Abstract
Practical relevance:
Feline mast cell tumors (MCTs) are frequently encountered in general practice. MCTs are the most common splenic tumor, second most common skin tumor and third most common intestinal tumor in cats. Treatment and prognosis can vary dramatically with location and histologic classification.
Clinical challenges:
While a cytologic or histologic diagnosis is often easy to obtain, the various histologic classifications, lack of a relevant grading scheme, and disparity in behavior depending on anatomic location make prognostication for cases of feline MCTs confusing. This is quite different from canine MCTs, where there is an established grading system which correlates clinically with prognosis and an accepted standard of care.
Audience:
Due to its prevalence, general practitioners encounter MCTs regularly. In many instances, referral for diagnosis and treatment is not necessary.
Evidence base:
Historically, there has been limited clinical evidence upon which to determine optimal treatment of MCTs in cats. Most recommendations are based on limited case reports or retrospective studies. With the recent introduction of receptor tyrosine kinase inhibitors to the veterinary market, there has been new research on the use of these drugs in cats, and new treatment options are on the horizon.
Histologic classification and associated behavior
Mast cell tumors (MCTs) account for 2–15% of all tumors in cats, and are classified anatomically into cutaneous and visceral forms.1–3 The cutaneous form of MCTs predominates in cats, with MCTs being the second most common skin tumor in this species.4,5 Cutaneous MCTs are most often located on the head, neck and trunk (Figures 1–3) and are divided histologically into two forms: mastocytic and atypical (previously histiocytic).2,4–6 The mastocytic form of the disease is the more common of the two, and is further subclassified into well-differentiated (previously compact) or poorly differentiated (previously pleomorphic or diffuse) forms.7–10
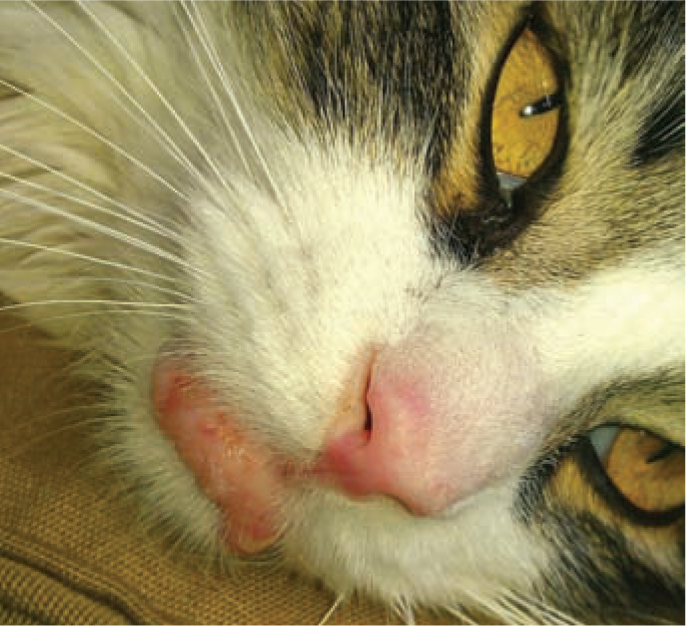
A 1.5-year-old domestic longhair cat with a solitary cutaneous MCT evident on the lower lip. Lymph node metastasis was detected and this cat developed diffuse skin lesions and circulating mast cells. Courtesy of Dr Sarah Kraiza

MCT affecting the lateral canthus. Periocular MCT may be associated with a lower recurrence rate than MCTs at other sites, although tumor size and location may limit surgical options. Courtesy of Dr Elizabeth Giuliano, University of Missouri

Diffuse distribution of poorly differentiated MCT affecting the head, neck and trunk. Courtesy of Dr Andrew Novosad
The cutaneous form of mast cell neoplasia predominates in cats, though visceral MCTs may be associated with secondary cutaneous lesions.
Mastocytic well-differentiated (compact) tumors are circumscribed, non-encapsulated masses that consist of solid sheets of uniform round cells with little mitotic activity.9,11 These are the most common histological type, accounting for 60% of feline cutaneous MCTs in one review. 12 Mastocytic poorly differentiated (pleomorphic) MCTs are less common, accounting for <28% of all feline cutaneous MCTs. They are characterized by pleomorphism, mononuclear or multinucleated giant cells and infiltrate the dermis and subcutis. This variant commonly contains eosinophils.9,11,12 The majority have a very low mitotic rate (<1 mitosis per 10 high power fields). The mitotic rate appears to be prognostic in this subset of feline cutaneous MCTs, as those cats with poorly differentiated tumors with a high mitotic rate have a more behaviorally malignant disease. 10
Canine cutaneous MCTs are graded histopathologically using the system proposed by Patnaik et al in 1984 in order to predict patient survival time and prognosis. 13 This grading system has not proven to be predictive of outcome in cats.13–15 In fact, several grading schemes have been proposed to categorize feline MCTs, but none has gained widespread acceptance or been shown to be reliably prognostic.2,5,8
The atypical form accounts for 10–20% of all feline MCTs and is most common in young (less than 4 years of age) cats.5,9,11 These tumors are not true proliferations of histiocytic cells, but atypical populations of mast cells that are not granular in appearance. 5 This variant has been described in two litters of Siamese kittens less than 6 months of age. 16 The lesions are usually small, non-encapsulated deep dermal or subcutaneous masses that may occur as a group of papulonodular lesions on the head.5,9,11 These lesions will spontaneously regress over time, with reports of regression 4–24 months after diagnosis.5,16 Topical or parenteral administration of corticosteroids does not appear to alter the disease course or speed regression. 16
Visceral MCTs most often affect the spleen (Figure 4), accounting for 15–26% of splenic disease in cats.17,18 The alimentary tract is another common location for visceral tumors, with MCTs being the third most common form of feline intestinal neoplasia. 19 Visceral MCTs may be associated with secondary cutaneous lesions.4,20,21 Other reported non-cutaneous MCT sites include the liver, lymph nodes and nasal cavity. 21

Enlarged, mottled spleen of a cat with splenic MCTs. Courtesy of Cuyahoga Falls Veterinary Clinic (fallsvetclinic.com)
Visceral MCTs most often affect the spleen; the alimentary tract is another common location.

Diagnosis
Diagnosis of MCT can usually be accomplished with cytological evaluation of a fine-needle aspirate of the mass. Cells typically contain metachromatic granules that are smaller than those seen in other species (Figure 5). For visceral tumors, ultrasonographic appearance of the affected organ can be useful for guiding fine-needle aspiration for cytology. The feline spleen may appear mottled, nodular or irregular, and lymph nodes are often hypoechoic, misshapen and enlarged, whereas affected intestines often have a loss of wall layering. 23 Caution is required when aspirating suspected visceral MCTs, as mast cell degranulation may occur, with fatal consequences. 24 Pretreatment with antihistamines, specifically diphenhydramine, is advised.
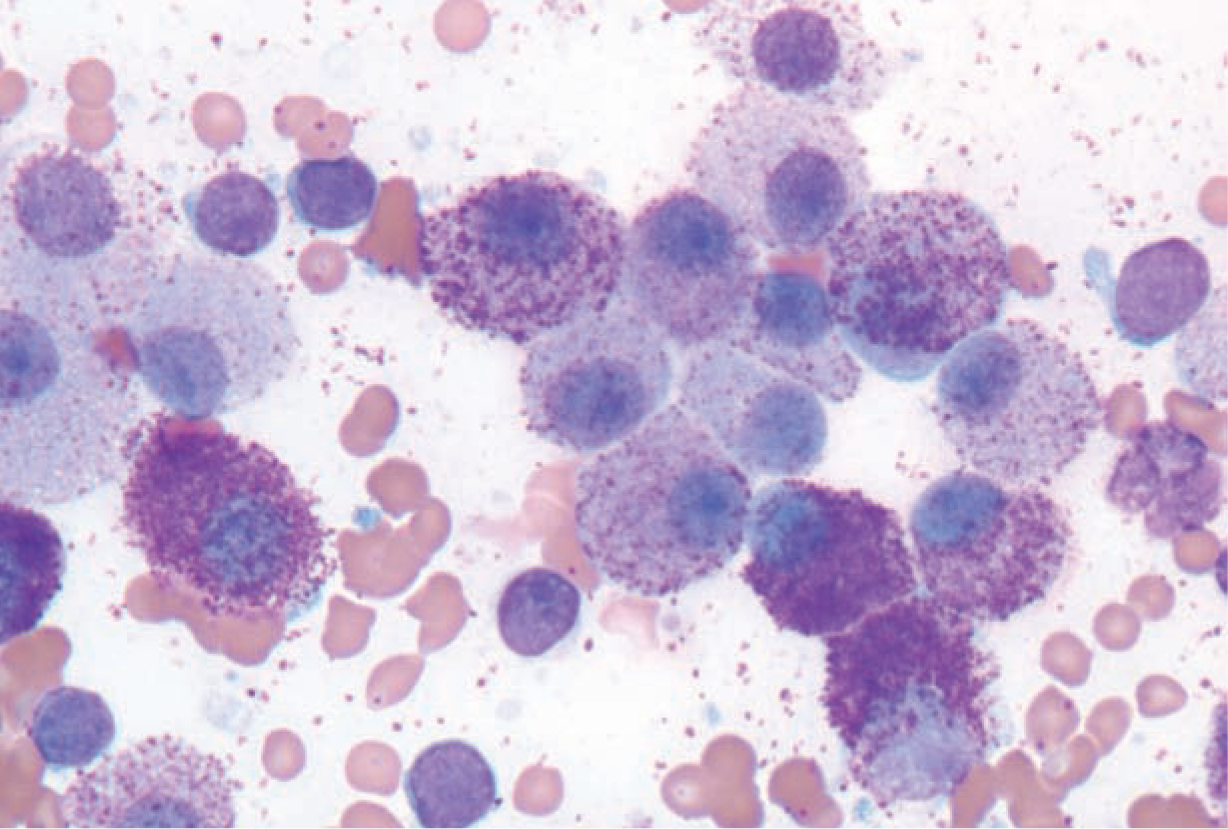
Cytological preparation typical of feline MCT, with cytoplasmic granules easily identified. Courtesy of Dr Marlyn Whitney, University of Missouri
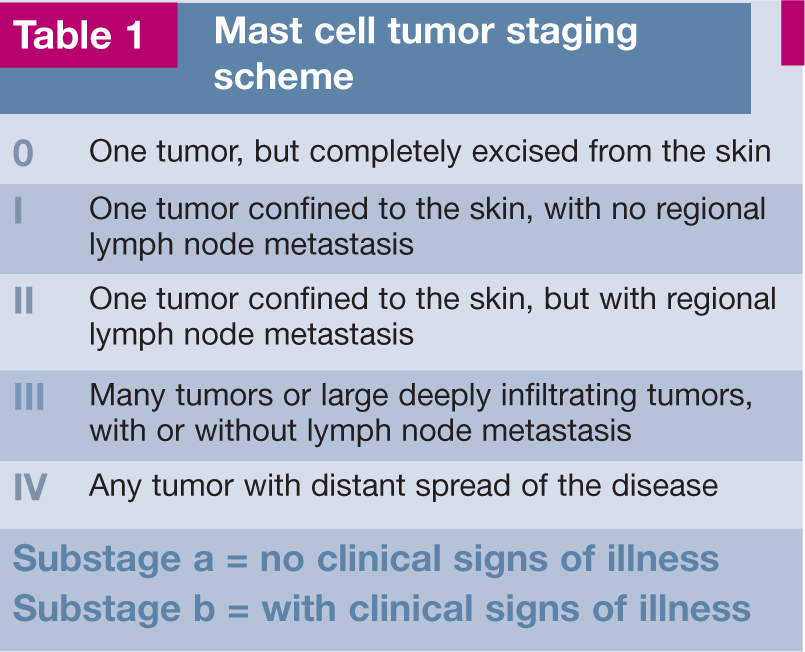
Complete staging (Table 1) should be performed in all cats with visceral MCTs, as well as those with multiple cutaneous masses, palpable abdominal abnormalities, clinical signs of systemic disease, or when the clinical behavior or histology of the tumor is atypical. Complete staging should include a complete blood count (CBC), serum chemistry panel, coagulation profile and bone marrow aspirate. Thoracic radiographs and abdominal imaging (radiography and ultrasound) are helpful for documenting peritoneal or pleural effusions, which are present in up to one-third of cats with visceral MCTs. 25 Abdominal ultrasound can also be used to identify organomegaly and guide fine-needle aspiration of organs with abnormal echogenicity. Ultrasound can furthermore identify concurrent disease that may alter therapy decisions, especially if the definitive therapy for the MCT is likely to be expensive or invasive.
Mast cell tumor staging scheme
Circulating mast cells are often a feature of MCTs in cats and can be documented with a buffy coat smear evaluation. Circulating mast cells may also be apparent on manual slide review of the CBC. A MCT should be considered in any feline patient with circulating mast cells, as clinically normal cats and cats without MCT-related disease are unlikely to have a positive buffy coat.26,27 One might argue that for cats with a single cutaneous MCT and no evidence of draining lymph node involvement, complete staging prior to surgical removal is unnecessary. However, positive buffy coat evaluations have been reported in cats with single cutaneous nodules and may signify a more extensive tumor volume than initial examination revealed. In these cases, further staging as described above is warranted. Based on the apparent disease specificity of a positive buffy coat evaluation for feline MCTs, and its potential to alter diagnostic and therapeutic plans, the authors and others recommend this procedure as part of the routine pretreatment work-up for all new cases of feline MCT. 27

Treatment
Histamine blockade
Medical management with histamine blockade (H1 and H2 blockers), without or without a proton pump inhibitor, should be considered in all cases where there is known gross or microscopic disease. Famotidine and diphenhydramine can be started at the time of cytologic diagnosis and can be discontinued once surgical excision has confirmed complete removal of the tumor. Animals with visceral disease should stay on these medications for their lifetime.
Surgery
Surgery, when feasible, is the treatment of choice for MCTs. Whereas completeness of surgical excision is predictive of case outcome in dogs with cutaneous MCTs, the same does not appear to be true for the feline form of the disease. Numerous reports have failed to find a correlation between completeness of excision and recurrence rate.14,31–33 Tumor recurrence has been reported in up to one-third of all cats with cutaneous MCTs, regardless of completeness of surgical excision.14,32 The reported recurrence rate for periocular MCT is lower (<5%), but also shows no correlation with completeness of surgical excision.31,33
Splenectomy is recommended for treatment of splenic MCT in cats and can be accomplished via laparotomy or laparoscopy.34–37 Although surgical excision has been suggested by some as the treatment of choice for intestinal MCT, 7 the overall post-surgical prognosis for this disease is poor. This is especially true if the intestinal MCT is of the sclerosing variant, where survival beyond 2 months is unlikely. 38
Plesiotherapy
In cats with single or multiple cutaneous tumors and no evidence of visceral metastasis, strontium 90 irradiation has been reported as an alternative to surgery. In a single case series, only 1/35 (3%) cats developed recurrence. 39 This is lower than the reported recurrence rates after surgery. The reason for this disparity is not apparent; however, due to the retrospective nature of the report, some tumor selection bias may have occurred.
Chemotherapy
The need for chemotherapeutic intervention is questionable in many cases of feline MCT. In published reports of case outcome, cats with stage I, II and IIIa disease (Table 1) rarely die due to their MCT and can enjoy long-term survival with surgery alone.14,32 Even cats with splenic MCT and evidence of systemic disease survive an average of 12–19 months with splenectomy alone.35–37
The oral alkylating agent, lomustine, has shown promise for the treatment of feline MCTs in a retrospective study of 38 cases. Lesions were cutaneous in 26 cats and the remaining 12 cats had MCTs of the mesenteric lymph nodes (n = 7), liver (n = 2), gastrointestinal tract (n = 2) or multiple visceral sites (n = 1). The target dosage of lomustine was 50–60 mg/m2 PO, with dosing intervals averaging every 4 weeks after the first two doses. Complete or partial responses were achieved in 19 (50%) of the cats, including 10/26 cats with primary cutaneous MCTs and 9/12 cats with non-cutaneous MCTs. 21 The dose-limiting toxicity of lomustine in cats is neutropenia. Pulmonary toxicity has also been reported for two cats receiving multiple doses of lomustine (200–400 mg/m2 cumulative dose). 21 Accordingly, cats undergoing therapy with lomustine should be monitored closely for both bone marrow and pulmonary toxicity.
Other chemotherapy agents that have been used in the treatment of feline MCTs include vinblastine, cyclophosphamide, chlorambucil and mechlorethamine.21,32,33 Although these agents have not been evaluated in a prospective study, tumor responses have been reported with vinblastine and mechlorethamine. 21
Receptor tyrosine kinase inhibition
With the introduction in 2007 of toceranib phosphate (Palladia; Pfizer), the first receptor tyrosine kinase inhibitor (RTKI) approved by the US Food and Drug Administration (FDA) for veterinary use in dogs, a new avenue for systemic therapy of MCTs may exist.
Tyrosine kinases are cell membrane-bound growth factor receptors that, when mutated, can result in uncontrolled cellular proliferation. A mutation of the c-kit (CD117) tyrosine kinase receptor, similar to that described in some dogs with MCTs, has been reported in 52–92% of feline cutaneous MCTs.12,40,41 There is also a case series in which several tumor-bearing cats responded favorably to the human RTKI product, imatinib mesylate (Gleevec; Novartis Pharma AG, Badel Switzerland). 41 Masitinib (Masivet; AB Science, Paris, France and Kinavet; AB Science USA, Short Hills, NJ) is conditionally approved by the FDA for use in dogs and has also been evaluated for safety in a group of healthy research cats. Side effects requiring dosage modification included proteinuria in 10% and neutropenia in 15% of cats treated. A large percentage of cats (65%) also developed grade 1 vomiting and/or diarrhea; however, these signs resolved without dosage modification. 42
No c-kit mutations were identified in a study of feline splenic MCT specimens, indicating that RTKI therapy for disease in this anatomic location may be of little benefit. 43
These findings suggest that RTKIs may have a role in the medical therapy of select feline MCT cases. However, to date, no large prospective trial has been performed to evaluate the true potential of these drugs.
Receptor tyrosine kinase inhibitors may have a role in the medical therapy of select cases of feline mast cell disease.
Prognosis
The prognosis for most cats with solitary cutaneous MCTs is good. Surgical excision of the mastocytic form is usually curative with low recurrence and metastatic rates, even in the face of minimal surgical margins on histopathology.2,14,32 The tumors that are most likely to result in metastasis are mastocytic poorly differentiated tumors with a high mitotic rate. 10 The treatment approach for these less common tumors should consist of wider surgical margins and a thorough assessment for metastasis. 7 Those tumors of the atypical subtype are more difficult to diagnose due to their unique cytologic appearance; however, these tumors are considered benign and will spontaneously regress.4,5,16 Caution should be exercised in cats with multiple cutaneous tumors, as this could represent metastasis from a visceral tumor. 44
The prognosis for cats with visceral MCTs is dependent on location. Those with splenic MCTs can enjoy long-term survival with splenectomy alone.35–37 Intestinal MCTs carry a poor prognosis, as metastasis is common.4,7
Footnotes
Key Points
Case notes
Physical examination
Joey was bright, alert and responsive. His vital signs were: temperature 101.0°F (38.3°C), pulse 160 bpm and respiratory rate 36 bpm. He had a body condition score of 8/9. Small flesh-colored, alopecic cutaneous nodules were diffusely distributed on his body. No abnormalities were noted on auscultation of heart and lungs. Abdominal palpation revealed no abnormalities.
Assessment and initial approach
Fine-needle aspiration was performed on several of the cutaneous masses. Cytology was consistent with mast cell neoplasia. Due to the diffuse nature of Joey’s cutaneous mast cell neoplasia, complete staging was performed. Results of a CBC, chemistry panel and urinalysis were within normal limits. There was no evidence of pleural or peritoneal effusion or other significant abnormalities on survey thoracic and abdominal radiographs. No evidence of visceral mast cell neoplasia was noted on abdominal ultrasound. A buffy coat smear was negative for mastocytosis. The patient was discharged with famotidine, omeprazole and prednisone (2 mg/kg q24h) treatment and scheduled for a recheck in 1 month.
Repeat staging and surgery
On recheck physical examination, many of the cutaneous masses had become more erythemic and ulcerated. Repeat staging was recommended. Buffy coat evaluation at this time was positive for the presence of mast cells, suggesting systemic dissemination of his cutaneous disease. Abdominal ultrasound revealed splenomegaly. Joey was premedicated with diphenhydramine and ultrasound-guided fine-needle aspiration of the spleen was performed. Results were consistent with splenic mast cell neoplasia. A splenectomy was recommended, given previous reports of long-term survival in cats undergoing splenectomy for MCTs, even with metastatic disease. At the time of surgery, the spleen was grossly abnormal and a liver nodule was noted and removed. Histopathology of both lesions was consistent with mast cell neoplasia.
Follow-up
Joey’s cutaneous lesions progressed following splenectomy. He was prescribed a receptor tyrosine kinase inhibitor but unfortunately experienced adverse effects from the drug and the owners opted to discontinue treatment. At the time of writing, 14 months after presentation to the authors, his disease had continued to progress cutaneously, although his quality of life was still good.
While the progression of disease in Joey is not typical of the feline cutaneous mast cell neoplasia described in many older reports, development of visceral disease in cats with cutaneous MCT has been reported more often in the current literature. As such, complete staging, including buffy coat evaluation, is recommended for cats with multiple cutaneous MCTs or for those presenting with signs of systemic disease including anemia and/or melena.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
Dr Henry has served on the PACE Oncology Advisory Board for Pfizer Animal Health.
