Abstract
Current understanding of etiology and pathogenesis
While the exact cause of inflammatory bowel disease (IBD) remains unknown, current hypotheses suggest that feline IBD, similar to IBD in humans and dogs, involves complex interactions between environmental factors (ie, intestinal microbial imbalances, dietary components) and the mucosal immune system, resulting in chronic inflammation in susceptible cats (Figure 1). 1

Chronic intestinal inflammation in feline IBD involves a complex interplay between the mucosal immune system and the enteric microbiota in a genetically susceptible host. Potential genetic factors affecting barrier function or innate and adaptive immunity may predispose susceptible cats to gastrointestinal (GI) signs, aberrant host responses and microbial imbalances (dysbiosis). Environmental factors (dietary constituents, exposure to enteropathogens, NSAID or antibiotic administration, etc) likely govern inflammation onset or reactivation (disease flares)
Genetic defects in the recognition of commensal versus pathogenic bacteria by the innate immune system play a pivotal role in disease pathogenesis in humans and dogs with IBD. Mutations in innate immune receptors of humans (NOD2/CARD 15) and dogs (TLR4, TLR5) have now been linked to IBD susceptibility; and in the presence of an enteric microbiota may lead to upregulated proinflammatory cytokine production (eg, IL-17, TNF-α) and reduced bacterial clearance, thereby promoting chronic intestinal inflammation.1–3 Whether or not similar pathomechanisms come into play in the development of idiopathic feline IBD has not been fully determined. However, antigens derived from commensal bacteria are likely to be important in driving disease pathogenesis, as increased populations of mucosa-associated bacteria (ie, Enterobacteriaceae) have been linked to clinical signs, cytokine mRNA and histopathologic lesions in cats with IBD. 4
Other studies have reported increased lamina propria myeloid/histiocyte antigen-positive macrophages, upregulated epithelial major histocompatibility complex (MHC) class II molecule expression, and increased antibody reactivity to components of the commensal microbiota associated with chronic intestinal inflammation. 5 Moreover, a potential role for dietary constituents is suggested by the clinical benefit of dietary therapy in some cats with IBD. While a genetic basis for IBD in some cat breeds is suspected, causal genetic defects have not been identified to date.
Taken together, these studies suggest that chronic intestinal inflammation of IBD may be due to overly aggressive T cell responses to enteric bacteria (or fungi) 6 in hosts with genetic defects that regulate microbial killing, mucosal barrier function or immune responses. Environmental factors likely govern inflammation onset or reactivation and modulate genetic susceptibility to disease. Future studies are needed to identify possible genetic predispositions in certain feline breeds that contribute to IBD development.
Further unraveling of the pathogenesis of IBD should facilitate the development of novel and more specific treatment options (eg, use of prebiotics or probiotics) for cats. This will require additional clinical trials (and more research funding) to elucidate which therapies are most efficacious in the prevention and treatment of chronic IBD.

Signalment, history and clinical presentation
The clinical manifestations of IBD are diverse and are influenced by the organ(s) involved, the presence of active disease, and nutritional (cobalamin) deficiencies.7–9 Feline IBD predominantly affects middle-aged animals but may also occur in cats 2 years of age or less. 10 Certain breed predispositions (Siamese and other Asian breeds) for IBD are recognized but any breed may be affected. 11
Vomiting and small intestinal diarrhea are most commonly observed and are often accompanied by decreased appetite and weight loss. Gastric and duodenal inflammation is usually associated with vomiting and small bowel diarrhea, while colonic involvement causes large bowel diarrhea with blood, mucus and straining. Cats with both small and large bowel involvement are usually evaluated for diffuse GI disease. Some cats exhibit concurrent inflammatory disease involving the liver or pancreas (feline inflammatory disease [FID], ‘triaditis’) that also contributes to clinical findings. 12
The clinical course of IBD is generally cyclical and is characterized by spontaneous exacerbations and remissions. Triggers for recurring signs are rarely identified but may include dietary indiscretion, transient exposure to intestinal pathogens or drug (eg, steroids, NSAIDs, antibiotics) administration. 8 Importantly, the clinical signs of IBD are not disease specific and share numerous overlapping features with other feline disorders.
Diagnostic approach to idiopathic feline IBD
A diagnosis of IBD is one of exclusion and requires careful elimination of IBD mimics. The possible causes for chronic intestinal inflammation may be excluded through the integration of history, physical findings, clinicopathologic testing, diagnostic imaging and histopathology of intestinal biopsies (see box on page 446). Importantly, a baseline complete blood count (CBC), biochemistry profile, serum total T4, urinalysis and survey abdominal radiographs are useful in eliminating the most common systemic and metabolic disorders (eg, renal disease, hepatopathy, hyperthyroidism) causing chronic GI signs in cats.
After the exclusion of infectious/parasitic agents, non-GI disorders, exocrine pancreatic insufficiency and intestinal structural abnormalities requiring surgery, the most common groups of intestinal diseases associated with chronic small bowel diarrhea include food-responsive enteropathy (FRE), idiopathic feline IBD and alimentary lymphoma (Table 1). A dietary trial using an antigen-restricted diet fed exclusively for at least 7 days will effectively rule out adverse food reactions causing vomiting and diarrhea in cats. 13 Differentiation of severe IBD from well-differentiated lymphoma may be especially problematic in cats. Diagnostic strategies that employ the endoscopic collection of ileal mucosal biopsies, the procurement of multi-organ biopsies during laparotomy, and/or the use of immunohistochemistry for B cell/T cell markers or polymerse chain reaction (PCR) for clonal expansion of lymphocytes may help to distinguish IBD from alimentary lymphoma.14–16
Comparative features of feline IBD, food-responsive enteropathy and alimentary lymphoma

Defining the presence and extent of inflammatory disease in other organs, such as the liver and pancreas, is aided by the performance of baseline laboratory tests (showing increased liver enzyme activities), specialized serologies (eg, increased feline pancreatic lipase immunoreactivity [fPLI] or thyroxine [T4], abnormal bile acids) and diagnostic imaging, especially abdominal ultrasound. There is no specific formula for the detection of these sometimes elusive disorders; rather, clinicians must rely on the completeness of their physical examination and the presence of laboratory abnormalities that are localizing to individual organs. Tissue biopsy is then the most definitive means of confirming a specific diagnosis.
Abdominal radiography is most helpful in defining extra-alimentary tract disorders causing gastroenteritis. Survey radiography might detect organomegaly (liver, kidney) unrelated to IBD or intestinal obstruction that might cause similar GI signs. Abdominal ultrasound is superior to radiology in defining focal versus diffuse mucosal disease, loss of layering, intestinal thickening and mesenteric lymphadenopathy seen with IBD as well as other infiltrative (eg, lymphoma) disorders. 17 Older cats with ultrasonographic evidence of muscularis propria thickening are more likely to have lymphoma than IBD. 18
Cats with severe small intestinal disease (ie, frequent severe diarrhea, decreased activity/appetite, excessive weight loss) often have decreased serum cobalamin concentration.19,20 Serum concentrations of both folate and cobalamin should be measured as they may identify the need for supplementation and guide disease localization (ie, folate is absorbed in the duodenum and cobalamin is absorbed in the ileum). Failure to recognize and correct hypocobalaminemia can delay clinical recovery even with specific therapy for IBD. 9 A subset of cats with chronic enteropathy (ie, IBD or FRE) will have mild-to-moderate elevations in fPLI suggestive of pancreatitis; however, this observation does not appear to be associated with a negative outcome. 21 Pancreatitis is an elusive diagnosis and clinicians should integrate all clinical data (including repeat measurements of fPLI if possible) to arrive at the most accurate diagnosis.
Index of inflammatory activity
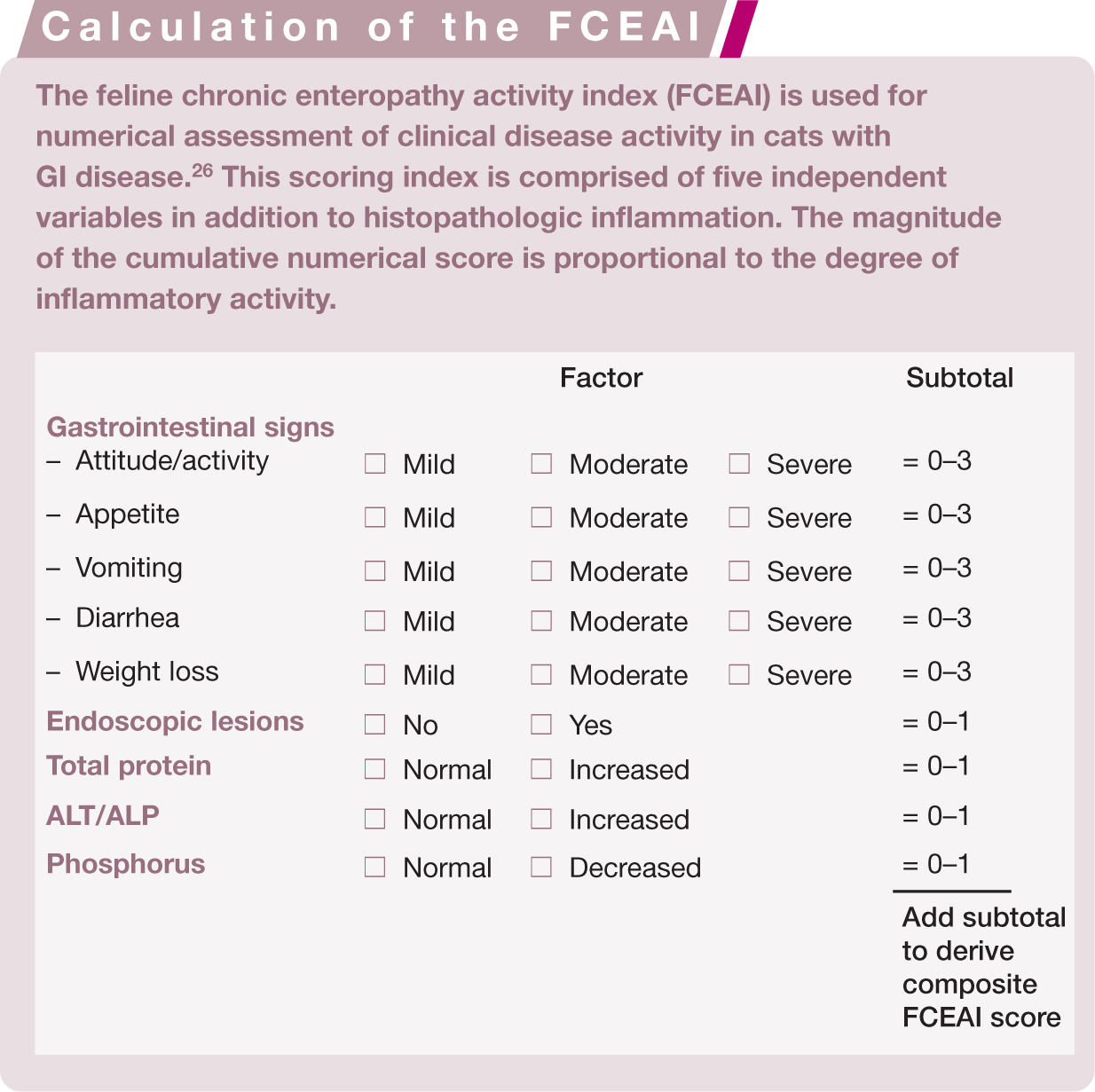
The measurement of clinical disease activity by means of quantifiable indices is well established in humans22,23 and dogs24,25 with IBD. An index for the assessment of inflammatory activity in cats with chronic enteropathy has been recently designed. The feline chronic enteropathy activity index (FCEAI) is comprised of five independent variables (along with histopathologic inflammation) which can be temporally assessed and compared when collected at different times (see box above). 26 Similar to other indices, the magnitude of the numerical score is proportional to the degree of inflammatory activity. This index serves as the principal measure of response to a therapeutic trial and may be used to tailor medical therapy for an individual patient’s need. Recent controlled studies indicate that the FCEAI is of value in defining disease activity in cats with either IBD or FRE.
Intestinal biopsy and histopathologic evaluation
Intestinal biopsies are required to confirm histopathologic inflammation and to determine the extent of mucosal disease. The decision about which organs to biopsy is largely dictated by clinical signs – with anorexia, weight loss, vomiting and small bowel diarrhea suggesting the need for gastric and small intestinal mucosal biopsies, while large bowel signs (ie, straining, fresh blood and mucoid feces) indicate the need for colonic mucosal biopsies. Cats with diffuse enteric disease will exhibit mixed bowel signs and require biopsy of both the small and large intestines for diagnosis. The author’s preference is for diagnostic endoscopy, as this technique allows for direct assessment of mucosal abnormalities and the acquisition of targeted biopsy specimens. Biopsy guidelines for maximizing diagnostic yield of endoscopic specimens in cats have recently been published (Table 2).27,28 Celiotomy with collection of mucosal biopsies from the stomach and intestines may be performed when GI endoscopy is not available. Caution is urged when full-thickness biopsies of the colon are collected due to the risk of bacterial contamination and subsequent infection.
Endoscopic guidelines for cats with chronic enteropathy

Clinicians should obtain this minimum number of samples per organ when performing GI endoscopy. It is assumed that tissue samples are at least marginal for histopathologic interpretation. 28 Increased mucosal cellularity is graded as described elsewhere. 30 Multiple colonic regions should be biopsied to maximize diagnostic yield
The association between endoscopic lesions and disease activity has been investigated to only to a limited extent. While endoscopic abnormalities may or may not correlate with outcome in dogs with IBD,24,29 in cats with IBD endoscopic abnormalities do correlate with both clinical activity and histopathologic lesions at diagnosis (Figure 2).4,26 Standard mucosal biopsies of the stomach and duodenum alone may occasionally miss more distal sites (eg, ileal mucosa) of cellular infiltration. Therefore, it is suggested that ileal biopsies should be obtained in cats to increase diagnostic yield whenever gastroduodenoscopy or colonoscopy is performed, especially since lymphoma is an important differential diagnosis.30,31 The need to perform ileoscopy may also be guided by the presence of hypocobalaminemia as cobalamin is absorbed in the ileum.

Increased granularity (a,b), mucosal friability (c) and erosions (d,e,f) are associated with GI signs and histopathologic lesions in affected cats
The rationale for performing intestinal biopsy with microscopy is to arrive at a definitive diagnosis with the potential for assessing long-term outcome. For example, the prognosis with IBD is generally good to excellent for control of GI signs and a normal life span. Conversely, cats with alimentary lymphoma, mast cell tumors or infectious causes for their GI signs will have a guarded-to-poor prognosis, which may be aggravated by a delayed or even missed correct diagnosis. A general treatment approach using prednisolone/metronidazole/prednisolone with chlorambucil (for potential lymphoma) might result in short-term remission but may mask more aggressive and potentially costly (ie, multiple clinician visits and repeat testing) disease.
The microscopic findings in IBD consist of minimal to pronounced inflammatory cell infiltration of the gastric and/or intestinal mucosa accompanied by varying degrees of mucosal architectural disruption (Figure 3). Unfortunately, biopsy interpretation is notoriously subjective and suffers from extensive interobserver variability, as well as technical constraints of specimen size and procurement/processing artefacts inherent in evaluation of endoscopic specimens.32,33

(a) Histopathologic inflammation in cases of feline IBD is predominantly lymphoplasmacytic in character, as seen in this endoscopic duodenal specimen. (b) Exfoliative cytology performed in the same cat shows a cluster of small lymphocytes embedded within a raft of normal small intestinal (epithelial) cells. This cytologic appearance is consistent with lymphocytic enteritis
Several grading systems for evaluation of endoscopic biopsies have been proposed but no standardized evaluation of intestinal histopathologic findings has been validated with respect to disease severity or outcome. One attempt to standardize interpretation of GI inflammation between pathologists has resulted in the design of a histology monograph that defines numerous morphologic and inflammatory features in endoscopic biopsies. 30 However, even with this standardized scheme there has been considerable disagreement between pathologists; moreover, simply summing unweighted numerical severity scores did not accurately reflect the microscopic severity in dogs with severe IBD (granulomatous colitis). 34 A subsequent analysis of these parameters has resulted in development of a ‘simplified model system’ for defining intestinal inflammation of IBD that is presently being tested in a separate clinical trial.
Histopathologic lesions of IBD are subjectively classified based on the predominant cellular infiltrate within the lamina propria. Intestinal infiltration with macrophages or neutrophils raises the possibility of an infectious process, and culture, special stains and/or fluorescence in situ hybridization (FISH) are indicated. While FISH is limited in its availability to a few academic/diagnostic laboratories, this molecular technique may be of clinical value by confirming the presence of increased mucosa-associated bacteria (with or without accompanying neutrophilic infiltration), which are best treated with antibiotics and/or probiotics. The presence of moderate to large numbers of eosinophils in intestinal biopsies (which, in the author’s experience, may be accompanied by mild circulating eosinophilia) suggests possible parasitic infestation or dietary intolerance. Increased numbers of lymphocytes and plasma cells –so-called ‘lymphoplasmacytic enteritis’ (LPE) – is the most frequently reported form of feline IBD.10,35,36 However, the appropriateness and clinical relevance of the term lymphocytic/plasmacytic enteritis is a contentious issue. For example, LPE may also be associated with intestinal parasites, dietary sensitivity and feline hyperthyroidism. 8 Moreover, cats with and without signs of intestinal disease have similar numbers of lymphocytes and plasma cells in tissues. 5 In hyperthyroid cats with LPE, successful treatment (via I131) of the hyperthyroidism has resulted in remission of clinical signs but follow-up biopsies were not performed to assess for eradication of mucosal inflammation (author’s unpublished observations).

Recent studies indicate that changes in mucosal architecture, such as villous morphology and fibrosis, are related to the presence and severity of GI disease. These studies have used quantitative observer-independent variables (eg, proinflammatory cytokines, mucosal bacteria) to identify histopathological correlates of disease. In cats with signs of GI disease, villus atrophy and fusion correlated with severity of clinical signs and degree of proinflammatory cytokine upregulation in the duodenal mucosa. In a separate investigation, histopathologic inflammation was correlated with clinical signs, endoscopic lesions (ie, mucosal friability, granularity and/or erosions), and clinicopathologic abnormalities (ie, increased total protein concentration, elevated ALT/ALP activities, hypophosphatemia, hypocobalaminemia, and/or increased fPLI) in cats with IBD. 26
Surgical versus endoscopically obtained biopsies
Biopsies for histopathologic diagnosis of IBD are obligatory and they may be obtained endoscopically (mucosal sample) or by laparoscopy or exploratory laparotomy (full-thickness sample). Controversy exists concerning the relative diagnostic accuracy of endoscopic versus full-thickness specimens for the diagnosis of IBD and alimentary lymphoma. Making a correct diagnosis is further complicated by the fact that these disorders share a variety of overlapping features (eg, clinical signs, physical examination findings, abdominal ultrasonographic abnormalities and histopathologic lesions) and that chronic mucosal inflammation (eg, gastric Helicobacter heilmannii infection) may progress to lymphoma in some instances. 40
Endoscopic biopsy specimens of the stomach and duodenum were considered inadequate compared with full-thickness biopsies for differentiating IBD from lymphoma in one study. 14 However, duodenal (endoscopic) assessment was limited to only 50% of the cats and mucosal biopsy was performed blindly (with only three specimens obtained per cat) in 8/22 (36%) of the cats. Because none of the cats in this study had endoscopic biopsy of the ileum performed, malignant infiltrates in this organ could only be confirmed in full-thickness specimens obtained by laparotomy. In another study, the likelihood for diagnosis of alimentary lymphoma was greatest in cats undergoing multi-organ biopsy from all segments of the intestine and the mesenteric lymph nodes. 15 Unfortunately, comparative data describing endoscopic biopsy results from different intestinal segments in these cats was not provided.
Overall, these studies emphasize that the quality of the endoscopic procedure and the adequacy of intestinal biopsies significantly impact accuracy in the diagnosis of feline lymphoma. Suboptimal endoscopic examination and failure to obtain adequate numbers of good quality specimens does not critically evaluate the diagnostic accuracy of thorough duodenoscopy and ileoscopy. 16 The ileum seems to be a consistently affected organ and should always be biopsied when lymphoma or IBD is suspected.14,31 Biopsy samples may be obtained by either ileoscopic examination (most ideal) using a smaller diameter endoscope or by passing the biopsy forceps blindly through the ileocolic sphincter (Figure 4). Histopathologic lesions may vary considerably between ileal and duodenal samples within the same animal.31,41 Finally, laparoscopy may be a useful procedure when full-thickness tissues are required, as this procedure is less invasive and has a shorter recovery time compared with laparotomy.

The ileum is a common site for GI inflammation in cats. Whenever possible, ileal biopsies should be procured by either direct mucosal assessment (ie, ileoscopy) or by passing the endoscope forceps blindly through the ileocolic sphincter
Cats in which there is a suspicion of multi-organ disease (see below) may be managed by celiotomy or laparoscopy, with biopsies of the liver and/or pancreas obtained along with full-thickness intestinal biopsies. Alternatively, GI endoscopy with procurement of hepatic biopsies aided by ultrasound guidance should detect inflammatory lesions affecting these organs.
Multiple organ inflammatory disease
Multi-organ inflammatory disease (ie, FID, triaditis) involving the liver, pancreas and possibly the kidneys has been previously reported with IBD. 12 Cats are at increased risk of the development of pancreatitis and cholangitis by virtue of the fact that the pancreatic duct enters the common bile duct before it opens into the proximal duodenum. This unique feline anatomy allows for reflux of bacteria (eg, Escherichia coli or other intestinal bacteria) and intestinal contents (eg, bile, pancreatic enzymes) into the pancreatic and biliary system. Moreover, small intestinal inflammation (ie, IBD) may also ascend the common bile duct and affect the pancreas and biliary tree due to the high bacterial numbers present in the duodenum. 42
In one retrospective study of 78 cats that had necropsy examinations of their liver, intestine, pancreas and kidney, the prevalence of IBD (15/18; 83%) and pancreatitis (9/18; 50%) was greater for cats with cholangiohepatitis compared with cats with and without lymphocytic portal hepatitis. 12 Thirty-nine percent of cats (7/18) with cholangiohepatitis had IBD and pancreatitis. Other investigators using similar retrospective approaches have shown that concurrent hepatic lipidosis and pancreatitis may be correlated with negative outcome, 43 and that subnormal serum cobalamin concentrations are common in cats with GI disorders affecting the small intestine, liver and/or pancreas. 20 A separate investigation evaluated histologic sections of 40 cats with acute pancreatitis and showed that numerous biochemical abnormalities suggestive of hepatic disease were present, including increased ALT and ALP activities and increased concentrations of bilirubin and cholesterol. 44 However, only mild histopathologic changes were observed in the intestine (13/40; 33%) and liver (5/40; 13%) of these cats.
Taken together as a whole, these studies indicate a potential causal association between small intestinal inflammation, pancreatitis and hepatic disease in cats. However, these earlier data were derived from retrospective studies, have focused on distinct subsets of cats, have used different indices for diagnosis of organ-specific disease, and have not confirmed a distinct temporal relationship between pancreatic, hepatic and intestinal inflammation (namely, that they occurred simultaneously). Furthermore, alterations in tissue morphology do not always indicate disturbances in organ function or the presence of clinically significant inflammation.45,46
More contemporary clinical reports indicate that some cats with IBD have pancreatic inflammation, as evidenced by increased fPLI concentrations and abnormalities in the pancreas detected on diagnostic imaging. While cats with IBD might have increased serum fPLI concentrations, this association does not appear to influence clinical outcome based on a recent report. 21 Clinicians should assume that there is a reasonable association between inflammatory diseases of the liver and pancreas in cats, while the association with intestinal inflammation is more tenuous.
Treatment options for cats with IBD
Nutritional therapy
The rationale for dietary therapy using an antigen-restricted or hydrolyzed diet is that restricting exposure to dietary antigens known to evoke sensitivity will reduce exaggerated host responses and attenuate intestinal inflammation. Other indications for specialized nutritional therapies include managing cats with a decreased appetite, impaired nutrient absorption or specific nutrient (eg, cobalamin) deficiencies.

The benefits of nutritional therapy (alone or in conjunction with pharmacologic therapy) in the clinical management of IBD are well documented. In one study, dietary therapy alone was successful in approximately 50% of referred cats with idiopathic IBD. 13 Moreover, most cats in this study responded quickly (within 2–3 days), suggesting that shorter dietary trials (minimum 7 days’ duration) are quite acceptable to gauge clinical response. While evidence-based observations indicate that most cats respond favorably to dietary intervention, the superiority of one novel protein source versus another, or the advantage in feeding an intact protein elimination diet versus a hydrolyzed protein elimination diet, has not been shown to date.

Characteristics of an ideal diet for IBD are that it should contain a novel intact (white fish, duck, venison, etc) or hydrolyzed protein source and a highly digestible carbohydrate, and be gluten-free, low in lactose and fiber, nutritionally balanced and highly palatable. 11 Traditionally, low-fat diets have been recommended in cats with chronic diarrhea in the belief that fat absorption is impaired in these patients and can aggravate clinical signs. The role of dietary fat in cats with chronic enteropathy has recently been questioned since low-versus high-fat diets resulted in a similar improvement in fecal scores. 49 A variety of commercial diets fulfills these requirements and is readily available.
Soluble fiber supplementation (ie, psyllium dosed at ¼ teaspoon at each meal) or the use of fiber-enriched diets such as Iams (Eukanuba) Low Residue cat food, which contains beet pulp, is useful in cats with colitis. Modifying the diet with omega-3 polyunsaturated fatty acids (PUFAs) may also modulate inflammatory responses by reducing the production of pro-inflammatory metabolites, such as leukotriene B4.50,51 Note that dosing is empirical in cats (ie, eicosapentaenoic acid at 17–25 mg/kg/day and docosahexaenoic acid at 8–18 mg/kg/day) and is extrapolated from dosages of enteric-coated PUFAs used in the treatment of human IBD. 52 Supplementation with parenteral cobalamin (250 µg/cat given subcutaneously weekly for 4–6 weeks while treatment for IBD is ongoing) is advised if serum concentrations are subnormal. A positive response to cobalamin treatment will include improved appetite, weight gain and a reduction in vomiting/diarrheic episodes in affected cats. 9
Pharmacologic therapy
Drug therapy for IBD includes the use of corticosteroids, antibiotics and various immunosuppressive agents. Practical drug treatment recommendations are determined by the clinical severity of disease, the segments of the GI tract involved, the character of the histopathologic lesions, and micronutrient (cobalamin) status. While some clinicians utilize a sequential therapeutic approach with diet and drugs, others propose concurrent therapy with diet, antibiotics, steroids and/or immunosuppressive drugs in cats with severe disease. The optimal drug or drug combination, as well as duration of therapy for induction and maintenance of remission of clinical signs, have not been determined for most protocols. 8

Glucocorticoids
Clinical data evaluating the efficacy of drug therapy for feline IBD is derived from only a few large case-based investigations. In separate studies it was shown that prednisolone alone (n = 14 cats) or used in combination with tylosin or sulfasalazine (n = 14 cats) resulted in resolution of clinical signs in cats with gastroenteritis or IBD colitis.35,36 Prednisone used alone or in combination with another drug resulted in partial or complete resolution of clinical signs in 39/47 (83%) of diseased cats in a different study. 54 A more recent trial reported excellent clinical responses with attenuation of GI signs and disease activity (ie, FCEAI scores) in cats with IBD treated with prednisolone as a single drug induction agent. 26
Taken together, these observations would suggest that corticosteroids such as prednisone and prednisolone are effective pharmacologic agents for treating IBD.
Metronidazole
Metronidazole has been advocated as a single drug agent or in combination with glucocorticoids for treatment of IBD. 8 The mechanisms of action of metronidazole in reducing intestinal inflammation might include antiprotozoan and antibacterial (including efficacy against mucosa-associated Clostridium species) activities, 4 and possibly immunomodulatory effects. 55 Metronidazole may be associated with poor patient compliance due to its bitter taste and tendency to promote inappetence. An alternative formulation is metronidazole benzoate, which is better tolerated by cats possibly due to a marked difference in taste. 52 As this latter formulation is only about 60% metronidazole by weight, it is dosed at a higher oral concentration to reach an equivalent amount of drug to 15 mg/kg of metronidazole, as shown in humans. 56
Caution is urged when using metronidazole as a long-term drug for feline IBD since it has been associated with the development of neoplasms in rodents 57 and in a subset of Crohn’s disease patients. 58
Chorambucil
Chlorambucil (an alkylating agent that crosslinks DNA) may also be used as an adjunct drug for IBD that is refractory to standard (ie, diet and prednisolone) therapy, for cats with severe lymphoplasmacytic IBD, or for cats with severe lymphocytic IBD that is difficult to differentiate from well-differentiated lymphoma. Chlorambucil is very well tolerated by most cats and is minimally myelosuppressive. It is ideally dosed at 2 mg/cat, administered q48–72h, although an alternative dosing regimen of 20 mg/m2 given every 14 days has also been used. 16 Clinicians should perform periodic monitoring (CBCs every 3 months) of cats receiving this 20 mg/m2 protocol.
Ciclosporin
Ciclosporin, an immunosuppressive agent used to treat steroid-refractory IBD in dogs, may also be effective in treating cats with refractory IBD. This drug’s actions include inhibition of T cell function and IL-2 production, which may reduce chronic intestinal inflammation. 59 Unfortunately, only anecdotal reports of clinical efficacy in feline IBD (at a dosage of 5 mg/kg q24h) are available.
Prebiotics and probiotics
Increasing evidence supports a potential therapeutic role for prebiotic and probiotic agents in human IBD. 60 If IBD in cats is similarly driven by impaired tolerance to components of the intestinal microbiota then prebiotics and probiotics may prove beneficial as primary therapies or in combination with dietary and drug therapy. A comparison of prebiotic and probiotic preparations is outlined in Table 3.
Prebiotics and probiotics presently in use for humans and cats with GI disorders
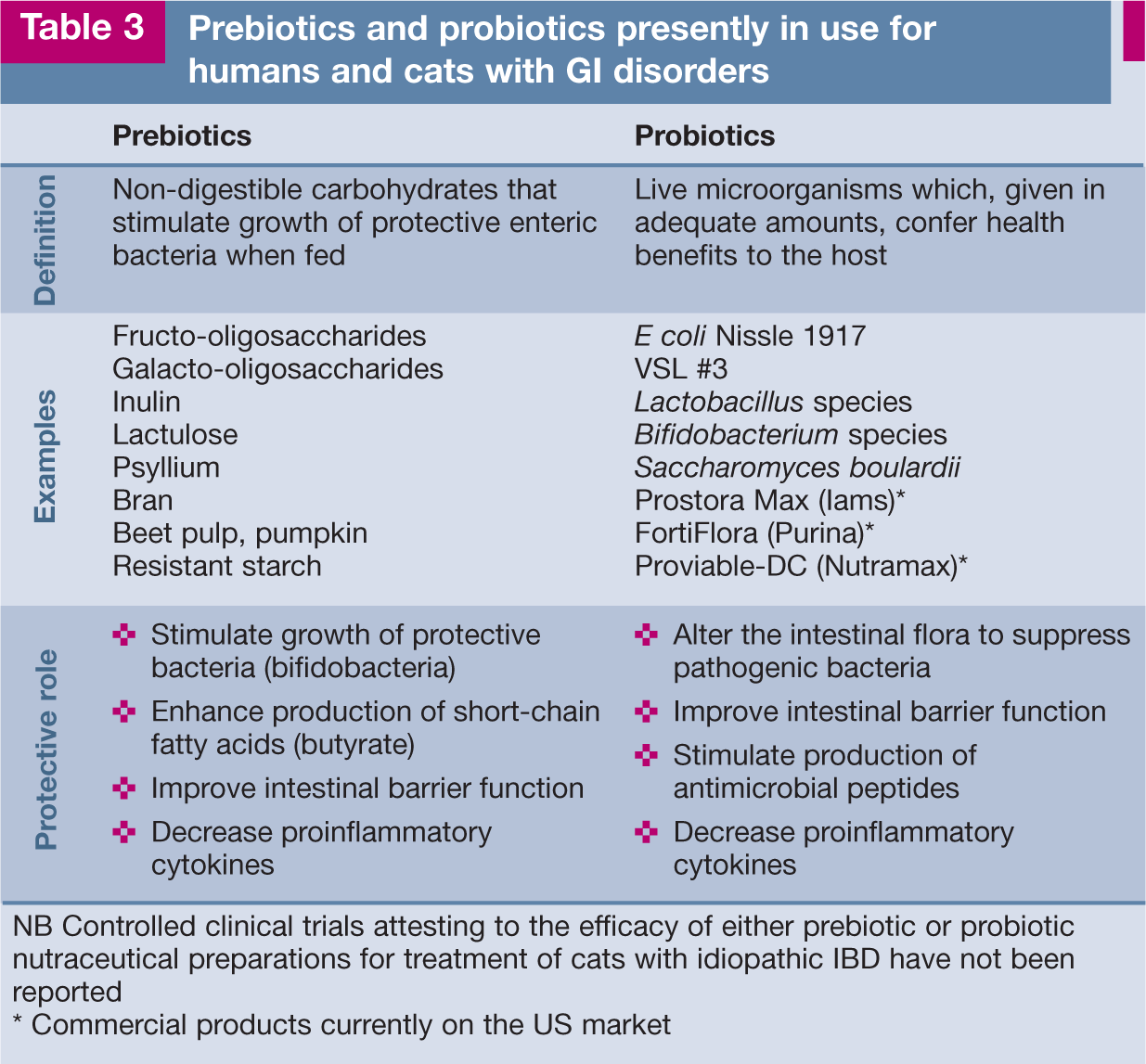
NB Controlled clinical trials attesting to the efficacy of either prebiotic or probiotic nutraceutical preparations for treatment of cats with idiopathic IBD have not been reported
Commercial products currently on the US market
There are only two published studies evaluating the effects of prebiotics on the intestinal microbiota in healthy dogs and cats. In one study, fructo-oligosaccharides (FOSs) supplemented at 0.75% dry matter produced qualitative and quantitative changes in the fecal flora of healthy cats. 61 Compared with samples from cats fed a basal diet, increased numbers of lactobacilli and Bacteroides species and decreased numbers of E coli were associated with the FOS diet. However, bacteriologic examination of the duodenal juice in these same cats showed wide variation in the composition of the duodenal flora across sampling periods, which was not affected by FOS supplementation. 62 Moreover, healthy Beagle dogs fed a 1% FOS diet over a 3-month trial showed inconsistent fecal excretion of Lactobacillus species and Bifidobacterium species. 63
Very little has been reported to date on the use of probiotics to treat IBD in dogs and cats. Recent in vitro studies have confirmed the capacity of a lyophilized probiotic cocktail (eg, three different Lactobacillus species strains) to modulate the expression of regulatory versus pro-inflammatory cytokines in dogs with chronic enteropathies. 64 However, a clinical trial using this same probiotic cocktail in dogs with food-responsive diarrhea failed to induce consistent patterns of regulatory (eg, beneficial) cytokine expression in spite of obvious clinical improvement. 65 One commercial probiotic (ie, FortiFlora – Enterococcus faecium strain SF68) was reported to be beneficial in controlling diarrhea in cats housed in an animal shelter. 66 Recently, a second probiotic (ie, Prostora Max – consisting of Bifidobacterium animalis strain AHC7) was shown to provide more rapid resolution of acute diarrhea than placebo in dogs. 67
Importantly, results from controlled clinical trials evaluating the efficacy of prebiotic or probiotic therapy in cats with IBD have not been reported. The author judiciously uses probiotics as an adjunctive agent along with diet and drug therapy in cats with severe clinical disease. A trial of at least 6–8 weeks’ duration is recommended and probiotic therapy may be continued indefinitely to maintain remission and to prevent relapse of signs.


Outcome
Little information is available regarding long-term outcome for cats with chronic intestinal inflammation. While many cats with IBD respond favorably to dietary and immunosuppressive therapy, treatment failures may still occur. Some of the more common reasons for inadequate clinical response include failure to adhere to dietary recommendations, the presence of severe intestinal inflammation, the possibility of concurrent disease (eg, hepatopathy, pancreatitis, occult hyperthyroidism, or hypocobalaminemia), and a missed diagnosis of GI lymphoma. 68 Low serum cobalamin concentration is one variable that has been associated with refractoriness to treatment in cats with chronic enteropathy. Both the FCEAI and serum acid glycoprotein (an acute phase protein indicative of inflammation) concentration have been shown to decrease in cats successfully treated for IBD or FRE, suggesting that acid glycoprotein may be suitable for laboratory evaluation of the effect of therapy in affected cats. 69
Footnotes
Key Points
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this review article.
Conflict of interest
The author declares that there is no conflict of interest.
