Abstract
Measurement of body temperature is a routine part of the clinical assessment of a patient. However, this procedure may be time-consuming and stressful to most animals because the standard site of temperature acquisition remains the rectal mucosa. Although an increasing number of clinicians have been using auricular temperature to estimate core body temperature, evidence is still lacking regarding agreement between these two methods in cats. In this investigation, we evaluated the agreement between temperatures measured in the rectum and ear in 29 healthy cats over a 2-week period. Temperatures were measured in the rectum (using digital and mercury-in-glass thermometers) and ear once a day for 14 consecutive days, producing 406 temperature readings for each thermometer. Mean temperature and confidence intervals were similar between methods, and Bland–Altman plots showed small biases and narrow limits of agreement acceptable for clinical purposes. The interobserver variability was also checked, which indicated a strong correlation between two near-simultaneous temperature readings. Results are consistent with auricular thermometry being a reliable alternative to rectal thermometry for assessing core body temperature in healthy cats.
Introduction
Assessing the vital signs constitutes a fundamental component of the patient examination. Measuring body temperature is certainly one of the simplest parts of physical examination, but can provide essential information to guide the clinician’s decisions. 1 For a long time, veterinary patients had their body temperature measured exclusively at the rectal mucosa, which is time-consuming and a potential source of cross-contamination, besides being stressful to many of them. 2
Since the development of infrared devices, there has been a move away from mercury thermometers toward the relatively inexpensive, safer to use infrared thermometers.3,4 These devices measure infrared radiation emanating from the tympanic membrane, which shares blood flow with the hypothalamus, providing readings thought to be an accurate representation of core body temperature. 5 In children, ear thermometry has widespread into clinical practice because it is a quick method of taking temperature and the ear is easily accessible. 6 Needless to say that temperature measurement in animals can be quite difficult, particularly when they are uncooperative or restless. This is likely the main reason why ear thermometry has also gained popularity among veterinarians in the past years.
In humans, most research has focused on the ability of these devices to detect fever in children, and, in general, the literature suggests infrared tympanic thermometers are neither accurate nor reproducible. 7 Studies in veterinary medicine have demonstrated contrasting results,1,5,8,9 with the majority of them concluding that auricular and rectal temperatures cannot be used interchangeably.
Although already in widespread use, the reliability of auricular thermometry has increasingly been called into question in both humans and animals. In this study, we hypothesized that a correlation would exist between auricular and rectal temperatures in a population of clinically healthy cats undergoing several repetitive temperature measurements over a 2-week period.
Materials and methods
Animals
Twenty-nine adult cats (3.6 ± 1.2 kg) of either sex and several breeds were studied. Prior to being enrolled in the study, every animal underwent a detailed clinical examination and ancillary laboratorial tests (complete blood count and biochemistry) to rule out any conditions that could preclude its inclusion in this investigation. Exclusion criteria included anemia, leukocytosis, elevated liver enzymes, elevated blood urea nitrogen and creatinine, as well as any part of the physical examination being considered abnormal, such as hyperthermia, hypothermia, lymphadenomegaly, atypical cardiac and/or respiratory auscultation and abdominal pain. The study was conducted entirely in accordance with guidelines outlined in the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Temperature measurements
Body temperatures were measured in the same room in which the cats were housed during the entire experimental period. The room’s mean temperature and relative air humidity throughout the study were 26.1 ± 1.3oC and 59.6 ± 13.9%, respectively.
Each animal had its temperature taken once a day for 14 consecutive days using commercially-available thermometers, including an auricular infrared device (Thermoscan IRC 4520; Braun), a mercury-in-glass thermometer (Accumed; G-Tech) and a digital equilibrium thermometer (Digital Soft Tip; CVS). After being positioned into the ear canal descending to the eardrum, the activation push-button was pressed and the auricular infrared thermometer provided readings within seconds. The rectal thermometers were inserted a minimum of 1 cm into the rectum and kept in contact with the rectal mucosa either for 3 mins for the mercury-in-glass device or until an endpoint reading audible beep was heard for the digital thermometer. Prior to the study, accuracy of both rectal thermometers was validated in a temperature-controlled water bath against a reference thermometer. A temperature-controlled thermal plate was used to validate the auricular thermometer.
A single experienced veterinarian obtained all temperatures in a randomized fashion. Additionally, another single, experienced observer blinded to the results of the first one performed a second, near-simultaneous measurement of ear temperature at the end of the three initial readings. All temperature readings were recorded at morning time.
Statistical analyses
The results are reported as the mean, standard deviation, 25 and 75 percentiles, and 95% confidence intervals (CIs). To assess the agreement between auricular and rectal temperatures we used Bland–Altman, a method in which the difference between two techniques is plotted against their mean and the limits of agreement calculated. 10 Pearson’s correlation coefficient was also used to check for correlations between all rectal temperatures and auricular measurements, as well as between the results of the two blinded measurements of auricular temperature.
Results
Table 1 gives the results of mean, SD, 25 and 75 percentiles, and 95% CIs of both auricular and rectal temperatures. All devices were used in every animal, producing 406 temperature readings for each thermometer. Compliance during the recording of body temperature was seen in approximately 52.9% and 72.4% of the readings obtained using either a mercury-in-glass or a digital thermometer, respectively, whereas auricular measurements were well tolerated in 93.1% out of the total measurements. Although overt signs attributable to fever were not documented in any of the cats, the maximal temperatures recorded were 40.0°C (auricular), 39.9°C (mercury-in-glass), and 39.8°C (digital).
Temperatures (°C) measured by auricular thermometer and by other rectal devices once a day for 14 consecutive days in 29 clinically healthy cats (samples = 406)

Agreement between auricular and rectal temperatures was satisfactory, as shown by Bland–Altman plots (Figure 1). For both rectal thermometers, the greater bias, which indicates the higher average difference between the two methods, was −0.0995°C for the digital equipment, with limits of agreement showing that the discrepancy between auricular and rectal digital temperatures for an individual animal ranged from -0.7608°C to 0.5618°C (Table 2). An even better agreement was demonstrated for mercury-in-glass thermometry, with discrepancy between the two methods ranging from −0.5060°C to 0.3838°C. With these limits, there could be cases in which auricular temperatures might be misestimated to a maximum of 0.5060°C and 0.7608°C compared with the mercury-in-glass and digital temperatures, respectively. However, one should consider that the biases and their standard deviations are much lower, therefore fitting most cases within acceptable limits for clinical purposes. In this regard, ear temperatures would be misestimated to a maximum of −0.0610 ± 0.2270 compared with the mean of mercury-in-glass and auricular thermometry, with the potential error in temperature readings being much lower than 0.5°C. If one examines the Bland–Altman plots carefully (Figure 1), it becomes quite obvious that the differences in temperature (auricular minus rectal) are close to zero for the majority of the samples, thereby indicating that the tested methods do agree between them.

Bland–Altman plots of temperatures measured by auricular thermometer and by other rectal devices once a day for 14 consecutive days in 29 clinically healthy cats (samples = 406). Average temperatures plotted against differences with temperatures obtained by either (a) a mercury-in-glass thermometer or (b) a digital equilibrium thermometer. Dotted lines represent upper and lower limits of agreement
Bias and limits of agreement (°C) between temperatures measured by auricular thermometer and by other rectal devices once a day for 14 consecutive days in 29 clinically healthy cats (samples = 406)

Pearson’s correlation coefficient was also calculated. In this case, moderate-to-strong positive correlation coefficients were documented between auricular temperature and rectal temperatures obtained by either a mercury-in-glass thermometer (r = 0.8359) or a digital equilibrium thermometer (r = 0.6657) (Figure 2). A strong positive correlation (r = 0.9675) was disclosed when near-simultaneous duplicate auricular temperatures performed by two different observers were compared (Figure 3), as measurements are considered more repeatable when coefficient approaches 1.

Scatter plot showing moderate-to-strong positive correlation coefficients between auricular temperature and rectal temperatures obtained by either a mercury-in-glass thermometer or a digital equilibrium thermometer in healthy cats (29 animals; 406 measurements). Means are indicated for mercury-in-glass (----) and digital (——) readings
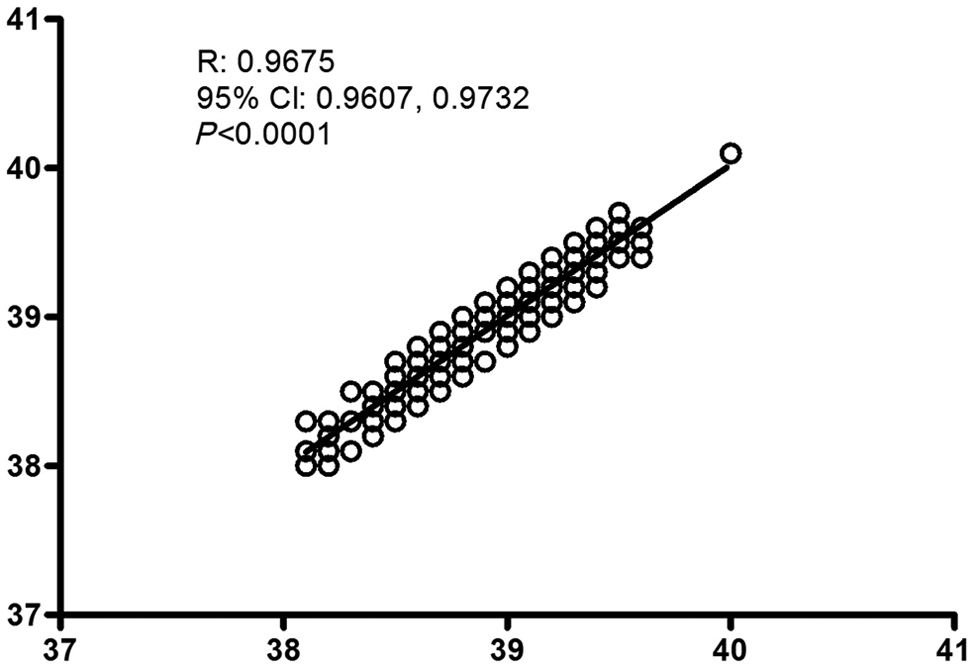
Correlation of two near-simultaneous auricular temperatures measured once a day for 14 consecutive days in 29 clinically healthy cats (samples = 406)
Discussion
Temperature measurement requires a reliable, precise and reproducible method of acquisition in clinical practice. It would be ideal to acquire body temperature at the hypothalamus level or at the thermoregulation center. 11 However, for obvious reasons, it is not possible to use these sites in a clinical setting, with the rectal mucosa remaining the gold standard for the assessment of body temperature in veterinary patients, possibly because of the good agreement that exists between this technique and core body temperature.12,13
Since tympanic thermometers became commercially available in human medicine back in 1986, 14 a great number of clinicians, particularly pediatricians, have introduced them into clinical use, probably because of faster readings, the very convenient site of temperature acquisition and the lower risk of cross-contamination. In veterinary medicine, auricular thermometry has been investigated in several species, including dogs, cats, non-human primates and various other laboratory animals.5,8,9,15,16 In general, these studies indicate that animals tend to tolerate the use of these devices more readily than rectal thermometers. However, the results regarding correlation with an established method of measuring temperature are somewhat contrasting. While Rexroat et al 17 documented very small biases supporting a good agreement between ear and rectal temperatures, in a previous study with euthermic dogs, we found the discrepancy between the temperatures obtained in these two sites to range from −1.335°C to 0.532°C, therefore indicating that either reading should not be interpreted interchangeably. 1
In a large review including 44 studies in children, the pooled mean temperature difference (rectal minus ear) was only 0.29°C, but the wide limits of agreement (–0.74 to 1.32) showed insufficient agreement with an established method of temperature measurement to be used when precise temperature readings are necessary. 6 On the contrary, the Bland–Altman plots of this investigation showed good agreement between auricular and rectal thermometry, with narrow limits of agreement acceptable for clinical purposes. When Pearson’s correlation was considered, our results also documented a correlation to exist between auricular and rectal measurements. Nonetheless, it should be stressed that this analysis is not appropriate to compare two methods of measurement. 10
Other studies comparing rectal and auricular temperatures in cats have been performed. However, most of them include only a small number of subjects and/or a few temperature readings. A recent study found 65% and 90% of the ear temperature values in cats to fall within 1.1°C and 1.6°C of the rectal temperature, respectively, which was considered acceptable by the author ‘if gaining a temperature is vital to shedding light on the potential diagnoses’. 9 An older study found limits of agreement ranging from −1.36°C to 1.43°C between the two methods, and was concluded by the authors as unacceptable for clinical use. 8 The literature regarding canine auricular thermometry is also conflicting.1,13,18
In this study, good correlations were found between near-simultaneous measurements performed by two observers. In dogs, previously published results 18 indicated that duplicate measurements were significantly repeatable (r = 0.999). Surprisingly, in our own study in dogs, we found a weak repeatability of duplicate auricular readings. This difference between studies may be ascribed to several factors, including the observers being more used to the technique of auricular thermometry, which allowed a better positioning of the thermometer probe into the ear canal, the cats being used to the veterinary hospital environment and not getting excited during multiple manipulations, as well as differences between canine and feline ear anatomy. Also, this close agreement was found when only looking at two people who were experienced at using the ear thermometer, and these techniques might not be expected to correlate so well if performed by many different staff (and students) with variable levels of experience of using the ear equipment.
The ear temperature was acquired in this study utilizing an infrared auricular thermometer designed for use in humans. We chose a human device because it is more widely available than veterinary infrared thermometers, therefore being much more common in clinical practice. Interestingly, a recent study in squirrel monkeys showed that the human tympanic thermometer readings did not differ from rectal results, while the veterinary tympanic thermometer measurements were significantly higher. 5 Nevertheless, the correct positioning of the thermometer probe into the ear canal descending to the eardrum is critical in ensuring it reads the tympanic temperature and not the temperature of surrounding tissues of the external ear canal. 4
Conclusions
This study found small differences between rectal and ear temperature measurements, indicating that auricular thermometry may represent an alternative to rectal thermometry for assessing core body temperature in euthermic cats. The documented limits of agreement are narrow enough to be acceptable for clinical purposes on a daily practice. Future studies are needed to assess the agreement between auricular and rectal measurements in hypothermic and febrile cats to better clarify the accuracy of this ‘novel’ technique in situations where body temperature needs to be measured with precision.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
