Abstract
One hundred female cats undergoing routine ovariohysterectomy under midazolam-medetomidine-ketamine anaesthesia were included in a blinded, randomised, prospective clinical study to compare postoperative analgesia produced by four analgesic drug combinations given preoperatively (n = 25 per group). A secondary aim was to assess the effects in kittens and pregnant animals. Buprenorphine 180 µg/m2 or butorphanol 6 mg/m2 were given with either carprofen 4 mg/kg (groups BUPC and BUTC, respectively) or meloxicam 0.3 mg/kg (groups BUPM or BUTM, respectively). Medetomidine was not antagonised. A simple, descriptive scale (SDS; 0–4), a dynamic and interactive visual analogue scale (DIVAS; 0–100 mm) and mechanical nociceptive thresholds (MT; 2.5-mm diameter probe) were used to evaluate postoperative pain. All pain scores were low (DIVAS <10 mm, SDS <2 and MT >10 N) and there were no significant differences between the groups. It was concluded that all protocols provided adequate analgesia and when used with midazolam-medetomidine-ketamine are effective for routine feline ovariohysterectomy.
Introduction
Management of pain in cats has improved considerably in recent years. However, they are still undertreated. 1 Many thousands of cats are neutered throughout the world, often in programmes involving large numbers in one session.2–4 It is essential that anaesthetic protocols that do not compromise safety or animal welfare, and are suitable for a high case load under relatively unsophisticated conditions are developed and disseminated.
Anaesthetic techniques using α2 adrenoceptor agonists, benzodiazepines and ketamine in various combinations have been very successfully used by feline neutering clinics, providing good surgical conditions and smooth recovery.2–4 However, these studies have not enabled postoperative analgesia to be rigorously evaluated.
It is universally acknowledged that surgical procedures cause postoperative pain, and both opioids and non-steroidal anti-inflammatory drugs (NSAIDs) have been shown to alleviate postoperative pain in cats.5,6 A combination of opioid and NSAID has been shown to be superior to either drug type alone, 7 providing the earlier peak effect of the opioid, followed by the longer duration of analgesia from the NSAID. 6
This study aimed to evaluate postoperative analgesia in cats after preoperative administration of a combination of opioid and NSAID before ovariohysterectomy under α2-benzodiazepine-ketamine anaesthesia. A secondary aim was to assess whether the effects were different in kittens and pregnant animals.
Materials and methods
The project was approval by the Animal Health Trust Clinical Research Ethics Committee (project number AHT 15 2010) and signed, owner consent was given in all cases.
Animals and anaesthesia
One hundred healthy, female, domestic cats admitted to the RSPCA Greater Manchester Animal Hospital or the RSPCA Oldham Animal Centre for ovariohysterectomy were studied. Food, but not water, was withheld for a minimum of 6 h before surgery in adults and a maximum of 3 h for kittens younger than 5 months of age. All cats were housed individually in cages prior to, and for 24 h after induction of anaesthesia. Free access to food, water and a litter tray was provided after surgery. After a physical examination to measure heart and respiratory rates, and to confirm that they were healthy and not in late pregnancy, the cats were randomly allocated to one of four groups (n = 25) (Graphpad Prism random number generator):
Group BUPC: 180 µg/m2 buprenorphine intramuscularly (IM) and 4 mg/kg carprofen subcutaneously (SC) (Rimadyl; Pfizer Animal Health). Group BUPM: 180 µg/m2 buprenorphine IM (Vetergesic Multidose; Alstoe Animal Health) and 0.3 mg/kg meloxicam administered SC (Metacam; Boehringer Ingelheim). Group BUTC: 6 mg/m2 butorphanol IM (Torbugesic; Fort Dodge Animal Health) and 4 mg/kg carprofen SC (Rimadyl; Pfizer). Group BUTM: 6 mg/m2 butorphanol IM (Torbugesic; Fort Dodge Animal Health) and 0.3 mg/kg meloxicam SC (Metacam; Boehringer Ingelheim).
Ten minutes before the start of surgery, each cat was anaesthetised using a combination of 60 mg/m2 (~ 3 mg/kg) ketamine (Ketaset; Pfizer Animal Health), 3 mg/m2 midazolam (~ 0.2 mg/kg) (Hypnovel; Roche) and 600 µg/m2 (~ 30 µg/kg) medetomidine (Sedator; Dechra) combined with the allocated opioid — buprenorphine or butorphanol — delivered as a single injection into the quadriceps muscles. The allocated NSAID — meloxicam or carprofen — was given SC.
Once the cat became unconscious the larynx was desensitised with lidocaine spray (Intubeaze; Dechra) and the trachea intubated with an appropriately sized uncuffed endotracheal tube. Anaesthesia was supplemented with isoflurane (0.25% inspired), delivered in oxygen via an Ayre’s T-piece with Jackson Rees modification. Surgery started 10 mins after anaesthetic injection and a routine ovariohysterectomy was carried out by one experienced veterinary surgeon (DY) using a ventral midline approach. Medetomidine was not antagonised.
Environmental conditions were kept as constant as possible throughout all surgeries and during recovery. The time of anaesthetic injection, the duration of surgery (from the first incision until the placement of the last suture) and the duration of recovery (from IM injection to sternal recumbency) were recorded. Surgical conditions were scored by the surgeon (good, adequate, poor). Additional note was taken of any abnormalities that may have altered the surgical procedure, such as pregnancy, mammary hyperplasia or excessive body fat. After surgery, the endotracheal tube was removed as the swallowing reflex returned and the cats returned to their cages for recovery from anaesthesia under observation.
Pain assessment
All assessments were made by a single observer who was unaware of each cat’s group (SP). Assessments were made before premedication and 2, 4, 8 and 24 h after surgery. Heart and respiratory rates were measured at the same time points by auscultation and observation of chest wall movement. Sedation was scored on a four-point descriptive scale (awake, sedated, very sedated or unconscious) determined by observation and palpebral response. Three methods of pain assessment were used:
A simple descriptive scale (SDS) with increasing pain from 0 to 4 based on a general observation of the cats’ appearance (see Table 1);7,8
A dynamic and interactive visual scale (DIVAS) using 100 mm line with 0 as no pain and 100 the worst pain imaginable; 7
Wound sensitivity by measuring the mechanical nociceptive threshold (MT) (force, N) close to the surgical wound, using a pneumatic device with a hemispherical 2.5 mm diameter tip (Prod; Topcat Metrology). As pressure = force/area, at 10 N (1 kgf) a 2.5 mm diameter probe tip (area 4.9 mm2) exerts a pressure of approximately 20 kgf/cm2.
Simple descriptive scale (SDS) used to score pain

Each cat was first observed in the cage without being disturbed. It was then gently handled, offered food, and its behaviour and posture assessed in order to award the SDS (1–4) and DIVAS (0–100 mm) scores. Finally, the cat was removed from the cage, held by an experienced assistant and MT measured by applying the tip of the Prod 10 mm from the wound edge. The force was increased at 2 N/sec, controlled by ‘traffic lights’ (green = go faster, red = slow down) on the unit. When the cat reacted by withdrawal, turning its head sharply towards the site or, occasionally, by vocalisation, the tip was removed immediately, thereby holding the peak reading, which was recorded as MT.
Buprenorphine 20 µg/kg was to be given as rescue analgesia in any cat deemed to be in unacceptable pain postoperatively (DIVAS >50 mm, SDS >1 or MT <3N).
Statistical analyses
Power calculations using unpublished data collected from 30 cats indicated that 100 cats (25 per group) would give 90% power to detect a clinically relevant difference in pain score (DIVAS 10 mm, SDS 1, MT 2N). Single factor data in the four groups were compared using one way ANOVA followed by Tukey’s post hoc test when appropriate (age, weight, duration of anaesthesia, duration of surgery, recovery time, body temperature). Non-parametric single factor data were compared using the χ2 test (surgical conditions). Parametric data collected over time were compared using two-way ANOVA and Bonferroni post tests when appropriate (MT and DIVAS). When group sizes were unequal (pregnant and age-grouped data) the area under the curve (AUC) was used as a summary measure and compared using Student’s t-test. SDS data (non-parametric) were compared using the Kruskal Wallis test. MT data at 2 h were also compared using the Mann Whitney U test (followed by Dunn’s post-test when appropriate) as many thresholds at 2 h went to cut-out (26–28 N) — necessitating ranking analysis. Within-group changes with time were compared using repeated measures ANOVA for parametric data (MT, DIVAS) or the Friedman test for non-parametric data (SDS). Individual opioids were compared by grouping BUPC with BUPM and BUTC with BUTM, and repeating the analyses. Similarly, to compare the NSAIDs, BUPC and BUTC were compared with BUPM and BUTM.
Results
The cats were aged 1 month–6 years and weighed 0.4–4.5 kg. The majority were domestic shorthairs. There were no differences between the groups in body weight, age, surgery time or recovery time (Table 2). Anaesthesia was uneventful and surgical conditions were excellent in all cases. Isoflurane supplementation was minimal and probably unnecessary, but precluded any need for incremental injectable anaesthetics which would confound the postoperative data. Surgery was unremarkable, although a few pregnant animals were less straightforward than the majority. In all cases, heart and respiratory rates decreased from the preoperative values, but remained within normal limits (70–200 and 14–40 per min, respectively) and were not different between groups (Table 3). There was no difference between groups in the degree of postoperative sedation. At 2 h 69% of the cats were still very sedated and only 17% ‘awake’. At 4 h 72% were awake and only 3% heavily sedated. All were ‘awake’ at 8 and 24 h.
Mean (SD) bodyweight, age, surgery time and recovery time of 100 cats undergoing ovariohysterectomy (n = 25 per group)

No significant differences between groups
Mean (SD) heart rate (beats per min) and respiratory rate (breaths per min) in all 100 cats undergoing ovariohysterectomy

No significant differences between groups
Significant change from pre (*P <0.05, **P <0.01)
There were no significant differences between groups in any of the measures used to assess pain and no cats required rescue analgesia. Taking all cats together, SDS was higher at 4 h than at any other time point (P <0.05), but the median never exceeded a score of 1, and no cats received a score above 2 (Figure 1). DIVAS was higher at 2, 4 and 8 h than preoperatively and at 24 h (P <0.05); the highest score recorded was 33 mm in one cat (group BUTC) but the majority had scores less than 10 mm (Figure 2). MTs were not measured preoperatively; at 2 h (all cats) it was 21.8 (8.1) N, decreasing to 11.4 (6.7), 9.8 (5.5) and 10.1 (5.4) N at 4, 8 and 24 h, respectively (Figure 3).

SDS score in 100 cats undergoing ovariohysterectomy after buprenorphine and carprofen (group BUPC), buprenorphine and meloxicam (group BUPM), butorphanol and carprofen (group BUTC) or butorphanol and meloxicam (group BUTM) (n = 25 per group). Pre = before anaesthesia and surgery. 2h, 4h, 8h, 24 h = 2, 4, 8 and 24 h after anaesthetic injection. No significant differences between groups. *4 h (all cats) significantly higher than preoperatively and 24 h (P <0.05)
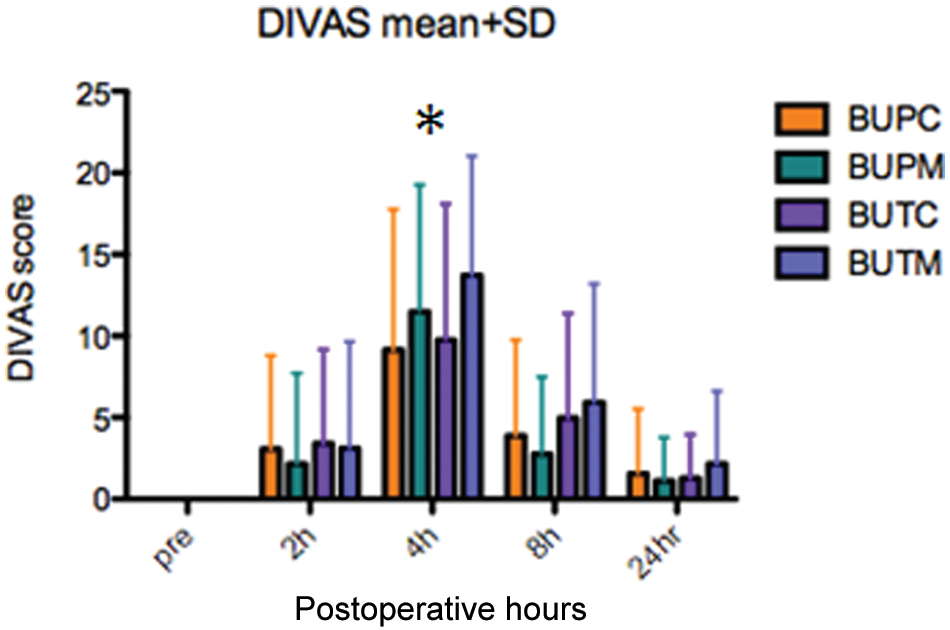
DIVAS [mean (SD)] in 100 cats undergoing ovariohysterectomy after buprenorphine and carprofen (group BUPC), buprenorphine and meloxicam (group BUPM), butorphanol and carprofen (group BUTC) or butorphanol and meloxicam (group BUTM) (n = 25 per group). Pre = before anaesthesia and surgery. 2h, 4h, 8h, 24h = 2, 4, 8 and 24 h after anaesthetic injection. No significant differences between groups. *2, 4, 8 h (all cats) significantly higher than preoperatively and 24 h (P <0.05)

Mechanical threshold [N, mean (SD)] in 100 cats undergoing ovariohysterectomy after buprenorphine and carprofen (group BUPC), buprenorphine and meloxicam (group BUPM), butorphanol and carprofen (group BUTC) or butorphanol and meloxicam (group BUTM) (n = 25 per group). Pre = before anaesthesia and surgery. 2 h, 4 h, 8 h, 24 h = 2, 4, 8 and 24 h after anaesthetic injection. No significant differences between groups. *2h (all cats) significantly higher than 4 h, 8 h and 24 h
Kittens were defined as cats aged 5 months or less. Twenty-five kittens underwent surgery (eight BUPM, one BUPC, nine BUTC and seven BUTM). Surgical time in kittens [9 (1) min] was shorter than in adults [10 (2) min] (P <0.05). DIVAS (AUC t-test) and SDS (Mann Witney U) in kittens and adults were not significantly different (Table 4; Figure 4), but when individual time points were compared, DIVAS was lower in kittens at 24 h (P <0.05). MT was also lower in kittens than in adults throughout the study (P <0.05) (Table 4; Figure 5).
Comparison of pain assessment data, mean (SD) DIVAS, MT and SDS median (range) in kittens (K, 5 months old or younger) and adults (A, older than 5 months)

DIVAS and SDS: no significant difference
MT: significant difference between kittens and adults (*P <0.05)

DIVAS AUC(2–24h) in adults (>5 months old) (n = 75) and kittens (5 months old and younger) (n = 25) after ovariohysterectomy. Pre = before anaesthesia and surgery. 2 h, 4 h, 8 h, 24 h = 2, 4, 8 and 24 h after anaesthetic injection. No significant differences between groups

Mechanical threshold AUC(2–24h) in adults (>5 months old) (n = 75) and kittens (5 months old and younger) (n = 25) after ovariohysterectomy. Pre = before anaesthesia and surgery. 2 h, 4 h, 8 h, 24 h = 2, 4, 8 and 24 h after anaesthetic injection. *Adults significantly higher than kittens
Eleven cats were pregnant. Surgery time was longer (13 min) than in non-pregnant animals (9.5 min) (P <0.05). However, there were no significant differences between the pregnant and non-pregnant animals in DIVAS [mean (SD) AUC: pregnant 126 (114) mm h, non-pregnant 88 (85) mm h], SDS or MT [mean (SD) AUC pregnant 223 (89) N h, non-pregnant 227 (108) N h].
No differences between the effects of either the two opioids or the two NSAIDs were detected in any of the measurements made.
Discussion
This study demonstrated that all the protocols used for anaesthesia and surgery fulfilled the essential requirements for neutering female cats and provided excellent postoperative analgesia. No placebo group was included as it has been sufficiently well established in a number of studies that ovariohysterectomy in cats causes pain, and absence of pain behaviour can be regarded as evidence that the anaesthetic and analgesic protocol produced an analgesic effect.9–11 Three methods of assessing pain were used. Although none is fully validated in the cat, DIVAS and SDS scoring have been widely used in placebo-controlled studies9,8 and MT testing is well established in many species as a method of assessing hyperalgesia following surgery. 5
DIVAS and SDS scores did not correlate with MT. DIVAS and SDS were lower at 2, 8 and 24 h, and highest at 4 h, whereas MT was higher at 2 h than at 4, 8 and 24 h (Figures 1–3). This may simply relate to the degree of sedation, as most cats were still heavily sedated at 2 h, hampering interpretation of the lack of pain behaviour or response to a noxious stimulus.
A limitation of this study was the lack of preoperative MT, so it was not possible to assess the degree of wound sensitivity or hyperalgesia. However, inference can be made from a study by Hunt et al 12 who used the same MT testing device (Prod; Topcat Metrology) with an 8 mm diameter probe tip. The nociceptive stimulus is produced by the pressure on the skin, and since Pressure = Force/Area, the smaller the area, the greater the pressure for the same force, so a larger probe tip will produce less pressure with the same force. The present study used a 2.5 mm diameter probe tip (~5 mm2, approximately one tenth of the area of the 8 mm tip, ~ 50 mm2) so the pressure at the same force must have been 10-fold that in the Hunt study. Hunt et al 12 report preoperative MT of 4–6 N in cats and dogs. This is considerably lower than the MTs recorded in the present study, even in terms of force alone; cut-out at over 25 N was commonly reached at 2 h; thereafter, most readings were around 10 N. These data suggest that all the protocols used in the present study resulted not only in preventing hyperalgesia, but also in producing analgesia even in the face of mechanical nociceptive stimulus. This interpretation is further supported by the observation that no rescue analgesia was required, suggesting that all the cats were very comfortable after the standard protocols alone. This is unusual in any investigation into postoperative analgesia, even when multimodal analgesia is used. 7
It is perhaps not surprising that analgesia was adequate. The anaesthetic protocol itself included medetomidine and ketamine, which both have analgesic properties, and, in combination, have been clearly shown to provide postoperative analgesia in contrast with thiopentone and halothane. 8 In addition to this, multimodal analgesia with a combination of NSAID and opioid was used, previously shown to be more effective than either agent alone. 7 It is also pertinent that in the present study all surgery was performed by one experienced surgeon. Surgery duration of around 10 mins (slightly longer in pregnant or more difficult cases) further serves to illustrate the limited surgical insult, which in itself will limit the degree of pain experienced when consciousness returns. Probably for the same reasons, no differences were detected between the opioids and the NSAIDs used in the study. If overall pain control is provided from a number of different agents, small differences between two that act by a similar mechanism will be hard to demonstrate, and any difference is unlikely to have clinical significance. Carprofen and meloxicam do not hold UK market authorisation for use in cats aged less than 6 weeks old or weigh less than 2 kg bodyweight, respectively — there are no NSAIDs licensed for use in such animals. There is no specific contraindication, the data simply have not been collected, and NSAIDs were used in accordance with the prescribing cascade. Kittens undergoing surgery require analgesia in the same way as adults, hence, it was important to include them in the evaluation.
Opioids are used in conjunction with sedatives and anaesthetics not only for their analgesic properties, but also for their sedative effects and ability to decrease the required dose of anaesthetic. Anaesthetic drug combinations, particularly of a2 adrenoceptor agonists and ketamine, to which opioids are added to reduce the dose of the anaesthetic drugs, are widely used in a number of species, not least the cat. For instance, the triple combination of medetomidine, ketamine and butorphanol has been licensed for anaesthesia in cats for many years, 13 and buprenorphine may be substituted for the butorphanol. 2 In the present study, a benzodiazepine — midazolam — was also included; this allowed reduction in the dose of medetomidine by more than half from the 80 µg/kg in the licensed ‘triple combination’ (NOAH Compendium of Data Sheets: http://www.noahcompendium.co.uk/Pfizer). Use of such anaesthetic combinations, while enhancing the quality of anaesthesia, may also prolong recovery. In the present study, times to sternal of over 2 h may seem unnecessarily long for a surgical procedure lasting 10 min. Animals recovering from anaesthesia require constant supervision until they can reliably sit up and maintain their airway. Brodbelt 14 reported that most anaesthetic-related cat mortality occurred during recovery and a shorter recovery period which does not compromise analgesia would reduce the required time of constant supervision. An a2 antagonist such as atipamezole may be used to shorten recovery time. 2 Although this would antagonise all the effects of the medetomidine, including analgesia, it is unlikely to compromise the overall adequacy of analgesia when opioids and NSAIDs are used. This approach is worth further investigation.
The data collected from kittens undergoing surgery demonstrated that the very young animals dealt with the surgery well. It may be argued that inclusion of animals up to 5 months old did not really allow the study of effects in the very young. However, too few very young kittens were included for useful analysis, and will be the subject of future studies. The study was powered to detect clinically relevant differences between the treatment groups in measures of pain, and there were insufficient very young animals to be sure that real differences between age groups were not missed through a type 2 statistical error. 15 Moreover, pain behaviour in cats is age-specific, 16 so the criteria used in DIVAS and SDS scoring may not apply in the same way to young, active, inquisitive kittens. However, even with a degree of misinterpretation of kitten behaviour, and even if differences in DIVAS and SDS were missed, the 24-h DIVAS score was lower in kittens, and all the scores are still very low. All circumstantial evidence suggests that the kittens were not experiencing serious pain.
In spite of the limited differences in subjective measures of pain, MT in kittens was lower than in adults. This suggests that the affective component of pain was limited and did not alter the kittens’ behaviour, whereas the sensory component, when the site was stimulated, was more evident. Again, the MT values are high in comparison with the data of Hunt et al, 12 suggesting that the sensory pain was not at all severe. The low MT in kittens may relate to the difficulties in detecting MT in very small animals with the same testing system as used in larger animals. Some kittens were less than 0.6 kg, so would simply be more easily moved by a force that would not affect a larger animal (6N force would move a 0.6 kg kitten). Hence, some MTs in kittens may have been artificially low. Further investigation in kittens should be undertaken with a smaller probe tip which allows a lower force range to be used.
There is considerable support for neutering young kittens as this is often the best time to be sure that all animals are available. 17 As there is little evidence that neutering young kittens is more harmful than in adults; 18 the present data indicating that pain relief can also be as good should further support the move for shelters and rescue organisations to promote neutering in younger kittens. This would also have the added benefit of reducing the number of pregnant cats to be neutered. Although in this study these cats did not appear to experience more pain, even in very skilled hands surgery was slightly longer and the risks of complications are likely to be greater where a more difficult and protracted surgical procedure is required. A larger study is required to elucidate this aspect further.
In conclusion, this investigation demonstrated that all four protocols provided good postoperative analgesia and are suitable for neutering female cats, although the slow recovery necessitated prolonged postoperative observation. Further light has been thrown on pain assessment in cats, demonstrating that MT testing is a valuable additional tool. Some insight was gained into the effect of surgery in both young kittens and pregnant cats. The data support the safety and feasibility of neutering kittens.
Footnotes
Acknowledgements
The Feline Advisory Bureau for providing the Prod. The staff at both RSPCA clinics for unstinting support during the study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest statement
Dr PM Taylor is a director of Topcat Metrology Ltd.
The investigation was the subject of a dissertation for a BSc at the University of Manchester (S Polson).
Accepted: 15 March 2012
