The patient was presented on day 133 to monitor serum calcium concentration and renal values. The azotemia and hypercalcemia had both resolved (Table 2), but there was a persistently decreased USG of 1.019. Calcitriol therapy was reinstituted at a dose of 250 µg PO twice weekly. At a follow-up visit on day 254 the patient was doing well clinically. Recheck CBC was unremarkable and the patient was once again able to concentrate urine USG 1.080 (1.015–1.060). However, the serum biochemistry profile revealed mild hypocalcemia (Table 2). Approximately 1 year after the diagnosis of the congestive heart failure (day 399), the patient remained asymptomatic, continued to receive twice-weekly calcitriol, and had no recurrence of heart failure or need for cardiac medications. A recheck CBC was unremarkable. The serum biochemistry profile revealed persistent mild hypocalcemia (7.1 mg/dl). As the patient was due for the twice-weekly dose of calcitriol later that same day, no dosage adjustment was made.
To our knowledge, this is the first reported case of hypocalcemia-induced cardiomyopathy in the veterinary literature. This clinical syndrome has been reported in several individual case reports in the human medical literature, with typical causes including hypoparathyroidism (often post-thyroidectomy) or Rickets disease due to vitamin D deficiency.3 –16 In most of these human cases, chronic hypocalcemia of months to years leads to congestive heart failure, which often improves with restoration of normocalcemia. We suspect that the cat in this current report had chronic hypocalcemia, which was documented 2 months prior to presentation by a mild (total) hypocalcemia. People with hypocalcemia-induced cardiomyopathy typically have a marginal response to standard heart failure medications, including diuretics, digoxin and angiotensin converting enzyme inhibitors, and demonstrate marked improvement once the underlying hypocalcemia and/or hypoparathyroidism have been identified and corrected.4,5,7,8,10,11,15 Hemodynamic improvement has been identified within weeks after initiating treatment for hypocalcemia, and cardiac function is often normalized within 2–5 months.3,7 Some patients with chronic hypocalcemia-induced cardiomyopathy may show a delayed response, likely due to structural myocardial changes, including fibrosis, muscle fiber degeneration and myofiber atrophy.3,7 In some human cases, the hypocalcemia has been identified at the time of initial diagnosis and the patient treated immediately with calcitriol and oral calcium salts along with diuretics and digoxin.13,15 In one case the patient was treated with calcium and dihydrotachysterol (vitamin D) alone and achieved both remission of clinical signs and improved myocardial function. 9 Many reported cases have achieved full and lasting recovery of cardiac function after supplementation with calcium salts and calcitriol,4,5,9,10,12,15 regardless of initial therapy and despite discontinuation of cardiac medications, as was seen in the cat reported herein. The requirement for calcitriol ± calcium supplementation is apparently lifelong, as two reports have documented relapse in patients who discontinued calcium or vitamin D supplementation and subsequent resolution after supplementation was reinstituted.8,13
Calcium is required for neuromuscular transmission and cell membrane stabilization during muscle contraction. Hypocalcemia increases the permeability of cell membranes to sodium, leading to increased excitability and subsequent spontaneous discharge of nerve fibers. This causes the muscle tetany associated with hypocalcemia. Generally, there must be a significant decrease in total serum calcium concentration to ≤6 mg/dl (or ionized calcium must decrease to ≤0.7 mmol/l) to develop clinical evidence of tetany.11,17 The involuntary muscle contraction of hypocalcemia-induced tetany results in an elevated CK enzyme activity. Skeletal myopathy or acute rhabdomyolysis may develop secondary to hypocalcemia and contribute to an elevated CK enzyme activity, which typically resolves after normalization of serum calcium concentration.
Myocardial contractility is determined by the amount of actin and myosin cross-bridge cycling within the cardiomyocyte, which requires binding of calcium to troponin C. Intracellular calcium concentration is regulated by the amount of calcium stored in the sarcoplasmic reticulum and mitochrondria, and by extracellular calcium entry into the cardiomyocyte through calcium channels. Adequate intracellular calcium concentration is necessary for myocardial contraction and relaxation.7,16 If the extracellular calcium stores (ie, serum calcium concentration) become depleted, this, in turn, depletes intracellular calcium resulting in decreased cardiomyocyte contractions and myocardial failure. Because the correction of blood (extracellular) calcium concentration does not restore intracellular calcium levels immediately, there is a delay before myocardial contractility improves after serum hypocalcemia is addressed, as was seen in this patient.
In addition to the effects of calcium on myocardial function, there is a direct effect of PTH on cardiomyocytes, including increased calcium entry into the cardiomyocytes and release of myocardial norepinephrine. Thus, decreased PTH may contribute to myocardial failure in patients with primary hypoparathyroidism. Additionally, hypocalcemia reduces renal excretion of sodium, which may exacerbate intravascular volume overload and contribute to the development of congestive heart failure. A ‘vicious cycle’ may develop when furosemide is instituted for treatment of congestive heart failure, as this increases calciuria and may aggravate hypocalcemia, thus worsening the degree of myocardial failure and increasing the severity of heart failure. Unless critical hypocalcemia is identified and corrected, standard heart failure therapies are typically unsuccessful in stabilizing a patient with hypocalcemia-induced myocardial failure.
Regulation of PTH secretion is dependent primarily upon changes in extracellular calcium concentrations. 1 There is an inverse sigmoidal relationship between extracellular calcium concentration and PTH secretion, meaning that, in the face of hypocalcemia, the PTH secretion is normally increased dramatically. However, there is a non-suppressible element to PTH secretion and this may explain the detectable levels of PTH present even with high serum calcium levels. 1 At normal calcium concentrations there is near-maximal suppression of PTH synthesis; thus, the parathyroid gland is set to up-regulate PTH synthesis in response to even minor degrees of hypocalcemia.1,17–19 In a patient with severe hypocalcemia, a measured PTH level that is in the lower half of the reference interval (as seen in this patient) is considered inappropriately low and indicates that the patient has clinical hypoparathyroidism. In cats, the most common cause of hypoparathryroidism and hypocalcemia is surgical thyroidectomy with compromise of adjacent parathyroid tissue. Primary or naturally-occurring hypoparathyroidism is a rare condition that has been reported previously in cats, though it appears to be more common in young cats with a mean age of presentation at 1.8 years and range of 0.5–6.7 years.17,18 The treatment of hypoparathyroidism is similar whether primary or surgically-induced. Initial stabilization with IV calcium gluconate is followed by maintenance therapy with oral vitamin D and calcium supplementation. Calcitriol is the one of the most common forms of vitamin D used for supplementation. Although lifelong calcitriol therapy is often required, it is generally possible to taper the level of calcium supplementation over the course of 2–4 months and eventually discontinue. 1
In the case reported herein, hypocalcemia-induced myocardial failure was diagnosed after consideration of other common causes of myocardial failure in cats. Taurine deficiency-induced myocardial failure was excluded after documentation of a normal whole blood taurine concentration. Idiopathic dilated cardiomyopathy was ruled out based on the rapid recovery of myocardial function and normalization of cardiac size within a few weeks of therapy for hypocalcemia, despite discontinuation of cardiac medications. Myocarditis was thought to be unlikely as the patient did not have a fever or demonstrate an inflammatory leukogram. Furthermore, the patient showed rapid recovery of function upon electrolyte normalization.
In retrospect, the interstitial pulmonary infiltrates and clinical signs of tachypnea at the time of initial diagnosis of hypocalcemia were the early stages of congestive heart failure secondary to severe myocardial failure, which progressed to fulminant congestive heart failure with development of pleural effusion and more pronounced pulmonary edema. Had vertebral heart size (VHS) measurements been used when the films were taken, it could have allowed for earlier detection of the patient’s cardiomyopathy. VHS was measured subsequently on all three sets of radiographs. 20 The VHS for the first two sets of films was markedly increased at 9.5–10 (reference interval 7.2–7.8 vertebrae), although it is difficult to measure cardiac size precisely owing to the presence of pulmonary infiltrates obscuring the edge of the cardiac silhouette. This difficulty alone could have provided early insight into the severity of this patient’s cardiac disease if VHS measurement had been measured on initial radiographs at the first onset of respiratory signs. On the third set of films, the VHS was 7.5, demonstrating a decrease in heart size into the normal reference interval. Unfortunately, diagnosis of myocardial failure in cats is often delayed until fulminant signs are present owing to the absence of a murmur in a majority (83%) of these patients. 21
The azotemia that developed during treatment of hypocalcemia was considered secondary to vitamin D intoxication from over-supplementation with calcitriol. Hypercalcemia is a potentially serious complication that may occur during treatment of hypoparathyroidism if calcium levels are not monitored carefully and medications adjusted appropriately. Hypercalcemia can lead to secondary nephrogenic diabetes inspidius and loss of the renal concentration gradient, and eventual metastatic mineralization in the kidneys. All three of these conditions cause polyuria and polydipsia. Left unchecked, these conditions may cause renal injury leading to renal failure.17,18 If renal injury is identified early, it can be reversed upon cessation or decrease in the dose of calcitriol administered. In the case reported herein, both the hypercalcemia and azotemia resolved after temporary discontinuation then subsequent lowering of the calcitriol dose.
A complete assessment of parathyroid function with testing of both total serum calcium and ionized calcium levels should not be overlooked in veterinary patients with myocardial failure. If hypoparathyroidism is identified, the patient may show only marginal improvement in response to standard treatment for myocardial failure, and could show dramatic improvement or even normalization of cardiac function with appropriate therapy for hypocalcemia.
There were a number of limitations of our case report owing, primarily, to client financial constraints in a private general practice setting. More extensive measurements for electrolyte abnormalities beyond those included on standard chemistry panels (ie, ionized magnesium levels, urine excretion of calcium and magnesium) were not pursued. In human patients, alterations in serum ionized magnesium concentrations may cause reversible hypoparathyroidism, which can occur in the face of normal total serum magnesium because total serum magnesium testing may not reflect intracellular magnesium levels. Hypomagnesemia typically causes decreased serum PTH levels, but only mild hypocalcemia. PTH secretion in these patients normalizes once normal magnesium levels are restored, and long-term supplementation is not required. Also, serum total magnesium eventually drops in human patients with persistent ionized hypomagnesemia.17,19 Although ionized hypomagnesemia cannot be ruled out in our patient as this was not tested, it is considered unlikely to be the primary cause of persistent hypoparathyroidism. The total serum magnesium was within the reference interval at all time points tested, the degree of hypocalcemia was severe and the serum PTH levels remained persistently low. In humans, urine excretion of calcium and magnesium is often used in the diagnosis and treatment of parathyroid disorders, but this testing is not performed routinely in veterinary patients owing to cost and complications associated with collection. Additional evaluation of the parathyroid glands with cervical ultrasonography or parathyroid biopsy was not performed. The PTH assay, in conjunction with calcium testing and evaluation of renal values, is considered the gold standard in the diagnosis of hypoparathyroidism in small animal patients. Financial constraints and the potential risk to the patient with anesthesia and biopsy also precluded the additional evaluation. The patient lived strictly indoors, with no potential exposure to flea preventatives as an alternative explanation for the tetany. While testing for pyrethrin intoxication could have been performed, it was considered likely to be of low diagnostic yield and the clients elected not to pursue it. A urinalysis was not performed at initial presentation or during the period of hospitalization (owing to oversight), but these results would have been useful. Evaluation of myocarditis by measurement of cardiac troponin I or myocardial biopsy, was not performed. Antemortem diagnosis of myocarditis in cats is limited, as endomyocardial biopsies are not feasible in this species owing to risk of death or severe complications. Cardiac troponin I may be measured as an indicator of myocyte damage or death, but is non-specific for a particular disease process. Typically, very high concentrations may be seen with acute myocardial infarction or myocarditis, but an elevated cardiac troponin I level may also be seen in other cardiac diseases, including dilated cardiomyopathy. While having serial measurements of cardiac troponin I would have been interesting in this cat to follow possible improvements as the myocardial failure improved, it would not provide a definitive diagnosis of the etiology of the heart disease, and was not essential for the diagnosis of myocardial failure secondary to hypocalcemia. Systemic blood pressure measurement was not performed when the patient had myocardial failure (owing to oversight); however, these results would have provided additional (complementary) hemodynamic data. Finally, electrocardiogram evaluation would have been interesting in this patient to assess for the known electrocardiographic abnormalities associated with hypocalcemia, 17 such as QT prolongation, but was not necessary to determine the diagnosis or treatment, and was not performed.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
