Abstract
We report an evaluation of the treatment and outcome of cats with suspected primary epilepsy. Phenobarbital therapy was used alone or in combination with other anti-epileptic drugs. Outcome after treatment was evaluated mainly on the basis of number of seizures per year and categorised into four groups: seizure-free, good control (1–5 seizures per year), moderate control (6–10 seizures per year) and poor control (more than 10 seizures per year). About 40–50% of cases became seizure-free, 20–30% were considered good-to-moderately controlled and about 30% were poorly controlled depending on the year of treatment considered. The duration of seizure events after treatment decreased in 26/36 cats and was unchanged in eight cats. The subjective severity of seizure also decreased in 25 cats and was unchanged in nine cats. Twenty-six cats had a good quality of life, nine cats an impaired quality of life and one cat a bad quality of life. Despite being free of seizures for years, cessation of treatment may lead to recurrence of seizures in most cats.
Introduction
Very little is known about the outcome of long-term treatment of seizures in feline primary epilepsy (PE). Phenobarbital is the first choice drug for cats with recurrent seizures. 1 Other anti-epileptic drugs (AED), such as potassium bromide, 2 levetiracetam 3 and gabapentin 4 can be used for feline epileptic patients; however, there are no published data regarding chronic gabapentin therapy. The use of potassium bromide has been associated with adverse respiratory effects and oral diazepam with fatal hepatotoxicosis.2,5 No long-term study has evaluated the efficacy of phenobarbital and its outcome in feline PE, and only expert recommendations exist.1,6,7 The aim of our study was to evaluate the long-term follow-up of cats with suspected primary epilepsy (SPE), mainly on the basis of number of seizures per year.
Materials and methods
The medical records of cats with recurrent seizures between 2001 and 2009 were reviewed retrospectively for the following criteria: SPE with follow-up information for at least 1 year after the start of therapy and anti-epileptic therapy. SPE was considered when the results of complete work-up [clinical and neurological examination, haematology, biochemistry, cerebrospinal fluid (CSF) analysis, intracranial magnetic resonance imaging (MRI)] were normal, or the work-up was not complete but more than 1 year had passed since the onset of seizures without any interictal neurological deficits. Apart from seizures, cats with SPE did not have any evidence of neurological disease at any time during or before the study period. All cats with SPE were re-evaluated 12 months after diagnosis by either AP or ML, and no abnormalities were identified on physical and neurological examination. 8 Cats were selected for the study on the basis of availability of data relating to therapy; seizure type (focal, generalised or both); seizure duration, severity and frequency; side effects; and quality of life (QoL).
The efficacy of treatment was evaluated by the number of seizures per year, the first seizure-free year (the serial number of the first seizure-free year after the start of treatment), the duration of seizure, the severity of seizure and side effects due to treatment.
The number of seizures per year was categorised into four groups: seizure-free (excellent control), 1–5 (good control), 6–10 (moderate control) and more than 10 seizures per year (poor control). The duration of obvious motor convulsion was considered to be the duration of seizure. The change in seizure severity due to treatment and the QoL were evaluated subjectively by the owners. Seizure severity was considered to be unchanged/decreased/increased when the intensity of ictal motor activity was, in most seizures, unchanged/less severe/more severe after the start of treatment. The QoL was defined as good when the cat’s life did not seem to be negatively influenced by the disease or the treatment. QoL was considered to be impaired when the disease or treatment had a significant or important negative influence, and to be bad when the owner considered euthanasia as a result of the disease.
Statistical analysis (Student’s t-test, U-test, Spearman’s correlation coefficient) was undertaken to determine the correlation between outcome (excellent/good/moderate/poor seizure control) and anamnestic data [age of onset, body weight, gender, treatment delay (time between disease onset and start of treatment) and number of seizures before the start of treatment]. All analyses were performed using SPSS v17.
Results
Thirty-six cats met the inclusion criteria. The breeds represented were European shorthair (n = 35) and Chartreux (n = 1). The mean age at onset of seizures was 4.81 years (SD ± 3.51, range 6 months–11 years). Twenty-one cats (58.3%) were castrated males and 15 (41.7%) were female (one intact and 14 spayed). Mean body weight was 5.33 kg (SD ± 1.6; range 4–10). The work-up [routine serum biochemistry and haematology (n = 36), CSF analysis (n = 24), brain CT (n = 9), MRI (n = 9) and pathohistological examination (n = 1)] was unremarkable.
The number of seizures before the start of phenobarbital therapy was 2–5 in 11 cats, 6–10 in seven cats and more than 10 seizures in 18 cats. The mean treatment delay was 144 days (range 2–730). The seizures were classified as focal in six cats and generalised in 17 cats; 13 patients showed both type of seizures.
Initial treatment was always phenobarbital twice a day, at a mean dose of 3.19 mg/kg/day (range 1.8–10 mg/kg/day). The dose remained unchanged during the study period in six (16.7%) cats, increased in 10 cats (27.8%) and decreased in eight cats (22.2%). Twelve (33%) cats received multiple changes to their dose. In four cats (8.3%), other drugs, such as diazepam, gabapentin or levetiracetam, were added to the treatment schedule (Table 1). The mean duration of treatment was 1490 days (range 365–4750).
Descriptive analysis of seizure type, treatment, treatment effect, side effects, survival rate and causes of death in the study

ALT = alanine transaminase; AP = alkaline phosphatase; pheno = phenobarbital
The mean proportions of seizure-free, well controlled cats, moderately controlled cats and poorly controlled cats in a particular year of treatment were 43% (range 0–67%), 31% (range 9–100%), 10% (range 3–14%) and 39% (range 29–64%), respectively. The distribution between seizure-free/good/moderate/poor control per year was 8%/22%/6%/64% in the first year, 42%/ 9%/12%/36% in the second year, 48%/14%/3%/34% in the third year, 45%/10%/15%/30% in the fourth year and 43%/21%/7%/29% in the fifth year, respectively. The seizure frequencies of the cats studied are summarised in Figure 1; Figure 2 shows the individual seizure frequencies per year during 10 years of treatment. Sixteen of the 36 cats (44%) reached seizure-free status in the last year evaluated.

Summarised seizure frequency of studied cats in different years of treatment. The number of evaluated cats is in brackets
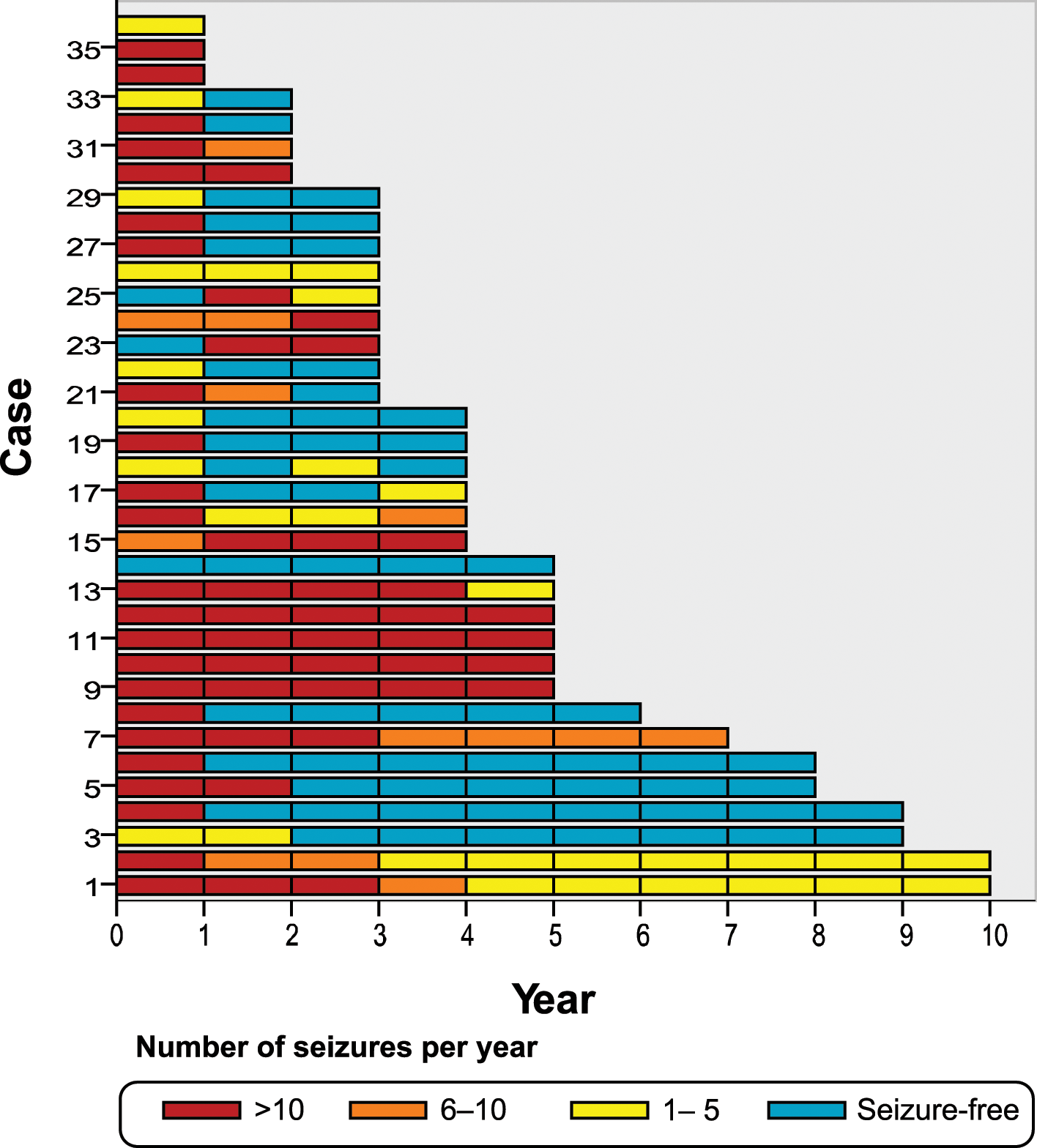
Individual seizure frequency of 36 cats in different years of treatment
Treatment was continued in 28/36 cats (77.8%) until their death or the end of the study. In eight cats (22.2%), the dose was reduced after one seizure-free year. The rate of reduction was 10–25% every 2–3 months. Phenobarbital was discontinued in two cats (5.6%) without return of seizure and, at the time of writing, more than 2 years have passed since finishing treatment. In another six cases (16.6%), seizures returned during dose reduction and thus the dose was increased again.
The mean duration of seizure was about 1.5 mins and ranged from a few seconds to 5 mins. The duration of seizure events (ictus) decreased in 26/36 cats (72.2%) and was unchanged in eight cats (22.2%) after the start of treatment. The subjective severity of seizure also decreased in 25 (69.4%) and was unchanged in nine (25%) cats. The change of seizure duration and severity could not be evaluated adequately in two cases (Table 1). Twenty-six cats (72.2%) had a good QoL, nine (25%) an impaired QoL and one (2.8%) a bad QoL.
The number of control examinations per patient ranged from 1 to 10 (mean 3.2) during the study period. At the beginning of treatment 34 cats showed sedation, ataxia and/or paresis, but the first 4 weeks were excluded from the evaluation as long-term adverse effects are more important for QoL. Twenty-two (61%) patients showed at least one adverse effect. The most common clinical side effects during treatment were mild sedation (n = 15), mild pelvic limb ataxia (n = 9), paraparesis (n = 6), polyphagia or polydipsia (n = 6), weight loss (n = 3), dermatitis and pruritus (n = 2) and behavioural changes (n = 1). Fourteen cats (39%) had no clinical side effects.
Routine biochemistry and haematological analysis during treatment was performed 64 times for 36 cats. Alanine transaminase was elevated 15 times (11 cats) (mean 210 U/l; range 112–821; ref<100), alkaline phosphatase was elevated six times (four cats) (mean 87 U/l; range 41–291; ref<30) and leucocytes were reduced five times (four cats) (mean 3768/µl; range 2590–4030; ref 6000–18,000). In three cats the neutrophil granulocytes were reduced (range 1540–2700 µl; ref 3600–12750). Thrombocytes were reduced once (140,000/µl; ref 180,000–430,000).
Information on the dose of phenobarbital at the final check-up revealed a mean of 6.76 mg/kg/day (range 1–20). Measurements of phenobarbital levels were available 63 times for 31 cats and the mean of the last measured level was 29.7 µg/ml (range 11–73). Five cats showed high phenobarbital serum concentrations (>35 µg/ml) at least once, although post-prandial bile acid determination was within the reference interval in all of these cats.
The concentration of phenobarbital in the serum was compared between cats with seizure-free status in the last evaluated year (available for 9/17 cats) and cats that were not seizure-free (available for 11/19 cats). The mean phenobarbital concentration was 27.9 (range 12–52) and 31.2 (range 11–73) µg/ml in seizure-free and not seizure-free cats; the difference was not significant (t-test, P = 0.624).
Nine of the 36 cats (25%) died during the study period. One cat was euthanased owing to poorly controlled seizures and a bad QoL. The cat was treated with high doses of phenobarbital, which resulted in severe sedation. In this case, neither additional levetiracetam (16 mg/kg q8h) nor gabapentin (16 mg/kg q8h) improved the control of seizures.
Another cat had an accident related to the adverse sedative effect of therapy. The cause of death in three cats was not related to epilepsy and the cause of death in the another four cats was unclear. Four of the nine cats that died during the study period had more than 10 seizures in the last year.
Age and bodyweight showed no significant correlation with the outcome (number of seizures per year) (Spearman’s correlation coefficients ranged from r = −0.026 to r = 0.203 with P-values from P = 0.880 to P = 0.291). There was no influence of gender to the outcome (U-test showed P-values from P = 0.842 to P = 0.472).
Spearman’s correlation analysis was carried out between treatment delay (time between disease onset and start of treatment) and number of seizures after treatment. Correlation analysis showed significant correlations between treatment delay and the number of seizures in the second year (r = 0.418; P = 0.015), but not in the first (r = −0.111; P = 0.520), third (r = 0.365, P = 0.051), fourth (r = 0.293; P = 0.211) or fifth (r = 0.255; P = 0.378) year of treatment. Only eight or fewer cats experienced the sixth and further years of treatment, which is too small a number to permit statistical analysis (see Figure 1). The analysis also revealed a significant correlation between treatment delay and the first seizure-free year (r = 0.888; P = 0.001).
The correlation between the number of seizures before treatment and the first seizure-free year (r = 0.271; P = 0.293) was not statistically significant. There was also no significant correlation between the number of seizures before treatment and the number of seizures in each of the years evaluated.
In summary, the correlation analysis suggests that the shorter the delay in treatment the lower the number of seizures in the second year, and the more rapidly cats become seizure-free for a year or longer.
Discussion
Our findings suggest that many cases of suspected feline PE show good-to-excellent control with phenobarbital treatment. About 40–50% of cases became seizure-free during treatment, 20% of cats were considered to be well controlled, 10–15% of cats were moderately controlled and only about 30% were poorly controlled, depending on the year of treatment considered. Sixteen of the 36 cats were free of seizures in the last year evaluated (44%).
Our findings are consistent with the results of Schwarz-Porsche and Kaiser, 6 who reviewed the use of diazepam for the long-term treatment of epileptic cats. According to these authors, phenobarbital has almost identical therapeutic results: 40% of cats become seizure-free, 40% show a decrease in frequency of seizures of more than 50% and 20% are resistant to treatment. However, the Schwarz-Porsche and Kaiser study 6 suffers from limitations in that the cats were not studied in detail, no follow-up was available and resistance to therapy was not defined. A further problem is that the reduction in seizure frequency does not fully describe the outcome of treatment. Quesnel et al 9 found that 17/30 cats had a good outcome, with 11 having no further seizure activity. However, differences in seizure etiology (meningoencephalitis of unknown origin 14/30, feline ischaemic encephalopathy 6/30, meningioma 2/30, polycythemia vera 2/30, post-traumatic epilepsy 1/30, abscess 1/30, undetermined 4/30) limit the conclusions that can be drawn from this study. Furthermore, the follow-up period was short (range 3–21 months), further hampering firm conclusions.
The term ‘drug-resistant epilepsy’ appears self-explanatory, although there is no precise definition for it and other terms, such as pharmacoresistant or intractable, have similar meanings. Even the human literature is inconsistent: 10 ‘Epilepsy is refractory when the patient’s QoL is compromised by frequent or severe seizures despite appropriate drug therapy’. 4 In other words, not only the number, but also the severity, of seizures is crucial. It is possible that one single seizure a year is harmful for patients because of its severity; while other patients may not be bothered by experiencing one seizure a week. 11 For this reason it is important that not only the number of seizure events, but also the adverse effects and the patient’s QoL are taken into account when evaluating control of seizures and effects of treatment, although, naturally, QoL is a highly subjective variable. It should also be borne in mind that classification as therapy-resistant epilepsy or excellently-controlled epilepsy are dynamic and the nature of the disease can change over time. To avoid the problems associated with the classification we do not use the term ‘pharmacoresistance’ in the present study. We believe it is clinically more meaningful to report the seizure frequency per year (Figure 2), adverse effects and QoL.
About 30% of cats had poorly controlled seizures, although, in most cases, the owners reported that the seizures were of brief duration and low severity. The duration of seizures is difficult to measure not only because owners are not sufficiently accurate, but also because the start and finish of seizures are not always obvious. Furthermore, some cats may have varying durations of seizure. To account for these limitations we evaluated the usual seizure duration. In many cases treatment influenced the duration and severity of seizures. The duration decreased in 26 cases and the severity in 25 cases. This means that, generally, the seizures were short and even in poorly controlled cases it seems that the disease did not severely influence most animals’ QoL. One exception was a cat with refractory seizures in which high doses of phenobarbital resulted in severe sedation. In this case neither additional levetiracetam (16 mg/kg q8h) nor gabapentin (16 mg/kg q8h) improved the control of seizures and so the cat was euthanased.
The follow-up of the cats showed an interesting tendency: if seizure-free status could be achieved for over a year, the cats remained free of seizures for a period of years (Figure 2). The exceptions were cases in which therapy was reduced or even stopped because the animals remained free of seizures for a longer period. Another interesting observation is that seizure-free status could be achieved only in the second or third year of treatment and not subsequently (Figure 2). This appears to suggest that after this period it is unlikely that the animals will respond to phenobarbital therapy with a substantial improvement, although this provocative conclusion could, unfortunately, not be analysed clearly.
There is no evidence in cats to define the best strategy for the start and duration of treatment or whether AED can be stopped in seizure-free cases. Our general therapeutic goal was to reduce the frequency and severity of seizures to a level that does not substantially compromise the quality of life for the pet and its owner while avoiding severe adverse effects. The frequency, severity and duration of seizures were thus the most important parameters to consider, although further factors, such as postictal changes, potential harmful peri-ictal phenomena (vomitus, risk for pneumonia, aggression) and the chances that the owner would be able to carry out the therapy in the long-term, were also taken into consideration.
There was no significant difference in phenobarbital concentration between seizure-free cats (in the last seizure-free year) and other cats. The mean concentration was even slightly higher in cats that did not reach seizure-free status over a year (31.2 µg/ml versus 27.9 µg/ml), which is likely to be a consequence of the dose elevation in cats with seizures. From this point of view it seems likely that some cases are resistant to the doses of phenobarbital that were used, while other cases respond very well. However, a firm conclusion could not be achieved as the phenobarbital serum level was not available for all cats.
Anti-epileptic therapy should not be terminated abruptly, but stopped slowly over a period of several weeks to months. 1 We reduced the dose of phenobarbital when a seizure-free status had been maintained for more than a year. We tried to reduce the dose or to eliminate phenobarbital in more than 20% of cases (eight) but in most (six) seizures recurred and so we recommenced or increased treatment. Based on this observation we conclude that the complete remission of epilepsy in cats is rare, which is consistent with Platt’s statement 7 that most cats should be maintained on anti-epileptic therapy.
The adverse effects of phenobarbital we observed were similar to those reported previously 12 and tended to be more severe at the beginning of treatment and at higher doses. It was not possible to examine the correlation between doses or serum concentration and adverse effects as not all cats were examined, but it seems likely that there are both dose-dependent and dose-independent effects.
Five cats showed high phenobarbital concentrations (over 35 µg/ml) in the serum. Higher concentrations in dogs are associated with increased risk for hepatotoxicity, but no similar correlation has been reported in cats. 2 Bile acid measurements did not suggest that any of the cats with high phenobarbital concentrations were suffering from significant hepatotoxicity.
As epilepsy is a disorder with episodic signs, veterinarians are dependent on the owners’ observations, which provide the most important guide for clinicians. However, the accuracy of the owners’ evaluation can differ in regard to the frequency and duration of seizures, to the adverse effects and to QoL. The subjective differences limit all clinical studies in the field. QoL was assessed subjectively as being good for 26 cats, impaired for nine cats and bad for one cat. Of all the results in our study, these are the most subjective and show the greatest bias. It is difficult to overcome this limitation in a retrospective study. Despite the problems with interpreting the data, though, we are confident in concluding that a high proportion of cats with SPE become seizure-free or have well-controlled seizures under phenobarbital treatment. Poor seizure control or bad QoL can lead to euthanasia, although in our study this was exceptionally rare. Theoretically, we cannot eliminate the possibility that cats whose owners were not satisfied with the animal’s QoL may not have completed the minimum 12-month follow-up period necessary for inclusion in the study, although we did not exclude any cats because of euthanasia before the end of the 12-month observation period.
Furthermore, the precise assessment of seizure control can be problematic. An anti-epileptic drug is generally considered to be effective if it reduces the seizure frequency by 50% or more. 4 Such efficacy is important when evaluating the effect of the drug, but might be too low for some patients. For example, a reduction of 30 seizures per month to 15 seizures would mean that the AED drug is effective, but the disease may remain harmful. For this reason we use the number of seizures per year as the most important parameter from the patient’s point of view. Additionally, owners may expect that clinicians are able to predict the frequency of seizures — at least on a statistical basis.
Human epileptologists have expended considerable effort in determining how to assess the efficacy or effectiveness of AED: ‘Efficacy is the ability of that medication to produce seizure freedom’. 13 Evaluating seizure freedom during a treatment is one of the easiest approaches; in our study 40–50% of cats became free of seizures. However, we do not really know whether AED therapy had a genuine effect or whether some cats with SPE simply have spontaneous remission. The high proportion of recurrent seizures after reducing the dose seems to make this possibility unlikely, although we cannot eliminate the possibility that it occurs in some cases.
The International League Against Epilepsy (ILAE) has developed evidence-based guidelines, with the quality of evidence broken down into four classes. 14 Class I, II and III studies include only randomised trials that are not carried out using feline pet animals. Only expert opinion and case reports have been published on the treatment of feline epilepsy, and these belong — in the ILAE classification — to the weakest class IV of evidence. A further weakness of studies on animal epilepsy is that there is no diagnostic test to confirm PE. The ante-mortem diagnosis is tentative and based on clinical examination and the elimination of other possible aetiological factors. This is an inherent limitation and cannot be addressed until other, for example genetic, tests become available. Important selection criteria were that cats had no previous evidence of neurological disorders other than seizure and were re-examined at least 12 months after the first seizure. It is important to note that not all cats in our study had a complete work-up therefore some of them could have been affected by an intracranial disorder, either non-progressively or slowly progressively. This is a fairly remote possibility and, in any case, it is unlikely to affect our results significantly.
Reports on the incidence of feline PE are controversial. Some authors suggest that PE is rare, or even exceptional, in cats,15,16 although other studies report a considerably higher incidence.6,17 A recent review suggested that PE is an important and not rare cause of recurrent seizures in cats, underlining the importance of long-term studies on the condition. 4 Our previous work supports this conclusion as PE was suspected in about 40% of cats with recurrent seizure disease. 8 Genetic epilepsy has recently been reported among labour cats. 18 However, the aetiological classification in cats is complicated by hippocampal necrosis, which may be a consequence of SPE or other events. 19 A further inherent limitation is that the epileptic origin of the seizures can only be confirmed by electroencephalography, which could record epileptic discharges during the event. This has not yet been attempted in feline clinical research, although a recent report described continuous intracranial EEG in dogs. 20 It is possible that similar studies could provide important knowledge on epilepsy in cats.
Our results indicate that an early start of phenobarbital treatment — after only a few seizures — may be associated with a better outcome than if phenobarbital treatment commences after many seizures and a longer time. However, significant association was only found between starting treatment a short time after the first seizures and a better outcome in the second year of treatment. This does suggest that the shorter the delay in treatment the lower the number of seizures in the second year and the faster a seizure-free year is reached. We recognise, however, that our provisional conclusion should be tested on the basis of additional data. Quesnel et al. 9 evaluated a further aspect of the initial treatment and suggested that more aggressive treatment is required to avoid permanent epileptic activity and to reduce the risk of refractory epilepsy.
Conclusions
The majority (50–80%) of cats with SPE show excellent-to-acceptable control of seizures with phenobarbital therapy, but resistant cases do occur and a second anti-epileptic medication may be indicated. Starting phenobarbital treatment early could be associated with a better outcome. A high proportion of cats (40–50%) may be seizure-free for years, although treatment withdrawal may lead to the recurrence of seizures in many cases. Further studies with a better design, a complete work-up and a more consistent follow-up will be necessary to enable more rigorous conclusions.
Footnotes
Acknowledgements
The authors are thankful to Graham Tebb for his kind assistance.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
