Abstract
Practical relevance:
Feline acute kidney injury (AKI) is a commonly recognized problem in small animal practice that requires prompt diagnosis and directed therapy. There are many treatment methods with which practitioners should be familiar, including medical options, surgical interventions and renal replacement therapy (dialysis). It is important to know which option is most appropriate for each cause and stage of AKI to deliver the most effective therapy.
Clinical challenges:
AKI can cause vague clinical signs, but a vast array of life-threatening sequelae. Rapid recognition of potential complications and knowledge of treatment options is imperative for successful management. Feline patients also require an understanding of their unique physiology as it relates to the therapeutic plan.
Audience:
This two-part review article is directed at small animal practitioners as well as specialists. Part 2 discusses the diagnosis of AKI in cats using physical examination findings, clinicopathologic results and imaging modalities. The treatment of AKI and its sequelae is also reviewed, with information on recent advances in this area.
Evidence base:
While there is very limited data comparing the outcomes of various treatment options, there is literature addressing the use of several medications, as well as renal replacement therapy, in cats.

Prompt recognition – a priority
Acute kidney injury (AKI) is a term used to describe the condition in which there is an abrupt reduction in renal function. This can occur due to several causes, as discussed in Part 1 of this article. In all cases prompt recognition of AKI is important to maximize the chance of a favorable outcome.
Diagnosis
Diagnosis of AKI should focus on attempts to identify an underlying cause and establish the severity of disease.
History and physical examination
A thorough history should be obtained from the owner regarding time course, previous therapies, medication history and potential exposure to toxins. Physical examination may reveal various degrees of lethargy and depression depending on the severity of systemic illness. With severe disease, patients may have oral ulceration and a ‘uremic breath’ odor (ammonia-like smell). Melena may be noted in patients with secondary gastrointestinal ulceration and bleeding. Attention should be paid to the size of the urinary bladder to evaluate for obstruction as well as hint towards urine production. Kidneys are often palpably normal or enlarged, and may be painful. A patient with a renal tumor or a ureteral obstruction may have asymmetry in renal size and shape. However, it should not be forgotten that patients with AKI may have pre-existing chronic kidney disease (CKD) as well and this must be considered when evaluating physical examination findings, as well as laboratory and imaging results.
Patients with AKI may have pre-existing chronic kidney disease as well.
Clinicopathologic assessment
Complete blood count results are often non-specific and may show an inflammatory or stress leukogram. Non-regenerative anemia is more typical of CKD, but could be found in an AKI patient, particularly if acute gastric ulceration is present. Chemistry profile results may be within reference intervals and show a rising serum creatinine over serial monitoring or may demonstrate varying degrees of azotemia and hyperphosphatemia. Hyperkalemia is common in patients with oliguria or anuria. However, potassium may also be increased with post-renal diseases (eg, urinary obstruction or urinary tract rupture), so this finding is not specific to AKI. Alterations in other biochemical values may occur, dependent on the underlying etiology (see discussion on ethylene glycol [EG] in Part 1). Isosthenuria is the most typical abnormality on urinalysis but other findings may be useful in identifying an underlying etiology, such as proteinuria, glucosuria, hematuria, pyuria, bacteriuria, crystalluria or casts.
Culture and sensitivity is recommended in all patients with an unknown cause of AKI prior to treatment with antibiotics. Unfortunately, a negative culture does not necessarily rule out pyelonephritis and it may be beneficial to repeat the culture, consider pyelocentesis or continue antibiotic therapy if a positive response is documented and pyelonephritis is suspected clinically.
Additional clinicopathologic testing, such as toxin/drug levels (eg, EG, non-steroidal anti-inflammatory drugs [NSAIDs], aminoglycosides), may be considered on a case-by-case basis. Doppler blood pressure measurement and a fundic examination should be performed in all patients with kidney disease given the high prevalence of hypertension in this patient population. Depending on the underlying cause and severity of disease, patients with AKI may also suffer from hypotension, which could exacerbate their kidney injury.
Imaging
Abdominal radiography and ultrasound imaging may be helpful to further characterize the etiology of injury. Radiography is useful for evaluation of kidney size and shape, and may also be used to identify radiopaque stones within the urinary tract. Abdominal ultrasound can be used to evaluate further for obstruction (particularly with non-radiopaque calculi), neoplasia or signs of pyelonephritis (Figure 1). Mild renal pelvic dilation can be detected in dogs and cats with clinically normal kidney function, but pelvic size will increase with kidney insufficiency, pyelonephritis or outflow obstruction. 1 Antegrade pyelography or computed tomography may further delineate the presence of a ureteral obstruction if not visible with ultrasonography, as is the case for dried solidified blood calculi or blood clots. 2
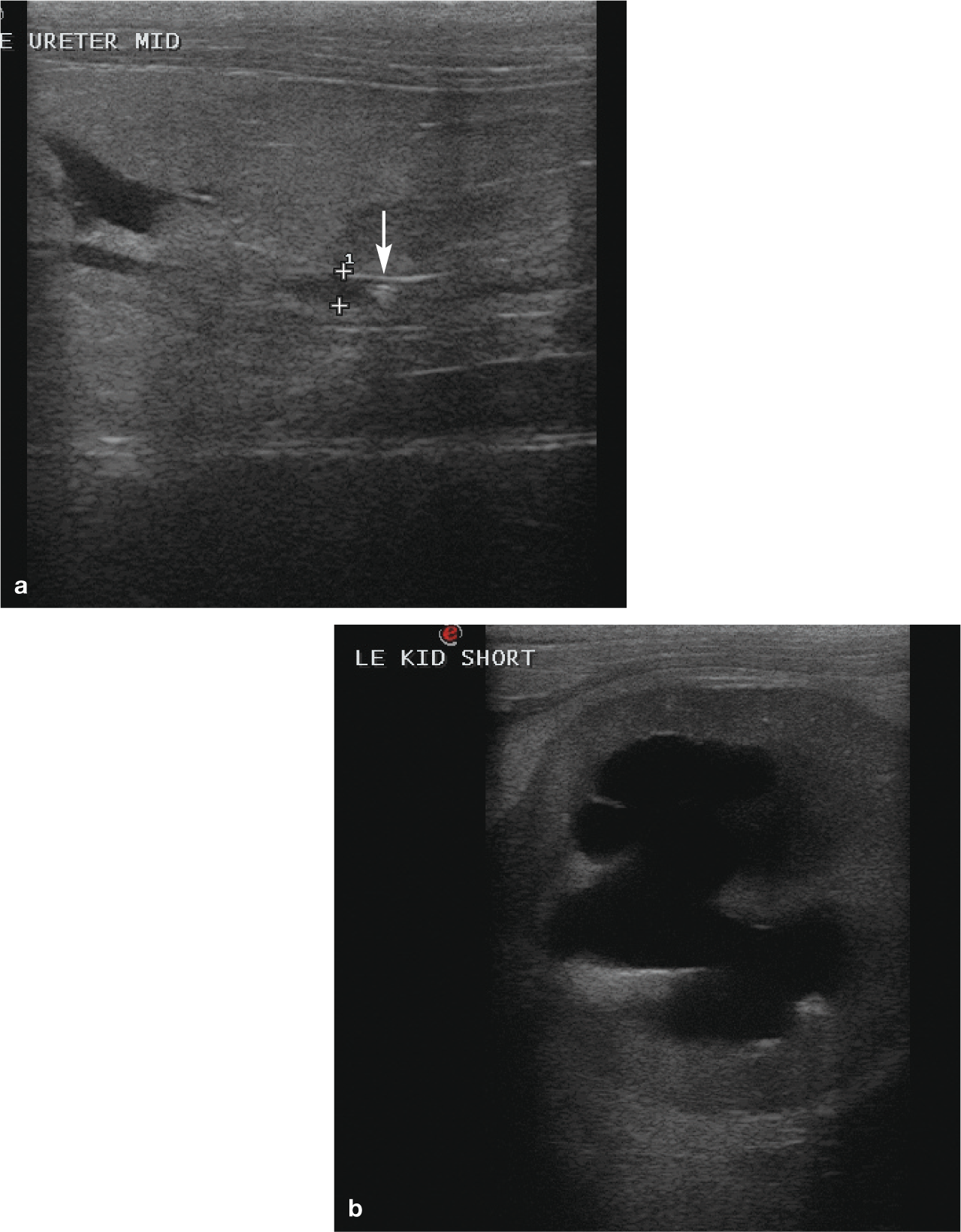
(a) Ureterolithiasis resulting in dilation of the ureter. (Ureter is between calipers; arrow indicates ureterolith.) (b) Short-axis view of the left kidney showing severe hydronephrosis secondary to ureteral obstruction. These ultrasound images are from a 10-year-old castrated male domestic shorthair cat that presented for acute severe exacerbation of CKD resulting from a ureteral obstruction. Traditional medical management was unable to resolve the obstruction and a ureteral stent was placed surgically

Biopsy and GFR estimation
Biopsy is not commonly employed in patients with AKI but may have potential benefit in determining the extent of insult, the prognosis, and whether the disease is purely acute in nature. It is likely required for the diagnosis of neoplasia; however, in the case of renal lymphoma a diagnosis is often possible through fine needle aspiration alone. Additionally, estimations of glomerular filtration rate (GFR) are rarely indicated in patients with AKI as GFR is difficult to determine in a patient that is not in a steady state, as is the case with this condition. GFR reduction in these patients is implied by an elevation in creatinine and alterations in urine output.
Treatment and monitoring
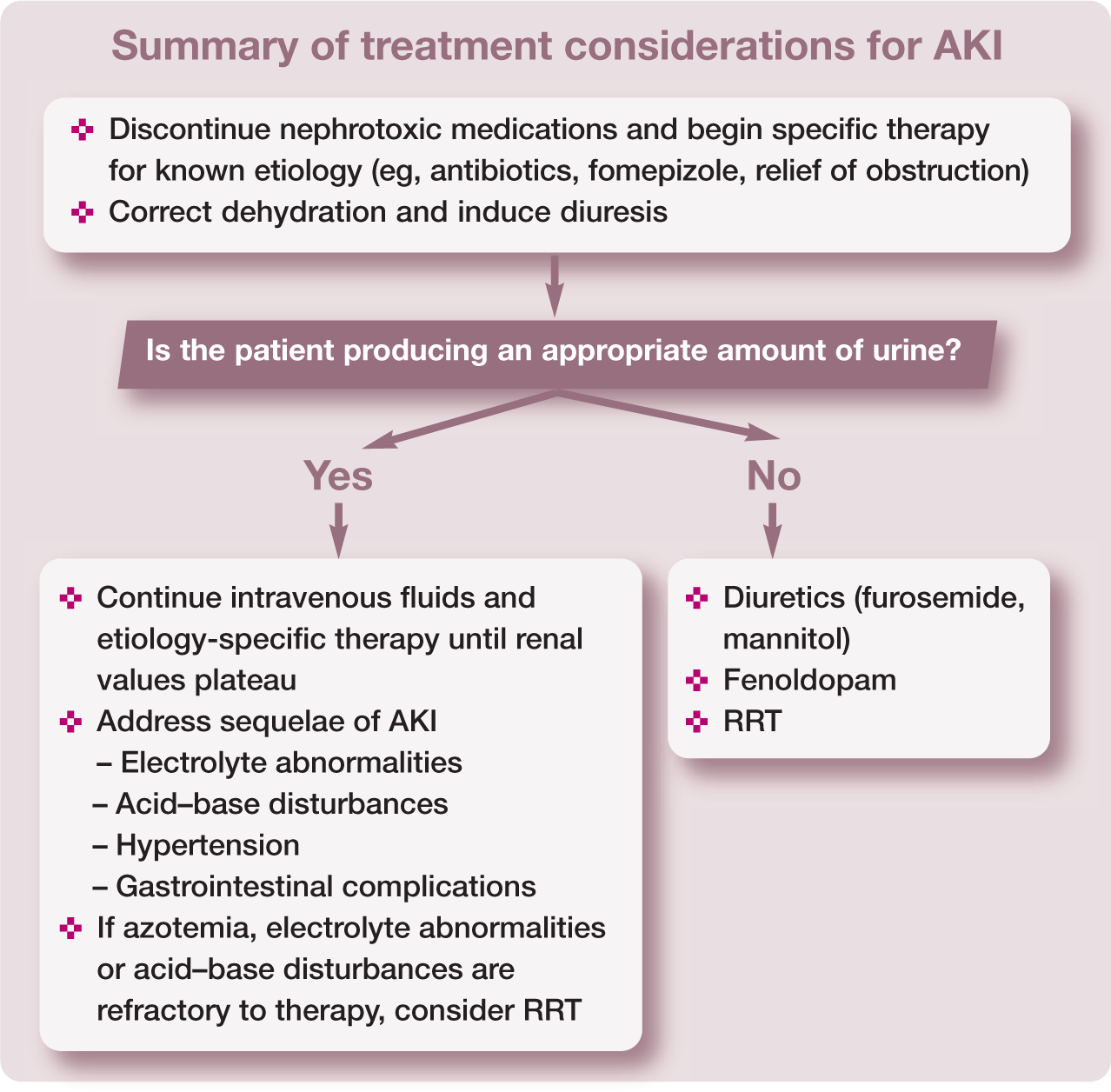
Treatment of AKI should be initiated as soon as possible and requires intensive patient monitoring. Any potentially nephrotoxic medications should be discontinued immediately. If the cause of AKI is known, specific interventions should be started immediately (eg, see Part 1 for discussion of treatments for EG toxicity).
Fluid therapy is the cornerstone of treatment for AKI, but close monitoring is essential to prevent volume overload.
Fluid therapy
Intravenous fluid therapy is the cornerstone of treatment for AKI, but it also requires careful monitoring to ensure appropriate use. Most commonly, a replacement solution (eg, PlasmaLyte A, lactated Ringer’s solution, Normosol-R) is chosen during initial management. Normal saline 0.9% can be considered in cats with severe hyperkalemia; however, caution should be exercised in using this potentially acidifying solution in patients that are frequently acidemic. As most cats with AKI also present with some degree of dehydration, it is important to correct this to ensure adequate renal blood flow.
Generally, fluid volume deficit can be assessed by multiplying the cat’s weight in kilograms by the estimated percentage dehydration based on clinical assessment to obtain an estimated deficit volume in liters. The time over which this volume should be replaced will vary depending on several factors such as cardiac status, the underlying disease process, and whether the dehydration is acute or chronic, but in general ranges from 4–12 h. After this is completed, intravenous fluids should be given to replace ongoing losses and potentially stimulate diuresis. Controversy exists regarding the benefit of forced diuresis in patients with AKI, as this may put the patient at increased risk of morbidity associated with fluid overload. Importantly, the fluid plan should be tailored to the individual patient’s maintenance needs and continued losses, and not necessarily aim to exceed that.
The duration of intravenous fluid therapy depends on the response of the patient. Ideally, the kidney values would normalize, but it is not uncommon to see partial improvement after several days of intravenous fluids with a plateau in azotemia. In general, the renal values are considered to have reached a plateau when no further change is noted over a 24 h period in a well-hydrated patient. Although renal recovery can take several months to reach its full extent, once the plateau is identified this is generally recognized as the point beyond which additional fluid diuresis has diminishing returns. Patient status and tolerance of tapering fluid therapy will help determine the next step.
Monitoring of fluid therapy should be undertaken, both to ensure adequate volume and to prevent overhydration (see box). Fluid overload is associated with increased morbidity and mortality in human AKI patients 5 and likely has similar impacts on veterinary patients.

Electrolyte disturbances
Electrolyte disturbances are common in AKI. The most frequently encountered and clinically important are hyperphosphatemia and hyperkalemia.
Hyperphosphatemia
Hyperphosphatemia is a result of decreased renal excretion and treatment consists of intravenous fluid therapy and administration of enteral phosphate binders to reduce intestinal absorption of dietary phosphorus if the patient is eating.
The terms oliguria and anuria are only applicable to a well-hydrated patient that is appropriately volume resuscitated.
Hyperkalemia
Hyperkalemia is a result of metabolic acidosis shifting potassium into the extracellular space, as well as decreased urinary excretion in patients with oliguria or anuria. Myocardial cells and other muscle tissue are the most clinically affected by hyperkalemia. Elevated potassium results in an alteration in resting membrane potential and deactivation of the sodium–potassium channels, with a resultant prolongation of the action potential and inability of myocardial cells to repolarize. 7 Consequently, there is a loss of cellular excitability. This can result in bradycardia, loss of P waves, a prolonged QRS complex, a peaked and narrowed T wave, a shortened QT interval, sinoventricular rhythm, ventricular flutter or fibrillation, and asystole. 7 Clinical severity is not at all predictable based on the degree of potassium elevation, so treatment considerations should be based on electrocardiographic (ECG) findings. 7
Treatment for hyperkalemia is aimed at cardioprotection and reduction of serum potassium levels. Calcium gluconate can be given intravenously to antagonize the membrane effects of hyperkalemia by decreasing the membrane threshold potential. This will immediately improve ECG changes, but the duration of effect is only about 20–30 mins. Calcium should be given over several minutes, with careful monitoring of the electrocardiogram during administration. Caution is required when administering calcium to patients with altered kidney function and hyperphosphatemia due to the risk of soft tissue mineralization.
Reduction of serum potassium can be achieved through a variety of means including administration of insulin, dextrose, sodium bicarbonate or terbutaline. Insulin and terbutaline (a β2 agonist) stimulate movement of potassium from the extracellular to the intracellular compartment by activation of Na+/K+-ATPase membrane pumps. Dextrose works in a similar way in that it induces release of insulin. Bicarbonate alkalinizes the blood, causing exchange of extracellular potassium ions for intracellular hydrogen ions.
If the patient is hyperkalemic secondary to urethral or bilateral ureteral obstruction, this should be relieved as soon as possible. If the patient does not produce adequate urine and hyperkalemia is refractory to treatment, dialysis should be considered.
Acid–base disturbances
Metabolic acidosis is common in patients with AKI due to a decreased ability to reabsorb and recycle bicarbonate and reduced excretion of hydrogen ions by the renal tubules. Other contributors to acidosis include lactic acid production due to decreased perfusion; decreased elimination of phosphate, resulting in decreased urinary excretion of acid; and EG metabolites, if present.
Treatment with sodium bicarbonate can be considered if the patient’s pH is <7.1 or TCO2 is <12 mEq/l despite improved perfusion through fluid administration. However, in most cases this is not necessary as diuresis often helps address acidosis. Sodium bicarbonate administration is associated with several potential complications, including iatrogenic alkalosis, paradoxical central nervous system (CNS) acidosis, hypokalemia and hypernatremia, and so this is an infrequently used therapy.
Oliguria or anuria
Oliguria is defined as urine production <1 ml/kg/h in a well-hydrated, normotensive, euvolemic patient receiving intravenous fluid therapy. Anuria implies zero urine production. Relative oliguria is another term that is frequently used to describe a patient that produces 1–2 ml/kg/h of urine despite infusion of higher volumes of intravenous fluids. These terms are only applicable to a well-hydrated patient that is appropriately volume resuscitated, and treatment interventions to convert to a non-oliguric state should only be attempted in such patients.

If oliguria or anuria persist despite correction of dehydration, hypovolemia and hypotension, there are various medications such as diuretics and dopaminergic agonists that are used in patients with AKI to attempt conversion to a non-oliguric state. There is currently no evidence that these medications improve outcome in human or veterinary AKI patients, but it is thought that if a patient is able to respond to these medical interventions, their kidney injury is likely less severe and, as such, they tend to have a better prognosis. If these therapies are unsuccessful, renal replacement therapy (RRT) is the only viable treatment option.


Treatments to consider
Dopamine is no longer recommended for treatment of oliguric or anuric AKI. Fenoldopam may be a more effective alternative.
Dopamine – the case against
Dopamine is a catecholamine that exerts an agonistic effect on dopaminergic receptors, β-adrenergic receptors and α-adrenergic receptors in a dose-dependent fashion. Low dose dopamine (0.5–5 μg/kg/min) has historically been recommended as a treatment for dogs and people with oliguric or anuric AKI due to its supposed effect on only D1 and D2 receptors. However, its use has become increasingly controversial due to a lack of proven benefit and possible deleterious effects. It has not been shown to positively influence morbidity, need for dialysis or mortality in human patients. 13 Additionally, in critically ill patients dopamine’s dose-dependent response may be less predictable and tachyarrhythmias, vasoconstriction or hypertension could result. 13 It is no longer recommended for human AKI.
Cats were previously believed to lack renal dopamine receptors, but it is now known that they possess a putative renal D1 receptor in reduced quantities compared with dogs and people. 11 This may explain their lack of response to dopamine infusion with regard to urine output, sodium excretion, heart rate, mean arterial blood pressure and creatinine clearance. 14 Given the lack of beneficial effect seen in feline patients, the potential for side effects and the changing recommendations in human AKI, dopamine cannot be recommended for feline oliguric or anuric AKI.
Hypertension
Systemic hypertension is a commonly recognized sequela in dogs with AKI, affecting 37–87% of this population.15,16 The prevalence of hypertension in cats with AKI has not been investigated to date but it is likely that this is an under-recognized condition that requires recognition and intervention. Blood pressure should be evaluated in these patients both at presentation and throughout their hospitalization (Figure 2). It is imperative that properly trained personnel utilize a standardized technique to ensure accurate assessment. A patient may present with a normal blood pressure which subsequently increases to a hypertensive range (>150/95 mmHg) with fluid resuscitation. Additionally, a fundic examination should be performed in all patients to evaluate for the presence of target organ damage of hypertension (eg, retinal hemorrhage or detachment) (Figure 3).

Doppler blood pressure monitoring in a cat. Here the cuff was measured and placed just above the right tarsus, with the transducer positioned over the dorsal pedal artery

Hypertensive retinopathy (a) and retinal hemorrhage (b) seen on fundic examination secondary to hypertension

A 6-year-old castrated male domestic longhair cat being treated with peritoneal dialysis for an oliguric acute exacerbation of chronic kidney disease secondary to severe pyelonephritis

A 14-year-old castrated male Siamese cat receiving intermittent hemodialysis using a Fresenius 2008H hemodialysis machine after experiencing an acute exacerbation of chronic kidney disease and subsequent volume overload. The cause of AKI was undetermined in this patient
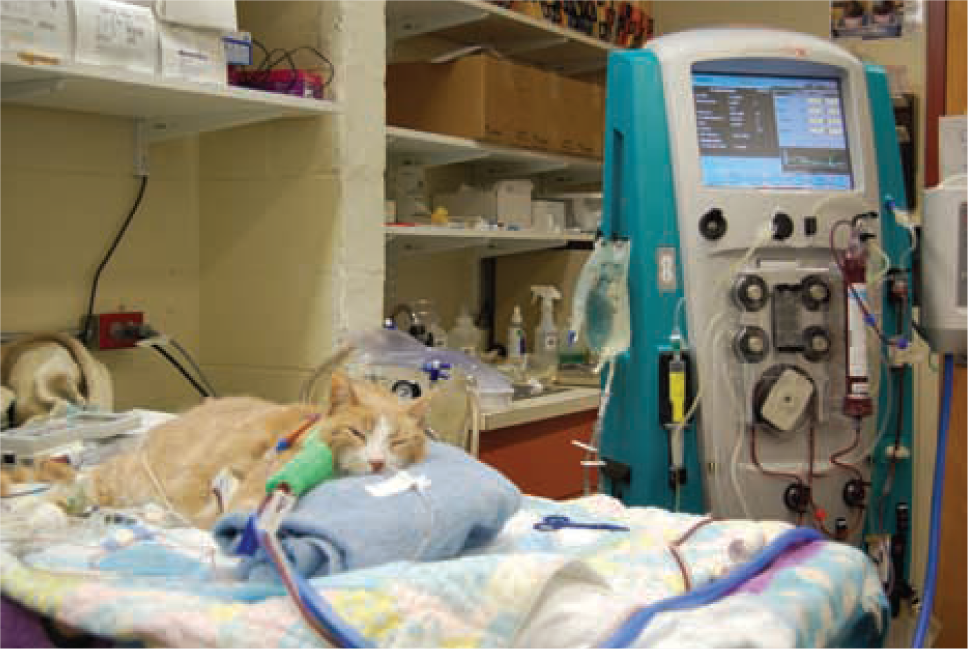
A 6-year-old castrated male domestic shorthair cat with lily-induced nephrotoxic anuric AKI receiving continuous renal replacement therapy using Gambro’s Prismaflex system
Amlodipine, a dihydropyridine calcium channel blocker, is considered the first-line treatment for hypertension in cats. 17 However, caution should be exercised when decreasing the blood pressure so as not to induce hypotension and compromise renal perfusion. The use of angiotensin-converting enzyme inhibitors such as enalapril or benazepril is not recommended in critically ill AKI patients due to their negative effect on GFR; these agents should be reserved for stable patients after recovery from AKI.
Blood pressure should be evaluated in AKI patients at presentation and throughout their hospitalization.
Gastrointestinal sequelae
Vomiting and anorexia are common complications associated with uremia due to AKI in cats and are frequently the reason for presentation. The cause of these sequelae is multifactorial. Uremic toxins stimulate peripheral and central receptors that trigger nausea, which can be treated by a variety of antiemetic medications (metoclopramide, dolasetron, ondansetron, maropitant). The kidneys are important for elimination of gastrin, a stimulator of gastric acid secretion; hypergastrinemia can cause gastric inflammation and ulceration is common in these patients. 18
As such, treatment with medications to inhibit gastric acid production may be beneficial. These medications include histamine-2 antagonists (famotidine, ranitidine) and proton pump inhibitors (omeprazole, pantoprazole). If uremic complications secondary to AKI persist, malnutrition is another potential problem and supplemental nutrition may be required either through placement of a feeding tube or via total parenteral nutrition. It is imperative that the fluid volume administered through parenteral or enteral feeding is taken into consideration with regard to volume status and risk of fluid overload, particularly in oliguric or anuric patients.
Renal replacement therapy
RRT is indicated for cats with AKI that are oliguric or anuric despite appropriate medical therapy, those with refractory hyperkalemia or acid–base disturbances, patients experiencing volume overload, or for removal of certain dialyzable toxins (eg, EG). These therapies essentially allow stabilization of the patient while awaiting renal recovery; or, in the case of toxin removal, they may prevent development of disease by removal of a harmful substance.
There are three different types of RRT — peritoneal dialysis (PD), intermittent hemodialysis (IHD) and continuous renal replacement therapy (CRRT) (see box on page 791). The latter two modalities are only available at a limited number of veterinary facilities (see Supplementary data) and require extensively trained personnel and a dedicated team.

Prognosis
The prognosis for cats with AKI is guarded to poor overall and there is a reported mortality of 47–64% if all causes of AKI are considered.20,21 Factors associated with decreased survival include elevated serum potassium, low serum albumin, low serum bicarbonate and decreased urine production. The severity of increase in the initial BUN and creatinine values is not prognostic.20,21 However, consistently decreased BUN and creatinine within 3 days has been associated with a lower case fatality rate. 21
The prognosis is considerably worse for oliguric or anuric patients as compared with non-oliguric cats and dialysis is required for treatment if they do not respond to initial medical management. 20 Reported survival for cats receiving RRT is 44–60%.19,22,23 However, it is important to keep in mind that the cats that received dialysis would likely die without that therapy. Despite appropriate therapy, residual kidney disease or incomplete recovery is common and affects about 50% of patients that survive an acute event. 20
Supplemental Material
1. Pathophysiology, etiology and etiology-specific management considerations
List of veterinary facilities offering dialysis
Footnotes
Key Points
Supplementary data
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
