Abstract

Cats and ATE
Arterial thromboembolism (ATE) occurs when a thrombus formed in one part of the circulation embolises to a peripheral artery (Figure 1). Arterial blood flow is decreased to tissues distal to the thrombus as a result of mechanical obstruction and vasoconstriction of the collateral blood supply. In cats, the source of the thrombus is generally the left auricular appendage (LAA) (Figure 2). Cats are particularly prone to ATE in comparison with other species, which is partly (but not entirely) explained by their high prevalence of myocardial disease.
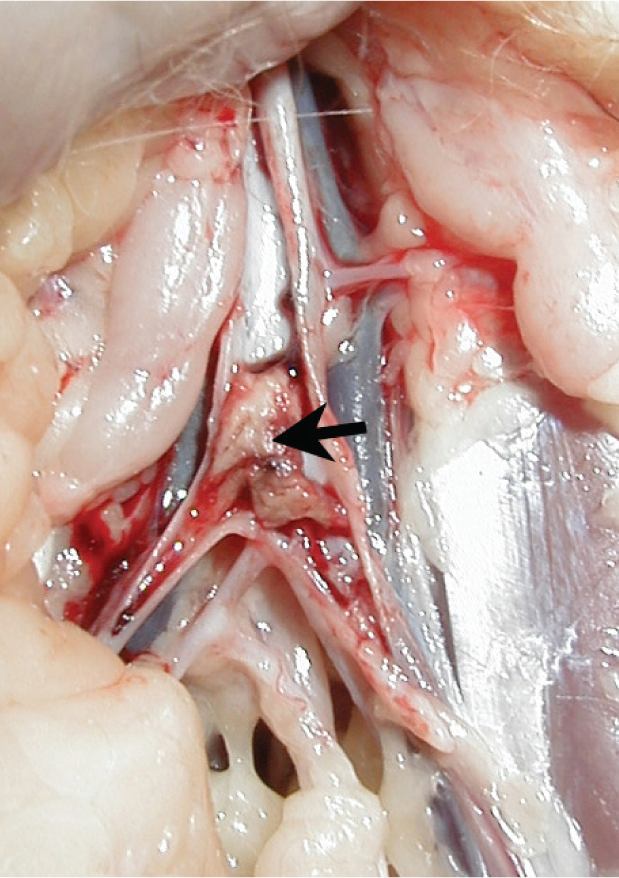
Thrombus (arrow) present in the terminal aorta in a cat presented with ATE

Echocardiographic image showing a thrombus (arrow) in the left auricular appendage in a cat with HCM and left atrial enlargement. This is a right parasternal short axis view. Ao = aortic valve, LA = left atrium
Associated conditions
Most cats presenting with ATE have underlying cardiac disease.1,2 Males are over-represented, but this probably reflects the male predisposition to myocardial disease. 3 Hypertrophic cardiomyopathy (HCM) is the most common underlying condition associated with ATE (being the most common type of myocardial disease), but cats with any form of cardiomyopathy (other than arrhythmogenic right ventricular cardiomyopathy) can be presented with ATE. The risk of ATE appears to be greatest with more severe forms of cardiomyopathy, irrespective of the specific type of myocardial disease. Cats with secondary myocardial disease are also at risk, which includes euthyroid cats with treated hyperthyroidism. 1 Some congenital heart defects such as supravalvular mitral stenosis have been associated with ATE, but this is an uncommon cause. 1 There is also a risk of systemic thromboembolism with septic emboli in infective endocarditis, but this is also rare.
The most common non-cardiac cause of ATE in cats is pulmonary neoplasia, although this is caused by tumour emboli rather than a true thrombus. 1 Rarely, no underlying condition is found.
Clinical importance
ATE is probably one of the most distressing conditions encountered in feline practice, particularly as there is often no advance warning. Owners experience the initial trauma of finding their cat paralysed and in pain, only to face the subsequent devastating news of the poor prognosis. Owners of affected cats are often advised that their pet may not survive the initial episode; or, even if it survives to discharge, may succumb to a future bout of thromboembolism. While both of these statements may be true, it is also true that some cats will regain completely normal motor function following an initial ATE episode, and ATE survivors are more likely to die of congestive heart failure (CHF) than ATE. 1 Fortunately, only a minority of cats with cardiomyopathy will go on to develop ATE, but HCM is sufficiently prevalent that ATE is still a commonly encountered problem in feline practice.
The true prevalence of ATE is not known, as most reports originate from referral institutions. Smith et al reported an overall prevalence of ATE of less than 0.6% of cats seen at the Veterinary Teaching Hospital of the University of Minnesota. 1 The reported prevalence in cats with HCM varies from 12–21%,4,5 although these are biased population samples that probably reflect a particularly high proportion of symptomatic cats. Recent studies of apparently healthy cats suggest that the prevalence of subclinical HCM may be much higher than previously thought (potentially up to 15% of adult cats),6,7 so the prevalence of ATE in cats with HCM is probably much lower than 12%, as ATE is usually seen only in cats with the most advanced cardiomyopathies. Conversely, many cats presenting with ATE in first opinion practice are euthanased, and are therefore not accounted for in prevalence estimates based on referral populations.
Clinical presentation
Cats with ATE typically are presented with a sudden onset of severe pain and distress. Affected cats often vocalise, and show unambiguous signs of pain. The exact clinical signs depend on the location of the thrombus, with the most common presentation being pelvic limb paralysis/paresis associated with embolisation to the distal aorta (Figure 3). In some cases, one pelvic limb is more severely affected than the other. Either forelimb may also be affected with embolisation to the brachial artery. The presentation is much more variable with other embolisation sites (brain, mesenteric arteries), with for example vomiting, abdominal pain or central nervous system signs, so that the underlying thromboembolic cause may not be recognised.

Typical stance of a cat with a ‘saddle thrombus’, showing bilateral pelvic limb paresis
Most cats presenting with ATE have no known history of cardiac disease, and peracute signs of pain and paralysis may be the very first indication of advanced cardiac disease.
Physical examination
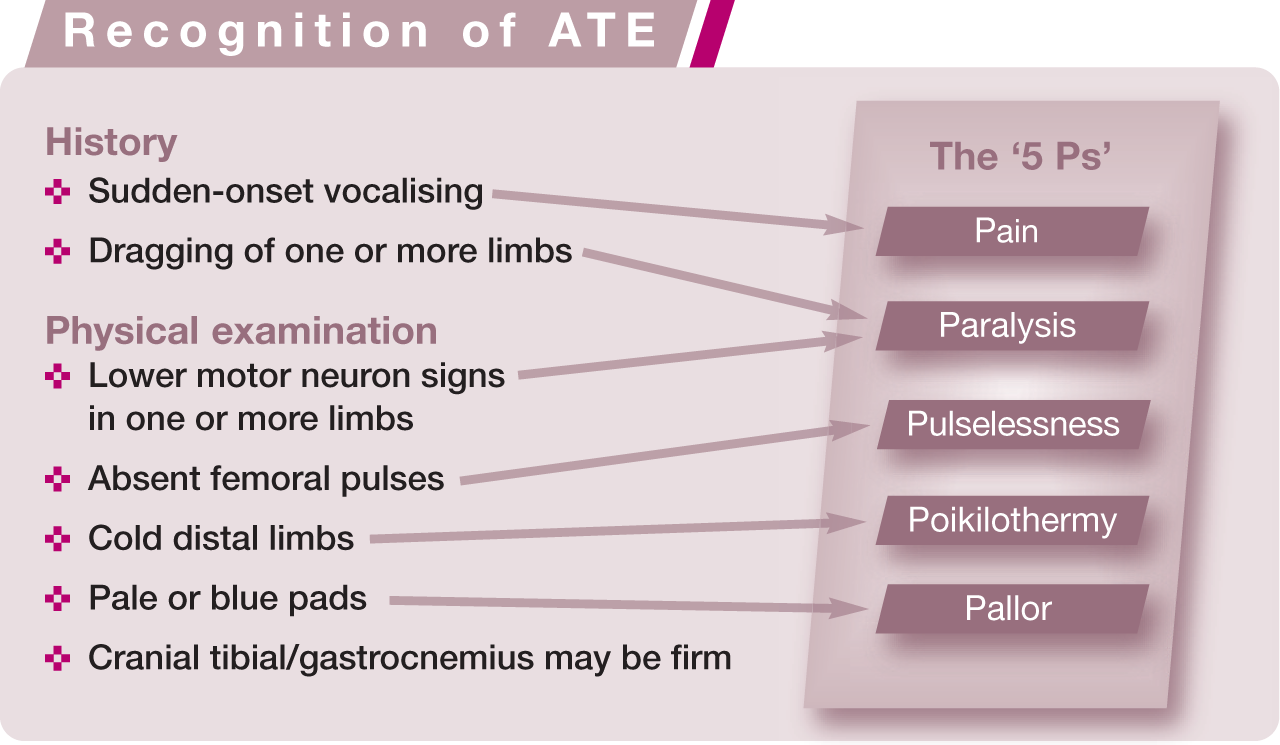
Physical findings vary according to the affected site. With the classic ‘saddle thrombus’ lodging at the aortic trifurcation, the diagnosis can be made from physical examination alone, based on the ‘5 Ps’ of pain, paralysis, pulselessness, poikilothermy and pallor (see box). Motor function is usually absent or reduced distal to the stifles, with skin sensation absent distal to the tarsus. 8 The combination of pelvic limb lower motor neuron signs with absent femoral pulses and cold extremities is pathognomonic for ATE.8,9 Lower motor neuron signs may be present in forelimbs with a brachial thrombus, where pulselessness may be more difficult to recognise. Foot pads are frequently pale or cyanosed in the affected limb, which is a particularly useful finding in forelimbs. In some cats, the ATE is only ‘partial’, with some motor function in the distal limb persisting or returning rapidly. If the embolised thrombus is small, rapidly lysed or collateral circulation is quickly re-established, motor function may be present by the time the cat is presented.
Rectal temperature is often reduced (see box below), and is considered a poor prognostic sign. 1 It is important to recognise signs of CHF, as this confers a worse prognosis and must be managed differently. Although a rapid respiratory rate is often interpreted as a sign of CHF in cats with heart disease, many cats with ATE will be tachypnoeic due to pain, whether or not they have CHF. It can be very difficult to distinguish rapid respiratory rate associated with pain or stress from tachypnoea due to pulmonary oedema, unless crackles can be identified on auscultation. Although myocardial disease is the underlying cause of ATE in the vast majority of cases, auscultation may be normal in up to 40% of cats presenting with ATE.1,10 Just over half of cats will have a murmur, gallop or arrhythmia on auscultation.

Laboratory tests
A wide range of biochemistry abnormalities may be present. Most cats will exhibit stress hyperglycaemia, and azotaemia and hyperphosphataemia are also common. Azotaemia is usually prerenal, although can also be associated with thromboembolism of a renal artery. Typically, serum creatine kinase concentrations are dramatically increased as a result of muscle ischaemia. Hypocalcaemia and hyponatraemia are also reported. Although hyperkalaemia is an important and potentially fatal complication of ATE, the rise in plasma potassium concentrations often occurs suddenly as perfusion is restored and some cats may actually be hypokalaemic on presentation. In older cats, thyroxine levels should be measured as hyperthyroid cats may be at increased risk of ATE irrespective of any cardiac changes. 1 Coagulation tests are frequently normal, although D-dimers may be elevated.
Radiography
In the absence of audible crackles, thoracic radiography may be the most reliable way to confirm the presence of CHF, but should not be undertaken in cats with severe respiratory distress. Thoracic radiographs are also helpful for identifying pulmonary mass lesions in those cats with an underlying neoplastic cause (Figure 4).

Lateral thoracic radiograph from a cat with ATE, showing a mass lesion in the caudodorsal thorax. Tumour emboli are probably responsible for the thromboembolic signs in cats with pulmonary neoplasia
Echocardiography
As the type of cardiomyopathy is not particularly relevant, it is not usually necessary to perform immediate echocardiography. Most cats have a dilated left atrium (LA), and some will have left ventricular systolic dysfunction.1–3,11,12 Spontaneous echo contrast (‘smoke’) is also frequently present, and is believed to be a marker of increased risk of ATE. 13 In contrast with most other scenarios, one cannot assume that CHF is the cause of respiratory distress simply because the LA is enlarged on echocardiography, as LA enlargement will be present in most cats with ATE.

In cats with a recent onset of signs, it may be possible to identify the thrombus in the terminal aorta using ultrasound imaging, but the diagnosis of ATE in cats with pelvic limb paresis is usually based on the distinctive clinical findings rather than imaging. Lack of a visible thrombus in the terminal aorta does not rule out ATE, particularly if signs are of greater than 24 h duration. Where no underlying cardiac disease is identified, further local imaging of the occluded artery is more justifiable.
Blood pressure measurement
A Doppler blood pressure measuring system can be used to identify the presence or absence of distal limb flow based on an audible Doppler signal when the crystal is positioned over the artery in question. Although the prevalence of systemic hypotension in feline ATE has not been reported, many cats can be expected to be hypotensive on presentation. Conversely, it should not be assumed that supranormal systolic pressures indicate primary systemic hypertension. Hypertension rarely causes ATE, and increased blood pressure is more likely to be caused by pain-induced stress.
Natural history and prognosis
Many cats are immediately euthanased on diagnosis. However, although ATE is widely perceived to be inevitably disastrous, some subpopulations of cats do much better than others. As many as 70–80% of cats with a single limb affected will survive to discharge, with up to 90% survival rates in cats presenting with some motor function. 14 This contrasts sharply with rates of survival of <40% in treated cats overall.1,14 However, in the study by Smith et al, rates of survival to discharge improved over the 10-year period evaluated, with 73% of cats surviving to discharge in the last year of study. Hypothermia appeared to be one of the most reliable markers of reduced initial survival in this study, with <50% chance of survival to discharge in cats with a rectal temperature <37.2°C on presentation. 1
The main mortality risks over the first 2–3 days are from CHF or hyperkalaemia from reperfusion syndrome. Pain is severe in the initial 24 h, but decreases substantially after the first 48 h. The cranial tibial and gastrocnemius muscles may become firm with severe ischaemic muscle damage, and ischaemic nerve damage may result in ‘dropped hocks’ as well as loss of distal sensation. Femoral pulses frequently return within 3–5 days. Where tissue ischaemia has been severe, there is a risk of skin and muscle necrosis, which usually manifests within the first 2 weeks (Figure 5). In severe cases, this may lead to loss of toes or distal limbs. Sometimes skin may necrose more proximally, sparing the extremities. Ischaemic nerve damage may be reversible, although this can take 8 weeks or longer.

ATE affecting the left pelvic limb of a Sphynx cat. A well demarcated region of ischaemia is evident as darker skin (a). Two days later, progression of ischaemic skin damage is evident over the lateral aspect of the tarsometatarsal area (b)
Reports of average long-term survival vary between 51 and 350 days. The most common cause of death is CHF, although ATE may recur in up to 50% of cats. 2
Pathophysiology
Thrombus formation is a complex process, but it is generally accepted that platelet activation, blood stasis and endothelial dysfunction all contribute in varying degrees to ATE in cats (Figure 6). Thrombogenesis involves multiple signalling pathways coordinating the interactions between platelets, coagulation factors and the endothelium, so that haemostasis can be safely achieved without risk of inappropriate thrombosis in the healthy individual.15,16 Resting platelets have surface glycoproteins which allow adhesion to the vessel wall where the endothelial layer is damaged or lost. Von Willebrand factor (vWF) is necessary for this adhesion, triggering platelet activation and production of adenosine diphosphate (ADP) and thromboxane A2 (TXA2). Local release of ADP and TXA2 activates additional platelets, leading to further recruitment. Tissue factor in the vessel wall results in thrombin production, which activates platelets via a different pathway. The growing platelet plug includes increasing numbers of platelets bridged together by fibrinogen, linking platelet integrin αIIbβ3 receptor sites.

The left panel of the diagram shows the normal state, where a healthy endothelium contributes to a microenvironment that inhibits thrombosis within a blood vessel (‘thromboresistant’). Endothelial production of nitric oxide (NO), antithrombin (AT) and prostacyclin (PGI2), and endothelial expression of thrombomodulin inhibit attachment and activation of platelets. The right panel represents an environment that promotes thrombogenesis, where the endothelium is damaged or missing, and collagen is exposed. Platelets attach to collagen-bound von Willebrand factor (vWF), resulting in platelet activation. Activated platelets release adenosine diphosphate (ADP) and thromboxane A2 (TXA2), which activate additional platelets. Tissue factor results in thrombin generation, which amplifies the effects of platelet activation and leads to further thrombin production, as coagulation factors such as Xa become involved. Platelet–platelet affinity is enhanced as attachments form with fibrinogen and vWF, leading to a more stable thrombus. The sites of action of aspirin, clopidogrel and heparin are shown
The endothelium normally produces factors that help maintain ‘thromboresistance’. These antithrombotic factors include antithrombin, thrombomodulin, tissue-type plasminogen activator (tPA), prostacyclin (PGI2) and nitric oxide. Fast rates of blood flow are associated with nitric oxide production mediated by shear stress, whereas blood stasis may reduce this antithrombotic effect.
Cats appear to be much more prone to intracardiac thrombus formation than dogs, and even humans. Human intracardiac thrombosis is associated particularly with heart failure and atrial fibrillation, and can lead to embolic stroke. Spontaneous echo contrast indicates increased risk of embolic stroke in human patients, and is believed to be associated with increased risk of ATE in cats.
Platelet function
Assessing platelet function in cats is particularly difficult as their platelets appear especially reactive compared with other species. The gold standard test for assessing platelet function (platelet aggregometry) requires operator skill and experience, and results will vary depending on the precise technique and agonist used.17,18 Problems are exacerbated in cats, as even venepuncture technique can affect the results. Evidence of a hypercoagulable state can be obtained by measuring circulating markers of thrombin generation such as thrombin–antithrombin complex concentrations. 19
Blood stasis
Spontaneous echo contrast is believed to be a marker of blood flow stasis and, as mentioned, is a known risk factor for human thromboembolism. Spontaneous echo contrast is commonly encountered in cats presenting with ATE, and this has been linked with low velocities of blood flow in the LAA. 13 A direct association between low LAA blood flow velocities and ATE has not been demonstrated in cats, however.
Endothelial function
The endothelial contribution to thromboregulation is difficult to test. Studies in rats have shown that an increase in atrial pressure reduces endocardial expression of thrombomodulin, leading to an increase in local generation of thrombin and risk of thrombus formation. 20 This suggests that LA stretch may itself be responsible for increasing thrombogenicity. LA enlargement is well documented as a risk factor for feline ATE.1,2,11,12,14 In human HCM patients, the degree of LA enlargement correlates well with plasma concentrations of thrombin–antithrombin complexes. 21

How to identify cats at risk
Our ability to identify cats at risk of ATE has greatly improved, but relies on access to echocardiography (see box above). Many cats at risk of ATE do not even have auscultatory abnormalities, and as these apparently healthy cats are not likely to be selected for screening by echocardiography, most cats presenting with ATE will have no known history of heart disease.1,14 Cats with a history of ATE are clearly at risk, as are cats with a visible atrial thrombus or spontaneous echo contrast. Any cat with myocardial disease and LA enlargement has an increased risk, particularly if LA systolic function is impaired. The risk is also increased in cats with left ventricular systolic dysfunction. 11 The short-term risk of ATE for asymptomatic cats with HCM and normal LA size is probably low.
Therapy for ATE
The range of drugs used in the treatment of ATE is summarised in Table 1.
ATE therapy
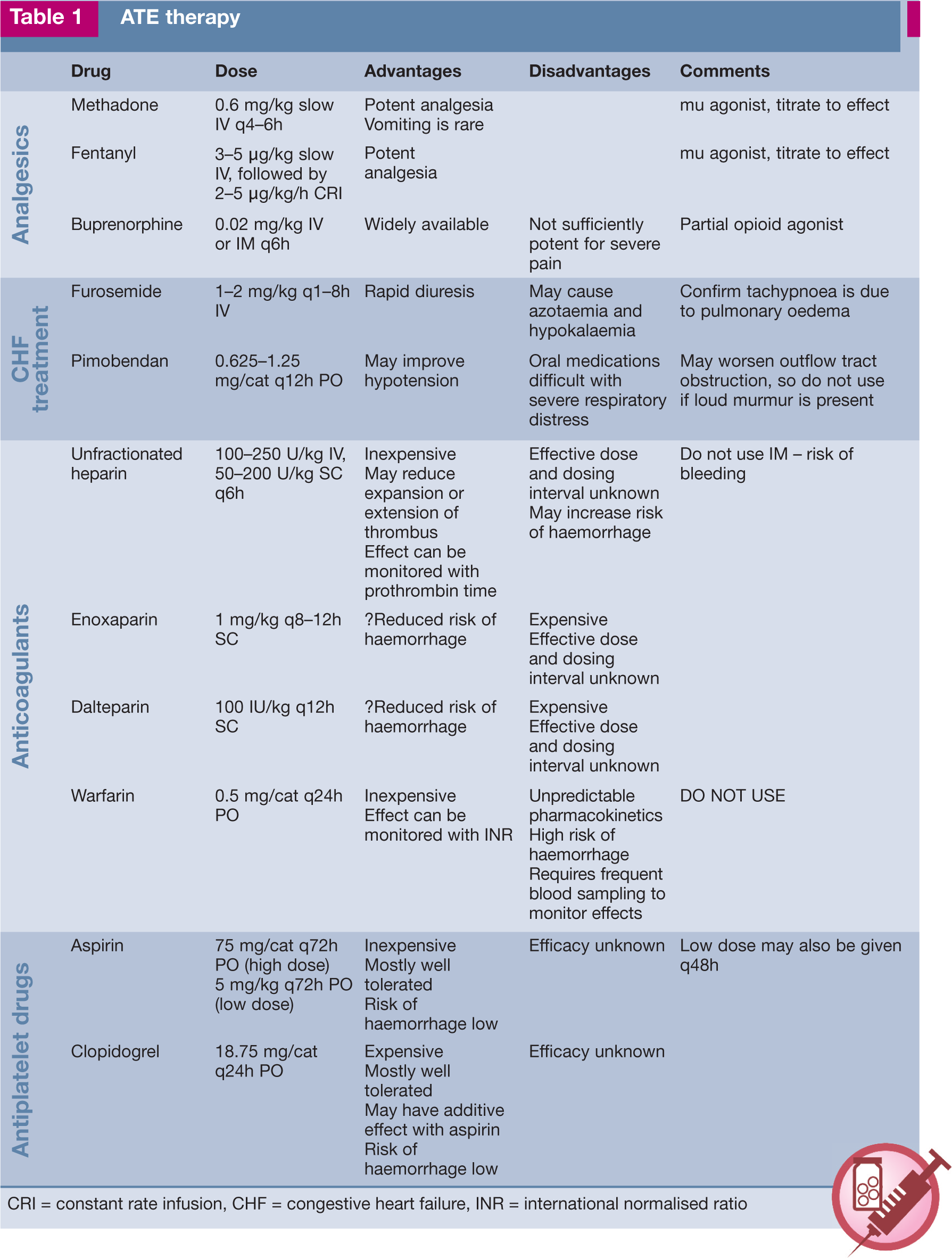
CRI = constant rate infusion, CHF = congestive heart failure, INR = international normalised ratio
Thrombolytic therapies
Although it seems logical to attempt to remove or lyse the thrombus in cats with ATE, this approach is not recommended. Surgery is not advocated because of the high mortality rates, and catheter removal of a thrombus is technically very challenging. More importantly, thrombolytic therapy with tPA or streptokinase results in mortality rates that are at best no lower than without thrombolytic treatment.3,22,23 This is because any method that results in sudden reperfusion of ischaemic tissue will risk the life-threatening complications of reperfusion injury. This occurs when ischaemic metabolites such as potassium and free oxygen species are flushed into the systemic circulation, causing arrhythmias, acid–base disturbances, renal dysfunction and death.
Anticoagulant/antiplatelet therapies
In general, anticoagulant therapy with vitamin K antagonists appears to be more effective than antiplatelet therapy in preventing stroke or peripheral arterial embolism in human patients, including those with HCM and atrial fibrillation.24–26 The preference for warfarin is less clear-cut in patients without atrial fibrillation, and trials are currently under way comparing aspirin versus warfarin in patients with dilated cardiomyopathy and sinus rhythm. 27
Warfarin
Despite the many difficulties associated with its use, the principal anticoagulant recommended to prevent human stroke is still warfarin. Warfarin blocks the effects of vitamin K necessary for coagulation factors II, VII, IX and X to be activated. Warfarin is highly protein-bound with unpredictable pharmacokinetics and pharmacodynamics, making it a difficult drug to use safely and effectively, even in human patients. Warfarin’s effects can be monitored by measuring the prothrombin time, and standardising the value for each batch of reagent to derive an international normalised ratio (INR).
Use of warfarin has been reported in cats with a history of ATE.2,3,28 The target INR for cats of 2.0–3.0 has been extrapolated from human guidelines, 28 but there are so many factors that can influence the effects of warfarin, both thrombus formation and haemorrhage are still possible. The intensive monitoring necessary with warfarin also compromises feline quality of life in terms of frequent clinic visits for blood sampling and an indoor lifestyle.
Unfractionated heparin
Unfractionated heparin is a mixture of sulfated glycosaminoglycans, requiring antithrombin for its anticoagulant effect. The heparin–antithrombin complex inactivates several coagulation factors, but particularly thrombin and Xa. The onset of effect is more rapid with high doses compared with low doses. 29
Heparin is often used early in the course of feline ATE to reduce extension of the existing thrombus, although its efficacy for this purpose has not been established.1,3,14,30 The risk of haemorrhage appears to be small, but the benefit is unknown.
Low molecular weight heparins
In prevention of human deep vein thrombosis, unfractionated heparin has been largely superseded by low molecular weight heparin (LMWH) because of its longer half-life and more predictable dose response. This allows dosing according to body weight in human patients, without a need to monitor the coagulation effects. In cats, the optimal dosing schedule for the LMWHs enoxaparin and dalteparin is not known. In a study comparing enoxaparin, dalteparin and unfractionated heparin in healthy cats, only unfractionated heparin showed adequate anti-Xa activity. 30 Anti-Xa activity does not always correlate with clinical antithrombotic effects, however. 31 Use of dalteparin has been reported in an uncontrolled retrospective study of cats that included 43 cats with cardiomyopathy. 32
Aspirin
Aspirin is well tolerated, and is associated with a 20% reduction in stroke in high-risk human patients. 33 Aspirin irreversibly inhibits platelet cyclo-oxygenase (COX)-1, a key step in converting arachidonic acid (AA) to TXA2. This inhibition lasts for the lifetime of the platelet, and is achieved at low doses. Aspirin also inhibits endothelial COX-1 at higher doses, which converts AA to (antithrombotic) prostacyclin. The association in human patients between aspirin dose and antithrombotic effect is therefore not linear. Treatment failure with aspirin may variously reflect poor patient compliance; platelet activation via ADP or collagen; increased production of new platelets unaffected by aspirin; genetic polymorphisms; or the presence of TXA2-independent mechanisms of thrombogenesis.
We know that pre-treating cats with very high doses of aspirin (650 mg) can prevent vasoconstriction of the collateral circulation that accompanies thromboembolism. 34 We also know that cats at risk can still develop ATE despite aspirin therapy, and we do not even know whether aspirin reduces ATE incidence in cats, as no placebo-controlled study has ever been undertaken. The incidence of ATE with low-dose aspirin (5 mg/cat q72h) was not different from high-dose aspirin (40 mg/cat q72h) in Smith et al’s study, although the incidence of side effects was lower. 1
In the absence of reliable outcome data, the only other means of assessing aspirin in cats is by resorting to measuring platelet function, which is often unsatisfactory in cats. A recent platelet aggregation study in healthy cats showed that aspirin had no effect on whole blood aggregation using ADP and collagen agonists, although plasma concentrations of TXB2 (a stable metabolite of TXA2) were decreased. 35 An older study had shown a similar lack of effect of aspirin on whole blood platelet aggregation in healthy cats with ADP and collagen, but marked inhibition of aggregation when AA was used as the agonist (AA is the more relevant agonist for testing the efficacy of aspirin). 36 Both studies suggest that aspirin is effective at inhibiting platelet COX-1 in healthy treated cats, but other TXA2-independent pathways may still result in platelet aggregation. Aspirin also appears to inhibit serum TXB2 production in cats with myocardial disease. 37
Clopidogrel
Clopidogrel is an irreversible ADP antagonist that further reduces the risk of vascular events in human patients when added to aspirin, at the cost of a slightly increased risk of haemorrhage. 38 ADP is a weak platelet agonist on its own, but is important in potentiating platelet activation in response to collagen, vWF and thrombin. 16 ADP also amplifies the effect of TXA2. Studies in normal cats showed that clopidogrel decreased platelet aggregation in response to ADP and collagen, and decreased platelet serotonin release. 39 Clopidogrel appears to be well tolerated in cats. 39
Approach to the cat with acute ATE
Initial presentation
Most cats presented with ATE in extreme distress and pain are euthanased. Unless adequate doses of suitable opiates can be provided (ie, methadone, oxymorphone or fentanyl), then euthanasia should certainly be discussed frankly with the owner. With partial ATE (ie, only one limb affected or some motor function persisting), the pain is less severe and the prognosis much better. This should also be discussed frankly with the owner.
Physical examination provides useful prognostic information: cats with a rectal temperature <37°C are less likely to be survivors. Confirmed CHF may also worsen the prognosis. The first 24 h are the most painful and distressing for the cat, so if euthanasia is the likely outcome, it is better to euthanase as soon as possible after the onset of signs.
First 60 minutes
It may be necessary to manage pain even before a decision is made to treat (see box at top of page 467). Cats presenting with the severe pain associated with bilateral pelvic limb ATE must be treated with potent analgesics titrated to effect, such as intravenous methadone or a constant rate infusion of fentanyl (fentanyl patches will take too long to take effect). For cats with partial ATE, buprenorphine may be adequate.
The next priority is to determine whether CHF is present. Oxygen should be administered to cats with respiratory distress. Inspiratory crackles on auscultation are highly suggestive of pulmonary oedema. Significant pleural effusion is less likely to develop acutely with ATE, although this can also be identified using physical examination when breath sounds are absent ventrally. For tachypnoeic cats with normal breath sounds, thoracic radiographs should be obtained. If pulmonary oedema is suspected, then intravenous furosemide should be administered at 1–2 mg/kg. Furosemide should be repeated to effect, at hourly intervals or more frequently depending on the severity of respiratory distress.
Once pain is addressed and CHF signs controlled, antithrombotic treatment can be started. The goal of antithrombotic therapy is to prevent extension of existing thrombus, and to prevent new thrombus formation, not to lyse existing thrombus. The value of unfractionated heparin or LMWHs for this use has not been established in cats. An alternative approach is to use oral agents such as aspirin or clopidogrel from the start, and not include heparin at all. The author uses both aspirin and clopidogrel together as early as possible.

First 24 hours
Further investigations/monitoring
Once pain and CHF have been addressed and antithrombotic treatment started, further investigations may be undertaken (see box below). Apart from monitoring vital signs, general demeanour and mobility, renal function and electrolytes should be monitored. Echocardiography will help determine whether the underlying cause of ATE is cardiac disease.

Management
Intravenous fluid therapy may be needed in azotaemic cats, but should be avoided where possible in cats with CHF. As there is no point in giving diuretics and intravenous fluids concurrently, the first approach with azotaemic cats with CHF should be to reduce diuretic dose and try other approaches to improve cardiac function. Although not authorised for use in cats, pimobendan (1.25 mg per cat q12h) may be helpful for cats with systolic dysfunction, and can be tried as long as there is no murmur present. Nursing care is extremely important to ensure that cats are kept comfortable and to watch for early warning signs of complications. Cats should be kept warm to promote circulation, with warm ambient temperatures preferred to direct heating pads. Physiotherapy may be helpful. Passive manipulation may help to prevent muscle contracture, although no studies have evaluated the effects.
24–48 hours post-ATE
Further investigations/monitoring
Improvement is generally easy to identify. Pain usually decreases over the first 24–36 h. An improvement in warmth, pulse quality and motor function of the affected limb(s) is an indication that limb perfusion is improving. Pulse strength will often improve within 4–5 days, reflecting improved circulation, even without specific thrombolytic measures. Where there is evidence of severe ischaemic nerve damage, this may take weeks to improve. 8
Identifying the warning signs of adverse effects or complications is much more challenging. The main complications are CHF, reperfusion injury, azotaemia and the local consequences of ischaemic tissue necrosis. As pulses return, reperfusion of ischaemic tissue can cause sudden life-threatening fluctuations in serum potassium concentrations and reactive oxygen species, as well as acid–base balance. Cats that are apparently making good progress can suddenly and dramatically deteriorate. This makes monitoring for such changes very difficult. Even with serum biochemistry testing q8h, critical electrolyte changes may develop between blood sampling periods. Fortunately, cats with partial ATE appear to be less vulnerable to sudden reperfusion syndrome.
Management
Therapy with aspirin and clopidogrel is continued. Analgesia is generally continued, but most cats will be more comfortable after 24 h. Pelvic limbs may still be hard, cold and paretic, and may benefit from gentle rewarming and passive manipulation.
>48 hours post-ATE
Affected cats will probably be happier and more comfortable at home after the initial 72 h post-ATE. If there is still any discomfort, oral buprenorphine can be administered by the owner. The owner should be warned that acute deterioration is still possible. The aspirin and clopidogrel can be continued for longer term prophylaxis. Cats with affected pelvic limbs tend to move with the distal limbs extended behind them; there is thus a risk of excoriation of the dorsal aspect of the limbs, which may require dressings to protect them. The owner should be instructed on how to administer physiotherapy at home, and how to identify early signs of distal limb necrosis. This occurs in a minority of patients, and is usually restricted to cats with very severe ischaemic damage.
Cats should be rechecked every 3–4 days for the first 2 weeks, initially to reassess limb function and the need for continued analgesia, presence of pulses, control of CHF signs, and renal function. In the second week, cats should be monitored to ensure there are no necrotic changes in the skin or digits secondary to ischaemic damage.
Prevention
Prevention of ATE is clearly preferable to attempting to treat it. The lack of controlled, prospective studies in cats makes it difficult to choose therapies on anything other than an empirical basis. A large multi-centre study is currently under way evaluating the relative risk of ATE recurrence in cats treated with aspirin versus clopidogrel. Dual antiplatelet therapy with aspirin and clopidogrel has become a standard antiplatelet combination in human patients, and is the preferred choice for therapy of ATE in cats in the author’s clinic, but has not been studied in a controlled, prospective manner. Warfarin and the LMWHs are associated with too many pharmacokinetic problems to be currently recommended in cats. Pimobendan is occasionally used off-label in cats with advanced cardiomyopathy, and may have some effect in reducing platelet aggregation. 40
Although there are a number of newer antithrombotic treatments available for thromboprophylaxis in human patients at high risk of ATE, there is no consensus as to whether these agents are superior to warfarin.41–43 Some of the newer alternatives to warfarin may ultimately prove useful in cats.
Sadly, even if an effective preventive therapy were to be identified, ATE will still continue to be a major problem until we can improve our ability to identify cats at risk.
Footnotes
Key Points
Case notes
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The author does not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
