Abstract
Objective
Trauma in pregnancy may cause placental abruption. Consequences of moderate placental injury on neurodevelopment are unknown. The aim was to evaluate placental histology after maternal trauma.
Methods
A prospective study was conducted at 2 tertiary medical centers in the Netherlands. Placentas from women exposed to maternal trauma ≥ 20 weeks’ gestational age were histologically examined. Neurological follow-up of the infants was performed at 1 year of age by means of Alberta Infant Motor Scale. Histological findings were compared to placentas from pregnancies without trauma.
Principal Results
Thirteen placentas were investigated in the trauma group. The control group consisted of 15 placentas. Placental pathology was seen more often in the trauma cases (11 of the 13) than in the controls (6 of the 15), P = .024. Neurological follow-up was normal.
Conclusions
In this small population, majority of the placentas showed pathology after minor trauma in pregnancy without consequences for neurodevelopment at 1 year.
Introduction
Trauma in pregnancy occurs in approximately 7% of all pregnant women in industrialized countries. 1 Minor and major trauma have been reported to cause fetal brain damage. 2 The prevalence of fetal brain injury with consequences for later neurological outcome after maternal trauma, however, is not yet known.
Fetal brain injury after mechanical maternal trauma can be elicited by direct injury to the fetal head or indirectly via (partial) placental abruption 2 or fetomaternal hemorrhage. 3 Fetal death has been reported after complete placental abruption following maternal trauma. 4
This study focused on the role placental pathology in the origin of neurodevelopmental problems after trauma in pregnancy.
In previous research by Redline 5 and Wu and Colford, 6 placental lesions (loss of villous or vascular integrity and/or chorioamnionitis) were reported to be related to abnormalities on brain ultrasound and/or neurodisability in infancy.
Placental pathology (such as chronic hypoxic changes or chorioamnionitis) has also been described in neonates with changes on cranial ultrasound and in children who developed cerebral palsy.6,7 Furthermore, the association between similar placental pathology and moderate grades of prenatal brain injury has been studied before birth in a population at risk of preterm delivery. 8 To our knowledge, there are no studies that performed placental histology after (minor) trauma exposure without evidence of placental abruption.
The aim of this study was to examine placental histology and obstetrical and neurological outcomes in pregnancies complicated by maternal trauma in a 3-year period. Histological findings were compared to placentas from pregnancies without trauma.
We hypothesize no differences between the cases and controls in case of minor trauma.
Methods
This is a prospective study, part of the Fetal Brain Imaging (FBI) study.
All consecutive women, with uncomplicated pregnancies (no hypertensive disorders and no other maternal or fetal disorders), seeking health care because of exposure to abdominal or high-energy trauma after 20 weeks’ gestational age (GA) at VU University Medical Center, Amsterdam, or University Medical Center Utrecht, Utrecht, the Netherlands, were eligible to participate in the Fetal Brain Imaging Study. In this study, serial fetal and neonatal brain imaging by means of ultrasound and magnetic resonance imaging (MRI) as well as histological examination of the placenta and a neurological follow-up of the infant until 5 years of age were performed. When trauma occurred after 38 weeks’ GA, women were not invited to participate, as fetal brain imaging was not expected to be feasible before delivery.
Approval from both local medical ethical committees was obtained. All women who participated gave written informed consent.
Histological findings were compared to those in placentas from women who delivered after an uncomplicated pregnancy between 2009 and 2014, matched for GA and mode of delivery (spontaneous or cesarean section). The secondary use of the coded data from the placental samples did not require informed consent according to Dutch legislation.
Cause of Trauma, Fetomaternal Hemorrhage, and Severity of the Injuries
Causes of trauma were classified as motor vehicle accidents, cycle accidents, falls, and other. Fetomaternal transfusion was tested by means of a Kleihauer–Betke test (<400 µL: no fetomaternal transfusion). Severity of maternal injuries was assessed with the Injury Severity Score (ISS). 9 The ISS is an international scoring system to provide an overall score (range, 0–75) of the severity of injuries following trauma. The body is divided into 6 regions (head, face, chest, abdomen, extremities [including pelvis], and external). Each injury is scored per region according to the Abbreviated Injury Scale (AIS), ranging from 1 (minor injury) to 6 (unsurvivable). In each body region, only the highest AIS score is used. The 3 body regions with the highest AIS are squared and summed to calculate the ISS. For clinical use and in accordance to other studies,1,10,11 an ISS of ≥9 was considered severe.
Placenta
During delivery, clinical confirmation of placental abruption (hemorrhagic amniotic fluid and placental birth directly after the infant) as diagnosed by the obstetrician was recorded.
All placentas were examined by experienced perinatal pathologists (JPvdV and PGJN), both unaware of the type of trauma, severity of maternal injuries, brain ultrasound findings, or neurological outcome of the infants.
Placentas were macroscopically examined for completeness. The membranes were resected, and a roll of the extraplacental membranes from the rupture edge to the placental margin was made. The umbilical cord was examined for the average diameter, obvious strictures, knots, length, and site of insertion. The coiling index (number of coils/10 cm) was determined, and abnormal coiling was recorded (coiling index > 0.30: hypercoiling and coiling index < 0.07: hypocoiling). 12
The cord was then resected from the placental disk, and the trimmed placental weight was compared to normal values. 13 A minimum of 2 cassettes containing full-thickness sections from the center of the normal-appearing placental disk, including the one close to the umbilical cord insertion, were taken for microscopic examination. Furthermore, sections were taken from the extraplacental membranes and from both the placental and fetal ends of the umbilical cord. Additional sections were taken from any macroscopically abnormal area.
Classification of Placental Pathology.

The presence of hemosiderin in decidua of basal plate, in placental membranes, or in the placental parenchyma was identified by light microscopy on hematoxylin and eosin-stained sections and confirmed by Prussian blue stain.
Ultrasound
Fetuses underwent biweekly brain ultrasound. Assessment consisted of echogenicities surrounding the ventricles and/or basal ganglia and peri- and intraventricular hemorrhage. 8 Echogenicity changes had to be present in at least 2 directions (coronal, sagittal, and/or axial) in order to be graded. In case echogenicity changes were seen in 1 plane only or in case the quality of the images was insufficient, the ultrasound was considered inconclusive.
Obstetric Outcome
GA, mode of delivery (spontaneous, vacuum extraction, and cesarean section), birth weight, and Apgar scores (<7 abnormal) at 1 and 5 minutes were recorded.
Neurological Outcome
The infants were seen for neurological examination according to Prechtl 18 at term-equivalent age and Touwen 19 and Alberta Infant Motor Scale 20 at 1 year.
Data Analysis
To compare placental pathological findings between the trauma and control group, the Fisher’s exact test was used. P < .05 was considered statistically significant. Statistical analysis was performed using IBM SPSS Statistics version 20.
Results
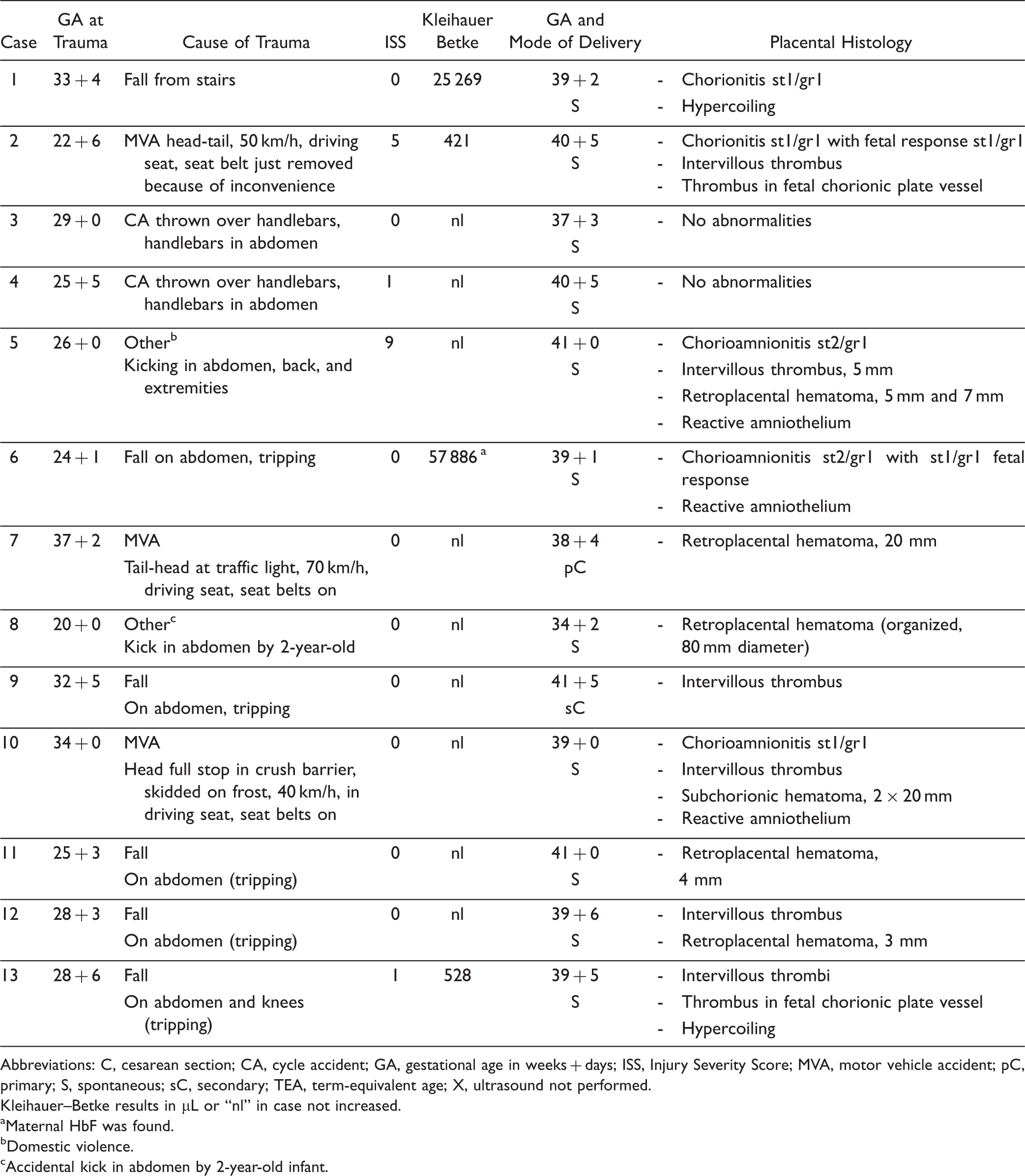
Seventy-five women were invited to participate in the Fetal Brain Imaging study between October 2011 and July 2014. Figure 1 depicts the number of women who accepted and declined participation. Almost half of the women, who were invited, declined to participate mainly due to anxiousness over unwanted test results or experiencing participation in scientific research too burdensome. Therefore, placentas of 13 women were available for pathological examination. GA at the time of trauma, type of trauma, ISS score, and Kleihauer–Betke outcome per case are shown in Table 2. Injuries were minor in all but 1 case, and 9 of the 13 women experienced contractions and/or abdominal pain. The 3 cases with increased Kleihauer–Betke test results were clinically stable, and pregnancy was not terminated. Controls: 15 placentas matched the criteria for the control group.
Patient inclusion characteristics. §Other reasons: one woman was admitted for psychiatric treatment and one other woman was admitted to the cardiac care unit, both not in a condition to be invited for scientific research. The third woman was a foreign woman who continued her way home after the admission. The fourth woman could not be invited because of practical difficulties. †FBI-study: Fetal brain imaging in fetuses at high risk of acquired anomalies. The study aims to investigate the value of advanced brain sonography and magnetic resonance imaging in a high-risk population for fetal brain damage (such as trauma in pregnancy, cytomegalovirus/toxoplasmosis infection, early fetal growth restriction). Characteristics at Trauma Admission and Placental Pathology per Case. Abbreviations: C, cesarean section; CA, cycle accident; GA, gestational age in weeks + days; ISS, Injury Severity Score; MVA, motor vehicle accident; pC, primary; S, spontaneous; sC, secondary; TEA, term-equivalent age; X, ultrasound not performed. Kleihauer–Betke results in µL or “nl” in case not increased. aMaternal HbF was found. bDomestic violence. cAccidental kick in abdomen by 2-year-old infant.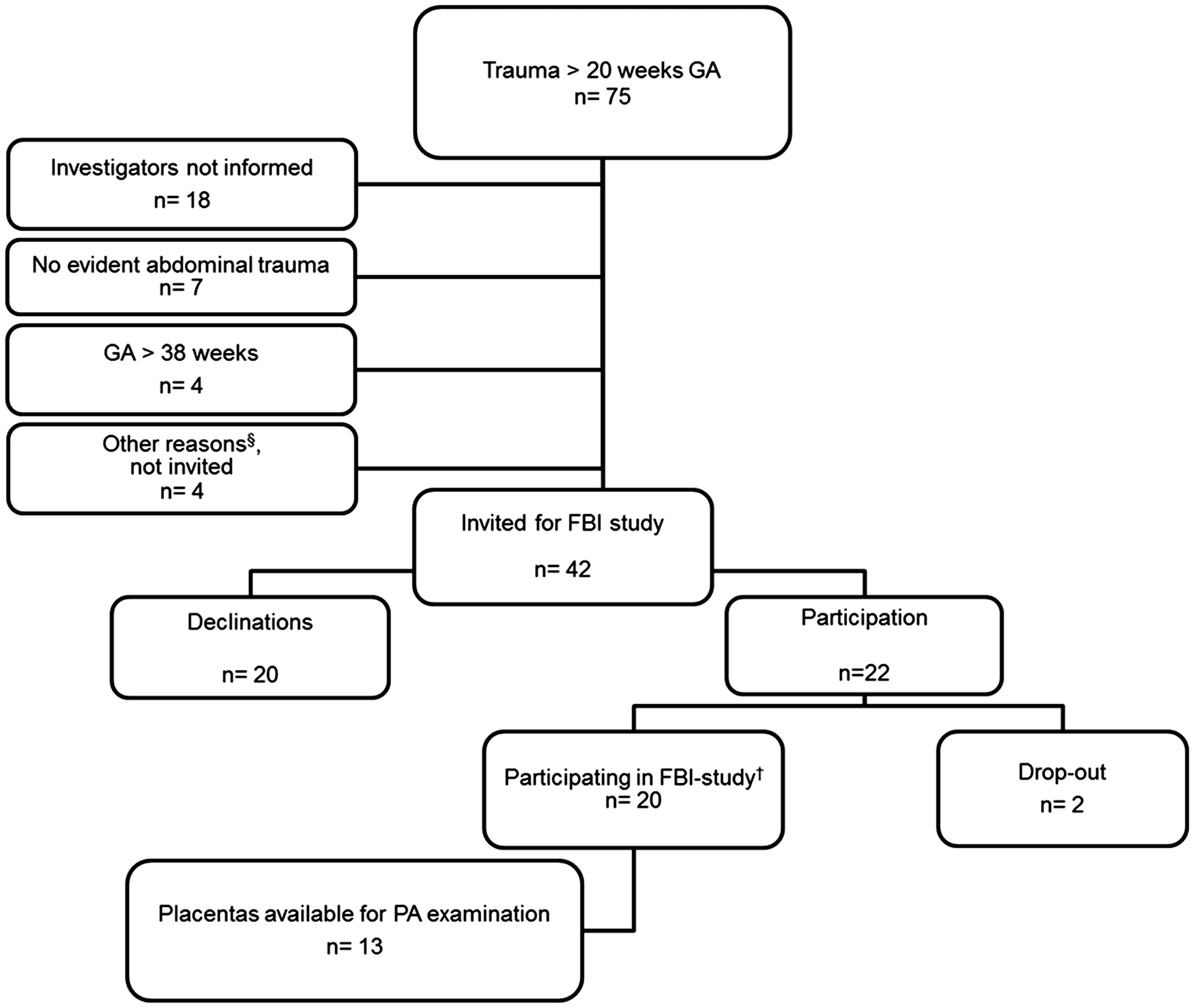
Placenta
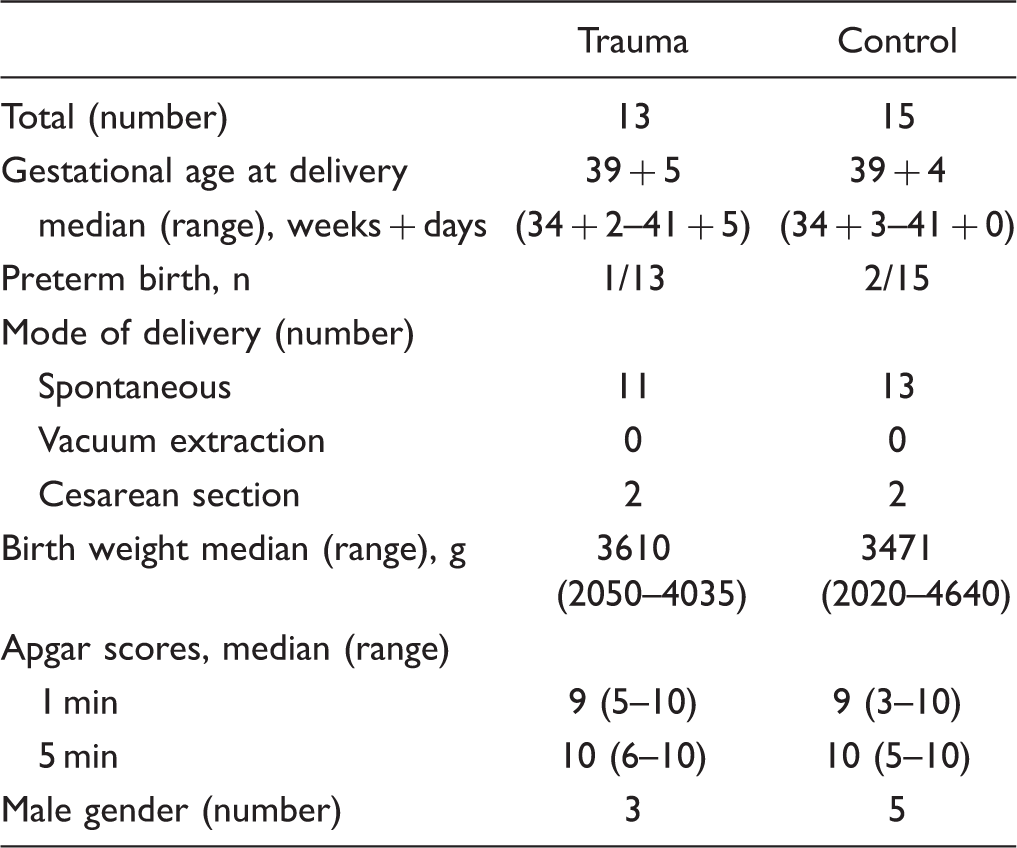
Characteristics at Delivery of Trauma and Control Group.
Placental Macroscopy and Histology of Trauma and Control Group.
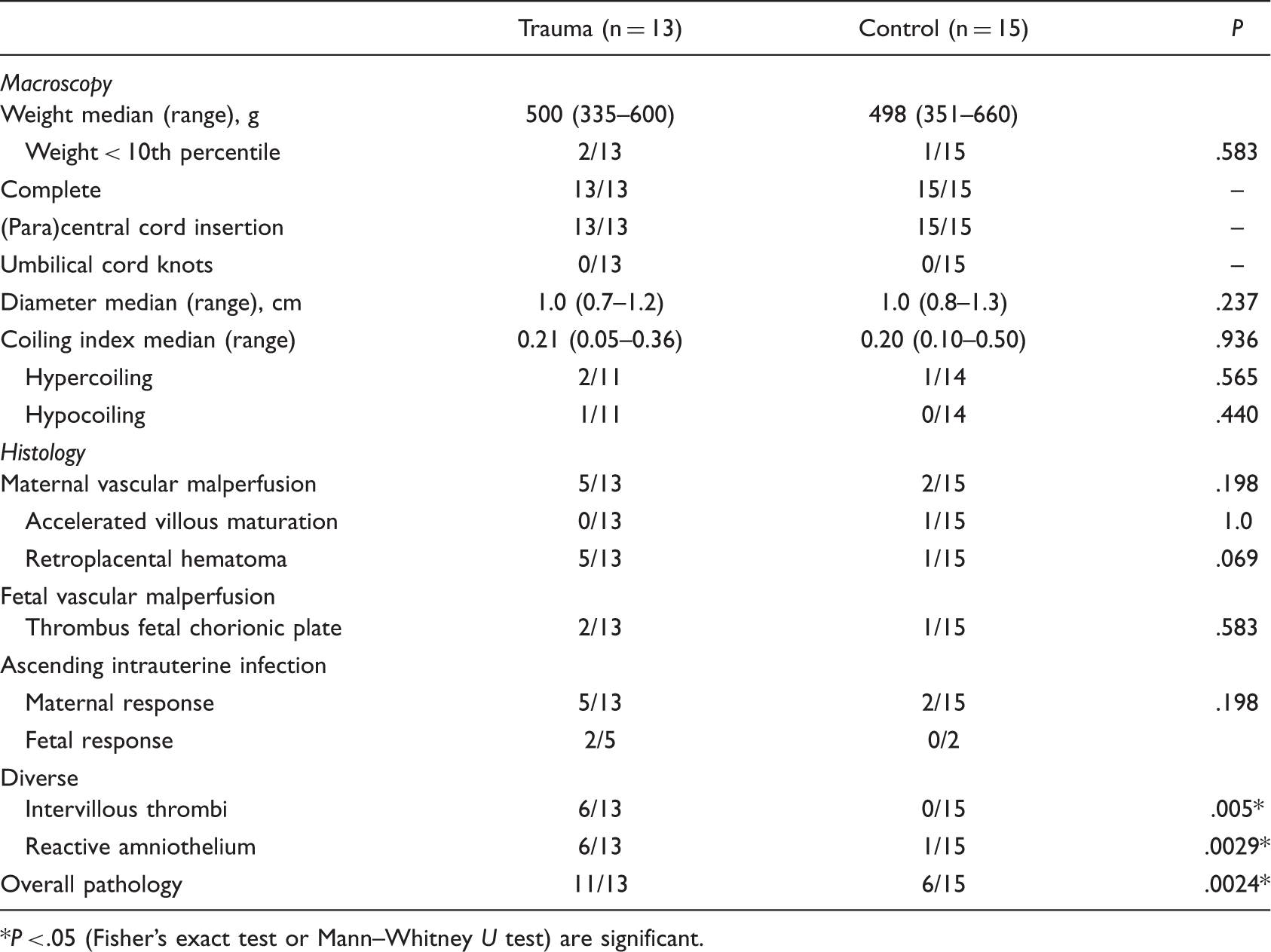
P < .05 (Fisher’s exact test or Mann–Whitney U test) are significant.
Three placental weights were below the 10th percentile: 2 cases and 1 control. The histology in case 6 was chorioamnionitis st2/gr1 with st1/gr1 fetal response and reactive amniothelium, and in case 13, 2 intervillous thrombi, thrombus in fetal chorionic plate vessel and hypercoiling; in the control, the histology showed a small thrombus in a chorionic plate vessel.
Main pathological findings were chorioamnionitis, retroplacental hematomas, and intervillous thrombi. In 1 of the 6 cases and in 1 control with reactive amniothelium, meconium-stained amniotic fluid was reported.
Ultrasound
Fetal ultrasound examinations were performed in 10 cases. In 4 cases, the assessment was inconclusive. Due to the limited number of conclusive ultrasound examinations, they were excluded from the analyses.
Obstetric, Neonatal, and Neurological Outcomes
Two infants had Apgar scores <7: the infant of case 1 (whose delivery was complicated by shoulder dystocia) was born with Apgar score 5 at 1 minute, recovering to 8 at 5 minutes and 10 at 10 minutes. The infant of case 8 was born prematurely with Apgar scores of 7, 6, and 7 at 1, 5, and 10 minutes respectively. She was noted to have hepatomegaly, hematomas, and epistaxis as well as respiratory problems for which she was admitted and treated with CPAP. Cases 7 and 9 withdrew from further study participation and therefore did not attend the follow-up examinations. Case 7 did give permission to obtain developmental information from the infants’ medical file in our hospital. Cases 5 and 13 were not able to attend one of the neurological follow-up examinations but agreed on retrieving information on their infants’ motor and neurological development from the well-baby clinic. In the well-baby clinic, motor development is monitored according to the “van Wiechen” schedule (a Dutch developmental milestone scheme for the assessment of the motor, language, and social development of the infant).
Neurodevelopment at term-equivalent age was normal in 10 of the 11 cases (case 11 suspect) and normal in 11 of the 11 cases at 1 year. The infant of case 7 visited a pediatric neurologist at 2 years of age, and a motor developmental delay was diagnosed. Additional testing (MRI and DNA microarray) did not reveal any abnormalities.
Discussion
To our knowledge, this is the first prospective study to assess histological pathology of the placenta after maternal trauma in pregnancy. We found placental pathology in 85% (11 of the 13) of our trauma population, with normal neurodevelopment at 1 year of age.
The prevalence of placental pathology in this prospective trauma study was higher than in the control group and consisted mainly of retroplacental hematomas, intervillous thrombi, chorioamnionitis, and reactive amniothelium. From the individual items, intervillous thrombi and reactive amniothelium were significantly more frequent in the cases. The degenerative changes of the amnioepithelium have been described in the presence of meconium in the amniotic cavity, elicited by fetal distress. Meconium, however, was not or no longer visible in 5 of the 6 cases with reactive amnioendothelium.
The prevalence of retroplacental hematomas in this prospective study was 38% and consisted of mainly small lesions, which is higher than the prevalence of placental abruption (8.5% without, 7.4% with nonsevere [ISS = 1–8], and 13.1% with severe injuries [ISS ≥ 9]) after motor vehicle accidents reported in a large retrospective cohort. 11 In this and other retrospective series reporting on the effects of trauma on obstetrical outcome, placental histology was not reported.1,10
In our population with minor trauma, almost half of the placentas showed small intervillous thrombi, whereas none of the control placentas showed intervillous thrombi. In a study on placental pathology in pregnancies complicated by fetal growth restriction, intervillous thrombi were found in 42% of the placentas from pregnancies with fetal growth restriction and in 12% of a healthy control group. 22 Intervillous thrombi are the result of leakage of fetal erythrocytes in the intervillous space, a process caused by the loss of fetal vascular integrity within the villi. 5 It is interesting that we found no correlation between the intervillous thrombi and the presence of positive Kleihauer–Betke test. Retroplacental hemorrhage is seen after the loss of integrity of the maternal vascularization. 5 Isolated retroplacental hematomas may be associated with fetomaternal hemorrhage.23,24 Thus, the presence of intervillous thrombi and retroplacental hematomas reflects hypoxic events and/or fetomaternal hemorrhage, which may explain the high prevalence after maternal trauma. In a previous study by Salafia et al., not related to trauma, the presence of hemosiderin (which derives from hemolized red blood cells, indicating old hemorrhage) in the basal decidua and/or extraplacental membranes was described in 64% of cases with nonhypertensive placental abruption. 21 In our study, 54% of the cases had hemosiderin depositions, almost all of them located around retroplacental and/or intervillous hematomas, compared to none of the controls. This indicates an event causing vascular damage (long) before birth.
The 40% chorioamnionitis in our trauma population is comparable to a 47% found in population with hypertensive disorders; 8 however, we found no difference between the prevalence in the trauma and the control group, which corresponds to the fact that chorioamnionitis has been reported as a common finding in normal populations in former research.5,25
We do not yet understand the findings of chorioamnionitis or hypercoiling, but both have been described as histological indicators of fetal hypoxia or ischemia.5,8,26 As chorioamnionitis occurs around the delivery, and it is considered unlikely that coiling density changes occur after 20 weeks’ GA, 26 we cannot explain the role of (minor) trauma in the finding of chorioamnionitis and hypercoiling, as the traumas occurred after 20 weeks’ GA and not directly prior to delivery.
Evaluating the histological abnormalities per type of accident, we could not find a clear difference in the distribution of the abnormalities. In the 3 cases with motor accidents, 2 had intervillous thrombi, 1 retroplacental hematoma, 2 chorioamnionitis, and 1 reactive amniothelium.
In the 6 cases with falls, 3 had intervillous thrombi, 2 retroplacental hematoma, 2 chorioamnionnitis, 1 reactive amniothelium, 1 thrombus in fetal chorionic plate vessel, and 2 hypercoiling. In the 2 cases with other trauma and in 1 case with domestic violence chorioamnionitis, intervillous thrombus, retroplacental hematoma, and reactive amniothelium were found, and in the case with accidental kick in the abdomen, a retroplacental hematoma of 8 cm was demonstrated 14 weeks after the incident after preterm delivery. The 2 women with cycle accidents had no anomalies in the placenta.
The number per type of accident is too small to correlate these findings; however, they demonstrate that even small incidents such as accidental kicking in the abdomen, falls, and also domestic violence can elicit histological abnormalities.
A relation between placental pathology and findings on brain ultrasound could not be made due to the limited availability of complete data of both brain imaging and placental histology.
Nearly, all fetuses in the trauma group were delivered at term and vaginally. Moreover, median Apgar scores at 1 and 5 minutes were normal, which underlines a lack of perinatal hypoxic ischemia, accentuating the fact that placental pathological findings most probably do not originate from intrapartum events, but from hypoxic events earlier in pregnancy.
Our finding of more placental pathology after minor trauma in pregnancy, together with previous reports linking similar pathological findings to abnormal findings on brain ultrasound and/or neurodevelopmental abnormalities,6,27 may elucidate a first step in the pathogenesis and therefore stimulates to conduct a larger study investigating the placenta after maternal trauma in pregnancy, together with follow-up of the neurological outcome of the infant.
Strengths and Limitations
The limited number of placentas and fetuses has to be considered as well as the fact that maternal injuries were mainly minor. This occurred despite the effort to invite women regardless of the severity of maternal injuries. The main reason for declining was the anxiousness for unwanted test results from brain imaging. Nonetheless, previous research focused on the effects of major placental abruption, often followed by preterm delivery (possibly influencing placental histology as well) and does not take into account that minor trauma may also cause placental pathology. Modesty about the severity of placental pathology in the trauma group is in order; however, the high prevalence after trauma compared to the control population did allow us to interpret the findings as abnormal.
In conclusion, the majority of the cases showed placental pathology after minor trauma in pregnancy in this small data set. This finding underlines the importance of placental histology and the need of a larger study. Narrowing the examination to placental histology only (instead of a broad observational study including brain imaging and a long infant follow-up until 5 years of age) may increase women’s willingness to participate in such study.
Footnotes
Acknowledgments
The authors are most grateful to LS de Vries, LR Pistorius, and JIML Verbeke for their help in the performance and/or assessment of brain ultrasounds. The authors would also like to thank PEM van Schie and VAM Schaaf for their contribution in carrying out the neurological follow-up examinations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
