Abstract
Borna disease virus (BDV) is a RNA-virus causing neurological disorders in a wide range of mammals. In cats, BDV infection may cause staggering disease. Presently, staggering disease is a tentative clinical diagnosis, only confirmed at necropsy. In this study, cats with staggering disease were investigated to study markers of BDV infection aiming for improvement of current diagnostics. Nineteen cats fulfilled the inclusion criteria based on neurological signs and pathological findings. In 17/19 cats, BDV infection markers (BDV-specific antibodies and/or BDV-RNA) were found, and antibodies in serum (13/16, 81%) were the most common marker. BDV-RNA was found in 11/19 cats (58%). In a reference population without neurological signs, 4/25 cats were seropositive (16%). The clinical history and neurological signs in combination with presence of BDV infection markers, where serology and rRT-PCR on blood can be helpful tools, improve the diagnostic accuracy in the living cat.
Introduction
A neurological disorder in cats known as staggering disease was first described in Sweden in the mid-1970s. 1 At that time the aetiology was not established, but a viral cause was suspected based on the histopathology, ie, non-suppurative meningoencephalomyelitis. Staggering disease was further characterised pathologically, showing that lymphohistiocytic or lymphoplasmacytic inflammation, accompanied by neuronal degeneration and neuronophagia, are predominantly found in the grey matter of the brain stem, basal nuclei and hippocampus. 2 Clinically, the major signs are gait disturbances, for example hind-leg ataxia, and behavioural changes.1,2 The clinical signs progress during the course of disease and in the final stage cats can develop pelvic limb paralysis. 2 Neurological signs are strikingly similar to those seen in horses with Borna disease (BD), and lesions in the same brain regions are found in Borna disease virus (BDV) infection of horses.1–3
Hence, initial studies to link BDV infection to staggering disease in cats were performed; BDV-specific antibodies were found in serum, 4 BDV-specific antigen was found in brain tissues 5 and a feline variant of BDV was isolated 6 from diseased cats. Cats experimentally infected with BDV developed clinical signs similar to staggering disease, histopathological lesions were similar, and BDV was detected by immunohistochemistry (IHC) and reverse transcription-polymerase chain reaction (RT-PCR). 7 Based on these studies, BDV, a neurotropic negative-sense RNA-virus, 3 is considered the causative agent of staggering disease. Other aetiological agents may also cause feline non-suppurative meningoencephalomyelitis,8,9 but with clinical signs and pathological lesions that seem to be more variable, 8 even for the same pathogen, than those present in staggering disease.1,2
For the clinician, staggering disease is a tentative diagnosis made by excluding other explanations for the clinical signs. The disease can be strengthened by necropsy and histopathological examination of the central nervous system (CNS), ie, the presence of lymphoplasmacytic or lymphohistiocytic inflammatory changes in the regions typically involved. Further confirmation of the presence of BDV antigen or RNA is not done routinely. 10 At present, there is no established gold standard for diagnosing BDV infection in the living animal. In horses, the most reliable ante mortem diagnosis is obtained by combining clinical signs, clinical history and epidemiology with detection of BDV proteins, BDV nucleic acids and/or BDV-specific antibodies, as well as presence of pathological changes in cerebrospinal fluid (CSF). 11
Several studies have revealed BDV-specific antibodies and/or BDV-RNA in cats from different parts of the world: Germany, Belgium, United Kingdom, Japan, Philippines and Indonesia, 12 Australia 13 and Finland. 14 However, these findings have not always been associated with neurological disease. In cats with unspecified neurological disease, the seroprevalence has varied between 0% and 67%, depending on the choice of method, size of the studied population, and geographical region.15–18 To which extent antibodies are present in a healthy reference population of cats is not fully known. The humoral immune response in cats with naturally occurring BDV infection has been considered weak or non-existing, whereas experimentally BDV-infected cats develop high antibody titres; 19 however, the value of serology as a diagnostic aid in cats has not been further evaluated. Conventional and real-time RT-PCR (rRT-PCR) assays have been developed, 20 but are not used in routine diagnostics.
Previous studies on staggering disease have focused on the initial characterisation of the aetiopathogenesis, clinical signs and epidemiology. Studies on potential diagnostic tools for making the clinical diagnosis in cats are lacking. In this study, we have thoroughly investigated 19 cats with staggering disease to study markers of BDV infection, aiming for an improvement of the ante mortem diagnostics for this disease.
Materials and methods
Animals
Cats referred to the University Animal Hospital [Swedish University of Agricultural Sciences (SLU), Uppsala, Sweden] were included in the study. Clinical inclusion criteria were presenting neurological signs consistent with staggering disease, ie, gait disturbances with or without behavioural changes.1,2 Clinical exclusion criteria were signs of chronic disease (bad fur quality and body condition score), distinctly asymmetric neurological signs, unilateral vestibular syndrome, and/or a history of epileptic seizures.
Cat owners with cats fulfilling the clinical inclusion criteria were asked to participate in the study and gave their informed, written consent. The owners also approved to necropsy of their cats. In all cats, euthanasia was performed because the neurological impairment was judged to decrease the quality of life. The study was approved by the local Animal Ethical Committee. Pathological inclusion criterion was presence of a non-suppurative inflammation of the CNS, consistent with staggering disease.1,2
Clinical examination and collection of samples
The cats were further examined including a standardised neurological examination, mostly performed by a board-certified neurologist (KHJ). In total, 21 cats were included in the study based on their neurological signs. The severity of neurological signs was graded into severe, moderate or mild, based on a combination of the degree of ataxia, how often the cat fell over spontaneously and the degree of postural deficits. Samples, as stated below, were taken at first presentation and/or at euthanasia.
Pathology
After euthanasia, gross and histopathological examinations were undertaken. Most of the examinations and all evaluations were performed by a board-certified pathologist (EK). The lesions were graded into severe, moderate or mild, according to the severity of inflammation. Tissue samples (olfactory epithelium, cerebral cortex, basal parts of the cerebrum and spinal cord) were fresh frozen and stored at -70°C for further virological studies. Two cats were excluded for not fulfilling the inclusion criterion; one was lost to follow-up and therefore not necropsied, and one had atypical histopathological lesions (senile lipofuscinosis).
Serology for differential diagnoses
To check for other possible reasons for the neurological signs, serology for some known feline and/or CNS pathogens were performed: antibodies for toxoplasma (ToxoScreen DA), feline immunodeficiency virus (FIV; ViraChek/FIV; Synbiotics ) and tick-borne encephalitis virus (TBEV: Immunozym FSME IgG all species; Progen), as well as antigen for feline leukaemia virus (FeLV: ViraChek/FeLV; Synbiotics), were analysed at the National Veterinary Institute (SVA, Uppsala, Sweden). Antibodies for feline coronavirus (FCoV) were also investigated. 21 Although feline infectious peritonitis (FIP) cannot be diagnosed by serology, most cats that develop FIP have high antibody titres.
Clinical pathology
For characterisation of the blood biochemistry of staggering disease and for differential diagnostics, serum, blood anti-coagulated with K3-ethylenediaminetetraacetic acid (EDTA), and CSF taken prior to and/or at euthanasia were analysed at SLU. Haemoglobin concentration and total white blood cell (WBC) counts were analysed using a Cell-Dyn 3500 analyser (Abbott Diagnostics), with leukocyte differential counts obtained automatically and used if they were considered correct after manual examination of cell morphology on blood smear by microscopy. A manual leukocyte differential count was performed if the automated differential count was not considered to be correct or if there was, for example, a left shift. Total protein in CSF and creatinine, alanine aminotransferase (ALT), alkaline phosphatase (ALP) and total protein concentration in serum were analysed using an auto-analyser Konelab 30 (Thermo Electron Corporation) and commercial reagents from Thermo. Total thyroxin (T4) concentration was analysed with Immulite 2000 (Siemens Healthcare Diagnostics) and reagents from Siemens. Leukocyte and erythrocyte counts in CSF were analysed by manual count in counting chamber (Bürker chamber). The samples for leukocyte count were mixed with Turks solution 1:2 for 20 min before counting. In most samples the type of leukocytes were also identified in the counting chamber and reported as mononuclear or polymorph nucleated leukocytes.
BDV serology
An indirect immunofluorescence assay (IFA) was used for detection of BDV-specific antibodies in serum and CSF. Briefly, Vero cells persistently infected with BDV (strain He/80) were seeded with non-infected Vero cells in a ratio of 1:3 on 10-welled glass slides. The slides were incubated at 37°C with 5% CO2 for 1–3 days before fixation with acetone for 10 min. Slides with non-infected Vero cells were used as negative controls. The slides were first blocked in 10% normal goat serum at 37°C for 30 min. Then, serum samples were incubated for 30 min at 37°C at a dilution of 1:20, and CSF samples at dilutions of 1:5 and 1:20. Finally, a fluorescein isothiocyanate (FITC)-conjugated goat anti-cat IgG antibody at dilution 1:64 (F4262; Sigma-Aldrich) was applied at 37°C for 30 min. The slides were analysed in a fluorescence microscope.
As a positive control, serum from a specific pathogen-free (SPF) cat, experimentally infected with BDV (strain V; cat 4), 7 was used. Serum from a SPF cat, intracerebrally inoculated with normal rabbit brain suspension, 7 was used as negative control. Two groups of cats without neurological signs were used to evaluate the seroprevalence in a reference population. The first group consisted of 51 cats, where serum had been taken after rabies vaccination for antibody titre control (serum bank of SVA). This group of cats had similar age distribution as the cats with staggering disease; however, pedigree cats were more common. Pedigree cats are more likely to be kept indoors and, for this reason, a second group of sera from 25 cats sent for analysis to the SLU were used. These cats were significantly older than those with staggering disease, but with a similar breed distribution. All cats were sampled at a veterinary clinic or animal hospital from the same regions as the cats included in the staggering disease study, ie, the counties of Stockholm, Uppsala and Västmanland, Sweden. Positive serum samples were further titrated in two-fold dilutions from 1:20 to 1:160.
Detection of BDV RNA
A previously described rRT-PCR assay was used for the detection of the phosphoprotein (BDV P) gene 20 with modified primers to increase the sensitivity.22,23 Total RNA was extracted using Qiagen Lipid Tissue kit, according to the manufacturers’ instructions (Qiagen), from the following samples: serum, blood anti-coagulated with K3-EDTA, urine, conjunctival, nasal, oral and anal swabs, as well as tissue samples taken at necropsy (olfactory epithelium, cerebral cortex, basal parts of the cerebrum and spinal cord). The extracted RNA was both processed into cDNA using gene-specific primers 24 and used in one-step rRT-PCR (Quantitect Probe RT-PCR master mix; Qiagen). For whole blood, urine and swabs, 8 µl of total RNA was used for cDNA synthesis, and for tissues 500 ng of total RNA was used. Two microlitres of cDNA was used in the following real-time PCR assay (Quantitect Probe PCR master mix; Qiagen). All analyses were performed in duplicate using Rotorgene 3000 and Rotor-Gene Real-Time Analysis Software 6.1 build 93 (Corbett Research). When at least one of the duplicates had an exponential amplification, that sample material was considered positive. Primers and probe were synthesised at DNA technology.
Statistical analysis
The different parameters (time from onset of signs to neurological examination, age at onset of signs, time from onset of signs to euthanasia, WBC, number of neutrophils and lymphocytes in blood, and number of erythrocytes, leukocytes, poly- and mononuclear cells, as well as protein content of CSF) were compared between cats grouped according to the degree of neurological signs or pathological lesions (mild versus moderate and severe, unless otherwise stated). Normality tests for each parameter were performed and, subsequently, either Student’s two-tailed t-test or the Mann-Whitney test was used.
Results
History and clinical signs
Sixteen cats were domestic shorthairs (DSH), one birman, one a DSH/Balinese and one Bengal/Ocicat. About half part of the cats (10/19, 52.6%) were neutered males, three were intact males, four were neutered females and two were intact females. One of the queens was pregnant. The median age was 4 years 1 month (range: around 1 year to 8 years 10 months), and the median time from onset of signs to first neurological examination was 15.5 days (range: 1 day to > 4 years). The most common signs were absent or decreased postural reactions and menace response, varying degrees of general proprioceptive ataxia, abnormal behaviour at nociception test and increased vocalisation (Table 1; Supplementary videos 1 and 2). All cats had outdoor access, either in the countryside or in an urban environment. The shortest noted time from allowance to be outdoors to onset of signs was 5 months (n = 2). The onset of signs most commonly occurred from December to May (n = 14, 74%), compared with five cases from June to November.
The major clinical signs of the studied cats are shown in percentage and as number of observations (n). N is the number of observed cats for that particular clinical sign. It was not possible to evaluate or judge all cats for some signs. To evaluate the ability to track objects visually (*), a cotton wool ball test was used

In seven cats, neurological signs were severe, four cats had moderate and eight cats had mild neurological signs (Table 2; Supplementary videos 1 and 2). There was no difference in time from onset of signs to first neurological examination or in age at onset of signs between the three groups (Table 3). Moreover, there was no difference in time from onset of signs to euthanasia, used as a measure for survival as all cats were euthanased (Figure 1).
Information on studied cats. Based on the neurological signs, the cats were divided into three groups: severe, moderate and mild neurological signs. Similarly, the pathological lesions were graded into severe, moderate and mild
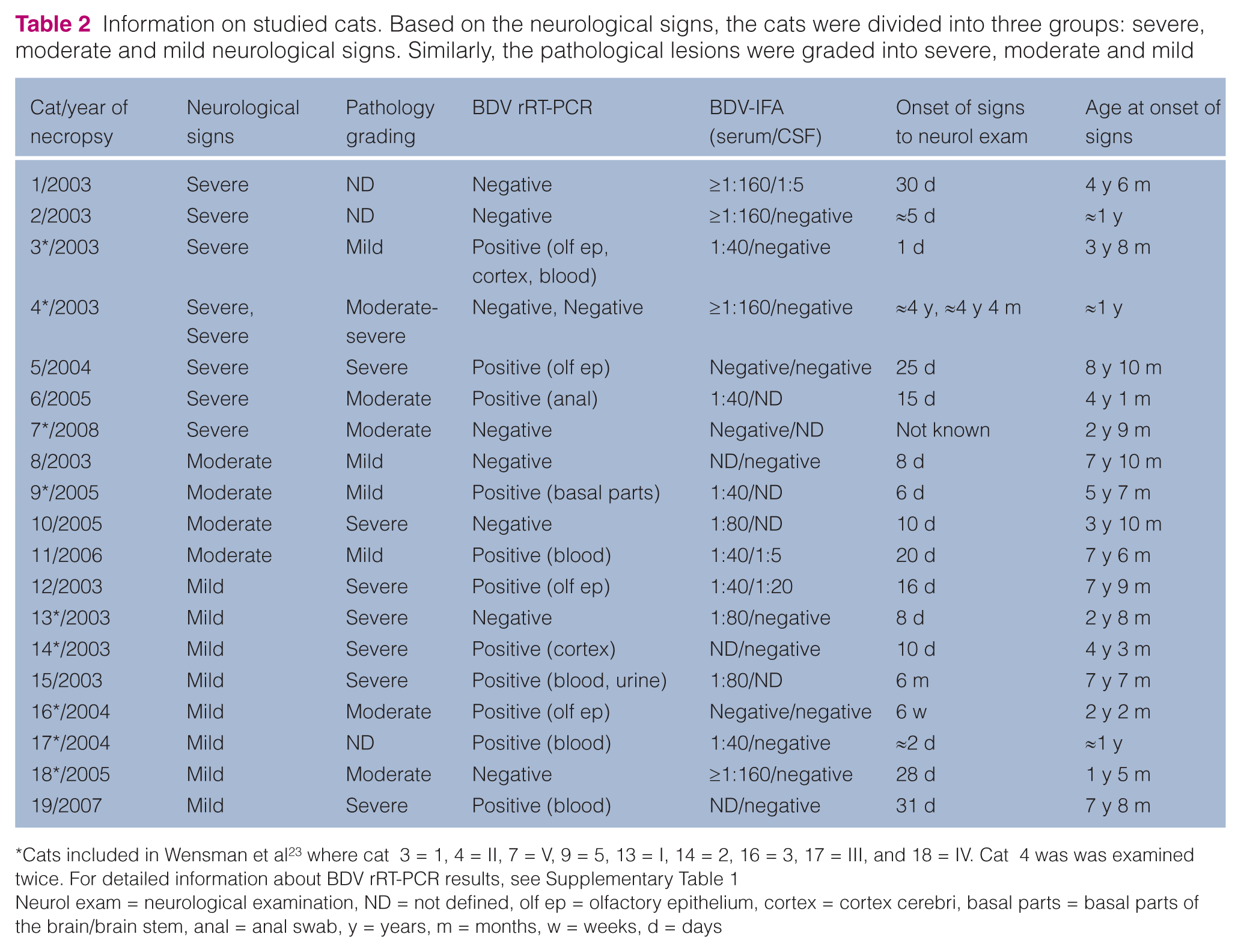
Cats included in Wensman et al 23 where cat 3 = 1, 4 = II, 7 = V, 9 = 5, 13 = I, 14 = 2, 16 = 3, 17 = III, and 18 = IV. Cat 4 was was examined twice. For detailed information about BDV rRT-PCR results, see Supplementary Table 1
Neurol exam = neurological examination, ND = not defined, olf ep = olfactory epithelium, cortex = cortex cerebri, basal parts = basal parts of the brain/brain stem, anal = anal swab, y = years, m = months, w = weeks, d = days
Comprehensive data based on neurological and pathological grading


A survival graph based on neurological signs (mild versus moderate and severe grouped together). The time from onset of signs to euthanasia is used as the measure of survival. No significant changes in survival between the two groups are seen. One of the cats in the moderate–severe group had a survival time of 1580 days
Pathology
All cats included had characteristic lesions consistent with staggering disease, ie, non-suppurative lymphohistiocytic or lymphoplasmacytic inflammation of the regions of the CNS typically involved (olfactory bulb, cerebral cortex, hippocampus, brainstem, and/or basal nuclei). Sixteen of the cats (84%) had a non-suppurative meningoencephalomyelitis, whereas two had non-suppurative encephalomyelitis and one had non-suppurative meningoencephalitis. In all cats, lymphocytes were the dominating inflammatory cell with additional plasma cells and/or macrophages. Neuronal degeneration and neuronophagia was sometimes observed. In addition, five cats (cats 10, 12–14 and 18) had a lymphohistiocytic or lymphoplasmacytic inflammation of an intra-abdominal ganglion, often close to the adrenal gland. Two of these cats (cats 10 and 14) also had a lymphocytic inflammatory reaction in the medulla of the adrenal gland, and cat 10 had non-suppurative neuritis of the femoral nerve.
Based on the degree of the inflammatory lesions, the cats were considered to have severe (n = 7), moderate–severe (n = 1), moderate (n = 4) or mild (n = 4) lesions (Table 2). In three cats, the lesions were not graded. The group with mild pathological lesions had a significantly shorter time from onset of signs to first neurological examination compared with the cats with moderate and severe lesions grouped together (Table 3; P ≤0.05). There was no difference in age at onset of signs depending on severity of pathological lesions. No cat had histopathological changes consistent with any other well-known feline inflammatory disorder within the CNS, such as FIP or toxoplasmosis.
Serology for differential diagnoses
None of the investigated cats had antibodies towards TBEV (n = 16) or FIV (n = 17). Five of the cats (5/17, 29.4%) had low titres of FCoV-antibodies (≤1:160); all other cats had no detectable titres. The overall seroprevalence of toxoplasma antibodies was 52.9% (9/17). One cat had low titres (1:40), two had moderately high titres (1:6000) and six had high titres (1:18000-1:162000). One of 17 investigated cats (5.9%) was positive for FeLV antigen.
Clinical pathology
All cats had low or low normal neutrophil and lymphocyte counts in blood (Table 4). Nine (48%) had neutrophil counts <3.0 × 109/l, and eight (42%) had low lymphocyte counts (<1.3 × 109/l). The cats with mild-to-moderate neurological signs grouped together had significantly lower neutrophil counts compared with the seven cats with severe signs (P ≤0.05). Three cats had mildly increased ALT (72–130 IU/l). No significant changes were found for haemoglobin, creatinine, ALP, total protein, albumin or total T4 results.
Blood leukocyte count and cell count and protein concentration of CSF. For WBC, neutrophil and lymphocyte counts in blood, the mean value ± SD are shown. For CSF cell count and protein concentration, the median value and range are shown. The * indicates reference values
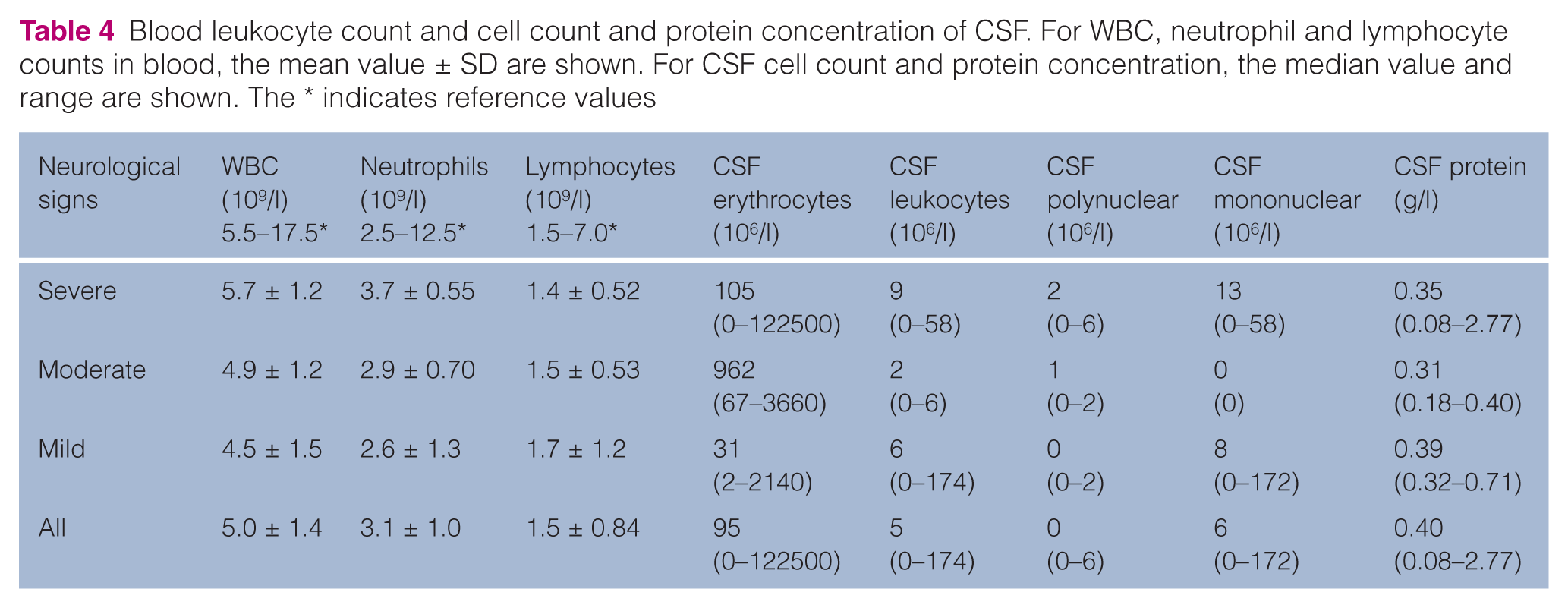
Increased erythrocyte numbers were found in the CSF samples from 14 of 18 sampled cats. One cat had xanthochromia (pink appearance of centrifuged CSF), indicating previous pathological bleeding. In three samples, the ratio between leukocytes and erythrocytes indicated that bleeding at the sampling procedure could be a cause of the increased erythrocyte count. Four (22%) of the 18 cats with CSF leukocyte count had increased leukocyte numbers in CSF, probably not caused by the sampling procedure, and were mainly mononuclear cells (Table 4). Three of these four cats had severe pathological changes and in one cat the pathological lesions were not classified. Four cats had CSF protein concentration above 0.5 g/l.
Detection of BDV infection markers
Thirteen of 16 tested cats (81%) had BDV-specific antibodies in serum and three cats had BDV-specific antibodies in CSF (Table 2). In the two groups of cats used as a reference population, 3.9% (2/51) of the rabies vaccinated and 16% (4/25) in the hospital group were positive for antibodies in serum.
In total, BDV-RNA was detected in 11/19 cats (58%). BDV-RNA was found in samples taken at the clinic (six cats), as well as in tissues taken at necropsy (six cats; see Table 2). In two cats, BDV-RNA was found in samples taken both at the clinic and at necropsy. The most common sample materials to be PCR-positive were blood (n = 5) and olfactory epithelium (n = 4; Supplementary Table 1).
Six of eight cats with mild neurological signs were PCR-positive compared with 5/11 cats with moderate or severe signs (Table 3). Time from onset of signs to neurological examination did not differ between PCR-positive and PCR-negative cats (median time 16 days and 10 days, respectively). However, the PCR-positive cats tended to be older than the PCR-negative cats (median age 5 years 7 months versus 2 years 9 months; P = 0.055). Age at onset of signs or time of onset to neurological examination did not differ between cats that were PCR-positive in a clinical sample compared with those that were positive in tissue samples taken at necropsy.
Discussion
At present, the tentative diagnosis of staggering disease is made on clinical signs (Table 1),1,2 clinical history and epidemiology consistent with the disease,1,2,25 and by ruling/other possible explanations. There is a lack of reliable methods for a clinical diagnosis of BDV infection and there is no established gold standard, even though the presence of clinical signs, viral nucleic acids, viral proteins and/or virus-specific antibodies, and pathological changes in accordance with BDV infection, together are considered to give a reliable diagnosis. 11 In this study, we report on 19 cases of staggering disease (based on presence of clinical signs and pathological changes consistent with the disease), which were thoroughly investigated, both clinically and pathologically, with the aim of finding and evaluating markers of BDV infection to improve the current diagnostics.
We show that serology can be a useful aid for the clinician, as a vast majority (81%) of cats with clinical signs of staggering disease were seropositive for BDV-specific antibodies. In comparison, the seroprevalence was 16% in the reference population with the highest risk of encountering BDV (the hospital group). This is similar to what is seen in horses in BDV-endemic regions in Germany when using the same serological assay. 11 Furthermore, three cats with staggering disease had BDV-specific antibodies in CSF. In horses, antibodies in CSF are only found in horses with BD. 11 Previous studies in cats with unspecified neurological signs have shown highly variable seroprevalence of BDV-specific antibodies, between 0% and 67%, depending on choice of method, size of study population and geographical region.15–18 Antibodies to BDV have also been found in healthy cats to a similar extent as in the current study (Turkey 16%; Japan 24%).15,18
In the present study, serology for possible pathogens was performed to exclude some other explanations for the clinical signs. For all pathogens studied, the seroprevalence was similar to that of the normal Swedish cat population.21,26,27 Although some of the cats had high titres of toxoplasma-specific antibodies, no parasites could be detected at necropsy, making toxoplasmosis an unlikely cause of the clinical signs. Other aetiological causes of feline non-suppurative meningoencephalitis have been considered,8,9 such as parvovirus, West Nile virus (WNV) and encephalomyocarditis virus (EMCV), although the clinical signs and pathological lesions seem to be more variable than those reported in staggering disease.1,2,8 For example, EMCV caused either pyogranulomatous (in two cats) or lymphohistiocytic (in two cats) inflammation, and one of the infected cats developed respiratory signs only. 8 Some of the potential aetiological causes, for example rabies virus and WNV, are not present in Sweden and can therefore be excluded. Previous studies have also ruled out FIV, FeLV, FIPV, feline herpesvirus, feline panleukopenia virus, TBEV, pseudorabies (porcine herpesvirus type 1), canine distemper virus, borreliosis and toxoplasmosis as aetiology for staggering disease in Swedish cats by negative serology, IHC, histopathology, virus isolation and/or by epidemiological features;1,2 however, as many non-suppurative meningoencephalitides have undetermined aetiology,8,9 hitherto unknown viral agents can not be totally ruled out.
From clinical material (mostly blood) BDV-RNA was detected from around a third of the cats. The majority of the positive blood samples were from cats with mild neurological signs.
The sensitivity and specificity of this test has been evaluated 20 and preliminary data from our laboratory on healthy cats (n = 28) tested were all PCR-negative (Eriksson, Berg and Wensman, unpublished data). Notably, the RNA-extraction method was slightly different compared with the current study, and the primers used for the healthy cats were not modified to include 5’-AT rich overhangs, which increases the sensitivity of the current study. 22 The presence of BDV-RNA in blood therefore strengthens the clinical diagnosis to a high degree. Still, not all cats in the present study were PCR-positive, which could be the consequence of low viral load and/or sequence dissimilarities. 23 In order to obtain the highest diagnostic efficiency, rRT-PCR should be complemented by serology. It can not be totally excluded that some cases of staggering disease are caused by other, hitherto undetermined, viruses.
A major observation of this study was that all cats showing mild neurological signs either had severe or moderate pathological lesions (Table 2). This indicates that even if there is a heavy inflammatory reaction in the CNS, this reaction does not necessarily cause severe neurological signs. BD has been regarded as an immune-mediated disease, 28 though there is also evidence for virus-mediated effects, as transgenic mice expressing BDV P show similar neurological signs as in BDV infection, 29 and newborn rats infected with BDV develop pathological and behavioural alterations without inflammation. 30 Furthermore, in a case of atypical staggering disease, BDV was detected in the CNS without inflammatory reactions, indicating direct virus-induced effects. 31 In vitro studies have also shown several direct virus–host protein–protein interactions, potentially affecting the normal neurophysiology.32,33 Recently, we have shown that such BDV–host protein–protein interactions are taking place both in experimentally- and in naturally-infected animals. 34 This could argue for a more direct effect of BDV in causing neurological signs also in natural infection, and would explain the lack of congruency between the degree of inflammatory reaction and the severity of clinical signs.
Moreover, we recently investigated interferon (IFN)-γ mRNA expression in cats with staggering disease. 23 Some of those cats were also included in the present study (for details see Table 2). Interestingly, cats showing mild neurological signs (n = 5) had significantly higher IFN-γ expression than cats showing moderate or severe signs (n = 4; P ≤0.05), and the expression of IFN-γ followed the degree of pathological lesions, ie, the IFN-γ expression was lowest in cats with mild lesions and highest in severe cases. Although this is to be expected, because a higher degree of inflammation should lead to higher expression of inflammatory cytokines, the reversed situation for neurological signs versus IFN-γ is somewhat surprising and confirms that a heavy inflammation is not the sole reason for severe neurological signs. Thus, these data further strengthen the arguments for a more direct, virus-induced effect on the clinical signs.
The proposed site of entry for BDV is the open nerve endings in the olphactory epithelium, and/or in the oropharyngeal mucosa. 35 Even in the early days of BDV research, the hypothesis of secretion of BDV in nasal secretions was proposed, investigated and concluded. 36 More recently, BDV-RNA has been detected in nasal secretions, as well as in saliva and lacrimal fluids, of subclinically-infected horses and sheep.37,38 In the present study we did not detect BDV-RNA in any of the nasal, oral or conjunctival swabs (Supplementary Table 1), but one of the sites most commonly PCR-positive was the olfactory epithelium. BDV-RNA was detected in the olfactory epithelium from cases with varying length of time from onset of signs to euthanasia (2 days up to 6 weeks). This indicates that BDV either persists at the site of entry or is spread centrifugally to the olfactory epithelium at a later stage. Upon experimental intranasal infection of rats, parts of the olfactory epithelium harbour BDV initially and later (3–7 weeks post-infection) BDV is spread centrifugally back to this site. 30
One minor clinical sign observed in the present study was constipation, which was seen in a quarter of the cats. In previous studies, constipation was either seen in around 50% of the cases 1 or in a similar proportion as in the present study. 2 Interestingly, constipation, as well as other gastrointestinal (GI) signs, is common in BD in horses,35,39 though it is not known whether these signs are caused by BDV. 11 In psittacine birds, avian Borna virus (ABV) causes proventricular dilatation disease (PDD), with GI and/or neurological signs.40–42 In PDD, ABV is detected in several tissues, including the ganglia of the autonomic nervous system, 43 and is shed in faeces. 42 BDV has previously not been detected in this organ system in horses and cats, but in the present study an anal swab from one of the cats was PCR-positive, indicating involvement of the GI tract. Five cats had a non-purulent inflammation of intra-abdominal ganglia, foremost in close proximity to the adrenal gland. In two of these cats inflammatory cells were also seen in the medulla of the adrenal gland. It is known from experimental infection that BDV spreads centrifugally to peripheral organs via peripheral nerves late in infection, for example to the adrenal gland. 30 This could indicate that these cats have had a long-lasting persistent infection, even though the duration of clinical disease is rather short (9–17 days).
In conclusion, the present study represents the first cases of cats with staggering disease, which were thoroughly characterised neurologically and, in parallel, investigated to find markers of BDV infection. The observations show that most cats with typical signs of staggering disease have pathological lesions in concordance with BDV infection in other species, as well as interesting findings suggesting several, more direct effects on pathogenesis of BDV, than previously been considered. The clinical history, epidemiology, neurological signs and pathological lesions of the cats in this study were consistent with previous reports,1,2,25 supporting the hypothesis that staggering disease is a disease entity of its own. Finally, BDV-infection markers (BDV-specific antibodies and/or BDV-RNA) were found in most of the cats (89%). When making the diagnosis, serology and rRT-PCR performed on blood samples of cats with neurological signs of staggering disease can be helpful in addition to clinical history and neurological examination; however, it is important to emphasise that a holistic view of all relevant information is needed before decisions are made on the prognosis, as further studies are needed for estimating true seroprevalence of BDV-specific antibodies in a representative cat population.
Supplemental Material
Click here for Supplementary Table 1
Detailed table of BDV rRT-PCR results
Footnotes
Acknowledgements
The authors would like to acknowledge Anna Lindhe, Louise Treiberg Berndtsson, Lena Renström and Dr Eva Osterman-Lind for assistance with serology. Dr Anne-Lie Blomström is acknowledged for her technical assistance with the BDV rRT-PCR.
Supplementary video 1
A representative example of a cat showing mild ataxia. In this short film sequence, cat 12 is shown as a representative example of the group of cats showing mild neurological signs.
Supplementary video 2
A representative example of a cat showing severe ataxia. In this short film sequence, cat 4 is shown as a representative example of the group of cats showing severe neurological signs.
Supplementary Table 1
Detailed table of BDV rRT-PCR results.
Funding
This study was supported by the research foundations of Agria Animal Insurance Company, Swedish Cat Owner Register, Rasehorn, and Intervet. SB is supported by an Award of Excellence provided by SLU. The study sponsors had no involvement in study design, collection, analysis and interpretation of data, in the writing of the manuscript or in the decision to submit the manuscript for publication.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Accepted: 4 April 2012
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
