Abstract
Purpose and Context:
Umbilical cord abnormalities with clinical signs of cord compromise are frequently associated with fetal vascular malperfusion (FVM). Single umbilical artery (SUA) has been reported to be associated with high-grade FVM in fetal growth restriction but not in an unselected population; our study aimed to address this issue.
Methods:
Clinical and placental phenotypes of 55 consecutive placentas with SUA (Group 1) were compared with those of 655 placentas with 3-vessel umbilical cord (Group 2) from patients who were in the second half of their pregnancy. The placentas were histologically examined using hematoxylin and eosin (H&E) staining and CD 34 immunostaining.
Key Results:
Several umbilical cord phenotypes and high-grade distal FVM, based on H&E staining and endothelial fragmentation by CD34 were significantly more common in Group 1, whereas decidual clusters of multinucleate trophoblasts were more common in Group 2. Notably, H&E staining or CD34 immunostaining evaluated separately showed that high-grade distal FVM was more common in Group 1 than in Group 2, but the difference was not statistically significant.
Conclusions:
SUA predisposes to remote, advanced, and recent high-grade distal villous FVM, with a pathogenesis partly different from that of stasis-induced FVM, likely related to fetal anomalies associated with SUA.
Introduction
Umbilical cord (UC) abnormalities are known to be associated with fetal vascular malperfusion (FVM), particularly in stillbirths, 1 and are more common in macerated stillbirths than in non-macerated stillbirths. 2 The likely stasis-induced FVM secondary to occult UC compromise should be considered during placental investigation in such cases. Hypercoiled UC is 9 times more frequent in macerated fetuses than in non-macerated fetuses. 2
Clinical cord entanglement and pathological cord abnormalities are significantly increased in placentas with FVM in patients who eventually develop cerebral palsy or other forms of long-term neurological impairment.3,4 Histologic examination of the placenta may reveal the -grade of FVM at various evolutionary stages in patients affected by perinatal arterial ischemic stroke. 5
The most common UC abnormality concerns its degree of coiling. 6 Frequencies of variable decelerations are significantly higher in cases with velamentous cord insertion, hypercoiled cords, and nuchal cords in the first stage of labor, 7 but some researchers believe that evidence from either unselected cohort studies or systematic reviews is insufficient to support the suggestion that abnormal cord coiling is associated with adverse clinical outcomes. 8 Patterns of acute and chronic placental injury are reportedly slightly more common with abnormal coiling of the UC and fetal vascular ectasia. 9 Except for a borderline significant association with maternal vascular malperfusion, peripheral insertion of UC is not associated with other placental lesions. 10
Single umbilical artery (SUA) is diagnosed in approximately 0.2–2% of pregnancies, though some case series have indicated rates up to 5%, with the rate of associated fetal structural anomalies ranging from 13% to 56% and that of aneuploidy up to 9%. 11 Genetic syndromes (VATER, Meckel-Gruber, and Zellweger syndromes) and non-chromosomal defects (bilateral and unilateral renal agenesis, imperforate anus, intestinal atresia, vertebral anomalies, sirenomelia, caudal regression syndrome, and body stalk anomalies) are also frequently associated with SUA. SUA occurs in 2–5% of twins and 1% of singletons. It is typically observed in the twin reversed arterial perfusion (TRAP) sequence, 12 as it is more common in high-risk pregnancies involving maternal diabetes mellitus (3–4 times more frequent), fetal growth restriction, stillbirths, or preterm births. 13 Thus, isolated SUA is independently associated with adverse pregnancy outcomes. 11 However, approximately 80% of SUA cases are incidental findings without associated fetal anomalies.
SUA is associated with other UC abnormalities such as thin and velamentous UC insertion.13,14 Placental abnormalities such as low placental weight, 15 circumvallate placenta, 13 chronic placental insufficiency, and abnormal villous maturation and differentiation have been associated with SUA. 16 A study reported lower odds of maternal vascular malperfusion in cases of fetal growth restriction with SUA. 17
Distal FVM diagnosed via hematoxylin and eosin (H&E) staining in SUA cases was previously reported more frequently in cases of fetal growth restriction (high grade) than in cases of cases of trivascular UC. 17 This study was aimed to analyze an array of patterns of placental injury in SUA cases, not limited to those associated with fetal growth restriction, through H&E staining and CD34 immunostaining.
Materials and Methods
This retrospective clinical trial was approved by the institutional review board (number 2016-7942). The placental examination, procedure, and definitions of placental lesions and patterns used in this study are consistent with the Amsterdam criteria18,19 and their later modifications.18,20 These include the use of E cadherin/CD34 double immunostaining to diagnose early FVM in distal chorionic villi (its CD34 component) 1 and clustered mineralization of chorionic villi to diagnose remote FVM in distal chorionic villi. 21 Therefore, for our analysis, we defined recent distal villous malperfusion as the presence of distal villous FVM featuring distal villi with clustered endothelial fragmentation by CD34; established distal villous FVM as avascular and hyalinized clusters of distal villi through H&E staining, and remote FVM as clusters of mineralized distal villi through H&E staining, or, in rare instances, iron and/or von Kossa histochemistry stainings. These lesions were classified as either low-grade or high-grade using the numerical criteria of the Amsterdam Consensus Conference for the numbers of clusters of avascular villi.
Of the 2254 consecutive placentas who signed up between 2006 and 2022, 111 had SUA UC (confirmed or diagnosed by H&E staining) and 2143 had 3-vessel UC. Among them, the gestational age was ≥20 weeks for 107 and 1977 patients, respectively. Further, in 55 and 655 of these patients with UC and 3-vessel UC (Groups 1 and 2, respectively), at least 1 grossly unremarkable full-thickness placental block was double immunostained with E cadherin and CD34. Independent clinical and placental phenotypes were evaluated using analysis of variance (ANOVA) or Chi-square, where appropriate.
Results
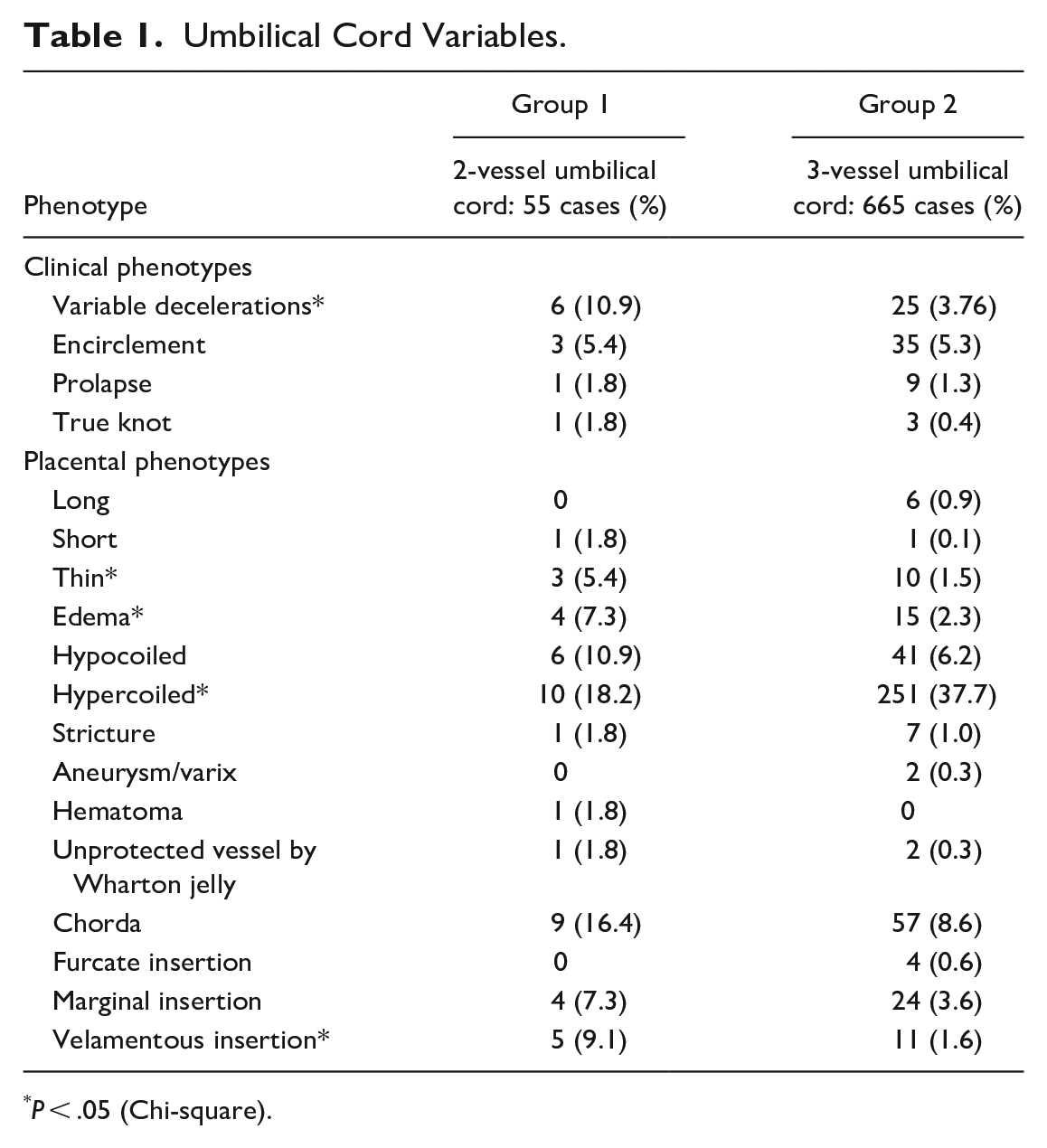
Variable decelerations, thin UCs, edematous UCs, and velamentous insertion of the UCs were approximately 2 times more common in Group 1 than in Group 2, whereas trivascular UC was 2 times more likely to be hypercoiled (coiling index >0.3) in Group 2 than in Group 1 (Figure 1). The frequencies of 20 other clinical and 11 anatomical UC phenotypes showed no statistically significant differences between the 2 groups (Table 1).
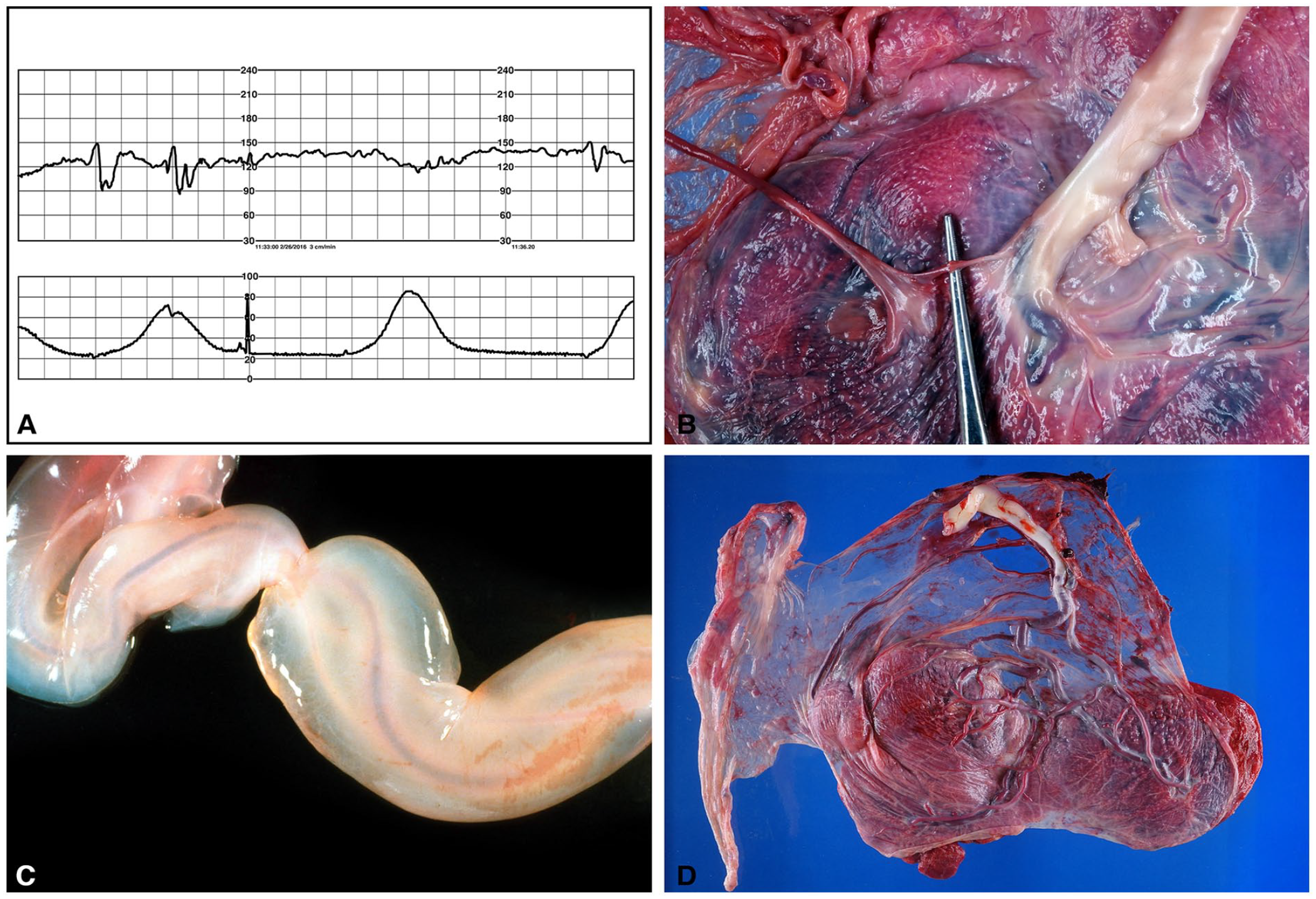
Umbilical cord related phenotypes associated with single umbilical artery. (A) Variable decelerations in labor, 38 weeks, nuchal cord x2. (B) Thin umbilical cord (left) of the perfused twin (acardius acephalus) of the twin reversed arterial perfusion sequence (TRAP) with its single artery anastomosed to an umbilical artery of the pump twin (right), 31 weeks. (C) Massive umbilical cord edema, 29 weeks, fetal demise. (D) Velamentous insertion of the umbilical cord, 35 weeks, gestational diabetes mellitus, polyhydramnios, right aortic arch, and bilateral superior venae cavae.
Umbilical Cord Variables.
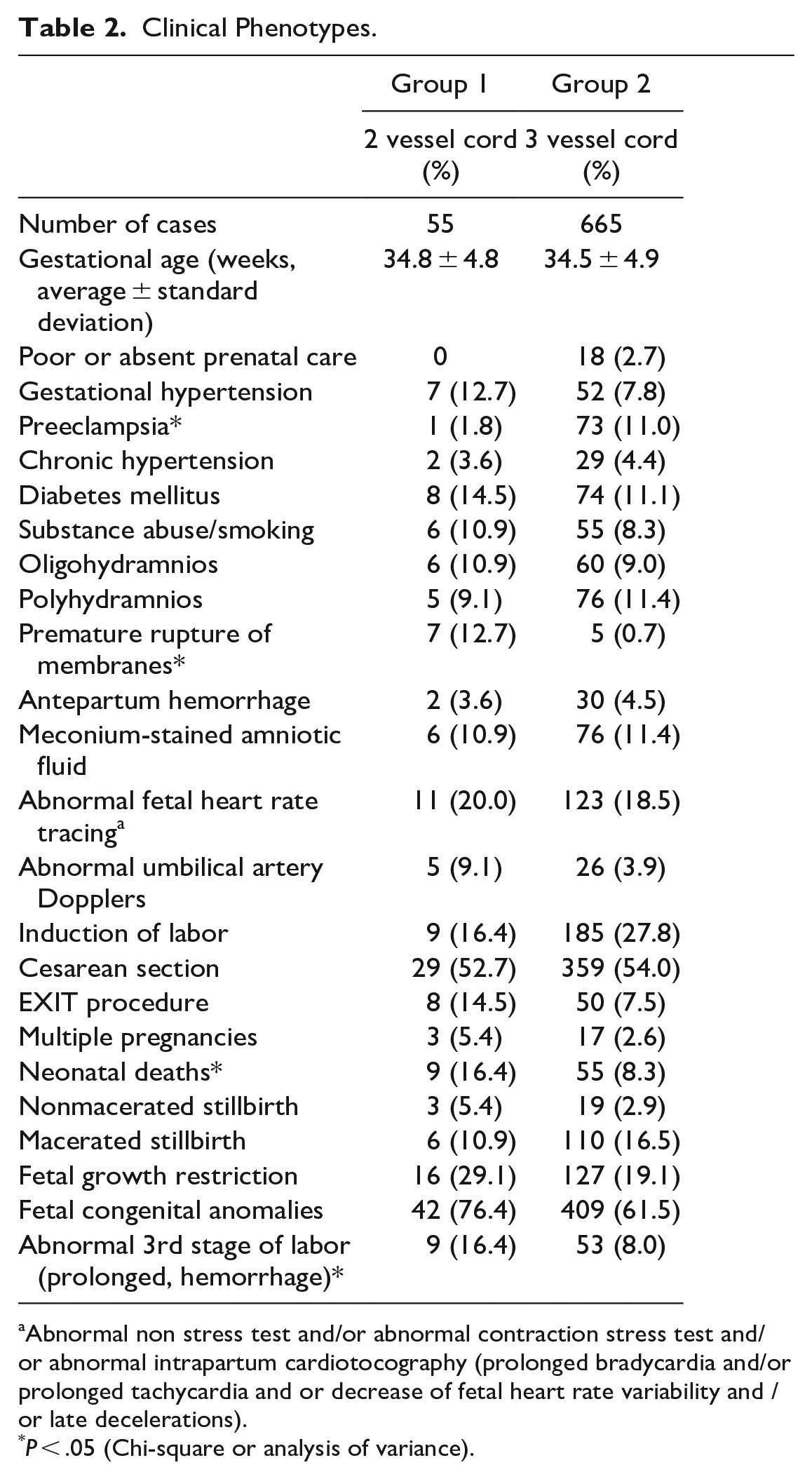
The average gestational age was similar in both groups. Fetal congenital anomalies were more common in Group 1 than in Group 2, although the difference was not statistically significant. This reflects the patient population treated at the Children’s Hospital. Preeclampsia was significantly less common in Group 1 than in Group 2, whereas premature rupture of membranes, neonatal deaths, and abnormal third stage of labor (prolonged, hemorrhage), were significantly more common in Group 1 (Table 2).
Clinical Phenotypes.
Abnormal non stress test and/or abnormal contraction stress test and/or abnormal intrapartum cardiotocography (prolonged bradycardia and/or prolonged tachycardia and or decrease of fetal heart rate variability and /or late decelerations).
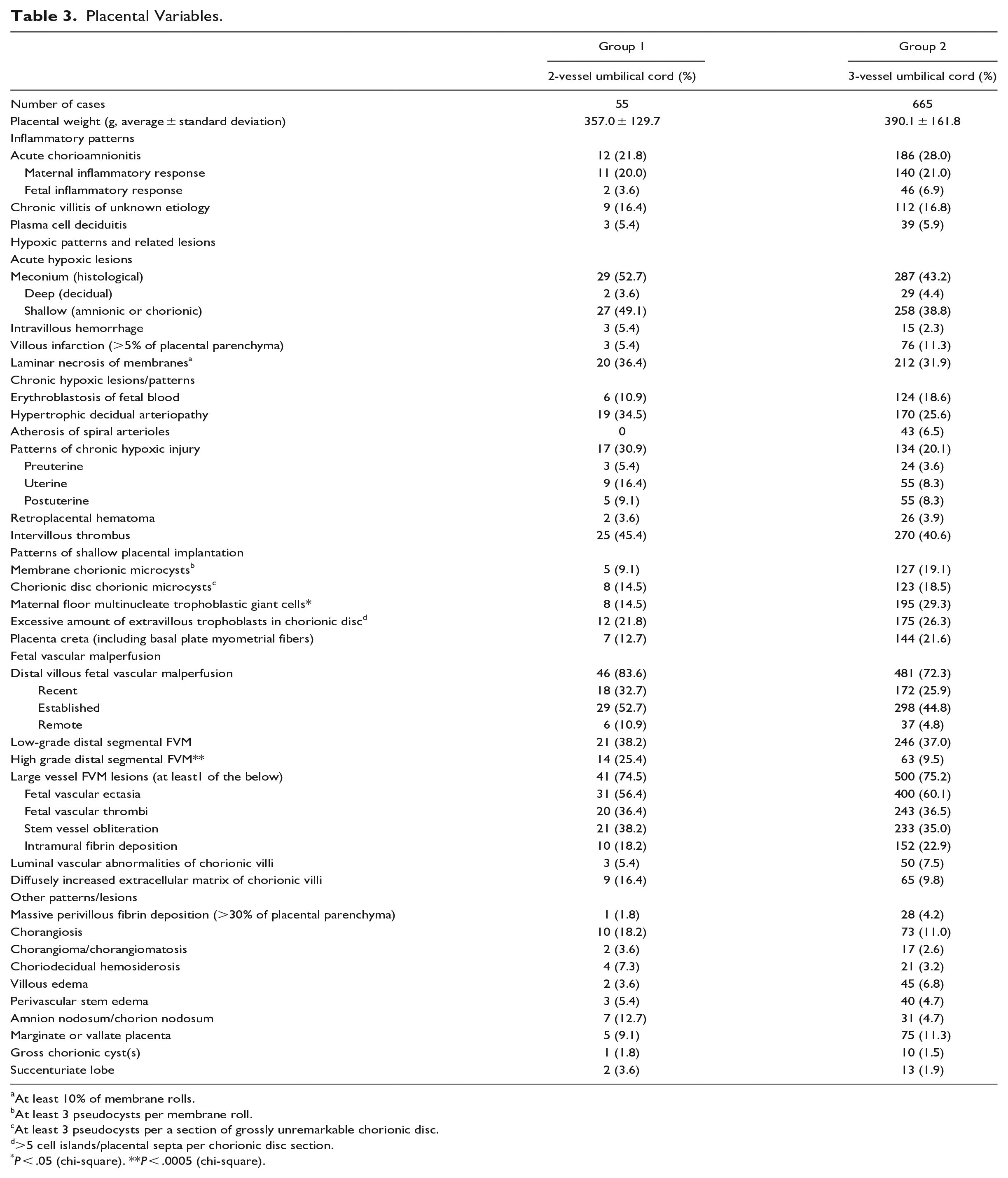
Comparing placental phenotypes (Table 3), Group 1 had a significantly higher frequency of high - grade distal FVM (more than 45 involved distal [terminal and/or intermediate] villi over more than 1 section of the placental parenchyma; combination of H&E staining 19 and CD34 immunostaining 22 ) than did Group 2 (Figure 2). However, the difference was not significant when H&E staining and CD34 immunostaining were considered separately; thus no statistically significant differences for recent, established, or remote distal villous FVM.
Placental Variables.
At least 10% of membrane rolls.
At least 3 pseudocysts per membrane roll.
At least 3 pseudocysts per a section of grossly unremarkable chorionic disc.
>5 cell islands/placental septa per chorionic disc section.
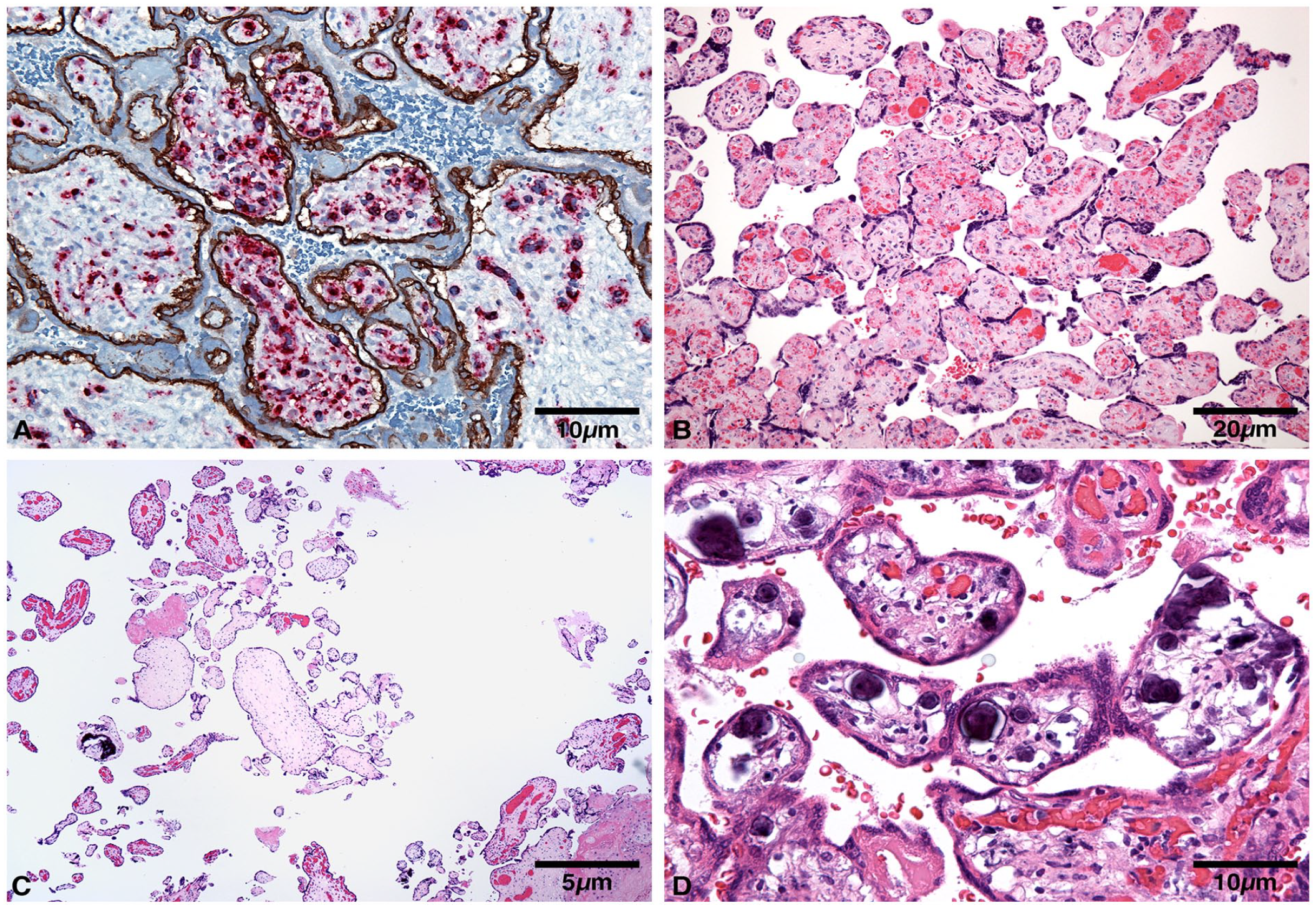
Distal villous vascular malperfusion. (A) Cluster of villi with endothelial fragmentation (CD34), 26 weeks, died on day 1 of life. (B) Clustered stromal vascular karyorrhexis, 39 weeks, double body, nuchal, and foot cord. (C) Cluster of avascular villi, 27 weeks. (D) Clustered mineralized villi, 26 weeks, died on day 1 of life.
There were no statistically significant differences in low grade distal FVM (3 or more foci of 2–4 of involved terminal villi, but fewer foci than the threshold for high grade distal villous FVM), or large vessel FVM or its components between the 2 groups. Maternal floor clusters of multinucleate trophoblasts 23 were less common in Group 1 than in Group 2, but there were no statistically significant differences in other lesions of shallow placental implantation. 24 Likewise, there were no statistically significant differences in inflammatory or hypoxic (acute or chronic) patterns/lesions of placental injury, or other placental lesions between the 2 groups. 20
Discussion
In the study population, SUA was associated with high-grade distal villous FVM only if the results of CD34 immunostaining 1 and clustered distal villous mineralization were analyzed in conjunction with the Amsterdam criteria for H&E staining (clustered sclerotic chorionic villi and stromal vascular karyorrhexis), 19 using the same numerical criteria for both stains. A similar pattern was observed in the placentas in cases involving neonatal intensive care unit stay—a short-term neonatal outcome—and neonatal neurological complications, which are more frequently associated with high-grade segmental FVM than with low-grade segmental FVM and global FVM in general and equally frequently in high-grade segmental FVM diagnosed on H&E staining and CD34 immunostaining and/or mineralization histochemistry. 25 This is true for all unselected cases, and not limited only to fetal growth restriction. 25 Lower odds of maternal vascular malperfusion than of fetal growth restriction birth with trivascular UC have been observed.16,17 Therefore, CD34 immunostaining increases the sensitivity of placental diagnosis of FVM not only in stillbirths, 1 but also in SUA. In our study, the combined number of cases with recent, established, and remote FVM was higher than the number of constituent placentas with recent (Figure 2(a) and (b)), established (Figure 2(c)) and remote (Figure 2(d)) distal villous FVM in both groups, which means that in several cases, the patterns were seen simultaneously in the same placenta (on-going FVM with temporal heterogeneity). 26
Among the analyzed UC abnormalities (Table 1), hypercoiled UC was the most common in this study. This and most other UC abnormalities, cause fetal blood stasis, resulting in a strong FVM predisposition, both large vessel and distal villous), which is referred to as stasis-induced FVM. However, among all anatomical UC abnormalities, SUA is not associated with fetal blood stasis or diminished placental blood flow separately because the single artery carries twice the blood volume of an artery in a trivascular umbilical cord.27,28 Therefore, fetal growth restriction in pregnancies with SUA is likely not caused by the decrease in the UC blood flow itself; rather it is due to wider pathologic processes resulting in an inherent defect in the placenta that is also associated with other fetal anomalies and high-grade distal villous FVM. The absence of a statistically significance difference in proximal FVM between the 2 groups (Table 3) indicates that high grade distal FVM in SUA pregnancies is most likely not stasis-induced. The associations shown in this analysis are particularly noteworthy as the cases in the studied population are predominantly fetal anomalies, which in the second half of pregnancy feature abnormal clinical phenotypes much more frequently than abnormal placental phenotypes. 29 When associated chromosomal abnormalities are also present, villous edema and erythroblastosis of fetal blood are likely due to fetal heart failure. 29 Mass-forming fetal anomalies feature placental histological lesions of shallow placental implantation, diffuse chronic hypoxic patterns, and FVM lesions that are likely stasis-induced. 29 The studied population of placentas (both Groups 1 and 2) featured a high prevalence of FVM, particularly large vessel FVM. This might be because the placentas were from patients with high-risk pregnancies, particularly fetal anomalies involving fetal heart failure and UC abnormalities. Individual types of large vessel FVM may also be less specific than distal FVM lesions. Some researchers claim that fetal vascular ectasia is an artefact of fixation. 30 Even if this is true, it is not relevant to this analysis because the overwhelming majority of placentas in our department are freshly grossed and photographed; only rarely for cases received from outside hospitals, the placentas are received in formalin. In addition, the difference in the prevalence of large vessel FVM between both groups was not statistically significant (Table 3). Hypercoiled UC, but not other UC abnormalities, were previously found in heart malformations and mass-forming fetal anomalies. 29 Most of such cases are in-house cases with congenital malformations, requiring EXIT procedures, and congenital heart disease in which, UC abnormalities range from 12% to 25%. 31 Our analysis indeed showed1 of the features of shallow placental implantation (decidual clusters of multinucleate trophoblasts) to be significantly more common in Group 2 than in Group 1, with trivascular UC. Decidual multinucleated giant cells are reportedly more common in UC abnormalities. 32
No features of fetal hypoxia of maternal vascular malperfusion origin were observed in placentas with SUA. The finding regarding clinical UC compromise (variable decelerations) in Group 1 is also meaningful, as confirmation should not be expected on placental examination if no morphological UC abnormality or abnormal UC insertion is found. 33 Lesions of shallow placental implantation are more commonly associated with distal villous and global FVM, abnormal UC coiling, and other UC abnormalities (excluding SUA). 24
The strength of this study is that all consecutive cases were analyzed and they were not from a specific clinical group of cases. In addition, CD34 immunostaining, which significantly increases the sensitivity of placental examination, was used for all cases. A limitation of this study is that the analyzed population consisted predominantly of fetal anomalies, and a smaller subset of other clinical conditions associated with FVM. However, UC abnormalities have been demonstrated to play a much larger role in the development of FVM than other clinical conditions (hypertensive conditions of pregnancy, diabetes mellitus, infections). 34 Additionally, placental vascular lesions (maternal vascular malperfusion and FVM) are inherently more difficult to reproducibly diagnose and grade than inflammatory lesions (villitis of unknown etiology and acute chorioamnionitis). 18
In summary, SUA is a specific cord anomaly predisposing to remote, advanced, and recent FVM, with a pathogenesis partially different from that of stasis induced FVM, likely because of the inherent conditions associated with SUA. CD34 immunostaining, by revealing recent FVM, increases the sensitivity of diagnosing distal villous FVM, as it is may diagnose the condition in H&E-negative cases.
Footnotes
Author Note
Presented as poster at the Spring Meeting of the Society for Pediatric Pathology, New Orleans, Louisiana, March 10–11, 2023.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
