Abstract
Introduction:
Chronic histiocytic intervillositis (CHI) is associated with adverse pregnancy outcomes and high recurrence risk. Recent studies suggest that CHI may represent a host-vs-graft rejection, and that C4d immunostain can be used as a marker for complement activation and antibody-mediated rejection in the CHI.
Materials and Methods:
This retrospective cohort study focused on 5 fetal autopsy cases associated with CHI (5 index cases) from 5 women. We analyzed placentas from the index cases (fetal autopsy cases associated with CHI) and placentas from the women’s previous and subsequent pregnancies. We assessed the presence and extent of CHI and C4d immunostaining in these placentas. We evaluated each available placenta and graded the severity of CHI as either <50% or ≥50%. Additionally, we conducted C4d immunostaining on one representative section from each placenta and graded the staining levels as follows: 0+ for staining <5%; 1+ for staining between 5% and <25%; 2+ for staining between 25% and <75%; and 3+ for staining ≥75%.
Results:
Three of the 5 women had pregnancies prior to their index cases (fetal autopsy cases associated with CHI). Despite the absence of CHI in their initial pregnancies, the placentas displayed positive C4d staining with grades of 1+, 3+, and 3+, respectively. These results suggest the presence of complement activation and antibody-mediated rejection in placentas from their prior pregnancies without CHI. Three of the 5 women received immunomodulatory therapy after experiencing pregnancy losses associated with CHI. After treatment, 2 of these women had live births at 35 and 37 gestational weeks, respectively, while the third had a stillbirth at 25 gestational weeks. The severity of CHI and the degree of C4d staining in the placentas decreased in all 3 cases following immunomodulatory therapies. Specifically, the level of C4d staining decreased from 3+ to 2+, 2+ to 0+, and 3+ to 1+ in these 3 cases, respectively.
Discussion:
In women with a history of recurrent pregnancy loss associated with CHI, C4d immunostaining was present in the placentas from their previous non-CHI pregnancies, suggesting activation of the classical complement pathway and antibody-mediated reaction in their prior non-CHI pregnancies before the development of CHI in subsequent pregnancies. Immunomodulatory therapy may improve pregnancy outcomes by reducing complement activation, as shown by the reduction of C4d immunopositivity in the placentas after immunomodulatory treatment. Although we believe that the study provides valuable insights, we acknowledge that there are limitations to the findings. Therefore, to further elucidate the pathogenesis of CHI, additional research efforts with a collaborative and multidisciplinary approach are necessary.
Keywords
Introduction
Chronic histiocytic intervillositis (CHI) is characterized by histiocytes in the intervillous space of the placenta. CHI is associated with adverse pregnancy outcomes, such as fetal growth restriction and fetal death, and has a high recurrence rate in subsequent pregnancies.1,2
The pathogenesis of CHI is not entirely understood, though there is increasing evidence that CHI may represent a host-vs-graft reaction akin to transplant rejection.3-5 Recently, Benachi et al 5 studied 2 women with a history of recurrent CHI. Both women had a normal first pregnancy, followed by recurrent pregnancy losses associated with CHI. In both women, very high levels of complement-fixing, fetus-specific antibodies targeting mismatched human leukocyte antigen (HLA) alleles, harbored by the 2 paternal haplotypes, were identified. In addition, the placentas with CHI demonstrated strong HLA and C4d immunopositivity along the microvillous surface of the syncytiotrophoblasts, which are the outermost cell layer of the chorionic villi and are thus in direct contact with maternal blood in the intervillous space. Based on these findings, Benachi et al 5 proposed that CHI could be a manifestation of placental alloantibody-mediated rejection.
Recipients of solid organ allografts may produce donor-specific, anti-HLA antibodies against HLAs found in the transplanted organs, which can lead to complement activation and antibody-mediated rejection. C4d, a byproduct of the classical complement pathway, is inactive. However, it binds covalently to target cells at the site of complement activation and becomes tightly attached to the tissue, serving as a marker of antibody-mediated tissue injury. As a result, C4d immunostaining is commonly used to identify antibody-mediated rejection in solid organ transplantation. 6 Since the placenta contains both maternal and paternal antigens, it is considered a semi-allograft. Therefore, the presence of C4d immunostaining in placentas affected by CHI could indicate complement activation secondary to the binding of maternal alloantibodies to mismatched antigens of paternal origin on the syncytiotrophoblasts.4,5,7,8
As there is increasing evidence pointing toward pathogenic immune mechanisms as possible causes of CHI, current treatments, while not standardized, typically involve the use of immunomodulatory agents to prevent CHI recurrence and reduce the likelihood of adverse outcomes in future pregnancies.9-11
This study has 2 main objectives. The first objective is to investigate whether there is any evidence of C4d immunostaining in placentas that were previously considered normal and did not have any signs of CHI, but belonged to women who later experienced pregnancy loss associated with CHI after a normal first pregnancy. The presence of C4d immunostaining in these prior placentas could suggest complement activation and alloantibody-mediated rejection during their previous pregnancies, despite the absence of histiocytic infiltrates in the placentas. The second objective of this study is to determine if immunomodulatory therapy can decrease the severity of CHI and C4d immunostaining in the subsequent pregnancies of women with a history of pregnancy loss associated with CHI.
Methods
Retrospective Cohort Study
This retrospective cohort study was approved by the University of Calgary Conjoint Health Review Ethics Board (REB 14-2391).
Fetal Autopsies and Diagnosis of CHI
Fetal autopsies performed at the Alberta Children’s Hospital/University of Calgary/Alberta Precision Laboratories from January 1, 2017 to October 30, 2021, formed the basis of this cohort.
All fetal autopsies were performed after receiving parental consent. The fetus and placenta were sent together to the pathology department at Alberta Children’s Hospital. Clinical information was collected from electronic medical records. A complete autopsy examination consisted of photography, whole-body X-rays, external examination, internal examination, cytogenetics analysis, microscopic examination of fetal tissue, placental examination, and ancillary studies, such as bacterial and fungal cultures and molecular studies for viral infections. Autopsy findings were entered into a single, final autopsy report. Protocols for placental examination were described previously. 12 Percentiles of birthweights and placental weights were assigned using previously published ranges.13,14 All autopsies were performed by anatomical pathologists specializing in perinatal pathology.
A pathologic diagnosis of “fetal death associated with CHI” was made when 2 conditions were met: (1) CHI was detected in the placenta, and (2) no other potential cause of fetal death was identified, such as congenital malformations, infectious etiology, or genetic aberrations.
A diagnosis of CHI was made when histiocytic infiltrates were observed in the intervillous space of the placenta (Figure 1(A)). However, since certain infections, including cytomegalovirus and SARS-CoV-2, are known to cause mononuclear inflammatory cells to infiltrate the placenta,15,16 an extensive investigation was conducted to eliminate any possible infectious causes of the histiocytic infiltrates observed in cases of CHI. As part of standardized care in Alberta, all pregnant women underwent routine prenatal screenings for syphilis, human immunodeficiency virus, hepatitis B, hepatitis C, gonorrhea, and chlamydia, as well as testing for immunity to rubella and varicella zoster. If there were any suspicion of an infectious disease in either the mother or fetus, further tests would be conducted, which might include serology for cytomegalovirus, toxoplasmosis, parvovirus B19, herpes simplex virus, and nucleic acid testing for SARS-CoV-2 in the mother, as well as bacterial cultures and viral nucleic acid tests and immunohistochemical stains on fetal and placental tissues. If no infections were identified during the infectious workup, a diagnosis of fetal death associated with CHI, but not associated with any infectious agent, would be established.
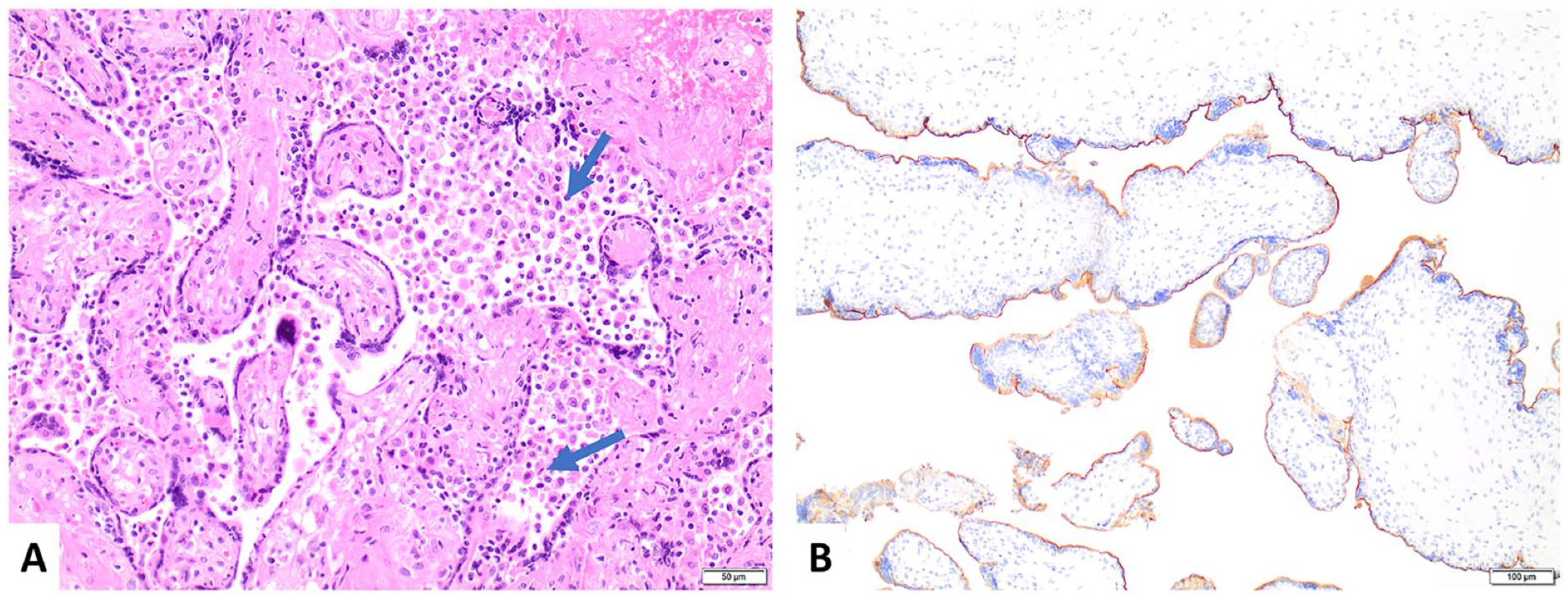
(A) Chronic histiocytic intervillositis is characterized by histiocytic infiltrates (arrows) within the intervillous space (hematoxylin and eosin). (B) C4d immunostaining along the apical microvillous surface of syncytiotrophoblasts in the placenta of a woman with recurrent pregnancy loss associated with chronic histiocytic intervillositis (C4d immunostain).
Identification of Index Cases, As Well As Previous and Subsequent Placental Pathology Reports
Fetal autopsy reports from January 1, 2017 to October 30, 2021, were extracted from the Cerner (Kansas City, MO, USA) Millennium Laboratory Information System (LIS), which included text-based diagnostic fields and the provincial healthcare number (PHN). A Perl function was used to scan the diagnosis field for “chronic intervillositis,” “chronic histiocytic intervillositis,” or “histiocytic intervillositis.” Fetal autopsy reports containing these terms were manually reviewed by a perinatal pathologist (ESC) to confirm a diagnosis of fetal death associated with CHI, and these formed the index cases.
To identify the mother’s placental pathology reports before and after the index case, we utilized the mother’s PHN and searched the LIS. Only mothers who had more than one placental pathology report available in the LIS were included in this study.
Additional Perinatal Data
Maternal age, gestational age (GA), gravida (G), parity (P), livebirth (L), and stillbirth (S) were obtained from medical records, as well as from the Alberta Perinatal Health Program (APHP), which collects information from registered midwives and hospital facilities across Alberta, regarding birth events in the province of Alberta, Canada.
Grading of CHI
A perinatal pathologist (ESC) reviewed slides of placentas obtained from the index cases as well as from the mothers’ previous and subsequent pregnancies. The severity of CHI was assessed and graded according to the grading scheme proposed by Ongaro and Terry, 17 which classifies the extent of CHI as present in either less than 50% (<50%) or equal to or more than 50% (≥50%) of the intervillous space. This grading scheme was selected for its high reproducibility.
Grading of C4d Immunostaining
A representative block was selected from each placenta for C4d immunostaining. Briefly, 4-μm sections of formalin-fixed, paraffin-embedded placental tissues were deparaffinized, rehydrated, and pre-treated with a target retrieval solution, followed by incubation with C4d rabbit polyclonal antibody (Biomedica, Vienna, Austria) diluted at 1:200 in Background Reducing Diluent (Agilent, Santa Clara, CA, USA). The signal was developed using a high pH, DAB+ Substrate Chromogen visualization system (Agilent, Santa Clara, CA, USA) on a Dako Omnis automated stainer (Agilent, Santa Clara, CA, USA). C4d immunostaining of the apical microvillous surface of syncytiotrophoblasts was given a score based on a grading scheme proposed by Bendon et al, 4 as follows: 0+ = <5%; 1+ = 5 to <25%; 2+ = 25 to <75%; 3+ ≥75% (Figure 1(B)). Note that there is no universally accepted grading scheme for C4d immunostaining in placentas. However, we chose to use the grading scheme that Bendon et al 4 proposed to maintain consistency with previous research. Five first-trimester, 5 second-trimester, and 5 third-trimester normal placentas without chronic or acute inflammation or other histologic abnormalities served as negative controls.
Results
The non-inflamed placental controls showed appropriately negative results for C4d immunostaining, with staining observed in less than 5% of the syncytiotrophoblasts’ apical microvillous surface.
Case 1
A 30-year-old G3P1L1 woman with no pre-existing medical conditions experienced a pregnancy loss associated with CHI at 13 weeks GA during her third pregnancy (index case). Examination of the placenta from this pregnancy revealed that it had ≥50% CHI and 1+ C4d staining. (Figure 2(C) and 2(D)).
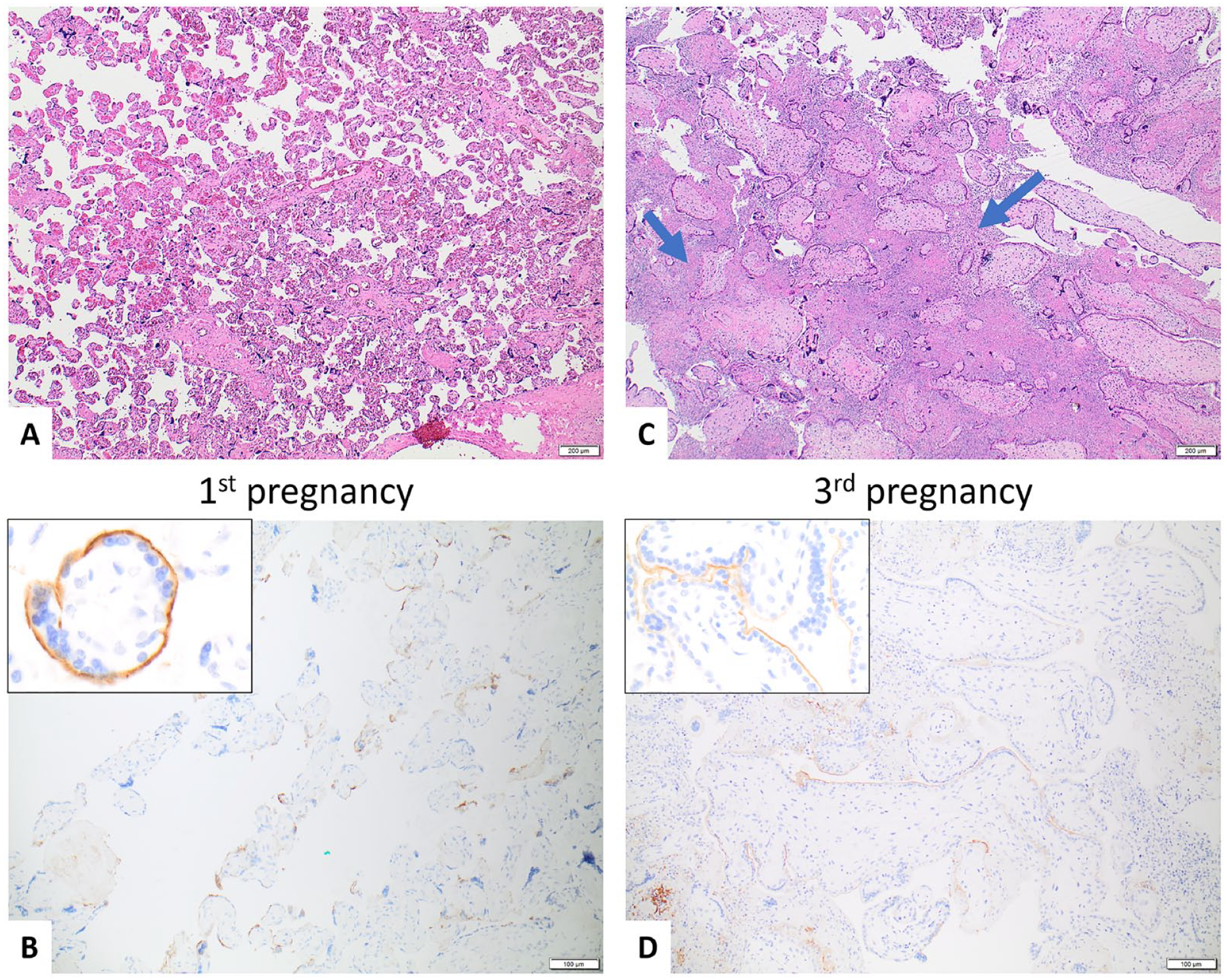
Case 1—30-year-old G3P1L1.
Upon further review of the woman’s medical history, it was found that her first pregnancy resulted in a liveborn at 38 weeks GA, and the placenta from that pregnancy showed no histologic evidence of CHI. However, 1+ C4d staining was observed in the placenta from her first pregnancy (Figure 2(A) and 2(B)). The woman’s second pregnancy resulted in fetal demise before 20 weeks GA, and unfortunately, the placenta was not available for review. Characteristics of pregnancies and placentas are listed in Table 1.
Characteristics of Pregnancies and Placentas From Case 1.
Case 2
A 41-year-old G9P2L2 woman with no pre-existing medical conditions experienced a pregnancy loss at 14 weeks GA associated with CHI in her ninth pregnancy (index case). Examination of the placenta from this pregnancy revealed that it had ≥50% CHI and 3+ C4d staining (Figure 3E and 3F).
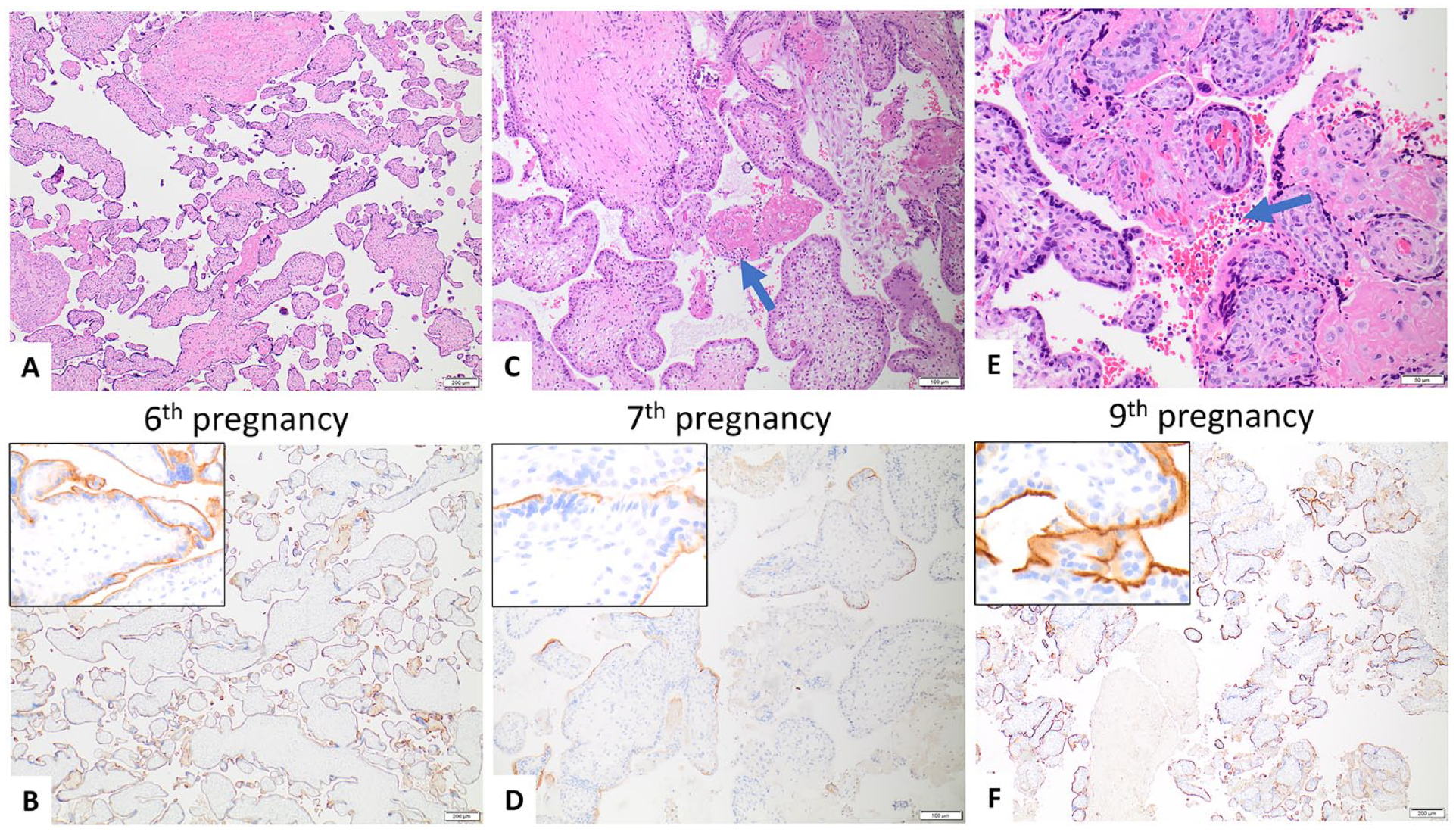
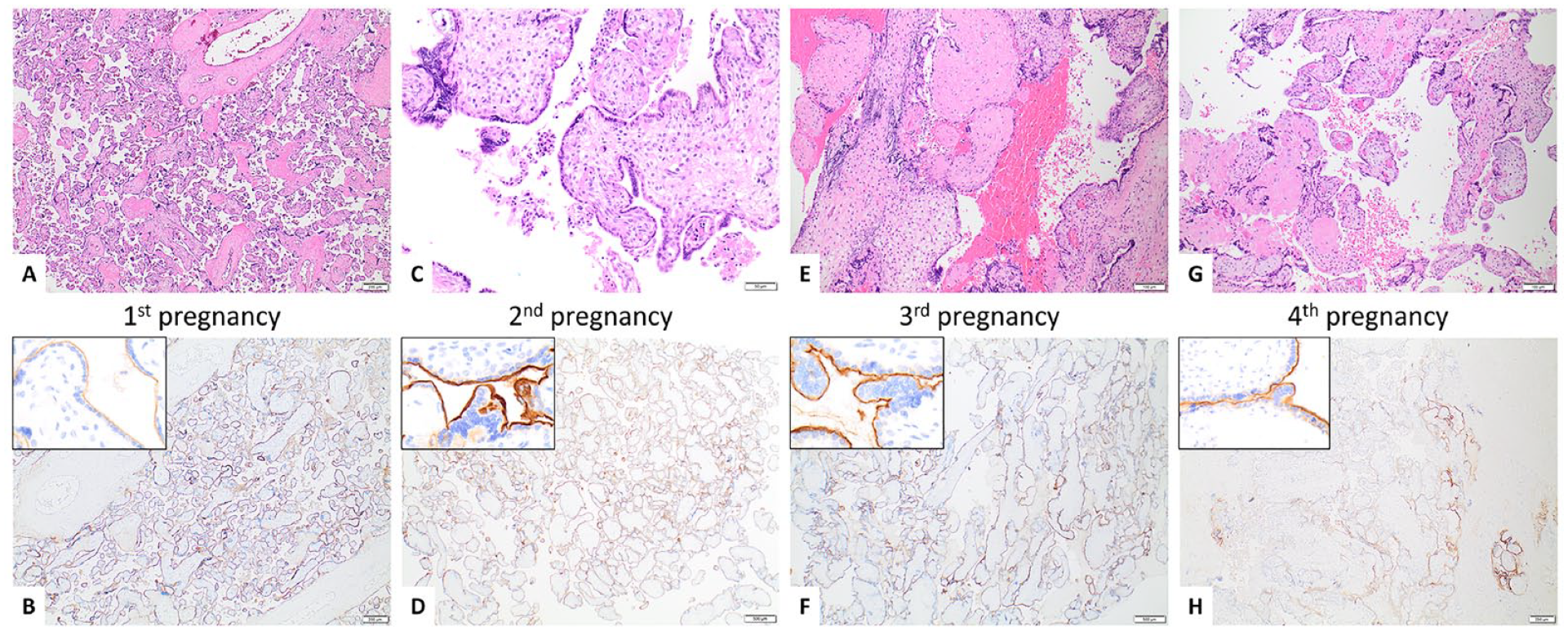
Case 2—41-year-old G9P2L2. After 2 term liveborns in her first 2 pregnancies, she had recurrent first- and second-trimester pregnancy losses. Only the placentas from her sixth, seventh, and ninth pregnancies were available for pathologic examination.
Upon further review of the woman’s medical history, it was found that her first and second pregnancies resulted in term liveborn, followed by 7 pregnancy losses in the first and second trimesters. Unfortunately, the placentas from the first 5 and eighth pregnancies were not available for pathologic examination. However, the placentas from her sixth, seventh, and ninth pregnancies were available for review.
The placenta from her sixth pregnancy did not show CHI, but C4d staining was diffusely positive (3+) (Figure 3(A) and 3(B)). The placenta from her seventh pregnancy showed <50% CHI and 2+ C4d staining (Figure 3(C) and 3(D)). Characteristics of pregnancies and placentas are listed in Table 2.
Characteristics of Pregnancies and Placentas From Case 2.
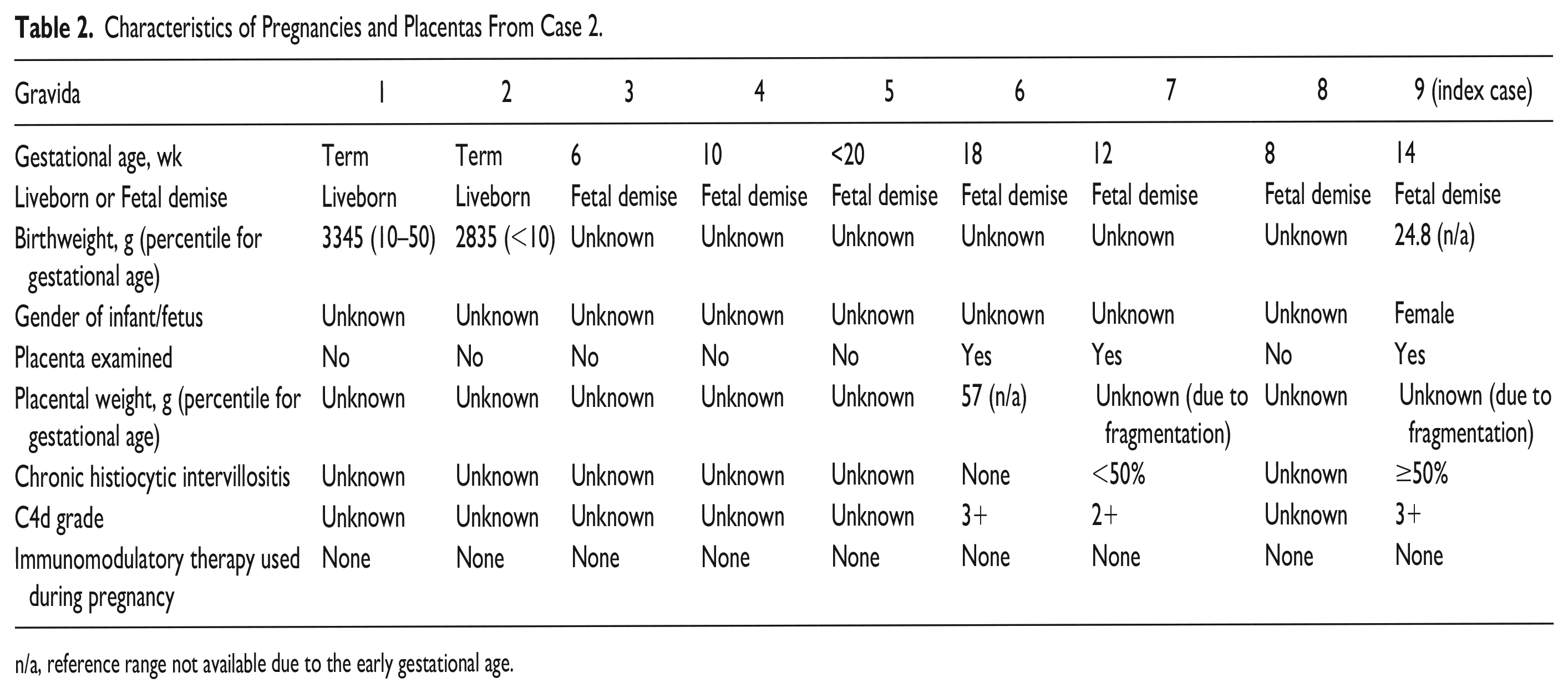
n/a, reference range not available due to the early gestational age.
Case 3
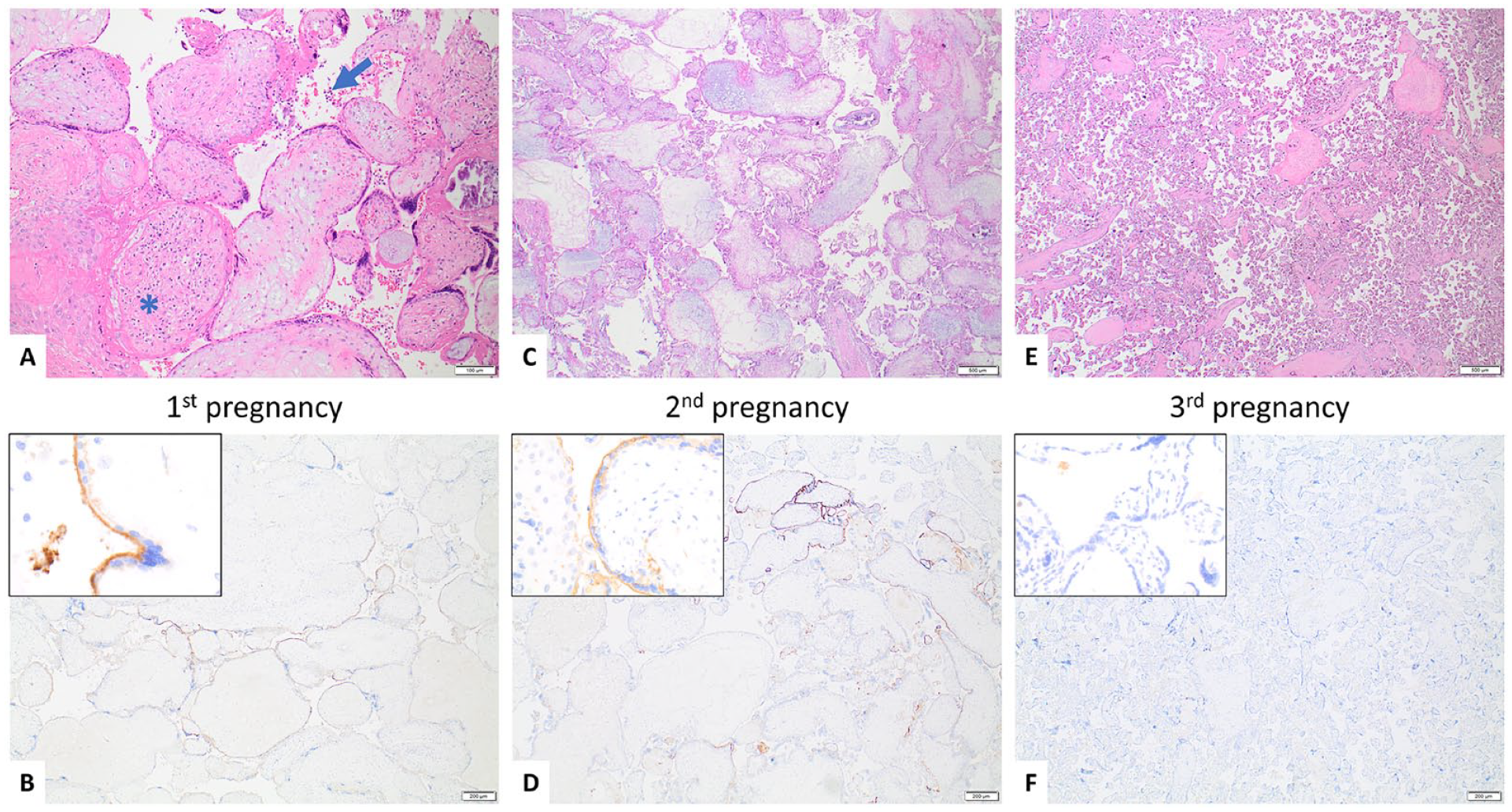
A 34-year-old G4P3L1 woman with no pre-existing medical conditions experienced a pregnancy loss associated with CHI at 21 weeks GA during her second pregnancy (index case). Pathologic examination of the placenta from this pregnancy revealed that it had <50% CHI and 3+ C4d staining (Figure 4(C) and 4(D)). During her third pregnancy, the woman suffered another pregnancy loss at 19 weeks GA, and the placenta from this pregnancy also showed <50% CHI and 3+ C4d staining (Figure 4(E) and 4(F)). Upon review of the woman’s obstetric history, it was noted that in her first pregnancy, she delivered a term male liveborn who was small-for-gestational-age. The placenta from the first pregnancy was small and had no CHI, but it showed diffuse positive staining (3+) for C4d (Figure 4(A) and 4(B)).
Case 3—34-year-old G4P3L1.
In her fourth pregnancy, the woman was given intravenous immunoglobulin (IVIG), prednisone, low-molecular-weight heparin (LMWH), and acetylsalicylic acid (ASA) starting at 11 weeks, GA due to her recurrent CHI-associated pregnancy losses. Unfortunately, despite receiving immunomodulatory treatment, she experienced another fetal loss at 25 weeks GA. However, the placenta from this pregnancy showed a reduction in CHI (<50%, though subjectively less severe compared to previous placentas) and in C4d staining severity (2+) (Figure 4(G) and 4(H)) compared to the placentas from her previous pregnancies, and interestingly, the post-treatment placenta showed diffuse subamniotic calcification, which was highly suggestive of immune complex deposition based on electron microscopy examination. Characteristics of pregnancies and placentas are listed in Table 3.
Characteristics of Pregnancies and Placentas From Case 3.
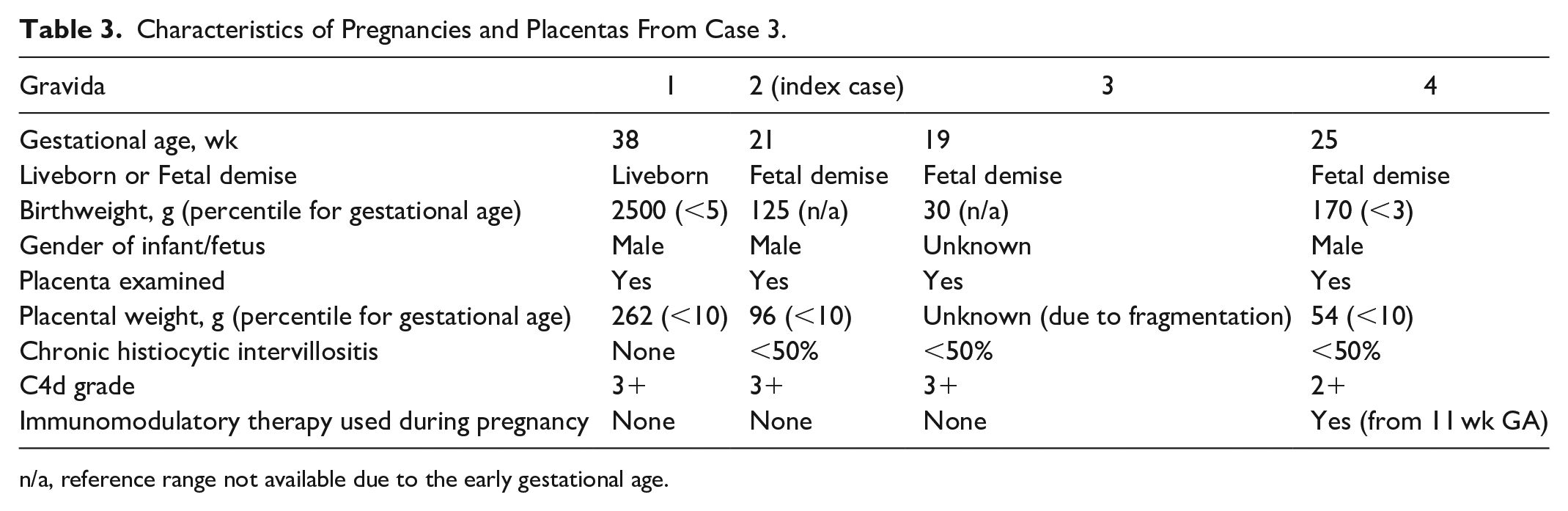
n/a, reference range not available due to the early gestational age.
Case 4
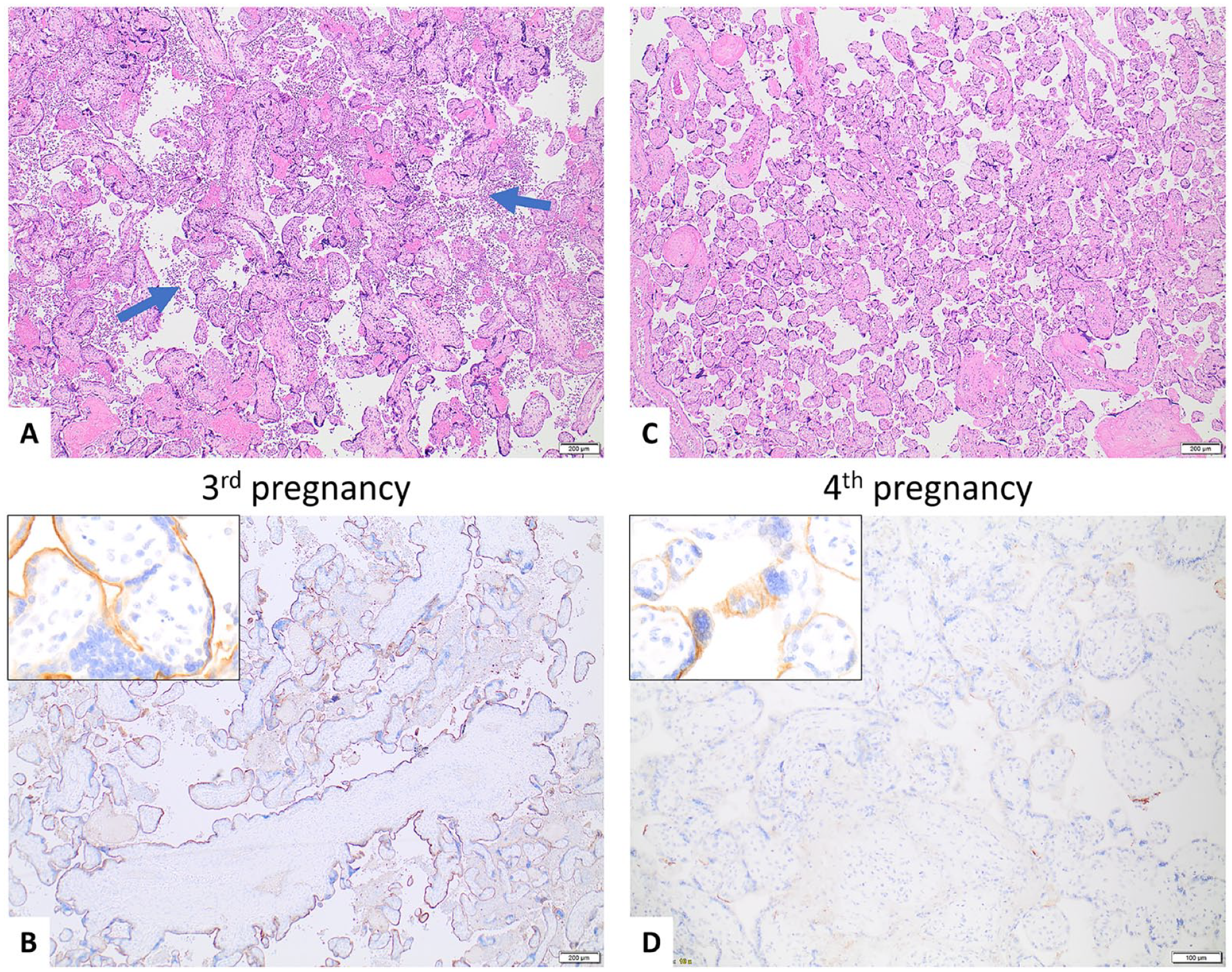
A 32-year-old G3P2L1 woman with a history of hypothyroidism suffered a pregnancy loss associated with CHI at 22 weeks GA during her first pregnancy (index case). Pathologic examination of the placenta from this pregnancy demonstrated hydropic changes in the villi, <50% CHI, and 2+ C4d staining (Figure 5(A) and 5(B)). Her next (second) pregnancy also resulted in a loss at 19 weeks GA associated with CHI (<50%), C4d staining (2+), and hydropic changes in the villi (Figure 5C and 5D).
Case 4—32-year-old G3P2L1 woman.
Due to her recurrent CHI-related pregnancy losses, she received immunomodulatory treatment in her third pregnancy. The treatment protocol included hydroxychloroquine, dalteparin, IVIG, and aspirin, starting from 10 to 11 weeks GA. Additionally, she took levothyroxine for hypothyroidism. Following treatment, she gave birth to a liveborn at 37 weeks GA, and the placenta from this pregnancy showed no CHI or C4d immunopositivity (Figure 5E and 5F). The placenta also demonstrated no hydropic changes. Characteristics of pregnancies and placentas are listed in Table 4.
Characteristics of Pregnancies and Placentas From Case 4.
n/a, reference range not available due to the early gestational age.
Case 5
A 34-year-old G4P3L2 woman with no pre-existing medical conditions had a pregnancy loss associated with CHI at 20 weeks GA during her third pregnancy (index case). The placenta showed ≥50% CHI and 3+ C4d staining (Figure 6A and 6B). The woman’s first pregnancy resulted in a term liveborn, followed by 2 pregnancies that ended in loss, one at 8 weeks GA (second pregnancy) and the other at 20 weeks GA (third pregnancy, index case). The placentas from her first 2 pregnancies were not available for examination.
Case 5—34-year-old G4P3L2 woman. She had a term liveborn in her first pregnancy and a fetal demise at 8 weeks GA in her second pregnancy. Placentas from her first 2 pregnancies were not available for pathologic examination.
Given the diagnosis of CHI in her third pregnancy, the woman underwent immunomodulatory treatment during her fourth pregnancy. The treatment protocol included aspirin (taken since preconception), Plaquenil (taken since preconception), prednisone (starting at 12 weeks GA), IVIG (starting at 13 weeks GA), and LMWH (starting at 12 weeks GA). Following treatment, she gave birth to a liveborn at 35 weeks GA; the baby had a normal birthweight. The placenta was small (<10th percentile) and continued to show CHI (<50%) and C4d immunostaining (1+) (Figure 6C and 6D). However, the degrees of CHI and C4d immunostaining were much less intense than in the previous placenta. Notably, the placenta from the fourth pregnancy also demonstrated acute necrotizing chorioamnionitis with fetal inflammatory response and patchy villitis of unknown etiology. Characteristics of pregnancies and placentas are listed in Table 5.
Characteristics of Pregnancies and Placentas From Case 5.
n/a, reference range not available due to the early gestational age.
Discussion
There is accumulating evidence that CHI may be a manifestation of placental antibody-mediated rejection,3,4,5 possibly caused by maternal alloantibodies targeting paternally inherited antigens in the placenta. 5
C4d immunostain is commonly used as a marker for detecting antibody-mediated rejection in solid organ transplantation. 6 In this context, when a recipient of a transplanted organ produces donor-specific anti-HLA antibodies, the classical complement pathway is activated, leading to the deposition of membrane attack complex on the transplanted organ. This, in turn, can cause tissue damage and rejection of the transplanted organ. The deposition of C4d, a degradation product of the classical complement pathway, is also observed in such instances. Although C4d has no known biological function, it has an internal thioester that can form stable covalent bonds with target cells. Therefore, the presence of C4d immunopositivity serves as a marker for antibody-mediated rejection.6,18,19
Previous studies have found that C4d immunostaining is typically absent in normal placentas3,20 but frequently positive on the apical, microvillous surface of syncytiotrophoblasts in placentas with CHI. 4 The discovery that C4d immunopositivity is present in placentas with CHI lends support to the idea that CHI could be a result of the placenta being targeted by antibodies from the mother.3,4,5 This is because the outermost layer of the placental chorionic villi (syncytiotrophoblasts) is in direct contact with the mother’s blood and any maternal antibodies present in the intervillous space (maternal circulation).
Additionally, in a recent study, 2 women experienced recurrent pregnancy losses associated with CHI after their first uncomplicated pregnancies. 5 These 2 mothers were tested after recurrent pregnancy losses, and both were found to have very high levels of complement-fixing, fetus-specific antibodies. These antibodies were directed against mismatched fetal HLAs of paternal origin, meaning that the mothers were making alloantibodies against the fetal HLAs that were paternally inherited. This suggests that CHI may be a manifestation of placental antibody-mediated rejection, secondary to maternal alloantibodies directed against paternally inherited antigens in the placenta. 5
Women who experience recurrent pregnancy losses due to CHI often have a live birth in their first pregnancy without any signs of CHI in their initial placenta, but subsequently suffer from multiple pregnancy losses associated with CHI.5,21 Despite the absence of any histologic evidence of CHI in the placentas from their first uncomplicated pregnancies, it is possible that the mothers may have already started producing alloantibodies against mismatched fetal antigens during their initial pregnancies. To test this hypothesis, we identified women who had pregnancy losses associated with CHI and examined the placentas from their prior pregnancies to determine if there were any signs of complement activation and antibody-mediated rejection, using C4d immunostain as a surrogate marker.
During the study period, 3 cases (Cases 1–3) were identified in which the mothers had uncomplicated pregnancies before experiencing fetal losses associated with CHI. In Cases 1 and 3, both mothers had a liveborn baby at 38 weeks GA in their first pregnancy, and the placentas did not show any signs of CHI. However, C4d immunostaining was unexpectedly positive (1+ and 3+, respectively). In Case 2, the mother had 2 term liveborn babies in her first and second pregnancies, followed by 7 pregnancy losses. The placentas from her first 5 and eighth pregnancies were not available for examination. In her sixth pregnancy, she experienced a fetal loss at 18 weeks GA. The placenta from this pregnancy did not show any signs of CHI upon examination, but diffuse positive C4d immunostaining (3+) was observed. The mother also had CHI with positive C4d immunostaining in her seventh and ninth pregnancies, both of which resulted in fetal demise. These cases highlight that C4d immunostaining could be positive in the initial “normal” non-CHI placentas from women who subsequently develop CHI.
Given the above findings, we speculated whether these women might have already produced alloantibodies against mismatched, paternally inherited antigens in the placenta during their initial “uncomplicated” pregnancy, causing complement activation, as evidenced by C4d immunopositivity, even before the appearance of histiocytic infiltrates in the intervillous space (i.e., CHI). It is worth noting that although there were no signs of histiocytic infiltrates in the previous “normal” non-CHI placentas, evidence of placental tissue injury was still observed, including accelerated villous maturation and increased syncytial knots. In addition, the babies and placentas from their initial “normal” pregnancies were smaller than expected for their gestational age, despite the absence of maternal medical conditions known to cause intrauterine growth restriction, such as maternal hypertension and diabetes.
Furthermore, we hypothesized that women who received immunomodulatory therapy for the treatment of CHI might have a decrease in histiocytic infiltrates and C4d immunostaining in their subsequent post-therapy placentas, compared to their placentas from previous, untreated pregnancies. During the study period, we identified 3 women (Cases 3, 4, and 5) who underwent immunomodulatory therapy following their CHI diagnoses. Two of these women (Cases 4 and 5) had live births at 37 and 35 weeks GA, respectively, after receiving immunomodulatory treatment. The third woman (Case 3) unfortunately had a stillbirth at 25 weeks GA, despite receiving treatment. The better clinical outcomes observed in the 2 women (Cases 4 and 5) who received immunomodulatory therapy align with the findings of earlier studies that demonstrated improved pregnancy outcomes after immunomodulatory treatment.11,22 In all 3 cases (Cases 3, 4, and 5), the severity of CHI and C4d staining in the placentas decreased following immunomodulatory treatments. These results indicate that immunomodulatory therapy could potentially lower the severity of CHI and the risk of CHI recurrence and fetal demise, perhaps by downregulating the classic complement pathway. Notably, in Case 3, despite a reduction in CHI severity and C4d staining in the placenta following immunomodulatory therapy, the pregnancy unfortunately resulted in a stillbirth at 25 weeks GA, and abnormal histologic findings were noted in the post-therapy placenta, which had been previously reported by our colleagues. 23 Specifically, the placenta showed diffuse subamniotic calcifications, which were highly suggestive of immune complex deposition based on electron microscopy. Our colleagues hypothesized that IVIG treatment may have contributed to immune complex deposition and subsequent dystrophic calcifications in this placenta. 23 We wonder if this abnormal response to immunomodulatory therapy may have played a role in the adverse outcome of this pregnancy.
Additionally, an interesting observation was made in Case 4. The woman in Case 4 experienced 2 pregnancy losses at 22 and 19 weeks GA, respectively, and both placentas showed similar histologic changes, including hydropic villi with myxoid stroma, increased perivillous fibrin deposition, CHI, and chronic villitis. Although the fetuses were not hydropic, the hydropic and myxoid changes in the chorionic villi were very pronounced. After receiving immunomodulatory therapy, the woman gave birth to a liveborn at 37 weeks GA, and the placenta showed no signs of CHI, C4d staining, or hydropic changes. Since hydropic villi with myxoid stroma is not typically seen in cases of CHI, there could be other factors contributing to the abnormal placental findings observed in the woman’s first 2 placentas. It is notable that immunomodulatory therapy was effective in treating this case of CHI, despite the uncommon histologic features in the placentas.
With the exception of the placenta from the fourth pregnancy in Case 3 which had subamniotic calcifications, and the placentas from the first 2 pregnancies in Case 4 which showed hydropic and myxoid villi, the remaining placentas in this study cohort did not present any unusual histologic abnormalities.
At present, there is no standard treatment for CHI. However, several studies have reported improved outcomes in patients who underwent immunomodulatory therapy.11,22 Given the serious consequences of CHI if left untreated, it is crucial for pathologists and obstetricians to be aware of this condition and have a high level of suspicion when a woman has a history of multiple unexplained early pregnancy losses. Early detection and appropriate management of CHI may lead to better outcomes for both the mother and the fetus.
Although other researchers and we have confirmed that C4d immunostaining is present in placentas with CHI, we do not believe that C4d immunohistochemistry is necessary for the daily clinical practice of placental pathology, even in cases of CHI. This is because CHI is a histological diagnosis based on the examination of hematoxylin-and-eosin-stained tissue slides. Furthermore, we found no consistent correlation between the severity of CHI and the severity of C4d immunostaining among different patients. However, C4d immunostaining might help identify women with recurrent pregnancy loss.
Our study has several notable strengths. First, all index cases underwent complete fetal autopsies by pathologists with expertise in perinatal pathology. This ensured that fetal demise was correctly attributed to CHI and not other underlying causes. Secondly, in Alberta, Canada, all pregnant women receive standardized and comprehensive prenatal care, and in cases of pregnancy loss, thorough maternal laboratory workup is frequently conducted. This comprehensive prenatal care and post-mortem investigation help prevent and rule out many confounding factors, including infections. Thirdly, Alberta maintains a well-established perinatal database, providing us with access to high-quality demographic data. These factors enhance the reliability and validity of our findings and lend greater confidence to our conclusions.
Our study also has several notable limitations. First, it is a small study. However, due to the rarity of fetal demise attributed to CHI and the even more infrequent occurrence of a complete autopsy being conducted, we believe that our study is reasonably sized. Nonetheless, we acknowledge that with such a small sample size, it is not possible to draw definitive conclusions and that this study could only serve as a pilot observational study. Secondly, not all placentas from all pregnancies of the women in our study were available for pathologic examination. Thus, in some of these women, it was not possible to follow their complete obstetric history and examine the placentas from all their pregnancies. Thirdly, we could not verify the father’s identity in each case. However, according to patients’ records, the pregnancies were believed to be conceived with the same partner for each woman.
Finally, although C4d immunostaining is commonly used as a marker for antibody-mediated rejection in pathology, particularly in organ-transplantation pathology, it is not a specific marker for antibody-mediated rejection. While activation of the classical complement pathway can be caused by antibody-mediated rejection, it can also be caused by pathogens such as cytomegalovirus and SARS-CoV-2.24,25 Nonetheless, C4d in CMV-associated placentitis has a different immunostaining pattern (cytoplasmic staining of the villous cytotrophoblasts) 25 than that seen in CHI (linear staining of the microvillous surface of the syncytiotrophoblasts). C4d immunostaining has also been found in other placental disease processes, including villitis of unknown etiology, placental infarcts, placentas associated with preeclampsia, and recurrent miscarriages.3,26,29 It is important to note that all women included in this study did not have pre-existing medical conditions, such as autoimmune conditions or a history of preeclampsia, except for the woman in Case 4, who had hypothyroidism. Additionally, placental infarcts were not observed in any of the placentas examined in the study cohort. Furthermore, the presence of C4d immunostaining in placentas with villitis of unknown etiology does not contradict the association between C4d and placental antibody-mediated rejection. This is because there is evidence that villitis of unknown etiology, which is frequently accompanied by C4d immunopositivity, may actually be a host-vs-graft response.3,27 In other words, the presence of C4d in villitis of unknown etiology cases supports the idea that there is an immune response occurring in the placenta, rather than contradicting it. Despite the potential relevance of C4d immunohistochemistry in detecting antibody-mediated rejection in placentas, we acknowledge that C4d immunohistochemistry is not a direct indicator of antibody-mediated rejection but rather a surrogate marker for it. Additionally, it should be noted that most laboratories optimize C4d immunohistochemistry for solid organ transplant biopsies. This means that C4d immunostaining in placentas could be variable and not optimized, as it is not the primary focus of most clinical immunohistochemical laboratories. Moreover, a study has shown that C4d staining on paraffin sections can vary considerably between different laboratories, 28 which could further impact the reliability of C4d as a marker for complement activation and antibody-mediated rejection in placentas.
Although C4d immunostaining has been used in studies investigating the pathogenesis of CHI,3,4,5,30 these studies have been limited by their small sample size and the fact that the causes of CHI may be multifactorial. To fully understand the role of antibody-mediated rejection in CHI-related fetal losses, further investigation is necessary. Specifically, a prospective longitudinal study would be needed to determine if mothers produce complement-fixing, fetus-specific alloantibodies during their initial non-CHI pregnancies and if these alloantibodies are responsible for subsequent CHI-associated fetal losses. Additionally, such a study could assess whether immunomodulatory therapy could reduce the production of these alloantibodies and potentially prevent CHI-related complications. This type of prospective, collaborative, multidisciplinary study would involve collecting blood samples from primigravida women during their first and subsequent pregnancies to detect the presence of maternal alloantibodies targeting fetal antigens of paternal origin. In addition, all placentas would need to be submitted for pathologic examination. While C4d immunostaining may be a useful tool in such a study, it alone cannot fully answer these complex questions. A more comprehensive investigation is needed to understand the underlying pathogenesis of CHI and to develop potential therapeutic interventions for this serious condition.
Conclusions
In this small retrospective cohort study, we found that in women who had an initial uncomplicated pregnancy followed by recurrent pregnancy losses associated with chronic histiocytic intervillositis (CHI), C4d immunostaining was present in the placenta from their prior pregnancy, even though CHI was absent during that pregnancy. This suggests that activation of the classical complement pathway, possibly triggered by antibody-mediated rejection, occurred in the initial pregnancy before the development of CHI and pregnancy loss subsequently. Moreover, immunomodulatory therapy may improve pregnancy outcomes in women with a history of recurrent pregnancy loss associated with CHI, likely by reducing complement activation, as shown by the reduction of C4d immunopositivity in the placenta after immunomodulatory treatment. However, this is a small observational study with several limitations as stated in the above discussion, and additional larger studies involving multidisciplinary efforts are necessary to fully understand the underlying pathogenesis and treatment of CHI.
Footnotes
Acknowledgements
The authors would like to thank Charlotte Monroe for her invaluable administrative support and Kathryn Buchanan for her expertise in transcribing the autopsy and placental reports.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
