Abstract
Clinical report
A 9-year-old, neutered female, domestic shorthair cat was presented to the Hospital for Small Animals, R(D)SVS, University of Edinburgh for management of a suspected feline injection site sarcoma (FISS). The mass had been noted 1 month after annual vaccination with a non-adjuvanted vaccine against feline viral rhinotracheitis (modified live), calicivirus (inactivated), infectious panleukopenia (modified live) and feline leukaemia virus (FeLV) (recombinant).
Referral findings and investigations
A non-painful, firm, irregular, cystic mass, of approximately 8 cm diameter, was located in the interscapular region. The mass was covered by normal skin and was broadly attached to the underlying musculature (Figure 1).

Appearance of the mass prior to surgery. A scar from a previous incisional biopsy is visible at the top of the mass. The cat has been clipped for extensive surgical excision
Serum biochemistry, total T4 and haematology revealed no significant changes. Feline immunodeficiency virus (FIV) antibody and FeLV antigen Snap test (Idexx) were negative. Computed tomography revealed the mass to be arising from the junction of the latissimus dorsi, trapezius and cutaneous trunci muscles, abutting the scapulae, thoracic dorsal spinous processes and the interscapular and epaxial muscles. Small contrast-enhancing tendrils extended caudally to the level of T12. Staging according to the World Health Organization was T3, M0, NX.
Tumour therapy
Neoadjuvant external beam radiation therapy was combined with aggressive surgical excision. Radiotherapy to the tumour and surrounding tissue consisted of 15 fractions of 3.2 Gy, Monday to Friday, for 3 weeks, using a single direct electron field at 9 MeV (Varian Linac linear accelerator) with the cat under general anaesthesia. The only overt acute radiation effect was patchy hair loss in the treatment field. As expected, no change in the size of the mass was noted post-radiation.
Four weeks after radiation therapy, wide local excision was performed, removing the dorsal margin of the right scapula, the nuchal ligament, the dorsal spinous processes and the epaxial musculature directly underneath the mass. Primary closure was achieved using a combination of undermining, layered wound reconstruction, tension-relieving walking sutures, anchorage of soft tissue to the bones at the periphery of the resection and placement of a closed suction drain (Figure 2). The wound was reconstructed without obvious tension along the suture lines. The drain was removed after 2 days and the cat recovered uneventfully.
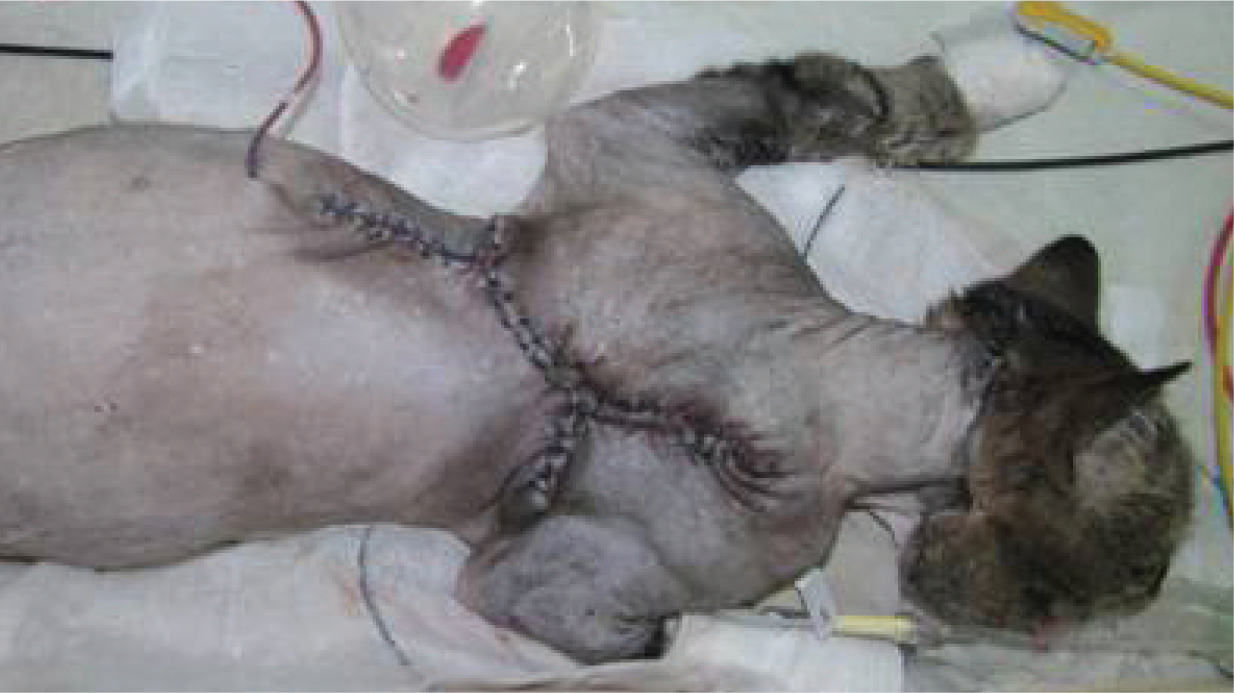
Complete excision was performed involving significant dissection. The wound has been reconstructed without tension and a closed suction drain has been placed
Fourteen days after surgery a central area of dehiscence was noted. Necrotic tissues were debrided, the area was lavaged and tension-free closure was again performed. One week later wound dehiscence recurred at the same site. Histopathology of the wound bed identified no residual disease or early tumour recurrence. The wound was debrided and closed using a stented horizontal mattress pattern to appose the skin following reconstruction of the deeper tissues. Five days later, discharge from the central portion of the wound was noted and a highly resistant Enterococcus species was cultured. Antimicrobial testing identified no effective drugs for systemic use. Instead, a high topical concentration of gentamicin sulphate (Genticin; Amdipharm) was applied, using paraffin-impregnated gauze dressings, and amoxicillin/clavulanate (Synulox; Pfizer) was administered systemically for synergistic effect.
Despite therapy, the infection continued and a large, open wound developed with underrunning over both scapulae (Figure 3).

Underrun pocket over the scapulae following wound dehiscence. Areas of red granulation tissue are apparent from application of the subatmospheric pressure therapy. The sealant paste is visible around the edges of the wound
Wound therapy
Progressive wound dehiscence, resistant bacterial infection and difficulty in applying topical therapy prompted the use of subatmospheric pressure therapy (SAPT). Under general anaesthesia the wound bed was sharply debrided and irrigated. A flexible, conforming, nanocrystalline silver dressing (Acticoat Flex 3; Smith & Nephew) was applied to the wound bed over the next 17 days and either gauze swabs or foam were used to maintain contact with the wound surface. An occlusive film dressing (Opsite; Smith & Nephew) was secured with stoma paste (Coloplast; Coloplast A/S) to provide an air-tight seal. A vacuum port was applied to the dressing and attached to a subatmospheric pressure wound pump (Vista; Smith & Nephew) (Figure 4). The pressure was initially set to a continuous suction of −50 mmHg and subsequently decreased in increments to −120 mmHg over the next 48 h. Analgesia was provided with parenteral buprenorphine (Vetergesic; Alstoe Animal Health) and oral meloxicam (Metacam; Boehringer Ingelheim). Antibiotic therapy was continued with amoxicillin/clavulanate.

(a) The subatmospheric pressure dressing in place. The nanocrystalline dressing and foam is filling the interscapular region. An occlusive dressing has been applied and sealed to the cat’s skin. A vacuum port is placed in the centre of the dressing and subatmospheric pressure is applied. (b) The patient in the hospital with the dressing in place. The dressing was well tolerated and easy to manage
Dressing changes were performed every 48–72 h with the patient anaesthetised. There was marked improvement in the wound bed, which contracted and developed a healthy layer of granulation tissue. Subsequently, the wound was dressed using an open-cell foam in direct contact with the wound.
Forty days after initiation of SAPT the wound pump was removed. The wound bed appeared healthy and had reduced in size to 30% of the original surface area. The underrun pocket over the right scapula had largely closed (Figure 5). Small numbers of Enterococcus species were isolated in pure growth from swabs taken from the surface of the wound, although there was no indication of active infection. Courses of cefalexin (Cephacare; Animalcare) and clindamycin (Antirobe; Pfizer) were initiated. Staggered wound closure was achieved by two procedures 10 days apart. Dead space between the granulation tissue and dermis was closed using a simple interrupted suture pattern (Monocryl; Ethicon) and a closed suction drain was placed. Subcuticular (Monocryl; Ethicon) and skin sutures (Ethilon; Ethicon) were placed (Figure 6).


Appearance of the wound prior to final closure. The wound has contracted to approximately 30% of its original size. The granulation bed appears healthy

The cat 2 days prior to discharge from the hospital. The wound has been sutured and a closed suction drain placed. There is no tension on the wound and hair regrowth is already occurring
The cat was discharged 7 days later and skin sutures were removed after a further 7 days. No further wound healing complications occurred and, at the time of writing, the cat continued to be disease free 12 months after the primary surgery.
Discussion
Management of FISS is difficult and early local recurrence is common, even after extensive resection. Multimodal therapy is recommended because of the invasive nature of this tumour. Improved disease-free intervals have been reported in cats when surgery has been combined with adjuvant radiotherapy.1–3 In humans, local recurrence rates are also significantly reduced in patients receiving adjuvant or neoadjuvant radiation in combination with surgery, although complication rates are higher with neoadjuvant radiation.4–6 Neoadjuvant radiation aims to destroy microscopic disease at the periphery of the tumour before surgery is performed to reduce the recurrence of tumour at the margins of resection. 7 It has advantages over adjuvant radiotherapy in that a smaller field is irradiated and the tissues are well vascularised and oxygenated at the time of irradiation, which improves the efficacy of radiotherapy. Wound complications following tumour removal and irradiation are common,8,9 prompting the recommendation to separate treatment modalities by a period of time. In the patient in this report, neoadjuvant radiation therapy was combined with surgical excision.
A number of factors probably contributed to wound dehiscence in this case. Histopathology following the primary excision identified extension of tumour cells to the deep margin, although no evidence of recurrence was documented on a second biopsy. Radiation injury to the tissues at the edge of the resection may also have contributed to the late wound dehiscence identified on day 14 in this case. While tension was not encountered during primary wound closure, continual scapular movement was likely to be a contributing factor.
Nanocrystalline silver dressings allow for sustained delivery of silver ions to a wound, causing bacterial cell wall damage via alterations in DNA and RNA transcription. The dressings are conformable and flexible, and can be changed on a 3- to 7-day basis without causing maceration to the wound bed. In comparative studies nanocrystalline dressings have been shown to reduce infection rates and incidences of bacteraemia. They are also active against resistant organisms, including meticillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas species,10,11 and act as a barrier to prevent further transmission. 12
In this case, SAPT was used because it allowed a nanocrystalline silver dressing to be kept in continuous contact with the wound surface without disruption by scapular movement and enabled the wound to be effectively sealed from further environmental contamination. SAPT has been reported to reduce wound management time prior to secondary closure and to accelerate closure by second-intention healing in human patients with incisional dehiscence. 13 Removal of wound exudates and oedema and deformation of the wound edges allowing contraction of the wound over time contribute to improved healing. 14
Although not extensively used in veterinary patients SAPT is reported to be simple to use, well tolerated and cost-effective and is gaining in popularity. Its use in dogs and cats has been reported for treatment of extensive avulsion injuries to both the body15,16 and extremities, 17 as well as urine-induced skin necrosis. 18 It has been described for management of wound dehiscence including infected sites 19 and, in combination with nanocrystalline silver dressings, for management of wound infection in a dog following an extensive burn injury. 20
Earlier reports expressed concerns over the use of SAPT at tumour excision sites, 21 as increased local blood flow associated with SAPT might stimulate cancer cell proliferation within the wound bed. However, it has been used successfully for the management of wound dehiscence at tumour excision sites in people, including following soft tissue sarcoma removal.22,23
Conclusion
This is the first report in the veterinary literature of the use of SAPT for the management of wound dehiscence following radiotherapy and tumour excision, and the first report of its use in combination with nanocrystalline silver dressings in a cat. The combination allows both direct bacterial cell kill through the use of the silver contact layer 20 and direct effects on wound contraction and healing through an increased blood supply, removal of oedema and exudates, and enhancement of granulation tissue.14,19 SAPT was highly effective at maintaining contact between the dressing materials and wound in this case, and the authors believe that this contributed directly to resolution of infection. Moreover, SAPT contributed to closure of dead space, promotion of granulation tissue and contraction of the wound, facilitating subsequent closure.
Footnotes
Acknowledgements
The authors would like to acknowledge Mr Colm Farrell (referring veterinary surgeon), and Dr Sionagh Smith, Karen Perry, John Ryan and all members of the R(D)SVS Division of Veterinary Clinical Sciences for their assistance in this work, especially the anaesthesia and nursing teams. They also thank Hilary Gossman and Lynsey Dey, of Smith & Nephew, for advice and assistance with using the SAPT and nanocrystalline silver dressings.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors declare that there is no conflict of interest.
