Abstract
Meningioma is the most frequently observed primary brain tumour in cats. Usually, it is associated with an intracranial expansion with consequent brain compression, oedema and brain herniation. Typical features of feline intracranial meningiomas are hyperostosis of the adjacent bone and intratumoral mineralisation. We describe a 13-year-old male neutered cat with a 1-year history of behavioural change. At clinical and neurological examination the cat showed signs consistent with right-sided forebrain lesion. Magnetic resonance images showed a right-sided extra-axial contrast enhancing mass in the region of the frontotemporal lobe. The overlying bone of the calvarium showed a marked defect with extracranial expansion of the tissue. Surgery was performed and the tumour could be exposed by a right-sided temporal approach. After extension of the bony defect the mass could be removed properly. The cat recovered well from surgery and a 12-month follow-up showed no persistent neurological deficits. Histopathological assessment of the tumour revealed a transitional grade 1 meningioma. Despite osteolysis and extracranial expansion of the tumour differentials should include menigioma in feline intracranial neoplasms.
Case Report
Meningioma (59%) is the most frequently observed primary brain tumour in cats. 1 Meningiomas typically arise from arachnoid cap cells or granulations. Intraventricular meningiomas can also originate from the tela choroidea, the pia mater or the choroid plexus. 2 Predilection sites for feline meningiomas are rostrotentorial (92.4% of cases), frequently within the third ventricle, over the parietal and the temporal lobes. 1 They grow initially into the subarachnoid space and subsequently lead to brain compression due to their expansive growth. A typical feature of feline meningioma is hyperostosis of the skull (73%) adjacent to the neoplasia. 3
Meningioma-associated skull osteolysis has only been described sparsely in humans.4,5 Two case reports refer to a feline intracranial meningioma associated with osteolysis and subsequent erosion of the adjacent bone.6,7 In both cases the tumour did not expand outside of the calvarium. Infrequently, meningiomas may extend into paranasal regions and metastasise outside the brain. 8 A special form of meningioma may occur as a primary extracranial mass as a result of embryonic displacement of arachnoid cells or meningocytes. 9
We describe a histologically benign, frontotemporal meningioma with bony osteolysis associated with extracranial extension of the tumour.
A 13-year-old male neutered domestic shorthair cat was referred to our institution with a 1-year history of behavioural changes. The first signs were episodic lethargy and reluctance to play, followed by episodes of aggressiveness. Three months prior to presentation the owner observed pain reactions when touching the head of the animal.
Complete blood count, serum biochemistry and ultrasound of the abdomen revealed no abnormalities. The cat was first treated with meloxicam (Metacam; Boehringer Ingelheim, initial dose 0.1mg/kg, maintenance 0.05 mg/kg q24h for 5 days) and also robenacoxib (Onsior; Novartis, 1.2 mg/kg q24h for 5 days) without success. An osteolytic lesion in the right temporal bone was detected on skull radiographs. Magnetic resonance imaging (MRI) was performed and a contrast enhancing mass with osteolysis of the skull and compression of the brain was diagnosed. The cat was treated with (Prednisolone; CP-Pharma, 0.5 mg/kg q24h PO irregularly during the following 30 days prior to surgery), resulting in mild improvement. The patient was subsequently referred to our clinic for treatment.
The clinical examination was unremarkable. Neurological examination revealed primarily behavioural changes. The cat was reluctant to manipulation and showed aggressiveness. The menace response was absent on the left and reduced on the right. The cotton ball test was normal on the right side and questionable on the left. The cat showed a hypersensibility to facial stimulation. There were no other cranial nerve abnormalities. In addition, the cat showed left-sided deficits when performing wheel-barrowing, hopping and the extensor postural thrust. Additional postural reactions and spinal reflexes could not be evaluated owing to the patient’s reluctance to being handled. Based on the history and results of the neurological examination, the lesion was localised in the forebrain with right-sided lateralisation. Differential diagnosis primarily included neoplastic and inflammatory lesions.
The MRI examination 6 weeks before presentation (Figure 1) showed a right-sided, round-shaped, space-occupying lesion in the calvarium, with a diameter of about 2.5 cm and a well-defined, but slightly irregular, contour extending in the right frontal and temporal lobe and outwards in the temporal musculature. At the location of the lesion, the bone was missing and the adjacent bone showed marked hyperostosis. The lesions caused a marked midline shift to the left. The lesion was heterogenous in T2-weighted images with an isointense periphery and a hyperintense centre, and slightly hypointense in FLAIR images. In some transverse T1-weighted and T2-weighted images a faint hypointense linear rim between the lesion and the brain parenchyma was visible. In the pre-contrast T1-weighted sequence the lesion was isointense with marked, almost homogenous, contrast uptake and a dural tail sign dorsally. In T2-weighted and FLAIR sequences there was a hyperintense signal in the surrounding white matter, interpreted as peri-tumoural oedema. The imaging diagnosis was an extra-axial, right-sided, space-occupying lesion expanding throughout the bone into the adjacent soft tissues. Differential diagnosis for this lesion included meningioma, lymphosarcoma, histiocytic sarcoma and germ cell tumour. Osteochondroma, osteoblastoma, fibrosarcoma, plasmacytoma or a granuloma were considered less likely. Owing to high suspicion of a meningioma in spite of the extracranial expansion and in agreement with the owner, surgery was planned for a complete resection or an excisional biopsy if resection was unsuccessful.

Transverse magnetic resonance images at the level of the thalamus (a–c) and more caudal at the level of the midbrain (d–f). The right-sided, space-occupying lesion shows heterogeneous signal intensity on T2-weighted images (a, d), and is isointense in T1-weighted images (b, e) with strong contrast enhancement (c, f) and a dural tail sign (f, black arrow head). The calvarial defect is visible and the mineralisation is visible as a faint hypointense line between the lesion and the brain parenchyma (a, c, black arrow). The dorsal hyperostosis is visible in all sequences (b, white asterisk)
The cat was anaesthetised for a pre-operative computed tomography (CT) scan and the subsequent surgery. It was premedicated with methadone (Methadon; Streuli, 0.2 mg/kg IV) and induced with propofol (Propofol 1% Fresenius; Fresenius Kabi, 4 mg/kg IV).After intubation the anaesthesia was maintained with 1.5–2% isoflurane (Attane; Minrad, 1–2%) and oxygen. Lactated Ringer’s solution (Plasma Lyte; 10 mg/kg/h IV) was administered throughout the whole procedure. For prophylactic antimicrobial therapy cefazolin (Kefzol; TEVA, 22 mg/kg IV) was administered before, and at the end of, surgery.
The pre-operative CT (Figure 2) showed the space-occupying lesion extending throughout the bony defect with slightly heterogenous contrast uptake in the described region delineated to the musculature laterally and to the brain medially by a faint mineralised and discontinuous rim. The adjacent bone was markedly thickened and sclerotic.

Transverse pre-operative computed tomography image shows the space-occupying lesion extending throughout the bony defect. There is a faint mineralised and discontinuous rim bordering the mass laterally to the musculature and to the brain medially. The adjacent bone is thickened and sclerotic
The cat was positioned sternally with the head tilted slightly to the left and elevated without impairment of the upper airways. The hair was clipped from the eyelids to the level of C2, and the skin scrubbed with chlorhexidine and disinfected with povidone–iodine. The cat was pretreated intravenously with methylprednisolone sodium succinate (Solu-Medrol; Pfizer, 30 mg/kg IV) and mannitol (Mannitol 20%; Bichsel AG, 1 g/kg IV) to minimise surgically-induced brain oedema and inflammation.
A right-sided temporal approach was performed to visualise the tumour within the temporal muscle mass. The bony defect was extended cranially and caudally removing the irregular and thickened bone allowing exposition of the whole external tumour circumference. After debulking and shrinking the tumour by enucleating its centre with the single window technique, the firm and irregularly-shaped mass could be removed en masse without extensive haemorrhage. The wound was closed routinely.
The postoperative CT scan showed subcutaneous soft tissue swelling and emphysema extending to the bone defect. The lesion with its mineralised borders was removed and there was no longer contrast-enhancing tissue within the cranium, but some hyperdense (HU 68) areas within the brain parenchyma were present, which were interpreted as haemorrhage.
The histopathological assessment of the tumour showed an ill-defined mass growing in the adjacent musculature (Figure 3). The diagnosis was a transitional grade 1 meningioma (Figure 4) (World Health Organization 2007 10 ).
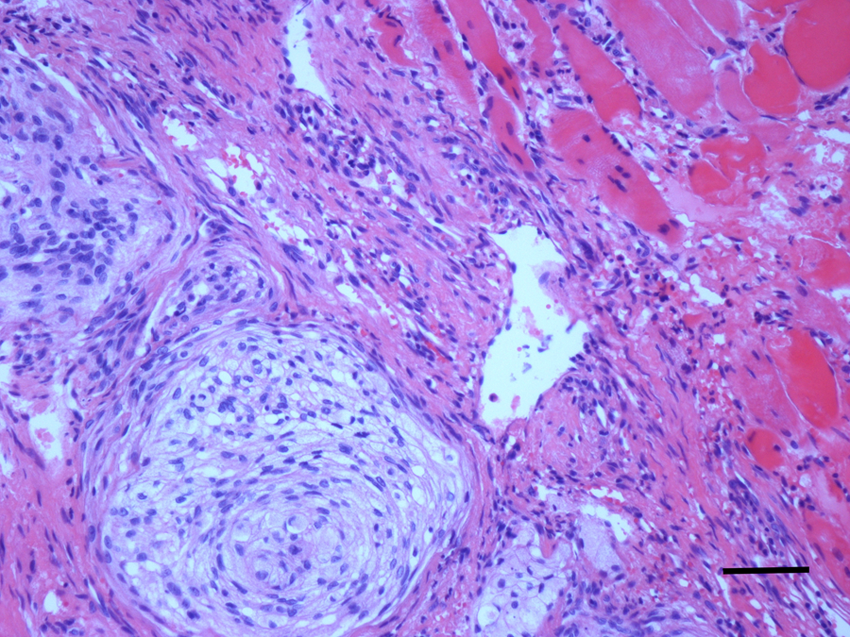
Typical architecture of a transitional grade 1 meningioma (World Health Organization 2007). Curl formation of densely-packed cells with a marked, but not clearly defined, cytoplasm. Adjacent to the mass there is normal striated muscle with minimal invasion of the neoplastic cells (haematoxylin and eosin)

Clear invasion of the tumour cells in the surrounding musculature. Deformation and reduction in fibre size of the remaining muscle fibres (haematoxylin and eosin)
Analgesia was maintained with fentanyl constant rate infusion (Fentanyl-Curamed; Actavis, 5 μg/kg/h) and after 12 h with buprenorphine (Temgesic; Reckitt Benckiser, 0.01 mg/kg q8h IV). One day postoperatively the cat showed a mild head tilt to the right and partial Horner syndrome on the right side with mild miosis and prolapse of the third eyelid. The menace response was still reduced, but the cat was under opioid therapy. The cat was calm and slightly apathetic, possibly as a result of the administration of phenobarbital and opioids, but responsive to external stimuli and reacting to the cotton ball test. It was discharged 4 days postoperatively. The cat was still treated with prednisolone (Prednisolone: Streuli, 0.5 mg/kg q12h for 7 days, and then tapered over 4 weeks), ranitidine (Ranitidin; Christoffel Apotheke, 1 mg/kg q12h for 7 days) and phenobarbital (Aphenylbarbit; Streuli, 2 mg/kg q12h). Because of phenobarbital-associated side effects, including depression, lethargy and apathy, medication was tapered down after 2 weeks of treatment. The cat improved immediately afterwards and became more active without any seizure episode.
Seventy days after surgery the cat’s postoperative deficits had resolved and the cat was clinically normal. At 12 months follow-up there were no neurological deficits or seizures.
Diagnostic imaging in our cat showed an extra-axial space-occupying lesion with many typical imaging features of a meningioma. Like most of the feline intracranial meningiomas (92.4%), the meningioma had a rostrotentorial localisation. The temporal region is reported as a predilection site for meningiomas in cats. 1 Also, the meningeal contrast enhancement visible as a dural tail described in feline intracranial meningiomas in 64% of histologically-confirmed cases was suspicious for a meningioma. 3 Small linear mineralisations were detected in the pre-operative CT study (Figure 2). As described in the study from Troxel, feline intracranial neoplasms with mineralisation were either meningiomas or olfactory neuroblastomas. 3
Hyperostosis has been frequently described in association with feline meningiomas and was focally present in this case. The pathophysiology of the hyperostosis in meningiomas is not fully understood, but there are several theories. The stimulation of osteoblasts by direct invasion of the bone by neoplastic cells seems to be one of the most reasonable theories. In contrast there were several cases with hyperostosis without invasion of tumour cells. Another possibility is the osteoblast stimulation by the bony isoenzyme of the alkaline phosphatase or other stimulating factors. 11 Vascular disturbances and stasis in the tumour stroma can give rise to hyperostosis and the tumour infiltration occurs later. 12 Direct bone production by the tumour, bony irritation without invasion of tumour cells and bone proliferation to prior trauma are other theories.
Despite the typical imaging features for meningioma, the extracranial tumour growth and the calvarial defect were misleading; however, the histological examination of the excised tumour material confirmed that the tumour was, indeed, a meningioma. Extracranial expansion of a meningioma is an extremely rare finding in animals and humans. There are three different ways for a tumour to extend out of the cranium: a neural foramen; pre-existing structures, such as skull sutures; and direct erosion of the skull. 13 As the former two seem unlikely in our case owing to the localisation of the tumour and the huge bony defect in the calvarium, erosion of the calvarial bone by the tumour seems the most likely mechanism.
The pathophysiology of meningioma-associated osteolysis is unknown. One of the mechanisms that could lead to bony destruction is simply invasion of tumour cells. As a minimally-invasive surgical technique was used, it was not possible to histologically assess the calvarial bone in our case in this respect, but, as mentioned previously and confirmed histologically in our case, feline meningiomas are typically benign and therefore rarely invasive.
Another explanation of bone infiltration and destruction is the production and increased activity of proteolytic enzymes by the tumour. Matrix metalloproteinases (MMPs) are zinc-dependent endopeptidases with degradating effects on extracellular matrix and the ability to breakdown basal membranes, as well as connective tissue. An increased, and perhaps synergistic, activity of MMP-2 and MMP-9 and invasion of meningiomas has been reported in human osteolytic meningioma.14,15 A more recent study in humans showed increased activity of MMP-2 in almost all extracranial masses than in the intracranial part of the tumour. 16 Whereas the correlation between MMP expression and malignant character in human intracranial meningiomas remains controversial, MMP activity in canine and feline meningiomas does not appear to correlate with malignant morphological patterns and their proliferative potential. Interestingly, feline meningiomas expressed significantly more MMP-2, although they are known to be less aggressive than canine meningiomas. 17 However, as osteolysis associated with feline intracranial meningioma is very rare with respect to the high prevalence of this tumour in this species, involvement of degradative enzymes and other factors causing osteolysis remains speculative. Other mechanisms like focal necrosis of the bone by pressure atrophy, ischaemia and secondary inflammation due to the tumour expansion could be considered.
In humans osteolysis does not appear to be a reliable prognostic factor and does not necessarily correlate with malignancy of meningioma. A few reports described osteolytic meningiomas to be malignant with a poorer prognosis because of higher proliferation activity and rapid regrowth. 4 In contrast, other publications show that osteolysis of the adjacent bone is not related to a more malignant pathology and a poorer outcome.5,16,18 The histology of our case showed a fibroblastic meningioma without any signs of malignancy. The two previously-described feline intracranial meningiomas with significant calvarial bone erosion were also classified as benign neoplasms — one being a fibroblastic 6 and the other a transitional meningioma 7 , the most frequently observed type. 17
Conclusions
The benign character of all three reported feline intracranial meningiomas causing osteolysis seems to indicate that, in cats, there is no correlation between malignancy and osteolysis. However, the invasion of the surrounding musculature, as seen in our case, would seem to increase the risk of residual tumour cells and recurrence. Malignant transformation after recurrence of initially benign meningiomas is also possible. Wide surgical excision of the bone and adjacent tissue seems to be the therapy of choice. Additional radiotherapy may complement the surgery, but was not performed in our case. Phenobarbital was used as a prophylactic antiepileptic drug for surgery in our patient. According to current knowledge, a non-sedative antiepileptic drug like levetiracetam, zonisamide or felbamate should have been used. Owing to the fact that at this time there was no injectable form of these drugs available, phenobarbital was used. In conclusion, differentials of a feline intracranial tumour exhibiting osteolysis and extracranial expansion should include meningioma. This behaviour is not necessarily associated with a more malignant character of the tumour.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
The case was presented in the poster session at the ECVN congress 2012 (13-15 September) in Ghent, Belgium
