Abstract
B-lymphoblastic leukemia/lymphoma (B-ALL) is the most prevalent childhood malignancy, with improved survival rates attributed to advances in diagnosis and treatment. Herein, we present a case of B-ALL in a 6-year-old female who presented with clinical symptoms of leukemia, including weight loss, fever, and lymphadenopathy. Molecular and cytogenetics study revealed a t(1;22) resulting in an EWSR1::PBX1 fusion. This case highlights diagnostic and prognostic implications of this recently recognized distinct EWSR1 entity.
Introduction
B-lymphoblastic leukemia/lymphoma (B-ALL) is the most common childhood cancer, with 5-year overall survival (OS) rates exceeding 90%. 1 However, outcomes vary by subtypes, which are often defined by genomic alterations. 2 These findings underscore the importance of cytogenetic and molecular subclassification in refining prognostication and in designing targeted therapy.
Here, we report a case of pediatric B-ALL harboring an EWSR1::PBX1 fusion, emphasizing the novelty of this molecular finding and its potential relevance in diagnostic hematopathology.
Case Report
A previously healthy 6-year-old female presented with several weeks of weight loss, fevers, fatigue, cervical lymphadenopathy, left arm pain, and petechial rash. Initial laboratory workup revealed leukocytosis (WBC: 58.7 × 103/µL) and thrombocytopenia (platelets: 32 × 103/µL). The peripheral blood flow cytometry showed that most of the leukocytes were lymphoid blasts. A morphologic review of the bone marrow biopsy demonstrated large blasts with open chromatin, convoluted nuclei with occasional nuclear cleaving, and basophilic cytoplasm containing rare clear cytoplasmic vacuoles. Flow cytometric analysis of the bone marrow sample showed a large cluster of cells (red) in the moderate range of CD45 expression on the CD19 versus CD45 plot, distinct from mature B cells (blue). These leukemic blasts showed strong CD10 and CD38 expression, moderate to absent CD34, no CD20 expression, and abnormally bright CD58 compared with the heterogeneous dim pattern expected for normal B-cell precursors (Figure 1).
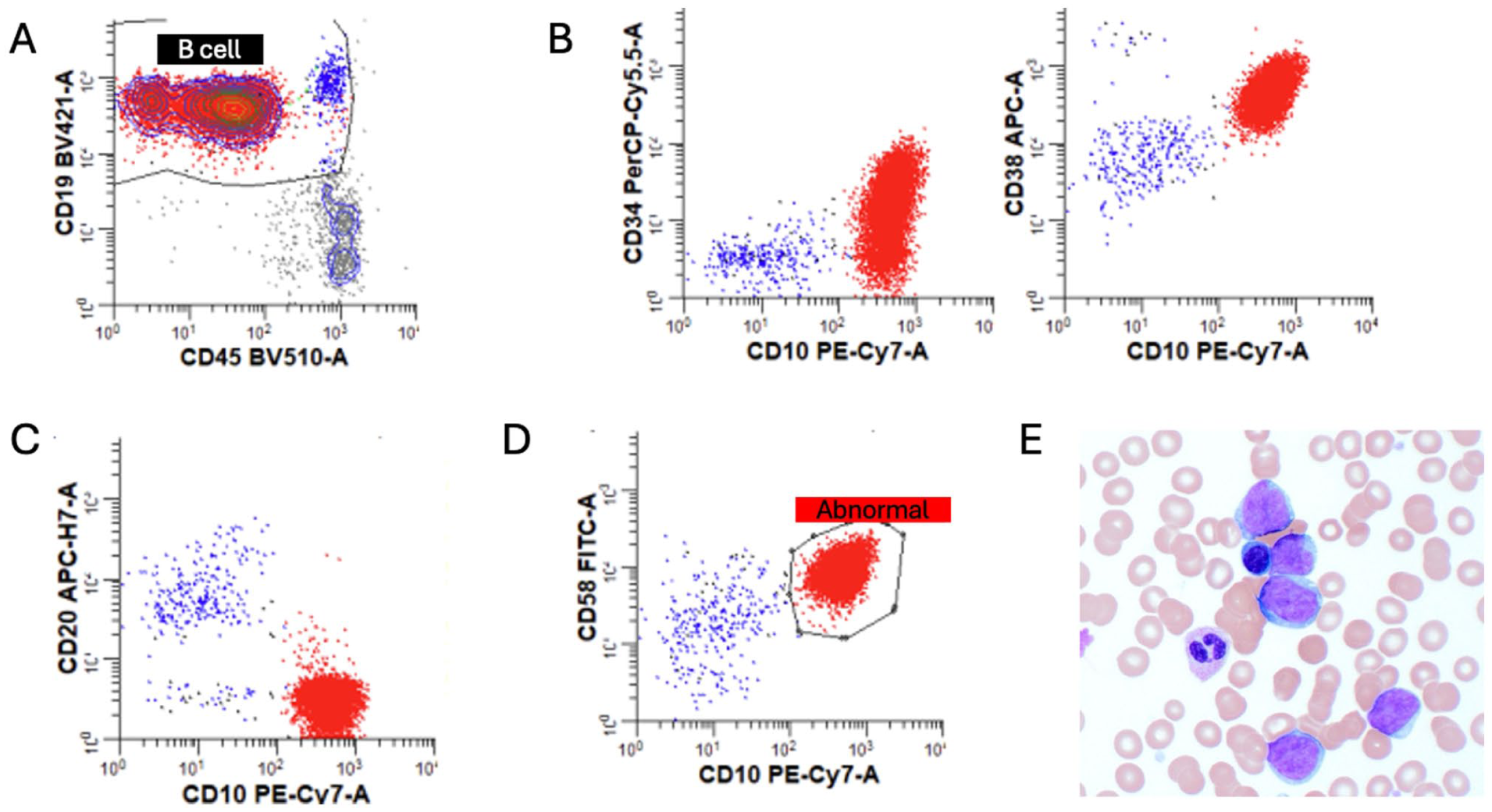
Bone marrow flow cytometry and morphologic findings, (flow plots A-D gated on B cells). (A) CD19 versus CD45 plot demonstrating a large cluster of leukemic blasts (red) with moderate CD45 expression, distinct from mature B cells (blue). (B) CD10 versus CD34 and CD10 versus CD38 plots showing moderate to absent CD34 expression and strong CD10 and CD38 expression on blasts (red) compared with mature B cells (blue). (C) CD20 versus CD19 plot demonstrating lack of CD20 expression on blasts compared with mature B cells. (D) CD58 plot showing abnormally bright CD58 expression on blasts relative to the heterogeneous dim expression typical of normal B-cell precursors. (E) Peripheral blood smear, Wright-Giemsa, stain showing blasts with open chromatin and basophilic cytoplasm (original magnification ×600).
Cytogenetic analysis was performed on metaphase cells prepared from unstimulated bone marrow (BM) cultured for 24 hours. Giemsa-banded metaphase cells were analyzed, and results were reported using the International System for Human Cytogenetic Nomenclature, 2020. 3 Results revealed a complex abnormal female karyotype: 50,XX,+X,t(1;22)(q23;q12),t(2;9)(p13;p22),?inv(13)(q12q34),+14,+18,del(20)(q13.1q13.3),+21[14]/46,XX[6] (Figure 2).
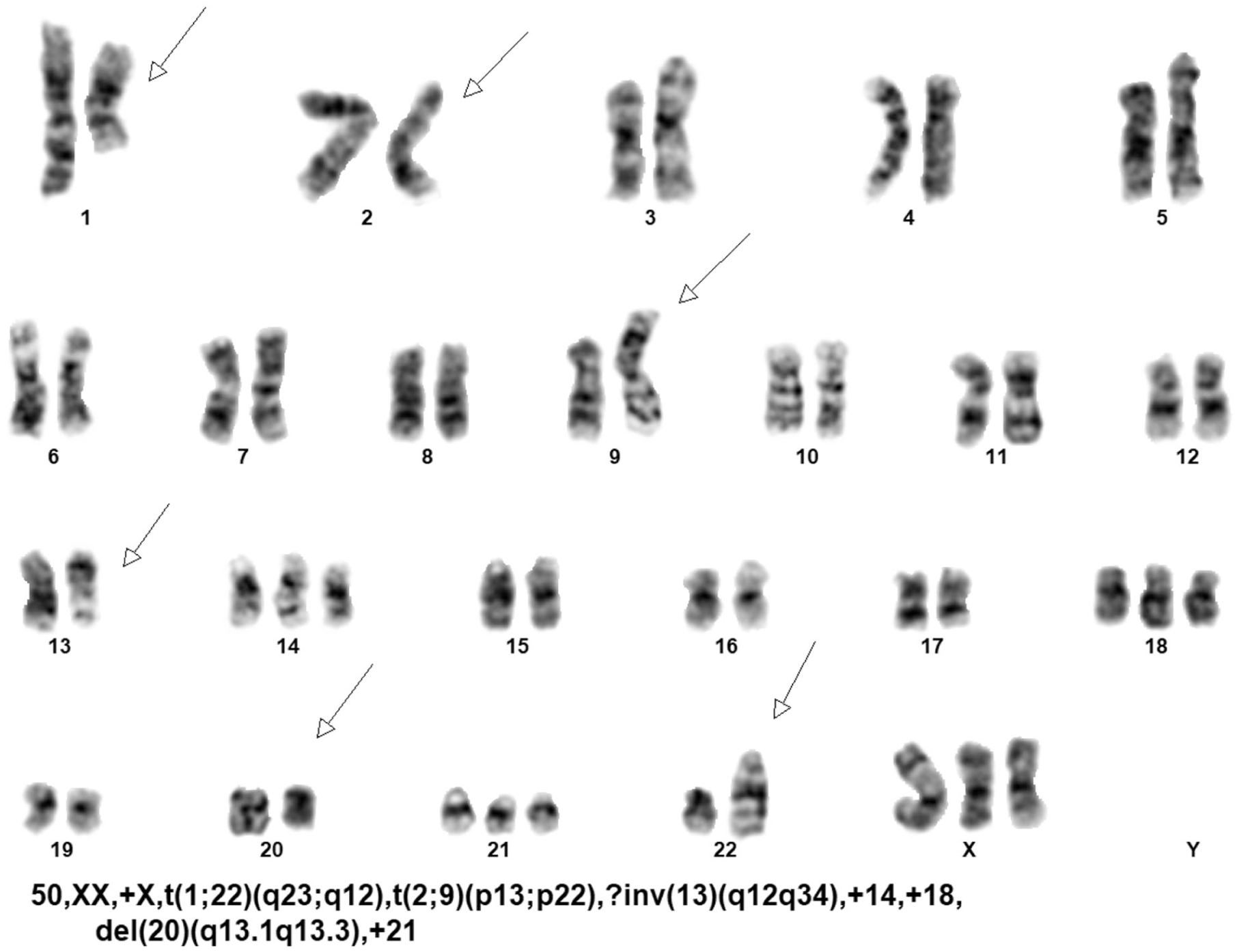
Representative abnormal G-banded metaphase cell.
Fluorescence in situ hybridization (FISH) was performed on interphase nuclei from the same BM culture used for the karyotype with specific probes for detection of B-ALL related genetic abnormalities using standard techniques. The PBX1 (1q23, spectrum orange) and TCF3 (19p13,3, spectrum green) dual-color dual-fusion translocation probe (Abbott Molecular, Downers Grove, IL) revealed 3 copies of PBX1 (1q23), IGH (14q32), and RUNX1 (21q22), each in 67%, 71%, and 81% of 200 nuclei analyzed, respectively. Karyotype and metaphase fish showed a t(1;22), and a PBX1 rearrangement with the 22q12 region (Figure 3). RNA sequencing on the bone marrow aspirate using a customized Archer FUSIONPlex™ Pan-Heme panel (Integrated DNA Technologies, Coralville, Iowa) revealed an EWSR1::PBX1 in-frame fusion between exon 14 of EWSR1 (NM_005243.3) and exon 5 of PBX1 (NM_002585.3). This fusion was confirmed by RT-PCR and Sanger sequencing. This result is consistent with the t(1;22) seen in the karyotype and the rearranged PBX1 by FISH. No other gene fusions or structural rearrangements were detected by the fusion panel, including no evidence of an intragenic IKZF1 deletion, although this panel cannot detect a whole gene deletion.
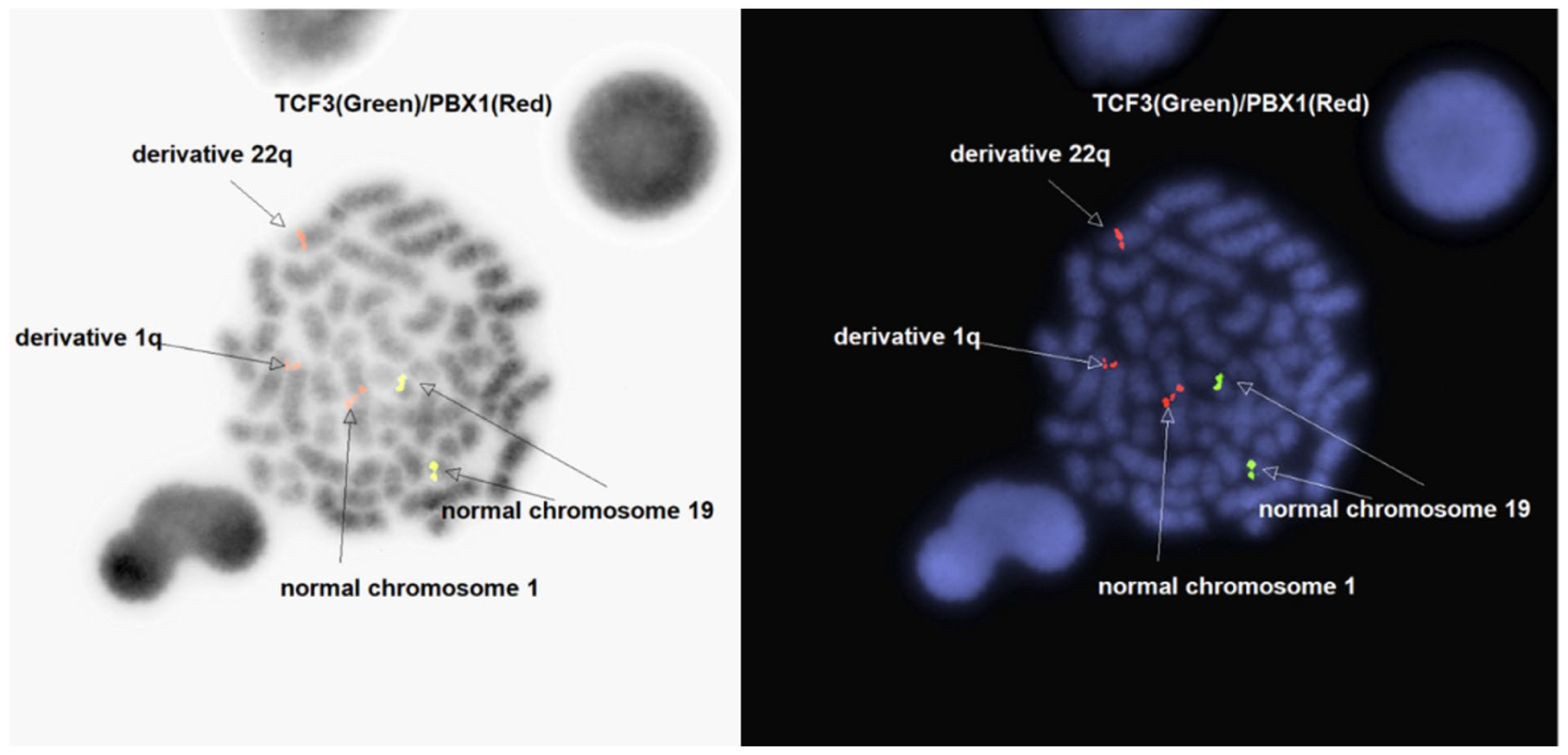
FISH metaphase cell result with rearranged PBX1 localized to 22q12.
Due to a presenting WBC >50 × 103/µL, the patient was treated according to the high-risk B-ALL chemotherapy regimen outlined in AALL1732, a Children’s Oncology Group (COG) study. A 4-drug induction (vincristine, dexamethasone, daunorubicin, and asparaginase) was given, and she achieved complete remission with minimal residual disease (MRD) <0.01% by end of induction. Therapy proceeded as per protocol with augmented BFM consolidation, interim maintenance, delayed intensification, and ongoing maintenance per AALL1732. At the time of diagnosis, the protocol did not include blinatumomab for high-risk patients. She remains in remission.
Discussion
Although EWSR1 rearrangements have infrequently been reported in hematologic malignancies, and TCF3::PBX1 fusions are common in B-ALL, an EWSR1::PBX1 fusion has not previously been reported in B-ALL. 4 In addition, while other 3′ gene partners (e.g., HLF) are seen with TCF3, variant 5′ fusion partners have not been identified with PBX1 in B-ALL.
EWSR1 encodes EWS, which is an RNA-binding protein involved in transcriptional regulation, RNA splicing, and mRNA transport. 5 Rearrangements of EWSR1 are well known in mesenchymal tumors such as Ewing sarcoma, desmoplastic small round cell tumor, clear cell sarcoma, and others, but are rare in hematologic malignancies.4,6 An EWSR1::FLI1 fusion was reported in a pediatric B-ALL case with massive extramedullary disease, initially misdiagnosed as Ewing sarcoma. 6 An EWSR1::ZNF384 fusion was also reported in a pediatric patient with B-ALL. EWSR1 fusions with different 3′ partners have also been described in other isolated cases of immature B-cell neoplasms, including a mixed phenotype acute leukemia (MPAL with B-, T-, and myeloid lineage antigen expression), and a high-grade B-cell malignant neoplasm with immature features.4,7 These findings suggest that hematopoietic cells may also harbor oncogenic EWSR1 fusion proteins, though their clinical relevance is not yet well defined, as there are too few cases reported to date.
PBX1 (Pre-B cell leukemia factor 1) was identified in 1990 as part of a fusion protein product of the chromosomal translocation t(1;19). PBX1 encodes a TALE-class homeobox transcription factor that partners with HOX proteins during development. 8 In B-ALL, the TCF3::PBX1 (formerly E2A:PBX1) fusion occurs in about 5% of cases and is recognized as a distinct WHO B-ALL subtype. Historically associated with inferior outcomes, TCF3::PBX1-positive ALL now responds well to contemporary therapy with good prognosis in children, though with higher CNS relapse risk. 9 In this canonical t(1;19), the chimeric fusion protein acts as an aberrant transcription factor. Similar to other fusions involving EWSR1 and PBX1, the EWSR1::PBX1 fusion combines the strong transcriptional activation domain of EWS with the PBX1 DNA-binding domain, potentially dysregulating PBX1 target genes and contributing to leukemogenesis.
Review of the literature identifies EWSR1::PBX1 fusions in myoepithelial neoplasms, and other rare soft tissue neoplasms.10-14 As summarized in Table 1, these cases span both benign and malignant behaviors, with most managed by surgical resection with good outcomes. 10 However, treatment details were not consistently described, and clinical follow-up was limited in several reports. These findings suggest that the biologic behavior of EWSR1::PBX1 fusions may be tumor-type specific. In contrast to localized solid tumors, pediatric B-ALL is treated with systemic chemotherapy. Whether the EWSR1::PBX1 fusion influences leukemia biology or treatment response remains unknown.
Reported Cases of EWSR1::PBX1 Fusions Across Tumor Types and Clinical Outcomes.
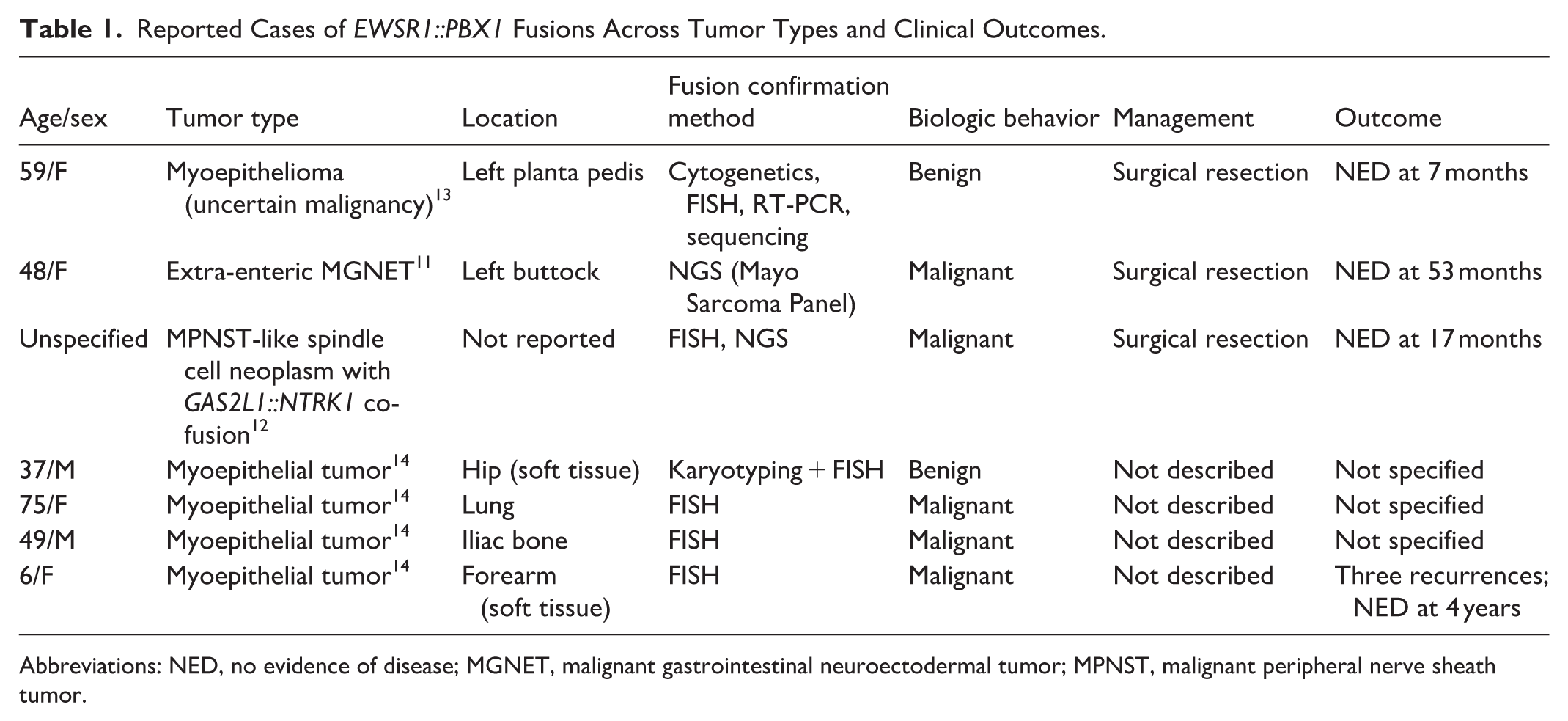
Abbreviations: NED, no evidence of disease; MGNET, malignant gastrointestinal neuroectodermal tumor; MPNST, malignant peripheral nerve sheath tumor.
The integration of genomic findings into the diagnosis and treatment of B-ALL has revolutionized diagnostic precision and risk stratification. While the clinical relevance of EWSR1::PBX1 fusion in B-ALL is unclear, broader sequencing efforts may identify additional cases and clarify its role. Prospective studies and long-term follow up are needed to determine whether this fusion portends unique prognostic or therapeutic implications.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MT is supported by the NIH-funded Vanderbilt Oncology Training Program (T32 CA217834). No other financial support was received for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB reports serving on a Data and Safety Monitoring Committee for Merck. The remaining authors declare no conflicts of interest in the research, authorship, and/or publication of this article.
