Abstract
A 4-year-old Ragdoll cat presented for dyspnea secondary to chylous pleural effusion to the University of Georgia Veterinary Teaching Hospital. Physical examination, complete blood count, serum chemistries, urinalysis, thoracic radiographs, abdominal radiographs, and thoracic fluid cytology and culture failed to identify an etiology for the chylous effusion. The patient tested negative for feline leukemia virus, feline immunodeficiency virus and heartworm disease. Respiration phasic influences on early diastolic trans-mitral, trans-tricuspid and pulmonary vein blood flow velocities during Doppler echocardiography were consistent with constrictive pericarditis. The cat underwent subtotal pericardectomy. The patient recovered without complication and is overtly healthy without radiographic or echocardiographic abnormalities 6-months post-surgery. Constrictive pericarditis should be considered in cats with idiopathic pleural effusion, with or without ascites, in which standard echocardiographic assessment is not suggestive of structural heart disease. If constrictive pericarditis is present, the Doppler characteristics outlined here may allow for this diagnosis to be made. Pericardectomy may be highly rewarding, although the specific etiology of the constrictive pericarditis may remain unknown.
Case Report
A 4-year-old, female Ragdoll cat was presented to the University of Georgia Veterinary Teaching Hospital with the complaint of dyspnea. The dyspnea had been previously identified as the result of chylous pleural effusion and treated on three occasions with thoracocentesis. The most recent thoracocentesis was performed 2 days prior to presentation. There was no additional history of illness. Physical examination was unremarkable with the exception of mild tachypnea. The diagnostic work-up was comprised of a complete blood count (CBC), serum chemistry profile, urinalysis, thoracic and abdominal radiographs, thoracocentesis with fluid cytology, aerobic and anaerobic cultures of the effusion, and echocardiography.
The CBC, serum chemistry profile and urinalysis results were unremarkable. Mild pleural effusion was present on thoracic radiographs. Evaluation of the thoracic fluid was consistent with a chylous effusion (fluid triglyceride concentration = 533 mg/dl; serum triglyceride concentration = 51 mg/dl). No organisms were isolated from the pleural fluid cultures. Abdominal radiographs were unremarkable. On echocardiogram, pleural effusion was present but pericardial effusion was absent. Cardiac chamber and wall thickness measurements were within reference intervals. Interventricular septal motion was visibly variable with phase of respiration, with a leftward shift of the septum during inspiration. Color-flow Doppler examination revealed no valvular regurgitation.
The trans-mitral and trans-tricuspid diastolic flow velocities varied with phase of respiration, as assessed from the left apical four-chamber view. Maximum early diastolic trans-mitral flow velocities and the maximum pulmonary vein flow velocities consistently occurred with the first cardiac cycle following the onset of expiration (Figures 1 and 2). In contrast, maximum early diastolic trans-tricuspid flow velocities occurred with the first cardiac cycle following the onset of inspiration (Figure 3).

Trans-mitral diastolic flow velocity assessed from the left four-chamber view. Notice the maximum velocity occurs with the first cardiac cycle following the onset of expiration (E)

Pulmonary vein flow velocity assessed from the left four-chamber view. Maximum pulmonary vein flow velocity occurs with the first cardiac cycle following the onset of expiration (E) with minimum flow velocity noted during inspiration (I)

Trans-tricuspid diastolic flow velocity assessed from the left four-chamber view. The maximum flow velocity occurs with the first cardiac cycle following the onset of inspiration (I) and minimum velocity noted during expiration (E)
These echocardiographic features were considered to be consistent with constrictive pericarditis. Based on the clinical and echocardiographic findings, the cat underwent thoracoscopic subtotal pericardectomy via a left lateral approach. The popliteal lymph nodes could not be found in other cats with chylothorax so a small, paracostal approach was used to access the mesenteric lymph nodes. Ports (3.5 mm; Karl Storz, Goleta, CA, USA) were placed in the caudal thorax in the 10th and 12th intercostal spaces. Methylene blue was diluted 1:10 and 0.2 ml was injected into an ileocecalcolic lymph node. Two large branches representing the thoracic duct were noted in the caudal thorax, and the mediastinum ventral to the sympathetic trunk and dorsal to the aorta was dissected then ligated with 3–0 silk suture by enlarging the caudal port site. After 20 min, the thoracic ducts were no longer blue, and another 0.2 ml of dilute methylene blue was injected. No coloration was noted cranial to the ligation site. The abdominal approach was closed routinely. The pericardium was approached from the left side using three ports (3.5 mm; Karl Storz) in the lateral thorax in the fifth through to the eighth intercostal spaces to allow for concurrent thoracic duct ligation. One port site required lengthening to achieve an assisted procedure to complete the pericardial excision because of the thickness of the pericardium and fear of atrial entrapment in a medium-sized window. The parietal pericardium was markedly thickened and was grasped to retract it away from the epicardium for incision with metzenbaum type scissors (3.0 mm; Karl Storz) and electrocautery. Elevation of the pericardium allowed incision without trauma to the epicardium, myocardium and coronary vasculature. Minimal hemorrhage occurred and the pericardium was not adhered to the epicardium. The pericardium was also incised twice longitudinally to fillet open as much as possible. The heart was subjectively larger upon completion of the pericardectomy and fillet. The heart was not compressed and its surface was apparently normal on gross examination. The musculature of the port sites was closed with 3–0 PDS in a combination of cruciate and simple continuous pattern, and the subcutis and skin were closed routinely. The excised pericardial tissue was submitted for histologic examination.
Histopathologic changes were consistent with chronic pericarditis. The parietal pericardium was severely thickened by granulation tissue and fibrosis along with multifocal to confluent infiltrations of lymphocytes and plasma cells (Figure 4). A layer of fibrin covered the surface in some areas. Infective organisms were not seen during microscopic examination. Results of aerobic and anaerobic bacterial cultures of the pericardium were negative.
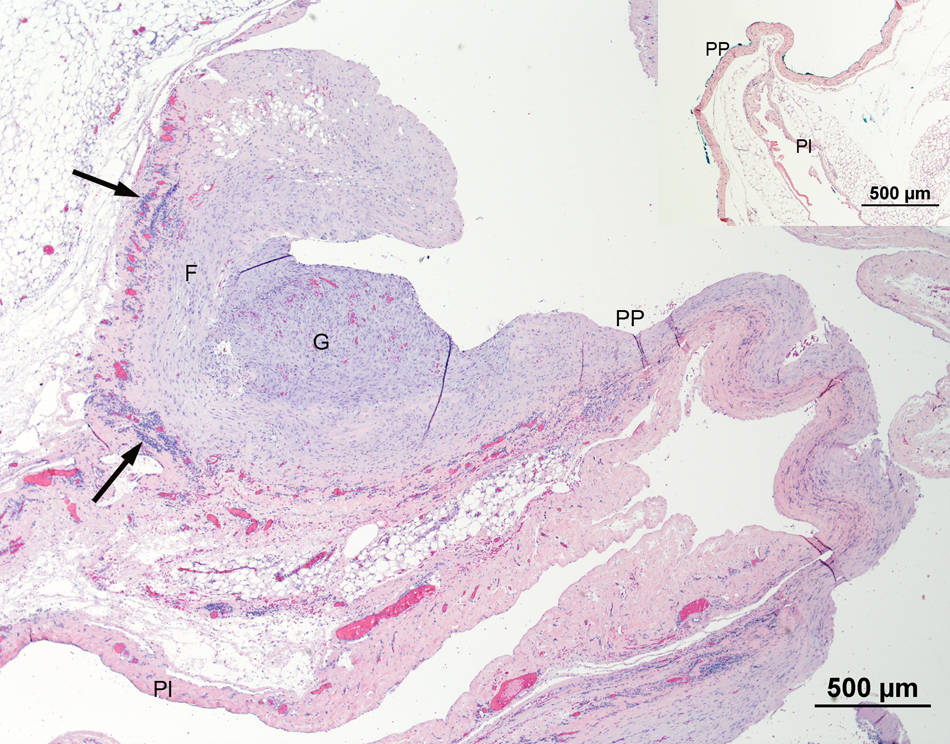
Histopathologic section of pericardial sac from a 4-year-old cat with constrictive pericarditis. The parietal pericardium is severely thickened by extensive areas of granulation tissue (G) and fibrosis of variable thickness (F). There are multiple aggregates of lymphocytes and plasma cells within the pericardium (arrows). Inset: Normal cat pericardial sac for comparison. PP = parietal pericardial surface; Pl = parietal pleural surface. Hematoxylin and eosin stain
We describe the successful surgical management of a cat with constrictive pericarditis diagnosed through the aid of Doppler echocardiography. Constrictive pericarditis in both humans and dogs is often of unknown etiology.1,2–7 In humans living in developed countries, constrictive pericarditis is usually thought to be the result of postviral chronic inflammation, but is idiopathic in almost 50% of patients.1–3 In dogs in our hospital, constrictive pericarditis has been associated with mesothelioma, but it is more often idiopathic. In certain geographic regions, constrictive pericarditis can result from coccidioidomycosis.4–6 Chronic bacterial pericarditis is a potential etiology. 8 An immune-mediated etiology was not discovered in a study of dogs with idiopathic pericardial effusion. 9 We are not aware of any reports of idiopathic constrictive pericarditis in cats.
The fundamental hemodynamic consequence of constrictive pericarditis is limited filling and enhanced interventricular dependence of the heart as a result of rigid encasement by the thickened, non-compliant pericardium. With constrictive pericarditis, the ventricle fills quickly during the early rapid filling phase of ventricular diastole. However, in mid-diastole, the chambers reach the maximum volume that the constraining pericardium will allow. At that point, filling abruptly stops. 1 In diastole, the pressures in both atria and ventricles are identical, or very similar. Obstruction of right ventricular filling causes elevation of central venous pressure. Thus, right-sided congestive heart failure can occur and manifest as pleural effusion and/or ascites. A modified transudate pleural effusion, with or without chylous characteristics, is common in dogs with constrictive pericarditis.4–6
In human patients with constrictive pericarditis, the most dependable echocardiographic findings are the result of normal phasic respiratory influences on venous return of blood to the right heart chambers. 1 In the face of abnormally decreased pericardial compliance (owing to a thick and stiff pericardium), there are hemodynamic consequences to the phasic variation in venous return.
For a heart with normal pericardial thickness and compliance, the left and right ventricles can fill with a relative lack of interdependence. During inspiration, when negative intrathoracic pressure and outward radial traction on the caudal vena cava increase venous return to the right heart, the right ventricular free wall can move eccentrically to allow for the increased filling.
In patients with constrictive pericarditis and decreased pericardial compliance, the increased right heart filling that accompanies inspiration occurs at the expense of the left heart. The right ventricular free wall is prevented from its normally accommodating eccentric movement. Instead, the interventricular septum is forced leftward, sometimes noticeable during two-dimensional echocardiographic examination. As a result, the left ventricular chamber’s ability to accept pulmonary venous and left atrial blood during diastole is compromised.
The corresponding measurable echocardiographic changes reflect the increased ventricular interdependence and compromised left-sided filling during inspiration. In patients with constrictive pericarditis, during inspiration relative to expiration, there is greater early diastolic trans-tricuspid valvular flow velocity, with lower maximal pulmonary venous flow velocity and early diastolic trans-mitral valvular flow velocity. The opposite changes occur during expiration when the left ventricle gets a relative reprieve from the compromising interventricular septum.
Changes in trans-valvular inflow patterns are not specific to constrictive pericarditis. For example, evidence of diastolic dysfunction is also present with pericardial effusion (irrespective of pericardial tissue compliance) and restrictive cardiomyopathy. However, an echocardiographic hallmark of restrictive cardiomyopathy is severe atrial dilation that is absent with constrictive pericarditis. Also, in human patients with restrictive cardiomyopathy, mitral inflow E-propagation velocity is decreased and respiratory variation in mitral inflow velocity is absent. 10 Doppler findings associated with constrictive pericarditis and pericardial effusion are similar. However, with cardiac tamponade, chamber collapse is a critical feature that is absent with constrictive pericarditis. With pericardial effusion, right atrial collapse is an early sign of tamponade, beginning late in ventricular diastole and extending into ventricular systole. 1
In humans, the prognosis for patients with constrictive pericarditis depends on the etiology; it is usually excellent for idiopathic constrictive pericarditis. 3 This has also been our experience in dogs with idiopathic constrictive pericarditis.
The incidence of constrictive pericarditis in cats is unknown. Constrictive pericarditis should be considered in cats with idiopathic pleural effusion, with or without ascites, in which routine echocardiographic findings are not consistent with primary myocardial disease, neoplasia or pericardial effusion with tamponade. Additional diagnostics, such as an echocardiogram to evaluate for ventricular interdependence and respiratory variation in transvalvular flow velocities as reported here, and/or hemodynamic studies, would be indicated for confirmation of constrictive pericarditis. Pericardectomy may be highly rewarding even though the etiology of the constrictive pericarditis may remain unknown. Although the pleural effusion in this case would have been expected to resolve following pericardectomy, thoracic duct ligation was undertaken to maximize the probability of successful outcome and to avoid repeat thoracotomy if pericardectomy alone was not successful at eliminating all the effusion.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
