Abstract

Alimentary lymphoma – and its three clinical entities
Alimentary lymphoma (AL), the most common anatomical form of lymphoma in cats, comprises a group of diseases centred on the gastrointestinal tract, with variable extraintestinal involvement. Three histological grades of AL are recognised: low (LGAL), intermediate (IGAL) and high (HGAL). A separate histological subclassification of AL, large granular lymphocyte lymphoma (LGLL), which can be of any grade, is also described. Although these different subtypes of lymphoma share features related to gastrointestinal dysfunction, such as weight loss, vomiting and diarrhoea, there are major differences in clinical presentation, techniques required for diagnosis, treatment and prognosis (Table 1). From a clinical perspective, LGAL and LGLL can be considered as separate entities and IGAL and HGAL can be considered together as a third entity because, other than histological grade, the clinical features of IGAL and HGAL are similar. 1 Accurate diagnosis is essential to differentiate these lymphomas from each other and from other primary and secondary gastrointestinal diseases so that appropriate treatment can be initiated.
Comparison of clinically relevant features of AL subtypes in cats
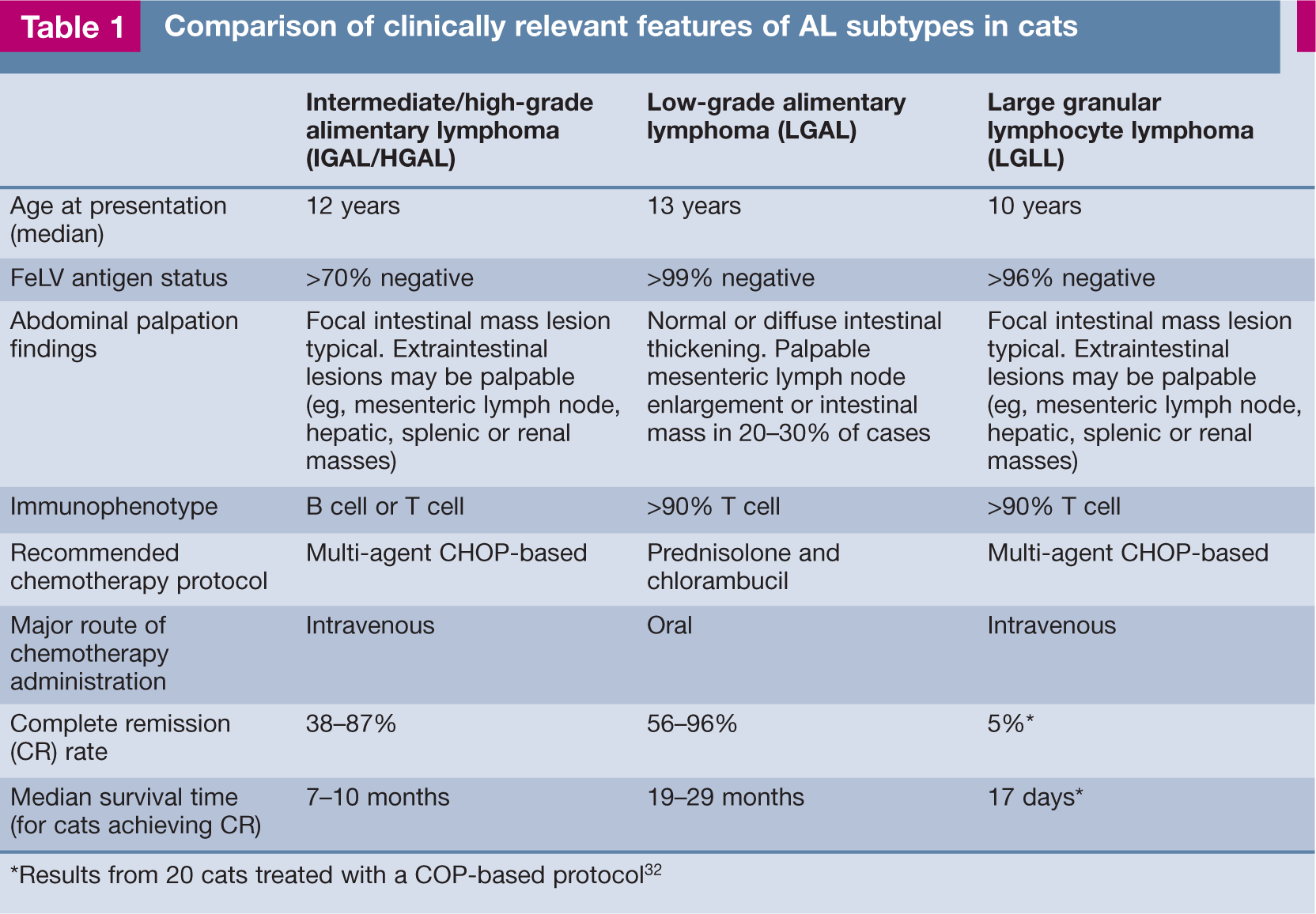
Results from 20 cats treated with a COP-based protocol 32
Classification and prevalence
Lymphoma is the most common intestinal neoplasm of cats, followed by adenocarcinoma and then mast cell tumour. In a study of 1129 feline intestinal neoplasms diagnosed histologically, 55% were lymphomas, 32% were adenocarcinomas and 4% were mast cell tumours. 2 Feline lymphoma can be classified by anatomical location, histological grade and immunophenotype.
Anatomical classification
The traditional anatomical classification recognises mediastinal, multicentric, alimentary and extranodal forms. Of these, AL is the most common anatomical form identified.3–10 The declining influence of feline leukaemia virus (FeLV) worldwide has resulted in an increase in the relative prevalence of AL, since AL has the weakest association with FeLV antigenaemia. Some studies suggest that the absolute incidence of feline lymphoma, particularly AL, is increasing.6,11 In one institution, cases of retrovirus-negative lymphoma increased by 78% in the 10-year period after 1994, compared with the previous 10 years; this could only be partly accounted for by an increase (29%) in the feline caseload. 6 Whether this trend reflects a true increase in the incidence of AL or increased demand for, and availability of, further investigation of feline patients is unclear.
Histological classification of AL subtypes
AL is characterised by infiltration of the gastrointestinal tract with neoplastic lymphocytes, with or without mesenteric lymph node involvement.3,12,13 The histological classification systems most frequently applied to feline lymphoma are the National Cancer Institute Working Formulation (NCIWF) and the Revised European–American Lymphoma/ World Health Organisation (REAL/WHO) schemes.1,9,14–19 The NCIWF scheme classifies lymphoma according to its natural rate of progression, recognising three histological grades (high, intermediate and low) based on the frequency of mitoses. The REAL/WHO scheme classifies lymphoma into specific disease entities based primarily on immunophenotype and morphological features. 20 These schemes are complementary since neither considers both histological grade and immunophenotype.
LGAL has been increasingly recognised in cats over the past 10 years.19,21–27 Synonyms include ‘well-differentiated’, ‘lymphocytic’ and ‘small cell’ AL. Using the NCIWF scheme, most cases of LGAL can be further classified histologically as ‘small lymphocytic lymphoma’.1,15,22–26 Using the REAL/WHO scheme, feline LGAL has been most frequently categorised as ‘epitheliotropic small T cell lymphoma’, ‘epitheliotropic T cell lymphoma’, ‘intestinal T cell lymphoma’ or ‘enteropathy-associated T cell lymphoma’.1,15,16,21,22,27–29 LGLL is a separate subclassification of AL recognised in the REAL/WHO scheme which can be of any histological grade.15,27,30–35

What does immunophenotyping tell us?
Over 90% of LGALs and LGLLs are of T cell immunophenotype, whereas I/HGALs are variably of B or T cell origin.5,9,12,14–17,21,25,27,31,37 A strong association between immunophenotype and location within the gastrointestinal tract has been identified.15,38 In two studies, B cell lymphoma predominated in the stomach and large intestine while T cell lymphoma was most common in the small intestine.15,27 In a retrospective study of small intestinal biopsies from 63 cats with lymphoma or inflammatory disease, 43/51 (84%) lymphomas were CD3-positive (CD3+), further supporting that primary small intestinal lymphomas in cats are typically of T cell origin. 21
T cell lymphomas arise from the diffuse, mucosal-associated lymphoid tissue (MALT) of the small intestine, which includes lamina propria and intraepithelial compartments populated largely by CD3+ T cells. Key immunophenotypic features shared by neoplastic LGLs and intraepithelial lymphocytes (IELs) in the cat (CD3+, CD8??+, CD103+) suggest that LGLLs arise from neoplastic transformation of natural killer cells and cytotoxic T lymphocytes within the intestinal epithelium. 31 Further, in contrast to lamina propria lymphocytes, 25–35% of normal feline intestinal IELs have LGL morphology. 39
B cell lymphomas appear to originate principally from organised lymphoid tissues, including Peyer’s patches and mucosal lymphoid nodules, which are concentrated in the distal small intestine, caecum and colon. 38 Gastric B cell lymphomas in cats are proposed to arise from diffuse gastric MALT colonised by Helicobacter heilmannii 40 or from gastric mucosal lymphoid nodules. 38
Risk factors
Feline leukaemia virus
FeLV is a directly oncogenic retrovirus and persistent antigenaemia confers a 60-fold increased risk of lymphoma development compared with antigen-negative status. 41 The strength of this FeLV association varies with anatomical type. Among thymic lymphoma cases, 80–90% test positive for FeLV antigen,8,42,43 whereas AL has shown the lowest association, with FeLV antigenaemia generally being detected in 0–12% of cases.3,5,8,12,13,19
While the ability to detect FeLV provirus may shed more light on its potential role in lymphomagenesis in exposed but antigen-negative cats with regressive infection, consensus has not yet been reached. In three studies where FeLV antigen was detected in 4%, 2% and 38% of cases of AL, FeLV proviral DNA was detected in 60%, 21% and 53% of tumours.10,36,44 In these studies, approximately equal numbers of T and B cell lymphomas were provirus-positive. In contrast, in 32 cats with AL, Stutzer et al detected much lower levels of FeLV infection and demonstrated concordance between tests; FeLV provirus was detected in only two cases, both of which were antigenaemic and expressed FeLV antigen in lymphoma tissue (6%). 8 The remaining 30 cats were negative for FeLV on all tests. A control group of 41 FeLV antigen-negative cats without malignancies all tested negative for FeLV provirus in the bone marrow. Immunophenotype was determined for 36/77 lymphomas of different anatomical forms. Lymphomas from FeLV antigen-positive cats were significantly more likely to be of T cell immunophenotype than those arising in FeLV antigen-negative cats. 8 This is consistent with previous studies which have demonstrated neoplastic transformation by FeLV of T lymphocytes, null cells and monocytes/ macrophages. 45
Feline immunodeficiency virus
Feline immunodeficiency virus (FIV) infection of cats increases the risk of lymphomagenesis by five-fold compared with uninfected cats. 41 An indirect role is favoured for FIV in lymphomagenesis. 46 FIV-associated lymphoma is typically extranodal, high grade and of B cell phenotype, including atypical lymphoma (eg, nasopharyngeal) and mixed lymphoma with involvement of multiple anatomical sites.47–50 Lymphoma was diagnosed in 21% of FIV-infected cats in one report and AL was the most common anatomical form. 51

Other risk factors

Differential diagnosis
The presenting signs of AL are common to many primary and secondary gastrointestinal diseases. LPE is a major differential diagnosis for LGAL in particular. A comparison between cats with LPE and LGAL found no correlation between clinical findings and the final diagnosis. 61 In cats with intestinal mural mass lesions, epithelial and mast cell neoplasia are major differentials. Diagnostic tests recommended to rule out other primary and secondary gastrointestinal diseases are listed in the box on page 186.
Non-invasive diagnostics
Routine laboratory testing

Weight loss is present in 80% or more of cats with low-grade alimentary lymphoma (LGAL)

Abdominal palpation can be normal in cats with LGAL, but common abnormalities include diffusely thickened intestinal loops or a palpably enlarged mesenteric lymph node


The frequency of perturbations in serum folate and cobalamin with other forms of AL has not been evaluated.
Abdominal ultrasonography
Abdominal ultrasonography facilitates evaluation of the gastrointestinal tract by assessment of wall thickness, layering, motility and luminal content. Normal intestinal wall appears as a five-layered image, with alternating hyper- and hypoechoic layers corresponding to the luminal surface, mucosa, submucosa, muscularis and serosa. Normal ultrasonographic intestinal wall thicknesses are: duodenum and jejunum ≤2.8 mm, ileum ≤3.2 mm and colon ≤1.7 mm. 65 Mesenteric lymph node diameter is generally ≤5 mm. 66 Mural thickening can be further characterised by symmetry, anatomical location and whether it is focal, multifocal or diffuse.
(a) Ultrasonographic and (b) gross appearance of a focal jejunal mass due to high-grade alimentary lymphoma (HGAL). The intestinal wall is thickened (1 cm) and has lost its normal alternating hyperechoic and hypoechoic wall layering pattern. The symmetrical concentrically thickened intestinal wall is visualised in (c) and vascularity identified using power Doppler. Extraintestinal involvement, such as concurrent renal (d) and hepatic (e) masses, is common in HGAL. Images (a), (c), (d) and (e) courtesy of Karon Hoffman, University Veterinary Teaching Hospital, Sydney
(a) Mild diffuse small intestinal wall thickening (wall thickness 3.4 mm) in a cat with LGAL; (b and c) severe small intestinal wall thickening in a cat with LGAL (wall thickness 4.9 mm in b, and 4 mm in c). Note that the alternating hyperechoic and hypoechoic appearance of intestinal layers seen in healthy cats is preserved in cats with LGAL
Footnotes
Key points
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this review article.
Conflict of interest
The authors declare that there is no conflict of interest.
