Abstract
The first part of the study aimed to describe prospectively the ultrasonographic features of the adrenal glands in 94 healthy cats and 51 chronically sick cats. It confirmed the feasibility of ultrasonography of adrenal glands in healthy and chronically sick cats, which were not statistically different. The typical hypoechoic appearance of the gland surrounded by hyperechoic fat made it recognisable. A sagittal plane of the gland, not in line with the aorta, may be necessary to obtain the largest adrenal measurements. The reference intervals of adrenal measurements were inferred from the values obtained in the healthy and chronically sick cats (mean ± 0.96 SD): adrenal length was 8.9–12.5 mm; cranial height was 3.0–4.8 mm; caudal height was 3.0–4.5 mm. The second part of the study consisted of a retrospective analysis of the ultrasonographic examination of the adrenal glands in cats with adrenal diseases (six had hyperaldosteronism and four had pituitary-dependent hyperadrenocorticism) and a descriptive comparison with the reference features obtained in the control groups from the prospective study. Cats with hyperaldosteronism presented with unilateral severely enlarged adrenal glands. However, a normal contralateral gland did not preclude a contralateral infiltration in benign or malignant adrenal neoplasms. The ultrasonographic appearance of the adrenal glands could not differentiate benign and malignant lesions. The ultrasonographic appearance of pituitary-dependent hyperadrenocorticism was mainly a symmetrical adrenal enlargement; however, a substantial number of cases were within the reference intervals of adrenal size.
Introduction
Adrenal glands are currently assessed by ultrasonography in feline medicine, based on two studies of healthy cats.1,2 Non-adrenal diseases may affect the ultrasonographic features of the adrenal glands. One study examines the measurements of feline adrenal glands in sick cats without endocrine diseases; 3 the authors did not find any statistically significant difference compared with the two studies of healthy cats. The sick cats only presented a wider range of values compared with healthy cats. During the 14 years between the first and the last studies about ultrasonography of the feline adrenal glands, there was a noticeable improvement in ultrasonographic equipment, and in scanning procedure and accuracy, which makes the comparison poorly reliable. The additional descriptions of changes in adrenal glands size in specific feline diseases are variable: significantly smaller adrenal glands were described in cats with idiopathic cystitis, and adrenal glands were not enlarged in cats with diabetes mellitus.4,5 The effect of hyperthyroidism is controversial, with studies showing mildly larger adrenal glands, but with significant overlap.6,7
Variation in ultrasonographic features and size of adrenal glands in non-adrenal diseases could affect the specificity of ultrasonographic examination of adrenal gland for the detection of adrenal gland hyperplasia or neoplasia associated with hyperaldosteronism or hyperadrenocorticism, and for the diagnosis of hypoadrenocorticism, sex hormone-producing tumours, pheochromocytoma or adrenal metastasis. 8 Moreover, there is no imaging study describing the ultrasonographic appearance of the adrenal glands in cats with adrenal gland diseases, for instance in hyperaldosteronism or hyperadrenocorticism. There are only multiple case reports and series describing the imaging findings with different precision levels (absent or subjective size evaluation to three-dimensional measurements; vague to very descriptive ultrasonographic features, eg shape, contour, echogenicity, homogeneity).
The first part of this report is a prospective study to assess adrenal gland features for healthy cats, but also for cats with chronic non-adrenal diseases, with a standard ultrasonographic protocol using high-resolution ultrasound. We aimed to describe more accurately the relative difference in adrenal size, shape and echogenicity that may appear during a non-adrenal disease.
The second part of this report is a retrospective study of ultrasonographic findings in cats diagnosed with hyperaldosteronism and hyperadrenocorticism in two referral centres. The aim is to compare these findings with the findings from the control population recruited in the prospective study.
Materials and methods
Prospective study: chronically sick and healthy cats
The chronically sick cats included in the study were evaluated at the Small Animal Clinic of the Veterinary Faculty of Ghent University and the Centre Hospitalier Vétérinaire Universitaire d’Alfort (CHUVA) of the veterinary school of Alfort from January to December 2010. Fifty-one cats with clinical signs for more than 10 days that had an abdominal ultrasonography and a precise, definite diagnosis of non-endocrine disease were included. Table 1 summarises the different types of diseases encountered in this group. Cats with suspected or confirmed feline idiopathic cystitis, hyperadrenocorticism, hypoadrenocorticism, diabetes mellitus, hyperthyroidism, primary hyperaldosteronism, hypersomatotropism or abnormal sexual hormone production were excluded. Mean duration of clinical signs was 112 days (SD = 116 days) with a range of 10–360 days (median = 90 days).
Overview of the different diseases diagnosed in the group of 51 non-endocrine sick cats

The control group was composed of 94 healthy cats screened at the small animal clinic of the Veterinary Faculty of Ghent for polycystic kidney disease (PKD) or that were participating in a study of geriatric screening. All cats were submitted to a complete physical examination, a urinalysis (specific gravity, dipstick, urinary protein/creatinine ratio, urine sediment and urine culture if indicated), a complete blood count and serum biochemistry profile. Cats older than 6 years also underwent systemic blood pressure measurement, an ionogram (sodium, chlorine, potassium and phosphorous), a feline immunodeficiency virus/feline leukaemia virus test and measurement of total thyroxine concentration. Cats were considered healthy and included if there were no significant abnormalities found on these examinations.
For both groups the ultrasonographic examination was performed with the cat in dorsal recumbency with only minimal manual restraint. The abdominal hairs were clipped if necessary and acoustic coupling gel was applied. Three ultrasound machines were used (ATL 3500§, Logic7 and MyLab30, with a multifrequency linear transducer (8–14 MHz and 10–15 MHz, respectively). Six ultrasonographers (supervised European College of Veterinary Diagnostic Imaging senior residents) were involved in the study (five in Ghent and one in Alfort). The adrenal glands were imaged according to a previously described protocol. 2
Three two-dimensional shapes of the adrenal gland were distinguished: bipolar (oval with a depression over the minor axis), oval (without depression) or fusiform (lower short to long axis with more pointed extremities). The echogenicity compared to the surrounding tissues (hypo-, iso- or hyperechogenic), the visualisation of layers (medulla/cortex distinct or not) and the presence of hyperechoic foci (number, uni- or bilateral, with or without presence of acoustic shadowing) were also assessed by the observers during the ultrasonography. The maximum length (cranio-caudal, L) and the height of the cranial (crH) and caudal (cdH) poles (dorso-ventral) were measured on a sagittal scan of each adrenal gland at the end of the examination by the ultrasonographer. All images were saved and reviewed during a remote examination by the first author.
Statistical analysis was performed on adrenal gland measurements. In order to assess the effect of different covariates on the maximum length (cranio-caudal, L) and the height of the crH and cdH poles (dorso-ventral) of the adrenal gland, a mixed model was fitted with cat as random effect and status, breed, gender, castration and age, and the two-way interactions as fixed effects. Hypothesis testing was based on the F-test at a significance level of 5%.
Retrospective study: cats with adrenal disease
Cats were recruited retrospectively from the database of two referral centers (Veterinary Faculty of Ghent and the CHUVA of the veterinary school of Alfort) from 2000 to 2011. Inclusion criteria for hyperaldosteronism were:
compatible history and clinical signs (at least two signs from the following: weakness, cervical ventroflexion, lethargy, depression, dysphagia, systemic hypertension, polyuria, polydipsia, periuria, weight loss, diarrhoea, polyphagia, abdominal mass, dehydration); 9
high plasma aldosterone concentration (>195 pg/ml) with concurrent hypokalaemia (<3.0 mmol/l); 10
abdominal ultrasound — adrenal echogenicity, shape and size available on recorded images and ultrasonography report.
Inclusion criteria for hyperadrenocorticism were:
compatible history and clinical signs (at least two signs from the following: insulin resistant diabetes mellitus, polyuria, polydipsia, fragile torn and thin skin, weight loss, lethargy, alopecia, polyphagia, potbelly, hepatomegaly, bruising, plantigrade stance); 11
diagnosis with low-dose dexamethasone suppression (LDDS) test (with 0.1 mg/kg IV of dexamethasone, plasma cortisol concentration >30 nmol/l after 4 h and 8 h) or urine cortisol:creatinine ratio (>3.6 × 10-5); 11
localisation with LDDS test (pituitary origin: >50% lowering of plasma cortisol concentration after 4 h or 8 h; adrenal origin: <50% lowering of plasma cortisol concentration after 4 h or 8 h) or plasma adrenocorticotropic hormone concentration (pituitary origin >80 pg/ml; adrenal origin <20 pg/ml) or pituitary mass on computed tomography; 11
abdominal ultrasound — adrenal echogenicity, shape and size available on recorded images and ultrasonography report.
Six cats with hyperaldosteronism and four cats with hyperadrenocorticism were included. No case of sex-producing adrenal tumour could be found in the database over the previous 10 years. The number of cats was too small to perform a relevant statistical analysis. We described the ultrasonographic appearance and size of the adrenal glands in these groups based on the review by the first author of a sagittal scan of the gland, the only plane recorded consistently and the plane used in the protocol of the prospective study. We made a descriptive comparison with the ultrasonographic appearance of the adrenal glands in the healthy and chronically sick cats described in the prospective study.
Results
Prospective study: chronically sick and healthy cats
The chronically sick feline group was composed of nine intact males, 22 castrated males, five intact females and 15 neutered females. The breeds represented were domestic shorthair (n = 29), British Shorthair (n = 10), ragdolls (n = 6), Persians (n = 2), Siamese (n = 2) and other breeds (n = 2; Scottish Fold, Maine Coon). Mean age was 7.4 years (SD = 5.5 years), median age was 5.5 years (range 0.7–19.0 years). Mean weight was 4.2 kg (SD = 1.2 kg); median weight was 4.0 kg (range 2.4–7.1 kg).
In the control group, mean age was 5.2 years (SD = 3.8 years), median age was 3.7 years (range 0.8–14.0 years). Mean weight was 4.2 kg (SD = 1.0 kg) and median weight was 4.0 kg (range 2.5–7.9 kg). There were 12 intact males, 19 castrated males, 27 intact females and 36 neutered females. The breeds represented were as follows: 48 domestic shorthairs, 21 ragdolls, nine British Shorthair, five Sphynx, three Maine coon, three Persians, two Norwegians and three other breeds (n = 3; Siberian, exotic shorthair, Angora).
Both right and left adrenal glands could be visualised in all cats (n = 145), although in 18/94 healthy cats and 12/51 chronically sick cats, the outline of one or the two adrenal glands was difficult to delineate precisely (close contact with vasculature, small size, isoechoic to surrounding tissue). In both healthy and sick cats, the most common appearance was a well-defined, bipolar hypoechogenic gland surrounded by a thin hyperechoic halo (Figure 1a). The adrenal gland shape was less frequently recognised as fusiform or oval (Figure 1b, c). The descriptive ultrasonographic features of the adrenal glands of the healthy and chronically sick cats are reported in Table 2. There was no difference in the shape and echogenicity of the adrenal glands between healthy and sick cats.
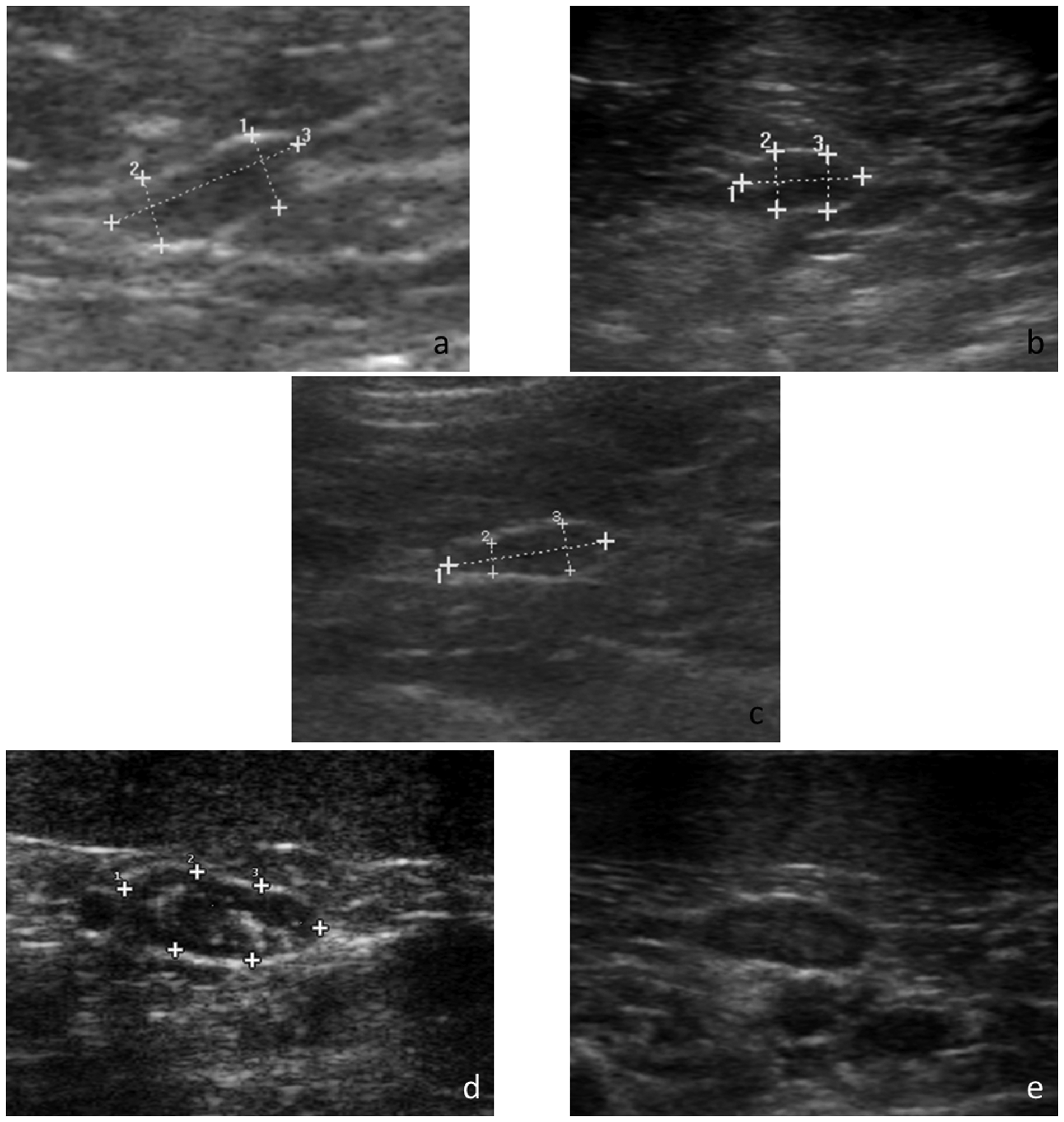
Ultrasonographic images of feline adrenal glands: (a) a bipolar left adrenal gland in a chronically sick cat (distances 1 = 3.0 mm, 2 = 2.7 mm, 3 = 7.7 mm); (b) an oval left adrenal gland in a healthy cat (distances 1 = 9.8 mm, 2 = 4.7 mm, 3 = 4.6 mm); (c) a fusiform right adrenal gland in a chronically sick cat (distances 1 = 8.1 mm, 2 = 1.5 mm, 3 = 2.4 mm); (d) hyperechoic foci in a hypoechoic left adrenal gland in a chronically sick cat (distances 1 = 11.0 mm, 2 = 4.5 mm, 3 = 4.2 mm); (e) concentric layers detected in a left adrenal gland in a healthy cat
Descriptive ultrasonographic features of the adrenal glands of the healthy and chronically sick cats

Eighteen cats presented hyperechoic foci in the otherwise hypoechoic adrenal gland, unilaterally in 11 cats and bilaterally in seven cats (25 glands were concerned). These hyperechoic foci were observed more often in sick cats (20%) than in healthy cats (9%). They were not creating a visible acoustic shadow distally (Figure 1d). They demonstrated variable shapes: multiple pinpoint, linear and central, single nodular and ill-defined heterogeneous. Cranial and caudal adrenal poles were equally involved. Hyperechoic foci were bilateral in 7/18 cats and unilateral in 11 cats (involving, similarly, the left and right adrenal glands).
Two distinct concentric layers could be distinguished in one healthy and two sick cats, with a hyperechoic center and a hypoechoic periphery (Figure 1e).
The measurements of the adrenal glands in healthy and sick cats are presented in Table 3. Adrenal measurements from the right or the left gland were not significantly different. There was no statistical difference in adrenal gland measurements between healthy and chronically sick cats. There was no significant effect of gender or breed on adrenal gland measurements. In both groups of healthy and sick cats, there was a moderate effect of castration on the caudal height measurement of the adrenal gland (P = 0.02). The gland was thicker in castrated cats (mean 3.7 ± 0.1 mm) than in intact cats (mean 3.4 ± 0.1 mm), in males and females combined. This mild difference is not clinically relevant. There was a small effect of age on the length in healthy cats (P = 0.03), with a slight decrease in length with ageing (estimate of the downslope = −0.1158), which was also not clinically relevant. There was no effect of age on the short axis measurements of the adrenal glands.
Ultrasonographic measurements of the adrenal glands in healthy and chronically sick cats

Data from the present study
Cartee et al 1
Zimmer et al 2
Zatelli et al 3
Retrospective study: cats with adrenal disease
According to the inclusion criteria, six cats were diagnosed with hyperaldosteronism. They were all domestic shorthair cats. There were three castrated males, one intact female and two neutered females. The median age was 12.5 years (range 11–17 years). The median weight was 4.4 kg (range 2.6–6.8 kg). The clinicopathological findings and ultrasonographic features of the adrenal glands of cats with hyperaldosteronism are summarised in Table 4. The most common ultrasonographic appearance was a unilateral mass (Figure 2), four concerning the left and two the right adrenal gland. Three patterns of echogenicity were recorded: hyperechoic heterogeneous in two masses, hypoechoic homogeneous in three masses and target-like (hyperechoic center and hypoechoic rim) in one mass. Vascular invasion was not detected in any of the cats. At the time of the ultrasonography, the contralateral gland was considered normal in size, shape and echogenicity. The histopathological analysis of the adrenal mass performed in 5/6 cats resulted in: one adrenocortical carcinoma, two adrenocortical adenocarcinomas, one adrenocortical adenoma and one adrenocortical adenomatous nodule. Malignant and benign tumours could not be differentiated according to their echogenicity pattern. The two largest masses were carcinoma and adenocarcinoma; however, one adenocarcinoma had adrenal mass measurements similar to the two benign tumours.
Clinicopathological findings and ultrasonographic features of the adrenal glands of cats with hyperaldosteronism

Reference intervals (laboratory data and Javadi et al 10 ): potassium = 3.5–5.8 mmol/l; PAC = 40–195 pg/ml; PRA (cat 1) = 60–630 fmol/l/s; PRA (cat 4) = no reference interval reported in cats in this laboratory; NE = not examined

Ultrasonographic images of the right (a) and left (b) adrenal glands in a cat with hyperaldosteronism caused by bilateral carcinoma. The left adrenal gland is roundened, hypoechoic, mildly heterogeneous with well-defined borders. The left adrenal gland length is 24.3 mm and its height 15.1 mm. The right adrenal gland has a normal bipolar shape, a well-defined contour and a hypoechoic parenchyma. The right adrenal gland length is 12.4 mm and its maximal height 6.1 mm
Histopathologically, the three malignant tumours were locally invasive, with cellular infiltration beyond the adrenal capsule, in the surrounding fat, even forming nodules in one cat. Phrenicoabdominal venous embolisation was noticed during surgery in one of the three cats. None of these local invasions was diagnosed ultrasonographicly. In 3/6 cats, a histopathological analysis of the contralateral adrenal gland had been performed (two necropsy and one bilateral adrenalectomy): the two carcinomas were bilateral with more malignant infiltration of the gland on the side of the ultrasonographic mass, but also a mild malignant infiltration of the contralateral adrenal gland. Retrospectively, these two contralateral adrenal glands were at the upper limit of adrenal measurements in healthy and chronically sick cats. The third cat had a unilateral adenoma and a normal contralateral gland, retrospectively, within the mid-range of adrenal measurements of healthy and sick cats.
Based on our inclusion criteria, four cats were diagnosed with hyperadrenocorticism — all were of pituitary origin. No case of hyperadrenocorticism secondary to adrenocortical secreting tumour could be found in the database over 10 years. The cats with pituitary-dependent hyperadrenocorticism were all domestic shorthair cats. There was one castrated male and three neutered females. The median age was 14 years (range 13–15 years) and the median weight was 3.2 kg (range 2.0–3.6 kg). The clinicopathological findings and ultrasonographic features of the adrenal glands of cats with pituitary hyperadrenocorticism are summarised in Table 5. The adrenal glands had mostly a bipolar shape, with the exception of one left adrenal gland, which was oval (Figure 3a). They were all heterogeneous with hyperechoic foci or hyperechoic medulla in an otherwise hypoechoic parenchyma compared to surrounding fat tissue. These foci had variable shapes and distribution: linear central, irregular central and cranial pole nodule. One cat showed an isoechoic nodule in the cranial pole of the right adrenal gland, mildly deforming the adrenal outline (Figure 3b). In 2/4 cats, the ultrasonographic measurements of both adrenal glands were within the range of the healthy and chronically sick cats. In 2/4 cats the short-axis measurement of both adrenal glands were in the upper range or slightly over the upper limit of the range of cranial or caudal height measurements in healthy and chronically sick cats. Only 1/4 cat had a histolopathological analysis of the adrenal glands and another had analysis of the pituitary gland.
Clinicopathological findings and ultrasonographic features of the adrenal glands of cats with pituitary hyperadrenocorticism

Reference intervals (laboratory data and Feldman and Nelson 11 ): UCCR <10; LDDS: T0 = 13.8–149.0 nmol/l, hyperadrenocorticism if T4 or T8 >40 nmol/l, pituitary origin if >50% lowering of plasma cortisol concentration after 4 h or 8 h; IGF1 = 48.4–544.0 ng/ml; GH: no reference values in animals in this laboratory; ACTH: pituitary origin if >80 pg/ml
ACTH = adrenocorticotropic hormone plasma concentration; GH = growth hormone; IGF1 = insulin-like growth factor plasma concentration; LDDS = low dose dexamethasone suppression test; SBP = systolic blood pressure; T0 = plasma cortisol concentration before dexamethasone injection; T4 = plasma cortisol concentration 4 h after dexamethasone injection; T8 = plasma cortisol concentration 8 h after dexamethasone injection; UCCR = urine cortisol:creatinine ratio

Ultrasonographic images of the left (a) and the right (b) adrenal glands in a cat with pituitary-dependent hyperadrenocorticism. Both adrenal glands are well-delineated, hypoechoic with hyperechoic foci and bipolar. They are mildly over the reference intervals in size: right adrenal gland length = 13.0 mm, height = 6.4 mm; left adrenal gland length = 12.2 mm and height = 7.4 mm
Discussion
This study confirms the ability to image and measure the feline adrenal glands with ultrasonography. Subjectively, it appears easier than in dogs. 2 In healthy and chronically sick cats, all adrenal glands could be imaged without sedating them. The feline adrenal gland was surrounded by hyperechoic fat, creating a very good contrast with the commonly hypoechoic gland.1,2,12 The presence of two distinct concentric zones in the gland was described as a hyperechoic inner zone and a hypoechoic outer zone, visible in vitro and sometimes in vivo. 2 The histopathological significance of these two zones and their correlation with cortex and medulla were not assessed. Although a high resolution ultrasound machine was used, two distinct zones could only be detected in one healthy and two sick cats in this study.
The three adrenal shapes described in cats — fusiform, oval or bipolar — were found in the combined groups of healthy and chronically sick cats in this study, although the bipolar shape was most frequently observed in about 2/3 of the glands. 2 The oval and fusiform shapes were equally observed in the last third of glands. The shape of the adrenal gland in a sagittal plane appeared influenced by the exact ultrasonographic scan plane. As in dogs, the feline adrenal glands are often slightly oblique compared with the sagittal plane of the body with a more lateral cranial pole and more medial caudal pole. 13 The adrenal gland appeared more bipolar if a real sagittal plane of the gland could be performed or more oval if the plane was more sagittal to the body.
The presence of hyperechoic areas in the adrenal gland creating an acoustic shadowing has already been described in healthy adult cats and interpreted as microscopic calcifications in up to 30% of normal adult cats.12,14 In the present study, these areas were not associated with acoustic shadowing. The different hypotheses for this appearance are too little mineralisation to create a visible acoustic shadow, fat deposition or small haemorrhage. The cats with these hyperechoic foci were of all ages, both genders and all breeds, and they had a moderate weight. Proportionally, more sick cats (20%) had hyperechoic foci than healthy cats (9%). More frequent hyperechoic foci were also described in hyperthyroid cats compared with healthy old cats. 6 The latter study emitted the hypothesis of a relationship between the hyperechoic foci and a stimulation of the hypothalamic–pituitary–adrenal axis by a chronic stress in the case of the chronically sick cats. Moreover, all four cats with hyperadrenocorticism presented hyperechoic foci, hyperechoic central line or mineralisation in both adrenal glands.
As the cats examined at the veterinary hospital are usually sick, the influence of chronic illness on the adrenal glands is an important factor. Health status and hospitalisation were, indeed, reported to influence the urine cortisol:creatinine ratio in cats, but not the dexamethasone suppression test and the adrenocorticotropin hormone stimulation test.5,15 –17 Compared with previous studies in healthy cats, the adrenal measurements were more recently described in a small group of sick cats without endocrine diseases.1 –3 They appeared subjectively similar in size. The limitations of the previous studies were the small number of cats, the absence of a control group in the study about sick cats and the absence of standardisation of the ultrasonographic protocol. Therefore, to confirm this impression, the present study examined a larger number of cats with a standard ultrasonography protocol and a similar generation of high resolution ultrasound materials. All mean adrenal measurements were higher in chronically sick cats compared with healthy cats. The difference was too small to result in a statistically significant difference. From a clinical point of view the ultrasonographic measurements of the adrenal glands can be considered similar in healthy and chronically sick cats. Therefore, the reference interval of adrenal measurements can be inferred from the values obtained in a combined group of healthy and chronically sick cats (mean ± 0.96 SD): the adrenal length was 8.9–12.5 mm; the cranial height was 3.0–4.8 mm; the caudal height was 3.0–4.5 mm. The mean, median and range of adrenal measurements obtained in this study were comparable with the previous studies.1 –3 In the present study a higher upper limit was, however, noticed in length and height measurements (Table 3). Differences in length may depend on the visibility of the gland, particularly the right adrenal gland. As described previously for the shape of the gland it appeared necessary to scan in a sagittal plane of the adrenal gland instead of in a sagittal plane of the body, to obtain the longest cranio-caudal length. An in vitro study with ultrasonographic examination in different imaging planes of isolated adrenal gland specimens should, however, be perfomed to test this hypothesis. The height measurement of this study and the thickness measurement described previously could also represent two different anatomical distances that could explain the difference in mean thickness. The authors strictly measured the dorso- ventral height of each gland to assess a more precise and repeatable measurement of the gland. However, considering the measurement accuracy of the ultrasonography machines, these values are most likely rather comparable.
According to previous studies, there was no effect of bodyweight, body surface area, body condition score, gender and side on the ultrasonographic measurements of adrenal glands in healthy or chronically sick cats.2,3 As described previously, we did not find any difference between male and female cats. 2 However, statistically, the adrenal glands of castrated cats were larger than those of intact cats in both groups of healthy and sick cats. A study described an increased aldosterone-to-renin activity ratio in healthy neutered cats compared with intact cats. 18 The authors speculated that oestrogens and luteinising hormone (LH) could increase the expression of the adrenal receptors for angiotensin II and then increase aldosterone production. This hypothesis is comparable to the pathogenesis of the hyperadrenocorticism in ferrets. 19 In ferrets, adrenal hyperplasia is suspected to be due to absent negative feedback from the gonadal steroids on the pituitary gland and then an increase in circulating gonadotropin levels. Gonadotropins stimulate the expression of LH receptors in the adrenal cortex leading to overproduction of sex-steroid hormones and adrenal hyperplasia. By reduced negative feedback from absent gonadal steroids due to castration, an enlargement of the adrenal glands may be explained in castrated cats. It has, however, a low clinical significance given the difference in adrenal size is mild compared to the resolution of ultrasound equipment. The effect of ageing on adrenal gland size had been studied in humans, dogs and rats.20 –23 These studies commonly described an adrenal hyperplasia and an increase in adrenal gland size or volume with ageing. This age effect was detectable by ultrasound in dogs.24,25 In one study, a weak, but significant, relationship between age and adrenal length was reported (P = 0.009). In the other study, a significant relationship was obtained between age and left adrenal length (P = 0.0388) and width (P = 0.0019). According to these data in dogs and clinical experience of rounder and larger adrenal glands in older cats, the weak statistical relationship between age and adrenal length was surprising as the adrenal length decreased with ageing. However, if they are not thicker, the decrease in length could explain the rounder shape of the feline adrenal gland with ageing. This apparent roundening of the gland should not have any clinical consequence concerning adrenal disease diagnosis if the short axis of the gland is measured accurately with ultrasound.
One limitation of the prospective study in healthy and chronically sick cats is the absence of histopathologic analysis of the adrenal glands. For ethical reasons, the characterisation of the health status and the exclusion of endocrine diseases in healthy and chronically sick cats were limited to clinicopathological data.
In the retrospective analysis of data covering 11 years, the ultrasonographers and the equipment were variable, although the most recent cases were examined by some of the ultrasonographers of the prospective study. This represents a limitation in the remote interpretation of the ultrasonographic images of adrenal glands as the scan plane could be inaccurate, thus changing the shape or the size of the gland as hypothesised previously. In reviewing the images of adrenal glands included in the retrospective study, image quality was checked to allow comparison with the prospective study data. However, inaccuracy in scan plane is difficult to judge on a fixed image and remains possible.
In the six cases of hyperaldosteronism of this study, the ultrasonographic findings were mainly a unilateral mass of various echogenicity and echotexture. The size of the mass was obviously larger than the size of adrenal glands in healthy or chronically sick cats. Vascular invasion by the mass was never detected with ultrasound. The five cases diagnosed histopathologically were compared with the 22 cases of hyperaldosteronism reported in the literature with ultrasound description and histopathological or cytological diagnosis.10,26 –33 As in our study, there is an equal proportion of benign and malignant adrenal neoplasms in hyperaldosteronism. The size (10–47 mm) or the other ultrasonographic descriptive features of the mass (echogenicity, homogeneity and shape) did not allow the differentiation between benign and malignant adrenal lesions. As severe compression or invasion of caudal vena cava could be difficult to differentiate, even the appearance of adjacent vessels was not more specific.
Two of the six cases of hyperaldosteronism in this study were diagnosed initially as unilateral adrenal mass with ultrasonography and further confirmed as bilateral carcinoma after necropsy. Another similar case report of a unilateral mass identified on ultrasound that was later diagnosed as bilateral adenoma on necropsy has been reported. 29 As unilateral adrenalectomy or unilateral fine-needle aspirate of the apparently abnormal gland are most often described in the literature, the contralateral gland is most often not sampled and then rarely confirmed as normal with histopathological analysis. The real prevalence of bilateral involvement in adrenocortical tumour is most likely underestimated. Additionally, two cases of confirmed bilateral hyperplasia did not show any ultrasonographic abnormalities. 10 Consequently, a normal contralateral gland to mass or bilateral normal adrenal glands on ultrasound does not exclude a benign or malignant adrenocortical infiltration. This finding can be relevant if surgical treatment is considered, as unilateral adrenalectomy may not be successful if the other gland is also infiltrated or hyperplastic.
With the inclusion criteria of this study, only four cats with pituitary-dependent hyperadrenocorticism (PDH) could be recruited. Moreover, no adrenal-dependent hyperadrenocorticism (ADH) could be found in the database at all. This most likely reflects the higher frequency of PDH (40/50) compared with ADH (10/50) reported in the literature.11,34 –42 One limitation is the low amount of histopathological analysis of adrenal glands and pituitary glands in this group, but the diagnosis and origin of hyperadrenocorticism was based on biological testing. We compared the four cases of the present study with the 50 cases reported with ultrasonographic description, and definite diagnosis and location of hyperadrenocorticism.11,34 –42 In PDH the majority of cases showed bilateral adrenomegaly, although a few cases had a normal adrenal size compared with the reference intervals. In ADH, the main finding is a unilateral mass with a normal contralateral gland or no visualisation of the contralateral gland. As ultrasonographic protocol and normal data about imaging the feline adrenal glands are getting more precise, the ultrasonographic detection and description of adrenal glands may become more consistent. For instance, adrenal size and asymmetry may help in the suspicion of endocrine diseases in cats, although the diagnosis is invariably based on results of endocrinological testing. Indeed, atypical cases like bilateral adrenal tumours or asymmetrical adrenal hyperplasia would remain misleading with imaging.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
