Abstract
Introduction
Coronavirus disease 19 (COVID-19) has been a global pandemic for almost 2 years. During this year, many of its mysteries have been unraveled. Interestingly, children seem less affected than adults and when infected, present with different symptomatology, that is, more gastro-intestinal symptoms and less pulmonary complaints.1,2 Reasons for this are largely unknown, but might be attributable to different expression of angiotensin-converting enzyme 2 (ACE2) between children and adults. ACE2 is a metallopeptidase also functioning as receptor for SARS-CoV and SARS-CoV-2,3,4 responsible for COVID-19.
Although in adults the presence of ACE2 mRNA has been established in virtually all organs, 5 studies on its age-related expression are limited6-11 and mainly used RNA sequencing to assess ACE2 localization. However, mRNA and protein expression patterns do not necessarily correspond due to processes as post-translational modification. Furthermore, the use of immunostaining is capable of showing ACE2 expression in different subsets of cells that make up the organ parenchyma. Insight into the ACE2 protein expression profile during development and childhood is of great importance in understanding SARS-CoV-2 pathogenesis and age-related symptomatology of COVID-19. Therefore, the present study qualitatively investigated ACE2 protein expression in various organs from fetuses, younger and older children, adolescents, and young adults.
Materials and Methods
Age and Cause of Death.

Abbreviation: GA: gestational age.
Hardy scale: (1) Violent and fast death (terminal phase < 10min); (2) fast death of natural causes (terminal phase estimated at < 1 hr); (3) intermediate death after a terminal phase of 1 to 24; (4) slow death after a long illness, with a terminal phase longer than 1 day; (0) ventilator case, all cases on a ventilator preceding death. (GTEx Consortium. Hardy scale description. [cited 6 Dec 2021]. Available: https://www.ncbi.nlm.nih.gov/projects/gap/cgi-in/variable.cgi?study_id=phs000424.v4.p1&phv=169092#:∼:text=Deathclassificationbasedonthe,phaseestimatedat%3C10min).
Results
ACE 2 Location and Subcellular Staining Patterns.
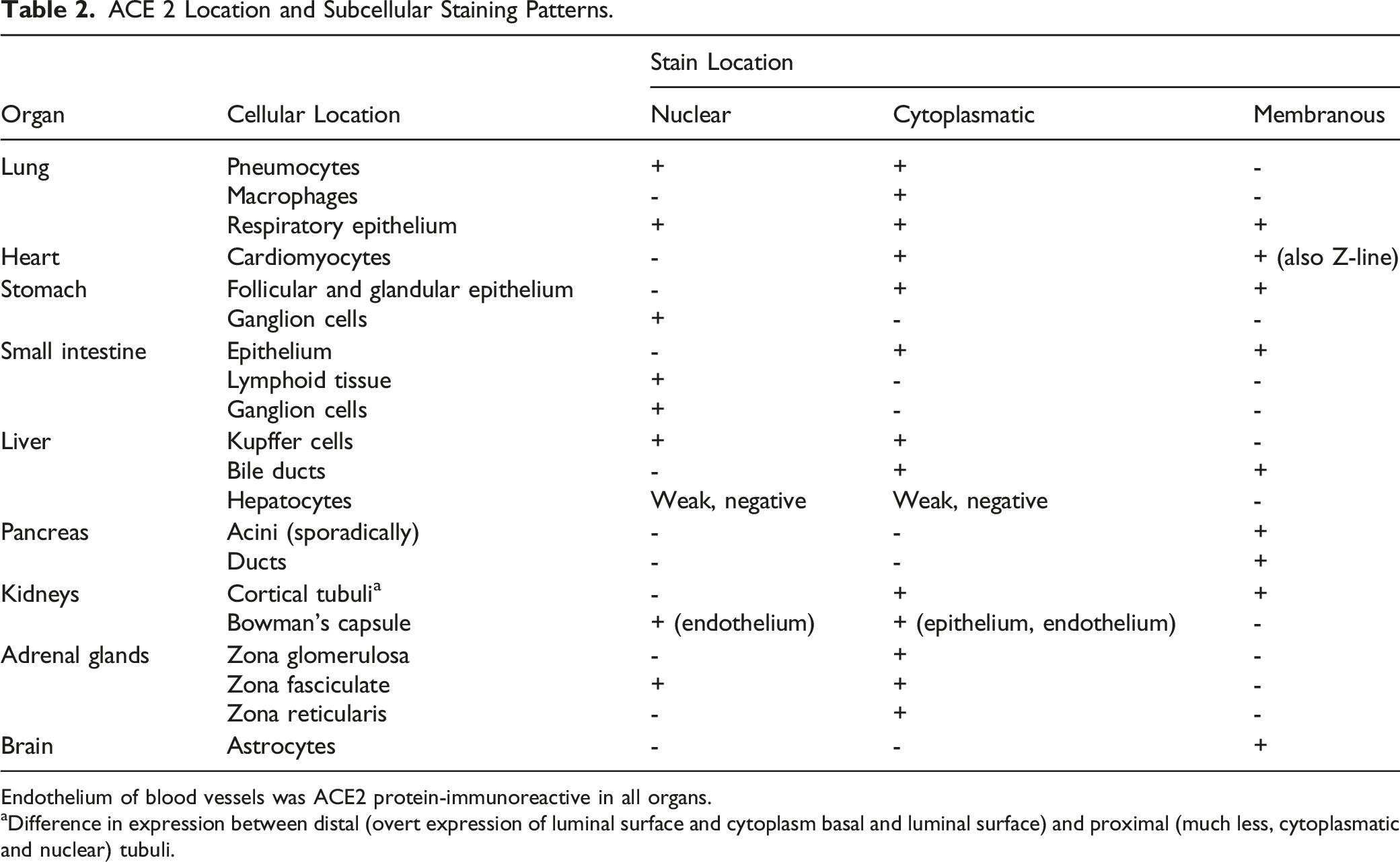
Endothelium of blood vessels was ACE2 protein-immunoreactive in all organs.
aDifference in expression between distal (overt expression of luminal surface and cytoplasm basal and luminal surface) and proximal (much less, cytoplasmatic and nuclear) tubuli.
In brain tissue, ACE2 was expressed in astrocytes and endothelial cells; no expression changes were detected amongst ages.
Discussion
Our study confirms that ACE2 is widely expressed during development and across all ages, paralleling findings of mRNA studies in adults. 5 Interestingly, we found previously unreported age-related differences in ACE2 expression in the lungs and gastro-intestinal tract, mainly present in cells in contact with the external environment (bronchiolar epithelial cells, enterocytes). ACE2 expression in the lungs increased during the first years of life, whereas ACE2 expression in the stomach and small intestine decreased with age. These age-related expression differences could be a substrate of the selective clinical vulnerability of the respiratory and gastro-intestinal system to SARS-CoV-2 infection during infancy.1,2 In other organs, expression of ACE2 protein did not change with age.
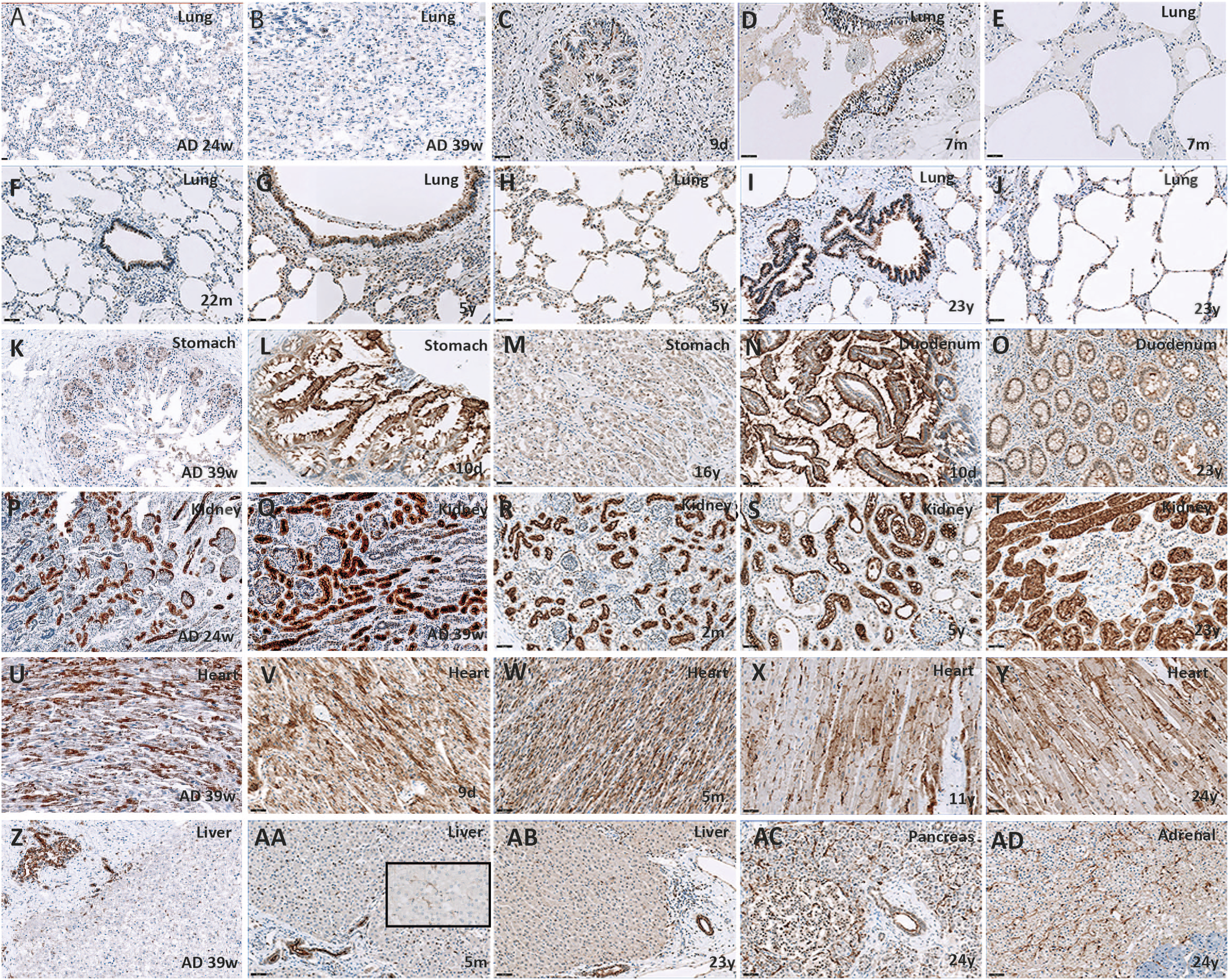
The majority of studies that evaluate ACE2 expression across ages were limited to the lungs. An age-dependent increase in ACE2 expression in the human lung was further found in this study, with absent staining during later gestational ages and an increase during the first years of life. This has also been reported by others6-8,11 and might partly explain the predominance of respiratory symptoms in older subjects infected with COVID-19. Although results are conflicting, it is important to take note of ventilation as a possible confounding factor11,13 in pulmonary ACE2 expression.
ACE2 expression in other organs in early life, childhood, and adolescence has not been studied before. Expression during fetal life has been investigated by few, and results are conflicting.7,9 This might be explained by the differences in techniques used (immunohistochemistry vs single-cell RNA sequencing) and the aforementioned difference between RNA and mRNA/protein expression. Therefore, although our sample size was limited, we are the first to present a comprehensive view on ACE2 expression during the different stages of life and illustrate that not only does ACE2 expression in the lung increase with age, ACE2 expression in the gut decreases with age. Our results could be one substrate of the selective clinical vulnerability of the respiratory and gastro-intestinal system to SARS-CoV-2 infection during infancy in comparison with adulthood. ACE2 protein expression through ages. All magnification ×200 (A-J). In the lungs, ACE2 protein expression increases with age in both bronchiolar epithelium and alveolar pneumocytes. (K-M) In stomach mucosal epithelial cells, ACE2 protein expression is enriched at the luminal surface and decreases with age. (N and O) The same occurs in the duodenum. (P-T) In the kidney, ACE2 protein expression is found in tubuli and epithelial blade of the Bowman’s capsule, and does not change with age. (U-Y) ACE2 protein expression in the heart is found in cardiomyocytes with enrichment at the Z-line. It remains stable through ages. (Z-AB) In the liver, ACE2 protein expression in the bile duct and Kupffer cells (insert) does not change with age. The same occurs in the pancreas (AC) and adrenal gland (AD). Note the nuclear localization in many cell types. ACE2 protein may modulate reactive oxygen species formation in the nucleus, providing a protective mechanism against oxidative stress and cell damage.
7
Footnotes
Acknowledgments
We thank the Mortuary personnel of the Expert Centrum for Post-mortem Diagnostics of the Amsterdam UMC for their assistance during autopsies and O. Bugiani for critically revising the manuscript.
Author Contributions
MB had full access to all of the data in the study and takes responsibility for the integrity and the accuracy of the data analysis.
BS, ER, and MB designed the study.
BS and ER performed the literature search. All authors acquired, analyzed, or interpreted the data. BS and MB drafted the manuscript. All authors critically revised the manuscript for important intellectual content and had final approval of the submitted and revised versions of it. MB supervised the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Amsterdam UMC Corona Research Funds (project number 2007794). The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
