Abstract
Dietary therapy plays an important role in the management of most gastrointestinal disorders. This study was designed to test the efficacy of a new therapeutic diet for cats with diarrhea, compared to the top selling brand. Sixteen adult cats with chronic diarrhea were grouped and assigned to diet X (Hill’s Prescription Diet i/d Feline) or diet Y (Purina Veterinary Diets EN Gastroenteric Feline Formula). Following baseline evaluations, cats were fed their assigned test diet for 4 weeks. Fecal scores (FS; 7 = very watery; 1 = extremely dry and firm) were recorded daily during the last week on each diet. Each cat was then switched to the alternate test diet and the procedure was repeated. Fifteen cats completed the study. Both therapeutic diets resulted in a significant improvement in average FS and diet Y also resulted in significantly better results compared with diet X. Average FS improved at least one unit in 40% of the cats while fed diet X and in 67% of the cats while fed diet Y, resulting in normal stools (average FS ≤ 3) in 13.3% of cats fed diet X and 46.7% of cats fed diet Y. This study confirms the value of dietary change in the management of chronic diarrhea in cats.
Introduction
Dietary therapy plays an important role in the management of most gastrointestinal (GI) disorders.1,2 The appropriate dietary modifications are dependent upon the type and expression of the disease, and on which part of the GI tract, or which associated organs, are affected. The presence of food in the GI tract provides a variety of trophic signals, including direct provision of nutrients to the cells of the GI tract, increased mesenteric blood flow and stimulation of the release of digestive enzymes and hormones — all of which can influence GI cell proliferation and differentiation. 2 In addition, dietary factors can induce immunologic and inflammatory responses, osmotic diarrhea, altered GI motility and alterations of GI microflora. 2 Dietary characteristics, such as the nutrient content or the digestibility of the diet, can influence the above effects.
Low fat diets have long been recommended for both acute and chronic diarrhea in dogs and cats.3,4 Recent research, however, suggests that dietary fat is not an issue in cats with diarrhea. 5 In that double-blinded clinical trial, both low fat (24% of calories) and high fat (45% of calories), highly digestible diets resulted in significant improvements in diarrhea: 75% of the cats improved, but dietary fat content did not affect the outcome. 5
Highly digestible, low fiber diets have been recommended for cats with GI diseases, including inflammatory bowel disease (IBD) not caused by food allergy.6,7 For both acute and chronic diarrhea, high dietary digestibility facilitates absorption in the proximal small bowel and reduces the antigenicity of bowel contents. 8 This is proposed to lessen the potential for the development of osmotic diarrhea and exposure to potential dietary or microbiological allergens. However, a diet that is too digestible will be almost completely absorbed in the small intestine and may not provide sufficient nutrients to the distal GI tract. This can actually worsen diarrhea, whereas inclusion of indigestible dietary fibers can be beneficial for GI health.9–12
Total dietary fiber (TDF) includes both insoluble and soluble fibers. Insoluble fibers provide increased water-holding capacity, and can reduce osmotic and other diarrheas.10,12,13 Fermentable soluble fibers and prebiotics have proven helpful in the management of various types of diarrhea, so may also be beneficial in cats with GI disease.4,9,11,14,15 Dietary fibers and prebiotics can influence the composition of the GI microflora. 16 It is increasingly recognized that the GI microflora has a strong impact on the health of animals and that microbial populations can be altered in GI disease.17,18 Some of the specific changes that have been documented in cats with GI disease, such as IBD, include increases in Clostridium species and decreases in Bidifobacterium species, Lactobacillus species and Bacteriodes species — all of which may be influenced by dietary characteristics, including fermentable fiber.17,19–23 In addition, the short chain fatty acids derived from bacterial fermentation of such fibers facilitate sodium and water absorption, and provide an important energy source for colonocytes. 24 The short chain fatty acid butyrate has been shown to enhance colon health in rats and humans with colitis, and thus may provide similar benefits for cats. 24 However, in our experience (Laflamme, unpublished) and as per published data, excess fermentable fiber can cause, or worsen, loose stools; thus, an appropriate amount and balance of fibers is important. 25
Carbohydrate digestion may decrease with intestinal disease. The disaccharidases that complete the digestion of carbohydrates are located in the small intestinal brush border, which may be damaged in GI disease. Increased carbohydrate fermentation, indicative of carbohydrate malabsorption, has been confirmed in cats with IBD. 26 Carbohydrate malabsorption may occur in IBD if inflammation inhibits the production of digestive enzymes or if inflammatory infiltrates inhibit nutrient absorption. 26 Carbohydrate malassimilation secondary to exocrine pancreatic insufficiency and other conditions can result in osmotic diarrhea, intestinal gaseousness, bacterial overgrowth and other adverse effects. 8 For these reasons, restriction of dietary carbohydrates may be beneficial for cats with GI disease.27,28 Carbohydrates that are included in diets for the management of GI disease should be highly digestible. 8
For cats with GI inflammation or IBD, diets with increased omega-3 fatty acids or reduced omega-6:omega-3 fatty acid ratios are recommended.8,26,27 Omega-6 polyunsaturated fatty acids, especially arachidonic acid, are precursors to inflammatory prostaglandins and leukotrienes. The omega-6 precursors can be replaced by dietary omega-3 fatty acids. The eicosanoids produced from omega-3 fatty acids are less inflammatory, and thus help to reduce excessive inflammation.29,30
A canned diet designed to incorporate key dietary characteristics recognized to be of value in the management of feline diarrhea was developed. The objective of the study reported here was to evaluate the clinical efficacy of this new canned diet in a controlled, crossover trial in cats with long-standing chronic diarrhea. Our hypothesis was that this new diet would result in a clinical improvement in cats with chronic diarrhea that would at least match that of the best-selling diet marketed for this purpose, which served as the control diet.
Materials and methods
Animals
Adult domestic shorthair cats of any age with chronic diarrhea were identified among cats at the Nestlé Purina PetCare Center in St Joseph, MO, USA. Cats were considered for the study if they had experienced chronic continuous or intermittent diarrhea for at least three months prior to entry into the study. Diarrhea was defined as a fecal score of 6 or 7 using a seven-point scoring system (Figure 1) where: 1 = extremely dry and firm; 2–3 = normal stools; and 7 = very watery. To be considered for inclusion into the study, cats with intermittent diarrhea had to have diarrhea at least 3 days per week for at least 3 months. Cats may have previously been treated for diarrhea with parasiticides (fenbendazole, metronidazole or ronidazole as indicated by positive fecal findings), cobalamin injections, intravenous fluids, antibiotics or corticosteroids to control their diarrhea. However, cats were excluded from the study if they had received any treatment for diarrhea in the 6 weeks prior to initiation of this study. Cats were to be excluded from the study if they had evidence of current infection with intestinal parasites. In addition, cats were to be excluded from the study if they had evidence of any infectious disease or if they had evidence of a systemic disease that may cause diarrhea including, but not limited to, renal failure, hyperthyroidism, hepatic disease or malignancy of any type. Cats were also to be excluded if their clinical condition was such that they required immediate medical care to control the diarrhea or its effects, such as those with hypoproteinemia or with recent weight loss in excess of 10% of their body weight or chronic, undesired weight loss in excess of 25% of any previous adult body weight.

Fecal scoring system for feline and canine feces. Per this system, a score of 1 is too hard, scores of 2–3 are preferred, and scores above 4 are excessively watery and may be considered diarrhea
Study design
During the study, cats were individually housed in comfortable facilities. Once cats were housed, the units were arbitrarily divided into two groups. In order to eliminate any differences in effects that might be influenced by the cats’ diets during the baseline evaluation, all cats entered into the study were scheduled to be fed the same canned maintenance diet (Fancy Feast Savory Salmon Feast Cat food; Nestlé Purina PetCare) during a 2-week baseline period. This period was considered sufficient as prior studies had documented that if cats are going to respond to a dietary change, they do so within 1 to 2 weeks.5,28,31 During the second week, fecal scores were recorded daily. Fecal scores throughout the study were assigned by one of three technicians trained in fecal scoring, with each stool being scored separately. Each group of cats was then fed either diet X (Hill’s Prescription Diet i/d Feline; Hill’s Pet Nutrition) or diet Y (Purina Veterinary Diets EN Gastroenteric Feline Formula; Nestlé Purina PetCare) as their sole diet for 1 month (period 1), after which they were switched to the alternate diet for an additional month (period 2). Diet changes at each period were done abruptly. Cats were allowed 3 weeks to adapt and respond to each diet before data were collected during the final week of each period. Blood (12 ml) samples were taken on the last day of each period following an overnight fast. Freshly voided fecal samples were collected daily during the last 3 days of each period. Fecal scores were recorded daily during the last week on each diet.
During the course of the study, cats were to receive no medications for diarrhea or GI disease. Cats had access to their assigned diet for at least 15 h daily and water at all times, unless fasted overnight prior to blood collection. The study protocol was approved by the Nestlé Purina Institutional Animal Care and Use Committee and complied with the Nestlé Purina policy on animal welfare.
Diets
All diets were canned products. The new diet (diet Y) was evaluated in comparison with the best-selling brand of veterinary-prescribed therapeutic diets (diet X) for cats with GI disease. The calculated metabolizable energy of all three diets was 4.6 kcal ME/g dry matter (1.09–1.14 kcal ME/g as fed). Protein and carbohydrate content, as well as dietary fiber and fatty acids differed among the diets (Table 1). Cats were individually fed once daily in sufficient quantities to meet their daily energy requirements.
Nutritional analysis of diets used in study

All diets contained 4.6 kcal metabolizable energy/g dry matter
Baseline = Fancy Feast Savory Salmon Feast Cat food, Nestlé Purina PetCare
Diet X = Hill’s Prescription Diet Feline i/d, Hill’s Pet Nutrition, diet Y = Purina Veterinary Diets EN Enteral Management Feline Formula, Nestlé Purina PetCare, NFE = nitrogen-free extract, determined as 100% - (% water + % protein + % fat + % ash + % crude fiber), n3 = omega-3, n6 = omega-6, TDF = total dietary fiber
Clinical observations and blood parameters
Evaluations completed at baseline and the end of each of the two treatment periods included complete physical examinations with body weight and body condition score (BCS), serum biochemical profile, thyroid profiles, hematology, serum cobalamin (B12), pancreatic lipase immunoreactivity (fPLI) and serum trypsin-like immunoreactivity (TLI). Fecal samples were analyzed for intestinal parasites using direct fecal smears, zinc sulfate fecal floats and Giardia species antigen tests (SNAP Giardia test; Idexx Laboratories). During the last 3 days on each diet, freshly voided fecal samples were collected, mixed with 10% w/w glycerol, flushed with CO2 and frozen at -80°C until analysis. Upon completion of the study, fecal microflora were quantified using real-time polymerase chain reaction (Roche LightCycler System; Idaho Technology) using oligonucleotide probes targeting Bifidobacterium species, Clostridium species and Lactobacillus species (TIB Molbiol, LLC). This assay, previously validated in our laboratory and contrasted against fluorescence in situ hybridization, as used by others,20,32 allows quantification of specific bacterial groups using frozen fecal samples.
Data analysis
Mean fecal scores (FSm) for each cat were determined by averaging all of that cat’s scores recorded during the last 7 days of each period. Repeated measure analysis of variance (Proc Mixed in SAS: Sigma Plot 11.0, Systat Software, USA) with two diets and two groups (cats grouped according to the order they received the test diets) was used to determine dietary effects. Linear regression analysis was used to examine the relationship between B12 concentrations and FSm, using SigmaPlot 11.0 (Systat Software). Data are presented as least square means ± standard error of the mean, unless otherwise indicated. Probability values of <0.05 were considered significant.
Results
Sixteen cats were identified and recruited into the study. With one exception, all cats had been experiencing diarrhea for more than 1 year. Two cats were removed during the baseline period when they refused to consume sufficient amounts of the canned diet. These cats were replaced by two additional recruits. The two replacement cats were fed the maintenance diet for only 1 week versus the 2-week baseline period for other cats. One additional cat was removed for unrelated medical reasons, leaving 15 cats that completed all periods of the study. Only data from these 15 cats [13 neutered males, two spayed females; mean age 10 years (range 6–17 years)] are included in the results. One of these cats was treated throughout the study for keratoconjunctivitis with ophthalmic cyclosporine (Optimmune Ophthalmic Ointment; Intervet/Schering-Plough Animal Health) and gentamicin sulfate ophthalmic solution. This cat also received subcutaneous lactated Ringer’s solution on two occasions (once during period 1 and once during period 2) for dehydration. During both periods, the fluids were administered at least 1 week before technicians began to record fecal scores. No other medications of any type were given during the study.
Initial body weights and body condition scores (nine-point system) averaged 4.5 ± 0.3 kg and 4.9 ± 0.3, respectively, and did not change (P >0.70) during the study. Metabolizable energy intake was significantly higher (P = 0.02) in cats when fed diet X compared with diet Y, but neither differed from baseline (182.27 ± 11.15, 196.32 ± 12.34 and 173.00 ± 11.82 kcal/day, for baseline, diet X and diet Y, respectively).
At baseline, and throughout the study, fecal evaluations for intestinal parasites were negative. Mean FSm improved after both diet X and diet Y, with significantly greater improvement after diet Y (P <0.01) (Figure 2). The benefit was not affected by the order in which the diets were fed (P = 0.65). However, cats switched from diet X to diet Y continued to improve (P = 0.022) while cats switched from diet Y to diet X showed deterioration in fecal quality (Figure 3). Individual FSm improved at least one unit in 40% of the cats while fed diet X and in 67% of the cats while fed diet Y, resulting in normal stools (FSm ≤3) in 13.3% of cats fed diet X and 46.7% of cats fed diet Y.
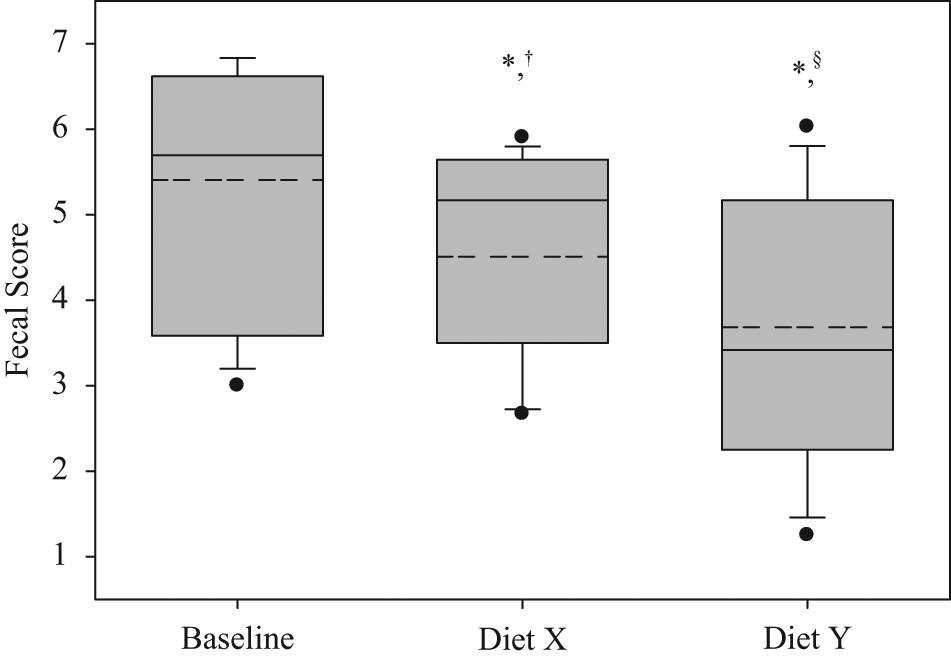
Box plot of average fecal scores in cats fed different diets. The dashed horizontal line represents the mean value, while the median is shown in a solid horizontal line. The box represents the central quartiles (25–75%) distribution of scores, and the upper and lower whiskers represent the 5–95% distribution. Additional data points are shown in filled circles. Data for each cat was the average of fecal scores recorded for each defecation during the recording period as defined in the text. *Differs from baseline, P <0.01. †§Different symbols differ, P <0.01

Average fecal scores (mean ± SEM) of cats fed different therapeutic diets by periods in a crossover trial. (●) Cats switched from the baseline diet to diet X for period 1, then to diet Y for period 2; (★) cats switched from the baseline diet to diet Y for period 1 then to diet X for period 2. Average fecal scores were not significantly different (P >0.10) between groups at baseline. A downward slope indicates improvement in fecal quality. The order of feeding effect was not significant (P >0.65). Cats switched from diet X to diet Y continued to improve (P = 0.022) while cats switched from diet Y to diet X showed deterioration in fecal quality
With the exception of TLI, fPLI and cobalamin, there were no patterns for abnormal blood values among cats with chronic diarrhea, but dietary effects were noted (Table 2). Relative to when cats were fed diet Y, those fed diet X had lower serum proteins, urea nitrogen, calcium, phosphorus, magnesium, aspartate transaminase and cholesterol, but higher serum glucose (P <0.05).
Serum biochemistry values showing abnormalities or significant differences among cats with chronic diarrhea fed different diets
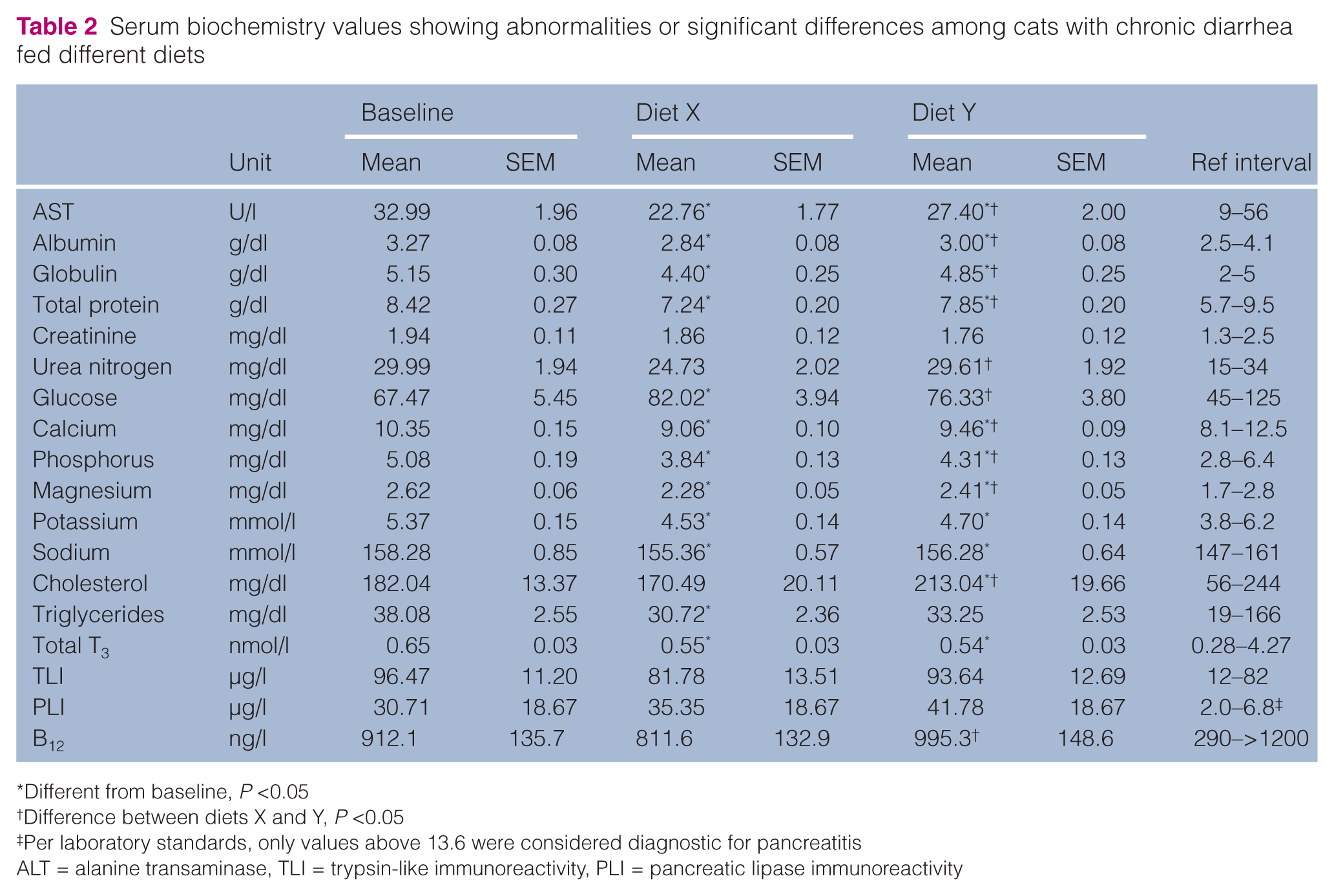
Different from baseline, P <0.05
Difference between diets X and Y, P <0.05
Per laboratory standards, only values above 13.6 were considered diagnostic for pancreatitis
ALT = alanine transaminase, TLI = trypsin-like immunoreactivity, PLI = pancreatic lipase immunoreactivity
The mean TLI and fPLI exceeded the upper limit of the reference interval for cats on this study at all time points, with all cats exceeding the upper limit for fPLI during at least one sampling period. However, only four cats exceeded the threshold (13.9 µg/l) established by the Gastrointestinal Laboratory at Texas A&M University as diagnostic for pancreatitis (Steiner JM, personal communications). Among these four, two cats vomited or regurgitated several times per week but showed no evidence of discomfort or other clinical signs of pancreatic disease. Additional procedures to diagnose pancreatitis were not performed. Three of the four cats developed normal stools while fed diet Y (Mean FSm: 5.0 on control; 4.5 on diet X and 2.75 on diet Y). No cats had TLI values below the reference interval which would have indicated exocrine pancreatic insufficiency.
Mean B12 was within normal limits and did not differ significantly with time, but did differ by diet (Table 2). Four cats (27%) had low (<290 ng/l) B12 concentrations at some point during the study: two cats throughout the study and two others developed low B12 during the study. Among all cats or only among cats with low B12, there was no detectable association between B12 concentrations and FSm (r2 = 0.00; P > 0.50). However, among the cats with low B12, mean FSm improved significantly only while cats were fed diet Y (FSm = 4.9 ± 0.8; 4.7 ± 0.6; 3.5 ± 0.8 for baseline, diet X and diet Y, respectively; diet Y differed from diet X and from baseline, P <0.05) .
No dietary effects were observed on fecal microflora, with the exception of a diet by period interaction where fecal Clostridium species were significantly lower in cats fed diet Y during the first period (Table 3).
Fecal microflora (log10 colony forming units/gram feces) from cats with chronic diarrhea fed different diets
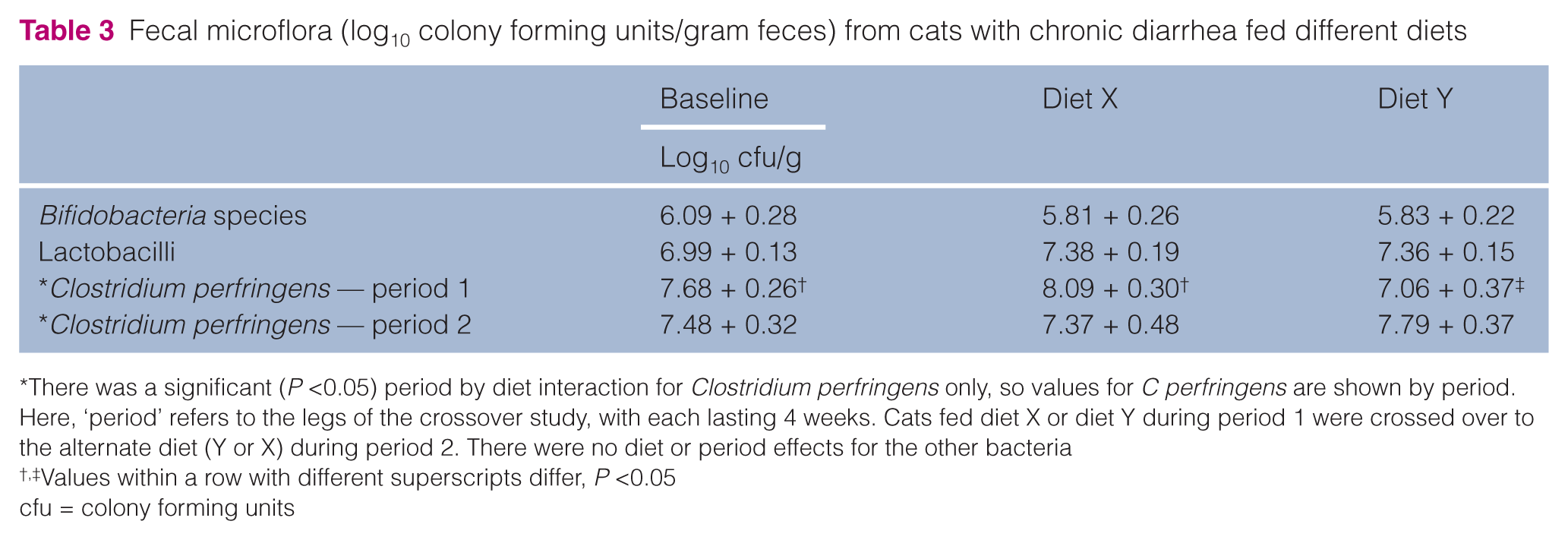
There was a significant (P <0.05) period by diet interaction for Clostridium perfringens only, so values for C perfringens are shown by period. Here, ‘period’ refers to the legs of the crossover study, with each lasting 4 weeks. Cats fed diet X or diet Y during period 1 were crossed over to the alternate diet (Y or X) during period 2. There were no diet or period effects for the other bacteria
Values within a row with different superscripts differ, P <0.05
cfu = colony forming units
Discussion
In this study involving cats with chronic diarrhea, stool quality improved when cats were fed therapeutic diets formulated for the management of cats with diarrhea. Given the long-standing nature of the condition in these cats, no spontaneous improvement would be expected. Therefore, it is assumed that any improvements observed were a result of the dietary change.
This study was not designed to identify or confirm individual factors beneficial in the management of feline diarrhea. The multiple differences in the diets studied precluded any such conclusions. Rather, this was an evaluation of a new diet designed to incorporate factors thought to be of value in the management of diarrhea in cats. Among the diets tested, cats responded best to diet Y, with more than three-fold greater proportion developing normal stools with diet change alone, confirming that this combination of factors can be beneficial for such patients. Diet Y resulted in improved stool quality even in cats with low B12 or laboratory evidence (elevated fPLI) suggestive of pancreatitis. It must be noted, however, that additional evaluations to confirm pancreatitis were not performed during the time of this study.
Diet Y was a high-protein, low-carbohydrate, moderate fat diet. Prior research demonstrated that low fat content does not appear to be an important factor in managing feline diarrhea. 5 Fat content varied only slightly among the diets tested in this study, although the specific fatty acid content did differ. At 1% of the diet dry matter, diet Y contained a higher amount of omega-3 fatty acids from fish than diet X, with an omega-6:omega-3 ratio of less than 5:1. Omega-3 fatty acids are recognized to provide a relative anti-inflammatory effect and are suggested to be beneficial in some types of GI disease.8,27,29,30 However, the maintenance diet fed at baseline contained even higher amounts of omega-3 fatty acids from fish, suggesting that this parameter alone may be insufficient to define an effective diet, at least when fed for a short time.
Vomiting and diarrhea are common signs associated with food allergy or intolerance, thus limited-antigen diets are often recommended for cats with GI signs.15,31 The diets tested in this study were not considered to be novel or limited in antigenicity. However, fecal scores improved for most cats fed diet Y with 47% of them developing normal stools. This is similar to the 49% reported by Guilford et al in cats fed novel protein diets. 31
Low carbohydrate diets may be beneficial in some cats with chronic diarrhea.3,22,28 diet Y contained less than half the carbohydrate content on an energy basis compared with diet X. Yet the maintenance diet was even lower in carbohydrates. While the maintenance diet was only fed for 1–2 weeks, this has been shown to be a sufficient time for diets to influence feline diarrhea.5,28,31 Thus, this suggests that, while low carbohydrate may be important in some cats, this parameter alone is insufficient to define an effective diet.
While crude fiber (CF) varied only slightly among the diets evaluated, TDF varied considerably (4.47%, 12.67% and 6.95% in baseline diet, diet X and diet Y, respectively). The CF and TDF assays detect different components of the indigestible carbohydrates and are not directly related: TDF includes both soluble and insoluble fibers, while the CF assay detects only part of the insoluble and none of the soluble fiber. 33 Although CF is a less useful analysis, pet food regulations require its use, so it is the measure found on the label of commercial pet foods.
Dietary fiber provides several benefits that promote GI health and may aid in the management of diarrhea.4,23 Insoluble fiber helps to normalize motility and reduces various types of diarrhea.12–14,34 Soluble fiber can serve as a prebiotic, enhancing the growth of beneficial bacteria such as Bifidobacteria species and Lactobacillus species, or reducing the growth of potentially pathogenic bacteria, such as Clostridium species, Salmonella species and Escherichia coli, and also can serve as a substrate for production of short chain fatty acids.9,16,18 In piglets, soluble fiber reduced bacterial-induced diarrhea while enhancing mucosal functions such as lactase activity and glutamine uptake. 9 In humans, soluble fiber significantly reduced diarrhea in patients fed enteral formulas. 11 Clinical symptoms from irritable bowel syndrome were significantly reduced in human patients consuming a soluble fiber supplement. 14
However, too much fiber can decrease diet digestibility and may have other, less desirable, effects.25,35 In dogs and cats, TDF in excess of 9% (about 2 g/kg body weight/day in dogs) to 10% (about 1.7 g/kg body weight/day in cats) reduced dietary digestibility.25,35 In addition, excess insoluble fiber contributes to increased stool volume, while excess soluble fiber can cause soft stools. 25 Past experience in our research facilities demonstrated that soluble fiber in excess of about 2% of the diet dry matter can cause loose stools in otherwise healthy cats and dogs. An optimum amount of fiber for cats with diarrhea appears to be provided by diets containing TDF at about 4–8% of the diet dry matter, with 15–35% of that amount coming from soluble fiber.
The lack of changes in fecal microflora measured in this study does not exclude the possibility that alterations in GI microflora occurred. There are hundreds of different organisms present in the GI tract, of which only relatively few are known, and these can differ between luminal, mucosally adherent and fecal populations.19,21,32 Prior research has confirmed alterations in intestinal microflora in cats with GI disease.19,21 However, further studies with more comprehensive methodologies are needed to determine if changes in GI microflora are associated with improvement in diarrhea in cats.
Hypocobalaminemia was detected at least once during this study in four (26.7%) cats with chronic diarrhea. This is similar to the 16.5% and 20% reported in two other studies, but considerably less than the 61% reported by Simpson et al in their studies of cats with gastrointestinal diseases.5,36,37 While an association between low B12 and non-responsive diarrhea has been reported, this association was not observed in the current study.5,37 On the contrary, the overall FSm of cats that had, or that developed, low B12 was slightly better than for the balance of the cats, and regression showed no association between FSm and B12. Likewise, in a previous study of feline diarrhea, the mean serum B12 concentration was actually greater in cats not responding to dietary change than in cats that improved, so the importance of low B12 is unclear. 28 Nevertheless, as low B12 can itself contribute to intestinal pathology, testing for, and correcting, low B12 may be important, especially in cats that do not respond to other forms of management for their gastrointestinal disease.
There were several weaknesses to this study. One weakness was that there was no definitive diagnosis for the cause of diarrhea in these cats. Also, the study was not truly blinded. Fecal scores throughout the study were assigned by one of three technicians trained in fecal scoring. This team of technicians also had responsibility for the care of these cats, including feeding, but also simultaneously cared for and collected data from cats on multiple other studies. Owing to the number of cats being cared for and the temporal separation between feeding and fecal scoring, technicians were unlikely to have an awareness of the specific dietary treatment of any individual cat during fecal scoring. Another potential weakness was the lack of a washout period between the two test diets so cats did not return to baseline fecal scores before beginning the second period. It was considered during planning the study that there would be no carry over effect as cats with diet-responsive diarrhea usually respond within 1–2 weeks and a 3-week adaptation time to both diets X and Y was allowed prior to recording fecal scores.28,31 The lack of washout did not appear to impact the results as the order in which the diets were fed did not have a significant effect on the results.
Conclusions
This study confirmed that a canned food formulated to contain high protein, low carbohydrate, moderate fiber with both soluble and insoluble fibers, and a source of long-chain omega-3 fatty acids was beneficial in the management of cats with chronic diarrhea. While cats fed both therapeutic diets showed an improvement over baseline in average fecal scores, the improvement was significantly greater when cats were fed diet Y. When cats with long-standing chronic diarrhea were fed diet Y, 47% of cats developed normal stools with dietary change alone.
Footnotes
Acknowledgements
The authors wish to thank the staff veterinarians and technicians at the Nestlé Purina Petcare Center for assistance with data collection and animal care.
Funding
This project was funded and conducted by Nestlé Purina PetCare, St Louis, MO 63164.
Conflict of interest
This project was funded and conducted by Nestlé Purina PetCare, St Louis, MO 63164. All authors are full-time employees of this company.
