Abstract
Background:
We previously identified placental lesions associated with stillbirths of varying gestational ages (GA) using advanced feature analysis. We further investigated the relationships between placental lesions and cause of death in stillbirths within these GA ranges.
Methods:
Using data from the Stillbirth Collaborative Research Network, we derived a sample of stillbirths who underwent placental examination and Initial Causes of Fetal Death (INCODE) evaluation for determining cause of death. We then compared the rates of causes of death within and among GA ranges (extreme preterm stillbirth [PTSB] [<28 weeks], early PTSB [28–336/7 weeks], late PTSB [34–366/7 weeks], term stillbirth [≥37 weeks]) according to the presence of these lesions.
Results:
We evaluated 352 stillbirths. In extreme PTSB, obstetric complications and infections were associated with acute funisitis. In early PTSB, uteroplacental insufficiency was associated with parenchymal infarcts. In term stillbirth (vs early PTSB), increased syncytial knots were associated with umbilical cord causes and infection.
Conclusions:
Placental lesions of high importance in distinguishing stillbirths at different GAs are associated with specific causes of death. This information is important in relating the presence of placental lesions and fetal death and in helping to understand etiologies of stillbirths at different GAs.
Introduction
Placental examination remains an important tool in investigating the causes behind adverse perinatal outcomes, particularly stillbirth. 1 This group of authors (Cersonsky et al. 2 ) previously tried to understand which lesions are most important in delineating stillbirths at different gestational ages (GAs). Using kernel principal covariates regression to assess feature importance, we identified the following lesions as important at different GAs: acute funisitis, accelerated villous maturity, and nucleated fetal red blood cells (RBCs) were important among extreme preterm stillbirth (PTSB, <28 weeks); parenchymal infarcts were important among early PTSB (28–336/7 weeks); and multifocal meconium and increased syncytial knots were important among both early PTSB and term stillbirths (≥37 weeks). 3
While this knowledge is powerful in helping relate placental features to etiologies of stillbirth, such information becomes even more vital when lesions are related to known cause of death. A diverse range of placental pathologies can contribute to intrauterine fetal death, including pathology of the placental bed, developmental placental dysfunction, and umbilical cord complications. 4 However, a single placental lesion can be related to multiple conditions; for example, while avascular villi can result from disrupted placental blood flow due to fetal vascular malperfusion, similar changes can also be found in the setting of fetal demise and complete vascular involution.5,6 Therefore, it is important to relate significant lesions to their respective causes of death in order to truly comprehensively characterize the differences in placentas associated with fetal demise according to gestational age.
We sought to further understand how those important lesions identified in our prior analyses related to specific causes of death within those GA ranges. This information may be valuable in determining both preventative measures that can be taken to prevent stillbirths and also to help reduce the high number of unexplained stillbirths through postmortem examination.7,8
Methods
Cohort Selection
Data were derived from the Stillbirth Collaborative Research Network (SCRN), a study of stillbirths and livebirths conducted in the United States from 2006 to 2009. Inclusion/exclusion criteria from our previous study (delivery of a singleton stillbirth >20 weeks GA, complete placental examination) were utilized to select participants for these analyses, in addition to the requirement that each have cause of death determined and recorded for inclusion. 2
Placental and umbilical cord examination has been described previously; these protocols were standardized across institutions to reduce inter-pathologist differences. 9 Lesions were assessed in these analyses according to our previous results, including acute funisitis, acute umbilical arteritis, acute chorionic vasculitis, diffuse terminal villous immaturity, parenchymal infarct, retroplacental hematoma, accelerated villous maturity, nucleated fetal red blood cells, placental disc (hemorrhage, increased syncytial knots, and trophoblast proliferation), decidual vasculopathy, and multifocal reactive amniocytes, calcifications, or meconium. 2 Cause of death was determined using the Initial Causes of Fetal Death (INCODE) system, which evaluates conditions as present, possible cause of death, or probable cause of death within the categories of hypertensive disorders, maternal health conditions, obstetric complications, fetal condition, placental causes (related to specific placental lesions), umbilical cord causes, and infection. 10
Statistical Analysis
Stillbirths were categorized according to GA at delivery using the same schema as our previous analyses: extreme PTSB (<28 weeks), early PTSB (28–336/7 weeks), late PTSB (34–366/7 weeks), and term stillbirth (≥37 weeks). Prevalence of each cause of death was first compared across GA ranges. Then, placentas with lesions unique to each GA range (according to the results of our prior analyses) were isolated within each range; prevalence of each cause of death was then compared within that GA range across the different lesions. For example, all extreme PTSB placentas with acute funisitis, accelerated villous maturity, and nucleated fetal RBCs were identified and the prevalence of each cause of death was compared across lesion groups. Placentas with more than 1 lesion were not included in the comparative analyses due to small sample sizes. Chi-Square tests were used for all assessments; P-values <.05 were considered significant. All analyses were conducted using R software (version 4.2.3).
Ethical Approval
Institutional Review Boards and Data Coordinating and Analysis Center approval was obtained for study procedures; participants provided written, informed consent. 11
Results
Of the 445 participants included in our prior analyses, 352 had cause of death determined by INCODE and were included in our assessments. Table 1 details causes of death across GA ranges. Extreme PTSB was associated more frequently with obstetric-related cause of death, umbilical cord causes, and extensive decidual vasculopathy. Early PTSB was associated more frequently with hypertension-related cause of death. Late PTSB was associated more frequently with death due to chromosomal anomalies. Term stillbirth was associated more frequently with death due to diabetes, umbilical cord entrapment, and infection (particularly chorioamnionitis on placental examination).
Cause of Death by GA Range.
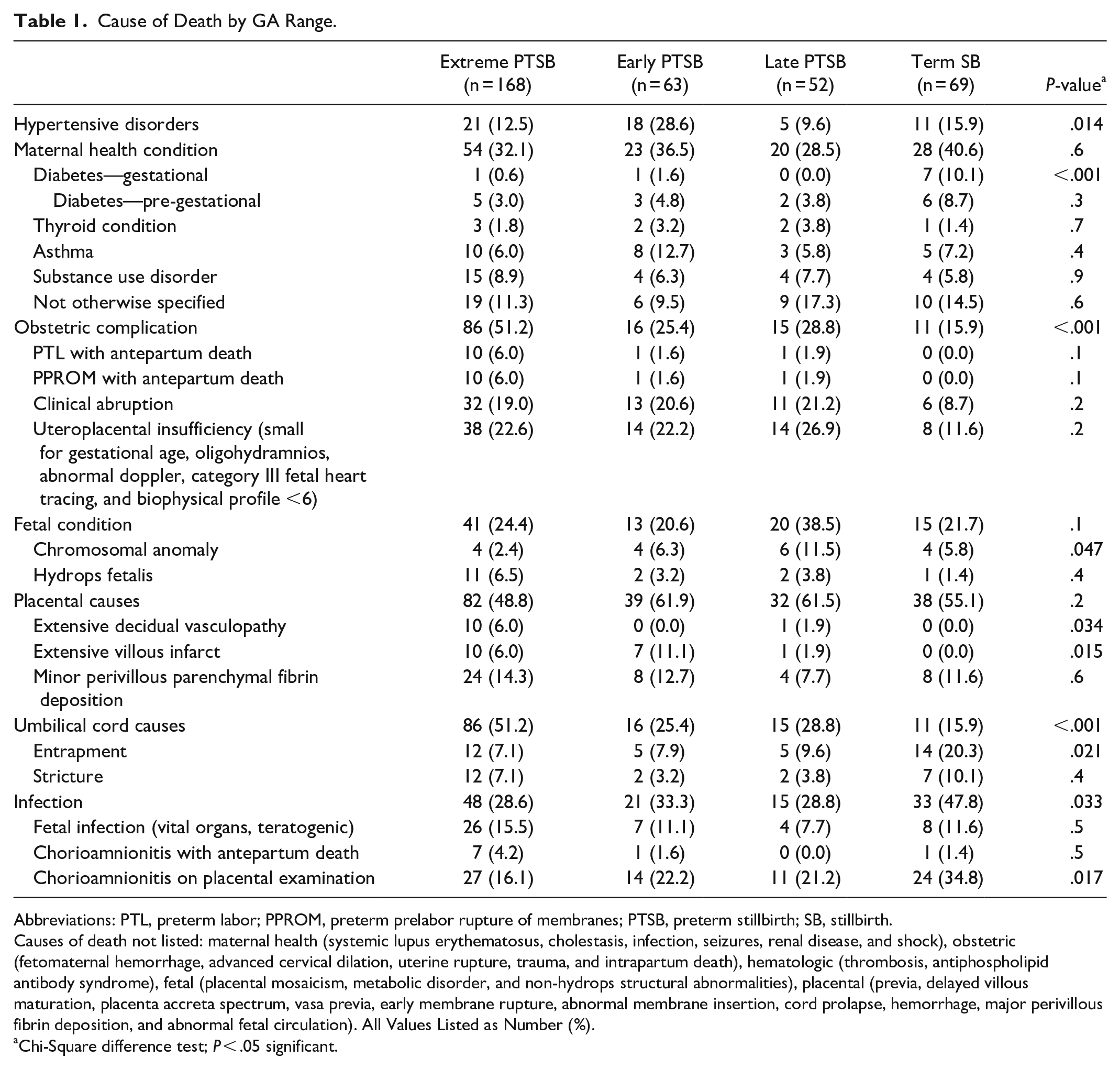
Abbreviations: PTL, preterm labor; PPROM, preterm prelabor rupture of membranes; PTSB, preterm stillbirth; SB, stillbirth.
Causes of death not listed: maternal health (systemic lupus erythematosus, cholestasis, infection, seizures, renal disease, and shock), obstetric (fetomaternal hemorrhage, advanced cervical dilation, uterine rupture, trauma, and intrapartum death), hematologic (thrombosis, antiphospholipid antibody syndrome), fetal (placental mosaicism, metabolic disorder, and non-hydrops structural abnormalities), placental (previa, delayed villous maturation, placenta accreta spectrum, vasa previa, early membrane rupture, abnormal membrane insertion, cord prolapse, hemorrhage, major perivillous fibrin deposition, and abnormal fetal circulation). All Values Listed as Number (%).
Chi-Square difference test; P < .05 significant.
Among extreme PTSB (Table 2), those with acute funisitis were more likely to have obstetric complications (85.7%, particularly preterm labor [PTL] or preterm pre-labor rupture of membranes [PPROM] with antepartum death) and infection (66.7%, fetal infection or chorioamnionitis on placental examination) compared to those with accelerated villous maturity or nucleated fetal RBCs. Though hypertension-related death was common among those with nucleated fetal RBCs (35.1%), this difference was not statistically significant. Placental causes were attributed frequently in those with accelerated villous maturity (57.6%), though not statistically significant. Among early PTSB (Table 3), parenchymal infarcts were associated with death due to uteroplacental insufficiency (37.0%) compared to increased syncytial knots and multifocal meconium. No other significant differences were observed across these lesions in this category.
Lesions and Causes of Death Among Extreme Preterm Stillbirths.
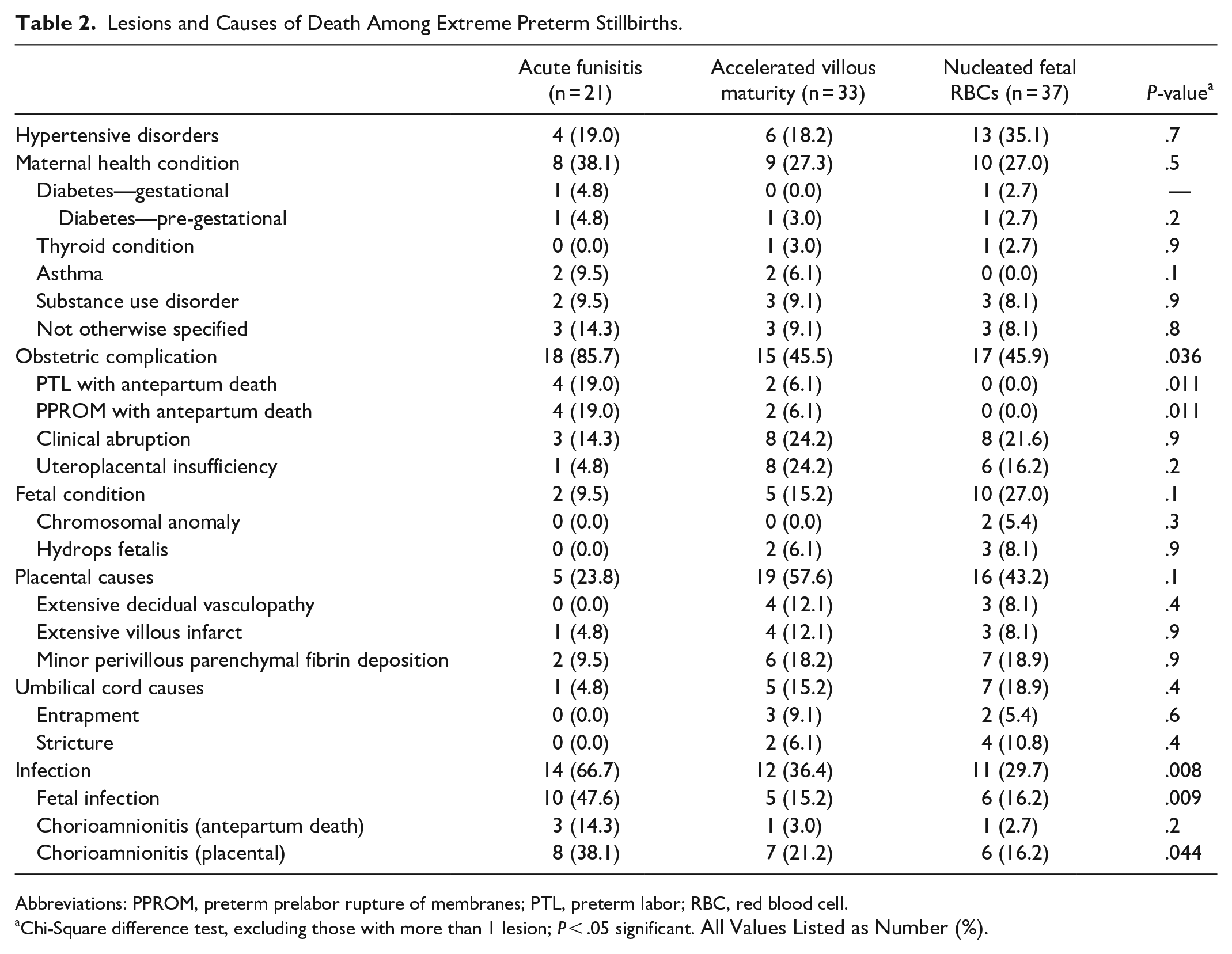
Abbreviations: PPROM, preterm prelabor rupture of membranes; PTL, preterm labor; RBC, red blood cell.
Chi-Square difference test, excluding those with more than 1 lesion; P < .05 significant. All Values Listed as Number (%).
Lesions and Causes of Death Among Early Preterm Stillbirths and Term Stillbirths.
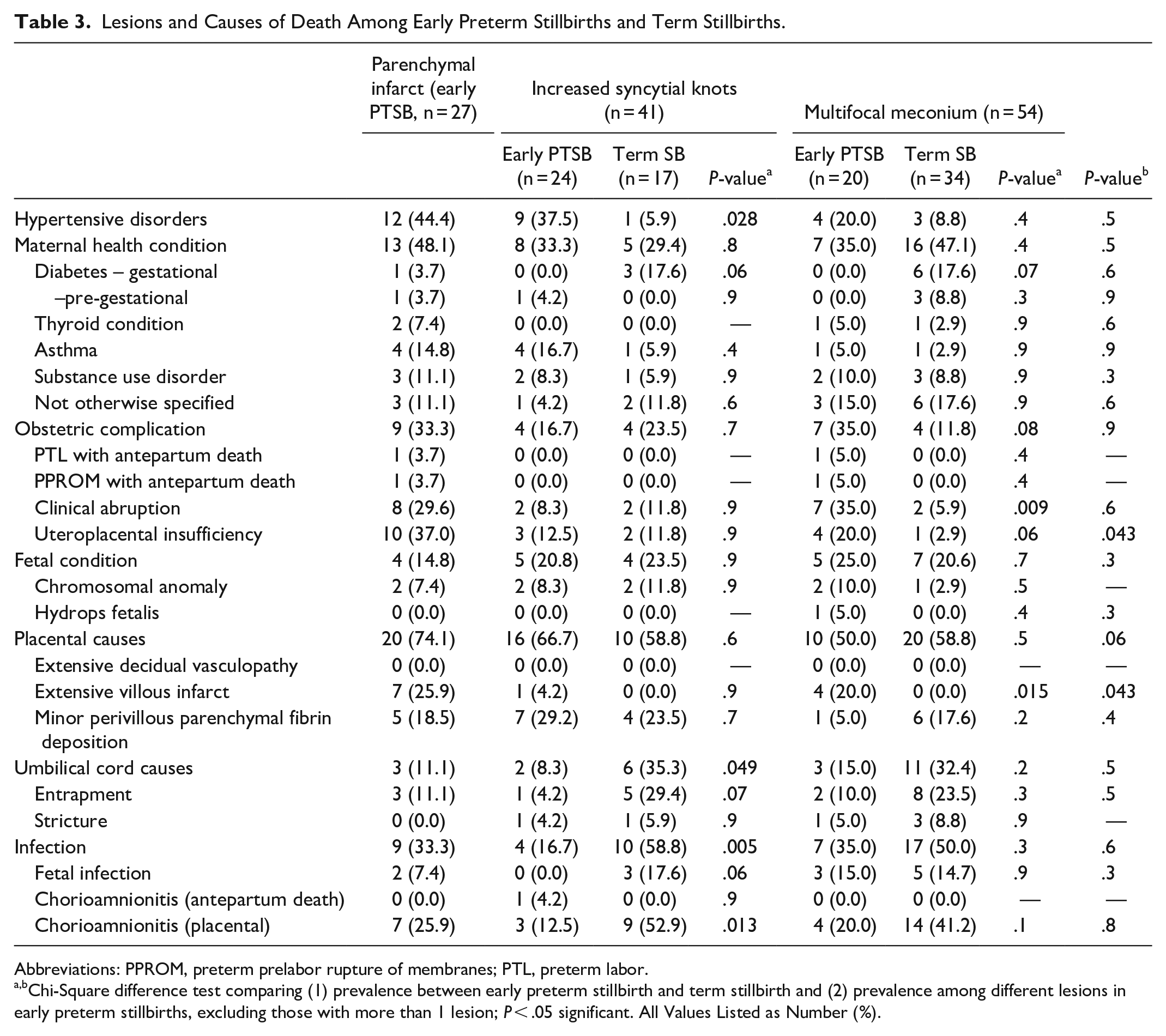
Abbreviations: PPROM, preterm prelabor rupture of membranes; PTL, preterm labor.
Chi-Square difference test comparing (1) prevalence between early preterm stillbirth and term stillbirth and (2) prevalence among different lesions in early preterm stillbirths, excluding those with more than 1 lesion; P < .05 significant. All Values Listed as Number (%).
We also compared causes of death across GA ranges within those with lesions identified to have similar importance in those ranges in our prior analyses. When comparing those with increased syncytial knots between early PTSB and term stillbirth (Table 3), hypertension-related death was more associated with early PTSB (P = .028), while umbilical cord causes (such as entrapment and/or stricture) and infection were more common among term stillbirth (P = .049 and .005, respectively). Clinical abruption and extensive villous infarcts were more common among early PTSB with multifocal meconium (vs term stillbirth with the same lesions; P = .009 and .015, respectively). We also compared causes of death between late PTSB and term stillbirth with diffuse terminal villous immaturity, trophoblast proliferation, decidual vasculopathy, and multifocal reactive amniocytes; there were no significant differences in these comparisons.
Discussion
In this follow-up study, we sought to characterize causes of death associated with placental lesions associated with stillbirth at different GAs. We found that, among extreme PTSB, acute funisitis is associated with obstetric complications and infection; among early PTSB, parenchymal infarcts were associated with uteroplacental insufficiency; and, among term stillbirth (vs early PTSB), increased syncytial knots were associated with umbilical cord causes and infection (vs hypertensive causes in early PTSB). While several of these associations confirm what is known about placental lesions and fetal death, they also add to our understanding of differing etiologies of stillbirths at different GAs.
Very early stillbirths (our extreme PTSB group, <28 weeks) were associated with obstetric complications leading to antepartum death; we now link those complications to potential underlying acute funisitis within this specific GA group. 12 This group also showed a strong relationship between acute funisitis and death attributed to chorioamnionitis on placental examination, which is common in deliveries at 21–24 weeks at gestation. 13 The association between acute funisitis and obstetric and infectious causes of death adds to the debate regarding the 2 mechanisms that potentially relate these findings: either ascending microbial infection or a sterile immune response stimulate extreme preterm labor leading to antepartum (not intrapartum) stillbirth.14,15 Regardless, we have identified chorioamnionitis and obstetric complications as causes of death distinct to extreme PTSB with acute funisitis, which is a clinical relationship important to identify in postmortem examination.
Our early PTSB group (28–336/7weeks) was distinct in our previous analyses for the presence of placental parenchymal infarcts, which were most associated with death due to uteroplacental insufficiency. Given the frequent findings of maternal vascular malperfusion (MVM) in placentas associated with severe early-onset preeclampsia (PEC, defined as onset of hypertension and proteinuria prior to 34 weeks) and clinical uteroplacental insufficiency, this is not unexpected.16,17 Though early-onset PEC is known to be associated with higher risk of stillbirth compared to late-onset PEC, in our analyses, stillbirths were most associated with parenchymal infarcts and uteroplacental insufficiency specifically in those at 28–336/7 weeks. This raises an important question in clinical management of such pregnancies diagnosed prior to 28 weeks, as the relationship between placental lesions of PEC and death due to uteroplacental insufficiencies and hypertensive disorders is weaker at this GA while neonatal morbidity is at its highest.
Finally, we found that term stillbirth with placental syncytial knots were more likely to have more frequent umbilical cord or infectious cause of death compared to early PTSB with these lesions. Increased syncytial knots are often associated with PEC (and are included in the definition of MVM), but findings in term stillbirths were not related to these etiologies. 16 Umbilical cord causes, usually entrapment, stricture, or knots, are more often associated with findings of fetal vascular malperfusion (FVM), particularly fetal hypoxia, rather than MVM. 6 It is more likely that the relationship between increased syncytial knots in term stillbirth and umbilical cord causes of death is instead attributable to increased syncytial knots present at placentas associated later gestational ages in general. Infectious deaths (chorioamnionitis on placental examination, in this GA group) are also not typically associated with increased syncytial knots or MVM. Possibly, histologic chorioamnionitis and MVM may coexist in term placentas that have concomitant chorioamnionitis and PEC.18,19
Our study is robust, offering a comprehensive examination of the relationships between important placental lesions and cause of death in stillbirths, but should still be interpreted in the context of several limitations. While we were able to investigate a range of causes of stillbirth, our sample size limited our ability to assess the differences among conditions that were merely “present” vs those identified as “possible” or “probable” cause of death as part of INCODE assessment. Furthermore, we excluded from our comparative analyses those placentas that had more than 1 lesion due to small sample size, which discounts the potential additive effects of multiple lesions of similar etiologies (e.g., multiple lesions within the category of MVM). These limitations may be mitigated in a future study with a larger group of stillbirths. We are furthermore unable to describe grade of MVM or FVM in the placentas in our study, which would provide detail regarding the degree of inflammatory response present. Finally, the study and pathologic assessments were conducted prior to the publication of the Amsterdam criteria; while we use these definitions to identify placentas with MVM and FVM, the results should be interpreted in the context of new standards. 20
We were able to identify causes of death that were strongly associated with important placental lesions at different gestational ages. This information offers valuable insights not just into the etiologies of stillbirths, but into the importance of placental examination for uncovering lesions related to these etiologies. This information may prove useful for targeting different etiologic pathways of stillbirth at appropriate times in gestation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development: U10-HD045953 Brown University, Rhode Island; U10-HD045925 Emory University, Georgia; U10-HD045952 University of Texas Medical Branch at Galveston, Texas; U10-HDO45955 University of Texas Health Sciences Center at San Antonio, Texas; U10-HD045944 University of Utah Health Sciences Center, Utah; and U01-HD045954 RTI International, RTP. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
