Abstract
The international cosmeceutical sector has experienced unprecedented expansion, compelling regulatory bodies to enhance and update systems to tackle consumer protection, product effectiveness, and ethical issues. This review critically evaluates and contrasts regulatory settings in key markets, such as the European Union, United States, Canada, Japan, China, India, and Brazil. The examination is concentrated on key features including product definitions, pre-market approval procedures, and ingredient regulation, labeling requirements, post-market surveillance, and integrating ethical and environmental considerations. The outcome shows significant advances in regulatory harmonization, especially in the area of ingredient safety and adverse event reporting; however, there are still considerable challenges. Pioneering among these are the lack of a standard definition for “cosmeceuticals,” highly variable ingredient limitations, and uneven application practice across jurisdictions. The speedy growth of e-commerce and cross-border sales additionally complicates regulatory control, adding to the possibility of non-compliant or counterfeited products reaching consumers. The review also identifies a shortage of empirical evidence to document the actual impact of recent regulatory reforms in the real world, as well as on innovation and market access. The present study recommends the promotion of international harmonization of standards, enhancement of post-market surveillance, convergence of ethical and sustainability dimensions, and targeted support for small- and medium-sized enterprises. Henceforth, while important progress has been achieved, the future of the industry rests on creating nimble, science-informed, and internationally harmonized regulatory systems that can keep pace with changing technologies and consumers’ and public health priorities, ensuring both consumer safety and industry innovation.
Introduction
Cosmetics and cosmeceutical business has become one of the fastest-growing and most dynamic industries globally, supported by ongoing innovation, changing consumer attitudes, and growing awareness regarding personal health and well-being.1,2 The industry has experienced exponential growth over the last few decades, driven by advances in formulation science, emergence of novel active ingredients, and extensive use of digital marketing and online platforms.3,4 As per recent market studies, the world cosmetic industry stood at around USD 419.8 billion in 2024 and can be expected to cross USD 550 billion by the early 2030s, with yearly growth rates between 4.6% and 6.6%. 5 This phenomenal growth reflects the industry’s importance not just as a significant economic driver but also as a core driver of consumer lifestyles and health. Still, this swift growth also gives rise to sophisticated challenges, most notably in the regulatory arena, where the imperative to provide product safety, efficacy, and honest marketing assertions must be weighed against the need to encourage innovation and support global commerce.
Cosmeceuticals, the neologism for products that sit at the boundary between cosmetics and pharmaceuticals, pose a specific challenging product category for regulators across the globe. 6 These cosmetics typically include biologically active compounds that purport to provide therapeutic or health benefits in addition to conventional cosmetic benefits, such as anti-aging, skin restoration, or color correction. 7 Even with their increasing popularity and market penetration, cosmeceuticals have no worldwide accepted regulatory definition, leading to a divergent international environment in which the same product can be referred to as a cosmetic in one country and as a drug or quasi-drug in another. 8 This regulatory uncertainty makes it difficult to develop, market, and distribute cosmeceutical products because firms have to contend with various and, at times, contradictory requirements concerning ingredient safety, labeling, substantiation of claims, and pre-market approval. The lack of harmonized requirements not only adds to the cost of compliance and delays new product introduction but also threatens consumer safety and erodes confidence in the industry. 9
The regulatory systems for cosmeceuticals and cosmetics are markedly different in important markets like the United States of America (USA), the European Union (EU), China, Japan, Canada, and Brazil. 10 The EU has set forth one of the most extensive and rigorous regulatory frameworks by Regulation (EC) No 1223/2009, harmonizing the cosmetics law among member states and mandating rigorous rules regarding ingredient safety, product notification, labeling, and the ban on animal testing. 11 The EU regime also features specific provisions for prohibited and restricted substances, obligatory safety evaluation by competent professionals, and compliance enforcement. The United States, by contrast, is governed by the Federal Food, Drug, and Cosmetic Act (FD&C Act) and the Fair Packaging and Labeling Act (FPLA), which have been amended relatively infrequently since they were enacted in the mid-20th century. 12 The US Food and Drug Administration (FDA) oversees cosmetics but does not exercise pre-market approval for most products, and manufacturers are therefore largely responsible for safety and labeling compliance. This more relaxed approach to regulation has been faulted for not being as rigorous and precise in its handling of the complexities that characterize contemporary cosmeceuticals.
China’s regulatory landscape has also evolved dramatically over the last few years, with the Cosmetic Supervision and Administration Regulation (CSAR) of 2021 representing a significant turning point toward enhanced stringent control. 13 The CSAR requires extensive safety assessments, registration or notification thresholds based on product classification, and increased post-market monitoring. China has also started to ease its traditionally obligatory animal testing requirements for ordinary cosmetics, tending towards more international cruelty-free standards, although with exceptions and prerequisites still in place. Japan and Canada offer hybrid regulatory frameworks that incorporate aspects of both cosmetic and pharmaceutical regulation. Japan categorizes products as cosmetics and quasi-drugs, where quasi-drugs are under stricter safety and effectiveness standards, while Canada is governed by the Food and Drugs Act and the Cosmetic Regulations, with proposals for implementing bans on animal testing. Brazil’s regulatory system is developing, including state prohibitions against animal testing and federal guidelines overseen by the Brazilian Health Regulatory Agency (ANVISA), but enforcement harmonization and conformity to world’s best practices are still in question. 14
The biggest regulatory challenge in cosmeceuticals is the absence of global harmonization. Whereas various international organizations like the International Cooperation on Cosmetics Regulation (ICCR), the Organization for Economic Co-operation and Development (OECD), and the International Organization for Standardization (ISO) have sought to advance mutual acceptance of testing procedures, safety evaluation guidelines, and labeling criteria, there are still considerable differences. 15 All of these differences are reflected in the divergent definitions of what a cosmetic or cosmeceutical product is, different lists of banned and restricted ingredients, mixed pre-market approval or notification requirements, and differing policies regarding animal testing. The EU’s total ban on animal testing for cosmetic ingredients as well as finished products stands in stark contrast to the quilt of state-by-state bans in the USA and the conditional exemptions that have been implemented recently in China. 16 Such regulatory fragmentation not only makes compliance more difficult for multinational businesses, but it also constrains innovation by introducing varying evidentiary requirements and safety standards that can hinder or block product introduction in some markets.
The regulatory divergence also affects cross-border trade, as products that are compliant in one place can be excluded or need expensive reformulation to suit another’s standard. This situation is particularly acute for emerging markets and small- to medium-sized enterprises (SMEs), which often lack the resources to navigate complex regulatory landscapes. Moreover, the rapid pace of innovation in cosmeceuticals—driven by advances in biotechnology, nanotechnology, and natural ingredient sourcing—poses additional challenges for regulators striving to keep pace with scientific developments while ensuring consumer safety. The addition of new ingredients, including peptides, plant stem cells, and microbiome modulators, frequently outpaces the ability of current regulatory systems to effectively evaluate for safety and efficacy, causing regulatory uncertainty and potential market fragmentation.
Another important point of concern is the convergence of environmental and safety regulations. Whereas cosmetic regulation targets mainly consumer protection and product performance, environmental regulations such as the EU’s Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) also take into account wider ecological effects of chemical substances. 17 Yet inconsistencies and overlaps between these regimes may result in incongruent restrictions for substance classes such as perfluorinated compounds (PFAS) and cyclic siloxanes that have both environmental and health hazards. 18 The failure of chemical safety legislation to align with the regulation of cosmetics could enable potentially dangerous ingredients to be left in cosmetics or make it difficult for manufacturers to comply.
Aside from the regulatory hurdles, ethical aspects such as animal welfare have come to the forefront, impacting not only consumer choices but also legislative developments. The campaign for a cosmetics animal testing ban has picked up momentum around the world, with the EU taking the lead on full bans and other nations incrementally implementing related measures. Yet the speeds and extents of the bans differ significantly, raising trade barriers and regulatory risks. The establishment and verification of new non-animal approaches are still a high priority for the industry and regulators, but the acceptance and adoption vary by region.
Against this dynamic and multifaceted environment, there is a strong need for continued research and discussion to determine areas of regulatory gaps, analyze the effect of recent reforms, and examine avenues to closer harmonization. A clear understanding of how differences in regulation influence innovation, market access, consumer protection, and ethical standards is necessary for consumers, industry players, and policymakers. The current global situation concerning the regulation of cosmeceuticals, with a focus on major challenges and opportunities, as well as the critical areas requiring research and further development of regulations, is the objective of this review. In doing so, it aims to assist in the evolution of a regulatory framework that reconciles safety, innovation, and ethics within the international cosmeceutical market.
Challenges in Regulation
Fragmented Global Standards and Definitions
Perhaps the most intractable and fundamental of the challenges facing cosmeceutical regulation is the absence of harmonized standards and definitions across national jurisdictions. Although the “cosmeceutical” term is used extensively within industry and in marketing, it is not officially utilized in the regulatory vocabulary of the majority of countries. Rather, products are designated as cosmetics or drugs based on what they claim and what they contain, creating a patchwork of regulatory obligations. A product sold as a skin-lightening cream would be regulated as a cosmetic within the EU, a quasi-drug in Japan, and a drug in the United States if it makes a claim about changing the function of the skin. This absence of one unifying definition creates regulatory uncertainty for manufacturers, who have to reformulate, reformulate labels, and reformulate claims for every market it is aimed at. 17 The absence of international agreement also imposes limits on trade across borders, as products acceptable in one area can be banned or forced to undergo substantial alteration somewhere else. 19
Escalating Regulatory Tightness and Ingredient Prohibitions
The regulatory landscape for cosmeceuticals is becoming progressively more stringent, notably in terms of ingredient safety and allowed claims. Within the EU, the regular revision of Regulation (EC) No 1223/2009 and its annexes has led to the prohibition or limitation of hundreds of ingredients, such as newly recognized allergens, endocrine disruptors, and nanomaterials. The addition of new 2025 regulations, including Regulation 2024/996 and OMNIBUS nano, will add more ingredients to the banned and restricted list, forcing companies to re-develop products and carry out more safety testing. 20 In the United States, the United States has enacted the MoCRA (Modernization of Cosmetics Regulation Act), now mandating full disclosure of ingredients, facility registration, and safety substantiation documentation for all cosmetic products. 21 These changing needs impose heavy compliance costs on manufacturers who have to invest in scientific research, testing of ingredients, and regulatory know-how to stay abreast of developing legislation.
Post-Market Surveillance and Adverse Event Reporting
The other significant challenge is the increasing focus on post-market surveillance and adverse event reporting. Both the EU and the US have enhanced their requirements for following product safety after launch. Under MoCRA, US manufacturers are required to report serious adverse events to the FDA within 15 business days and maintain detailed records for 6 years. 22 The EU requires reporting of SUEs to national authorities, who then notify each other across member states. These requirements call for strong internal systems for monitoring, analysis, and response to consumer complaints and safety signals. For small- and medium-sized businesses, the development and maintenance of such systems can be costly, and non-compliance can lead to product recalls, penalties, or market withdrawal.
Transparency, Labeling, and Consumer Communication
Increased transparency in ingredient listing and labeling of products is being subjected to greater scrutiny by regulators and consumers. 23 In the US, MoCRA currently requires disclosure of all ingredients, including those in their secret fragrance mixtures, and labeling for clear identification of allergens and instructions for reporting adverse events. The EU has also extended labeling demands, such as mandatory use of International Nomenclature of Cosmetic Ingredients (INCI) and clear warnings for high-risk substances. 24 These standards are created with the intention of equipping consumers with the knowledge to make the right decisions and for speedy action in case of safety issues. But the requirement of harmonization of labeling and claims across a number of jurisdictions with distinct standards and languages makes things more complicated and expensive for global companies.
Ethical and Environmental Issues
Ethical issues, especially animal testing and environmental sustainability, are determining regulatory agendas globally. The EU’s total prohibition of animal testing on cosmetics has established an international standard, but other parts of the world, including China and Brazil, continue to permit or mandate animal testing for some products, particularly those with new ingredients or special claims. This divergence generates barriers to trade and moral conflicts for brands that adhere to cruelty-free policies. Environmental regulations are also becoming more stringent, with prohibitions on microplastics, PFAS (and other long-lasting pollutants such as Mercury, PCBs, and dioxins “forever chemicals”), and other long-lasting pollutants. 25 Businesses are being pushed to implement “green chemistry” practices, re-formulate products, and show environmental responsibility, all of which entail significant investment and innovation.
Fragmentation of Regulation and the Difficulty of Global Compliance
Maybe the most ominous challenge facing multinationals is dealing with the broken regulatory environment that exists globally and regionally. 26 Each regulatory environment has its own set of banned and restricted ingredients, safety evaluation procedures, and labeling regulations. At the state level in the US, California’s ingredient bans and animal testing prohibitions create an additional layer of complexity on the top of federal regulation. 27 The absence of international harmonization not only enhances compliance costs but also brings uncertainty to manufacturers and consumers alike, potentially inhibiting innovation and narrowing consumer choice. Businesses need to be aware of regulatory evolution, transform their products and processes, and make compliance investments in order to retain access to world markets.
The Emergence of Digital Commerce and New Forms of Enforcement
Increased e-commerce and cross-border sales bring with them new regulatory challenges. 28 Online products may readily avoid conventional regulation, enhancing the risk of non-compliant or counterfeit products reaching consumers. Regulators are starting to confront these challenges through increased digital labeling requirements and greater attention to online platforms, but enforcement is still spotty and resource-intensive. 29 As online commerce continues to grow, regulators and industry must work to create new mechanisms for monitoring, enforcement, and consumer protection online.
In short, the control of cosmeceuticals is growing more complicated, stringent, and dynamic. The interaction between splintered world standards, tighter regulatory requirements, heightened monitoring, ethical and environmental demands, and the emergence of e-commerce poses an intimidating series of challenges to regulators and industry alike. Navigating this changing landscape successfully will involve continued investment in compliance, scientific knowledge, and open communication, along with a new focus on international discussion and concordance.
Beneficiaries in Developing Regulatory Environment
Consumers: Greater Safety and Empowerment
The most important beneficiary of a developing regulatory environment in the cosmetics and cosmeceutical industry is the consumer. As regulatory systems become mature and harmonized with global standards, consumers have greater confidence that the products they consume are safe, effective, and properly labeled. 30 In most developing countries, the lack of effective regulation has traditionally enabled the spread of counterfeit, substandard, or even dangerous products. The imposition and implementation of mandatory safety testing, complete ingredient transparency, and adverse event reporting systems offer an essential safety net. Consumers are better safeguarded against health hazards such as allergic reactions, skin irritation, or poisoning from toxic chemicals. In addition, more transparent labeling and the demand for supported claims give consumers the ability to make informed decisions, generating faith in both local and foreign products. 31 Public education campaigns and open complaint processes also enhance consumer rights, particularly among vulnerable populations who might otherwise have been denied access to credible information or remedy in case of injury.
Industry: Opportunities for Growth and Innovation
The industry of cosmeceuticals and cosmetics itself will gain considerably from having clear and foreseeable regulatory environments. 32 For distributors, manufacturers, retailers, and formulators, the harmonization of local regulations to international standards minimizes uncertainty and compliance costs stemming from dealing with fragmented or archaic legal systems. This is especially beneficial for multinational businesses, as it helps market entry smooth and reduces the need for expensive reformulation, while enabling the creation of integrated global supply chains. Local and regional companies enjoy increased competitiveness with better regulatory certainty, which leads to investment in research and development and creates innovation. 33 The use of best practices like obligatory safety evaluations and ingredient listing uplifts industry standards and bolsters consumer confidence. Also, with increasing regulations mandating or promoting the use of alternate, non-animal testing techniques, the industry is driven to invest in newer technologies and research, further spurring innovation.
Regulatory Authorities: Enhance Overseas Regulation and Global Integration
Policymakers and regulatory authorities are major beneficiaries since they receive more efficient market surveillance, risk assessment, and enforcement tools. The use of centralized product notification platforms, based on models such as the EU’s Cosmetic Products Notification Portal (CPNP), facilitates more effective monitoring and quick reaction to safety issues. 34 Greater regulatory capability not only enhances public health outcomes but also increases the credibility and legitimacy of regulatory institutions. Taking part in global harmonization efforts, the ICCR or the ASEAN Cosmetic Directive creates new avenues for cross-border trade and foreign investment, further enriching national economies. 30 As regulatory systems evolve, governments are better able to keep pace with new risks, technology, and changing consumer demands.
Scientific and Professional Community: Widening Expertise and Research
The scientific and professional workforce comprising toxicologists, dermatologists, regulatory experts, and academic researchers also gains with the development of regulatory landscapes. As regulations grow more intricate and science-based, requirements for product safety evaluation expertise, toxicological risk assessment, and regulatory compliance expertise expand. This spurs expansion in specialty training programs, professional development, and industry-academic collaborations. The shift toward alternative testing methods, prompted by bans on animal testing in many jurisdictions, stimulates innovation in in vitro and computational toxicology, creating new research opportunities and advancing global scientific knowledge. 35
Vulnerable and Marginalized Groups: Greater Protection and Inclusion
Modernizing regulatory frameworks can have a profound impact on vulnerable and marginalized populations, who are often at greater risk from unregulated or unsafe products. 36 They are disproportionately exposed to hazardous cosmetics in developing countries, where they have less access to quality products as well as information. Enforced regulations, along with outreach and education targeting these groups, defend them against harm and enable them to make safer decisions. Reporting mechanisms for adverse events and redress further underpin their rights and welfare. Besides, as regulations more and more consider environmental and ethical factors like prohibitions on toxic substances and green packaging requirements, communities victimized by pollution or immoral production methods may see their own standard of living improve as well.
Challenges and the Call for Inclusive Implementation
Although the rewards of regulation elaboration are considerable, the shift to stronger norms can be challenging, particularly for SMEs that have few resources. 37 Adherence to new testing, documentation, and labeling requirements could require major technology investment, training, and external advice. This could lead to market concentration, with the exit of smaller players from the market, unless carefully controlled. To ensure that regulatory modernization benefits are widely enjoyed, it is necessary for regulators to make transitional assistance, capacity-building programs, and transparent guidance available. International partnership and public-private collaboration have the potential to close resource gaps and facilitate fair access to the benefits of better regulation.
Dynamic Beneficiaries in a Changing Landscape
The roll call of beneficiaries in an evolving regulatory framework is by no means fixed. As scientific breakthroughs, consumer behavior, and technological advances remain the driving forces of the sector, new stakeholders form. The expansion of e-commerce and cross-border trade brings into view new beneficiaries like e-retailers and online platforms, but also poses enforcement issues. 38 Emerging attention to sustainability and humane sourcing present opportunities for green ingredient suppliers, environmental packaging suppliers, and cruelty-free certification services suppliers. As regulation evolves, opportunities for beneficial impact exist, as long as implementation is inclusive and responsive to new requirements.
In conclusion, building strong regulatory frameworks in the cosmetics and cosmeceutical industry brings benefits across a broad spectrum for consumers, industry, regulators, scientific experts, and marginalized groups. To guarantee equitable distribution of these advantages, continuing dialogue, stakeholder involvement, and dedication to capacity-building and cross-border cooperation are necessary. As an increasing number of nations adopt regulatory modernization, the potential for beneficial impact both domestically and in the global market increases.
Case Studies
Case Study 1: Harmonization of Cosmetics Regulation in ASEAN
The ASEAN harmonization of cosmetics regulation is a shining example of regional regulatory cooperation. ASEAN nations originally had different regulatory requirements, presenting obstacles for manufacturers and exporters. 39 Industry leaders, especially those already exporting to the EU with strict standards, campaigned for convergence in regulations. The process started with voluntary cooperation between governments and industry groups, aimed at harmonizing standards where the economic and consumer safety benefits would be most substantial. The second step was formal government commitments to harmonize the regulations, which resulted in the ASEAN Cosmetic Directive. The directive set common requirements for product safety, labeling, and post-market surveillance among member states. The process of harmonization not only opened up market access for large exporters but also brought focus to minor local producers, even drawing attention to the requirement of strong systems to track undesirable effects and safeguard consumers. The experience in ASEAN shows that although government requirements give legitimacy, industry participation and staged implementation are essential for effective regulatory convergence. Notably, this procedure enhanced consumer safety and facilitated manufacturers’ access to greater markets with less regulatory resistance.
Case Study 2: Cosmetovigilance and Regulatory Reform in India
India’s fast-evolving cosmetics market is characterized by special regulatory challenges, considering its enormous population and the popularity of traditional products like kajal and kumkum. 40 Adverse reactions to these products, such as dermatitis, have been reported with considerable frequency. Realizing the necessity for greater consumer protection, Indian authorities have banned the importation of animal-tested cosmetics and stressed the significance of cosmetovigilance regular monitoring and evaluation of undesirable reactions to cosmetic products. Vigilance systems and adverse event reporting are required by the Drugs and Cosmetics Act to detect and control dangerous ingredients. 41 This method has enhanced public health risk detection and guided regulatory decision-making, including ingredient prohibitions or labeling requirements. India’s experience underlines the value of incorporating post-market surveillance into regulatory systems, particularly in settings with extensive product diversity and prevalent use of indigenous cosmetics.
Case Study 3: Regulatory Compliance and Public Health in Kenya
Kenya’s cosmetics industry has grown fast, thanks to urbanization, a growing middle class, and rising consumer knowledge. Despite this growth, ensuring product quality and safety has been difficult, with several instances of adverse effects—especially from skin-lightening creams containing mercury recorded between 2018 and 2023. 42 Regulation, in terms of ingredient testing, microbial contamination analysis, and labeling, falls under the auspices of the Pharmacy and Poisons Board (PPB) and Kenya Bureau of Standards (KEBS). 43 Notwithstanding these measures, disparities in enforcement, scattered ingredient regulations, and the absence of centralized approval of products have created gaps in regulation. Small-scale production and counterfeits complicate quality assurance further. The Kenyan experience emphasizes the importance of strong enforcement regimes, regular ingredient evaluation, centralized approval mechanisms, and targeted consumer awareness on product safety and the risk of counterfeits. Constant product surveillance and increased vigilance are suggested to ensure public health protection and meet international standards.
Case Study 4: Tanzanian Regulatory Complexity and Market Access
The interaction between regulatory complexity and market opportunity is best illustrated in Tanzania. 44 The cosmetics industry is governed by various ministries and agencies, leading to duplicative mandates and cumbersome product registration, manufacturing establishment, and importation procedures. Consultations with stakeholders demonstrated that such regulatory barriers disproportionately impact SMEs, constraining them from growing and competing. The research further established that transferring some of the regulatory activities to a subsequent point in the value chain more than at market entry would make processes simpler and more conducive to domestic manufacturing and employment creation, with continuing protection for consumers. Efforts to streamline and clarify regulatory steps, eliminate duplication, and enhance coordination across agencies continue. Tanzania’s experience demonstrates how regulatory fragmentation can hinder both public policy objectives and private sector development, and underscores the importance of stakeholder involvement in regulatory reform.
Case Study 5: Global Regulatory Knowledge and Commercial Strategy
For most small- and mid-sized cosmetic firms, a lack of global regulatory knowledge remains a key impediment to going global. Every one of the world’s leading cosmetic markets has its own distinct regulation covering the use of ingredients, labeling, and packaging. 45 Major groups tend to have specialized regulatory staff to handle these issues, but small companies might not be able to do so. This imbalance can result in business plans going by the wayside as it becomes too difficult to overcome regulatory challenges, hence hindering innovation and limiting consumer choice. The requirement for competent local representatives in certain markets contributes to operating expenses, which makes it challenging for SMEs to make a business case for market entry unless clearly identified commercial opportunities far exceed compliance costs. This example supports the value of regulatory knowledge and early incorporation of compliance planning into product and market strategy.
Together, these case studies illustrate that regulatory difficulties in the cosmeceutical and cosmetics industry are complex, involving harmonization, enforcement, market entry, post-market monitoring, and the necessity for continuous stakeholder interaction. They also highlight the value of adaptive, situation-specific regulatory approaches to balance consumer safety and business development.
Comparative Analysis
Comparative Analysis.
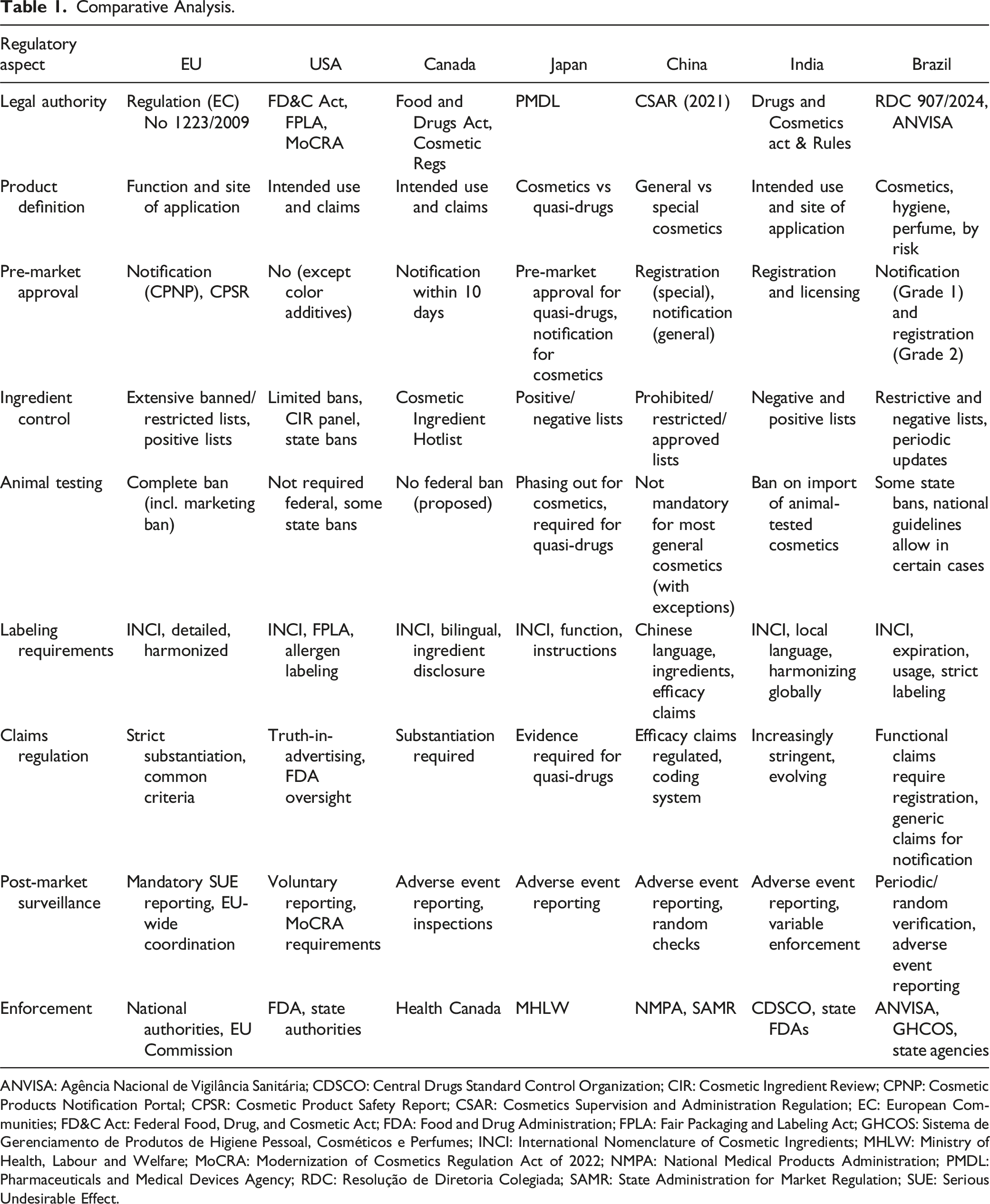
ANVISA: Agência Nacional de Vigilância Sanitária; CDSCO: Central Drugs Standard Control Organization; CIR: Cosmetic Ingredient Review; CPNP: Cosmetic Products Notification Portal; CPSR: Cosmetic Product Safety Report; CSAR: Cosmetics Supervision and Administration Regulation; EC: European Communities; FD&C Act: Federal Food, Drug, and Cosmetic Act; FDA: Food and Drug Administration; FPLA: Fair Packaging and Labeling Act; GHCOS: Sistema de Gerenciamento de Produtos de Higiene Pessoal, Cosméticos e Perfumes; INCI: International Nomenclature of Cosmetic Ingredients; MHLW: Ministry of Health, Labour and Welfare; MoCRA: Modernization of Cosmetics Regulation Act of 2022; NMPA: National Medical Products Administration; PMDL: Pharmaceuticals and Medical Devices Agency; RDC: Resolução de Diretoria Colegiada; SAMR: State Administration for Market Regulation; SUE: Serious Undesirable Effect.
The EU is usually quoted as the gold standard for cosmetic regulation due to its Regulation (EC) No 1223/2009, which sets out a harmonized regime across all member states. 11 The EU requires pre-market notification, thorough safety evaluations, and the designation of a responsible person for every product. Ingredient safety is the top priority: the EU has a very long list of prohibited and restricted substances (more than 1,300 chemicals), imposes a complete prohibition on animal testing on finished products as well as ingredients, and demands strong post-market surveillance and adverse event reporting. This strategy provides strong consumer protection but can erect barriers for non-EU producers entering the market.
On the other hand, the United States traditionally had a less stringent approach, with the FDA regulating cosmetics pursuant to the FD&C Act and the FPLA. 6 The recent MoCRA, 2022 is a major turning point, bringing with it mandatory facility registration, report of serious adverse events, and GMP for the first time. Whereas the FDA now possesses greater powers to demand safety information and pull products from the shelves, pre-market approval is still not mandated for all cosmetics (apart from color additives), and the US list of prohibited ingredients is much shorter than that of the EU. Animal testing is not federally prohibited, although many states have implemented their own bans, and pressure from consumers is encouraging the transition to cruelty-free testing.
Canada’s regulatory system, operated by Health Canada under the Food and Drugs Act and Cosmetic Regulations, is similar to both the EU and US models. 46 Canada demands notification of cosmetics within 10 days of initial sale, has a dynamic Cosmetic Ingredient Hotlist of forbidden and limited substances, and insists on bilingual labeling. Although Canada has not yet legislated a federal prohibition on animal testing, a pending bill is under debate. Post-market surveillance and spontaneous reporting of adverse events are rigorously enforced, and the system is formulated to reconcile industry flexibility and consumer safety.
Japan’s system, regulated by the PMDL (Pharmaceuticals and Medical Devices Agency), separates cosmetics from quasi-drugs. 47 Quasi-drugs, which constitute products with low therapeutic effects, need pre-market approval and proof of efficacy, whereas cosmetics are notified. Animal testing for cosmetics is being phased out in Japan, although it is still mandatory for some quasi-drugs. Ingredient control is based on positive and negative lists, and labeling regulations are stringent and harmonized with international INCI naming.
China’s regulatory landscape has been significantly transformed by the CSAR, which became fully implemented in 2021. 48 The products are categorized as general or special cosmetics, with pre-market registration being mandatory for special cosmetics (including hair dyeing products, sunscreens, and new efficacy-claiming products) and notification for general cosmetics. From May 2025, all registrants and notifiers will need to submit full safety assessment dossiers, aligning China more closely with international standards. 49 China has also relaxed mandatory animal testing for most imported general cosmetics, provided certain documentation is supplied, though exceptions remain for special categories and children’s products. Ingredient management is strong, with an explicit prohibited, restricted, and approved substances list, and a separation between “existing” and “new” ingredients, where the latter must have comprehensive safety data.
India’s regulatory framework under the Drugs and Cosmetics Act and Rules is changing quickly. India has a system of registration and licensing for cosmetics, with greater convergence towards global norms of ingredient safety, labeling, and post-market monitoring. 50 Animal-tested cosmetic importation has been prohibited, and adverse event reporting systems are being consolidated, but enforcement and implementation can be patchy, demonstrating ongoing development of regulatory capability.
Brazil, overseen by ANVISA, recently harmonized its regulations under RDC 907/2024, adopting a risk-based system which categorizes between Grade 1 (lower risk) and Grade 2 (higher risk) cosmetics. 14 Notification is demanded of Grade 1 products, but Grade 2 products need complete registration and technical dossier. Brazil adopts restrictive lists of ingredients and state-by-state prohibitions on animal testing, although national guidelines continue to permit it in specific instances. Labeling, claims substantiation, and post-market monitoring are more harmonized with global standards.
This comparative overview illustrates that, although harmonization continues to move forward most notably with regards to ingredient safety and notification procedures, considerable differences still exist in pre-market conditions, policies on animal testing, claims substantiation, and enforcement. These regulatory variations pose challenges and opportunities for global makers, highlighting the continued necessity of global cooperation, regulatory expertise, and responsive compliance strategies.
This close analysis highlights that although the global business is trending toward more regulatory maturity and harmonization driven by international cooperation, consumer activism, and scientific progress substantial regulatory philosophy, administrative capability, and enforcement differences remain. Regulatory professionals, policymakers, and manufacturers must comprehend these subtleties to facilitate successful global access to markets, risk management, and ongoing protection of consumer health and safety.
Conclusion and Future Recommendation
The international regulatory framework for cosmeceuticals and cosmetics has seen dramatic change over the past decade, as scientific progress, consumer demands, and globalization of the beauty market have compelled regulatory adjustment in both developed and developing markets. This review has shed light on the advancements made in enhancing consumer protection, harmonizing ingredient controls, and incorporating ethical and environmental issues in key jurisdictions like the EU, United States, Canada, Japan, China, India, and Brazil. Yet, the examination also identifies ongoing challenges such as a lack of an internationally accepted definition of “cosmeceuticals,” wide variations in pre-market requirements and ingredient prohibitions, and uneven enforcement of post-market monitoring and adverse event reporting. Among the main conclusions is that regulatory fragmentation still hinders global market access, makes compliance more difficult for manufacturers, and leads to inconsistency in consumer protection. The expansion of electronic commerce and the growing sophistication of supply chains have served to amplify areas of deficiency in regulation, which enable non-compliant or spurious goods to find their way into the hands of consumers. In spite of these challenges, there are encouraging tendencies towards regulatory alignment, as global organizations and regional blocs strive to standardize, and as recent legislation in a number of states evidences a willingness to prioritize consumer protection, innovation, and moral responsibility.
Limitations
Although this review does offer a thorough comparative regulatory framework analysis of the major markets, it is not perfect. To begin with, the research is largely founded on published regulatory reports, official guidelines, and secondary sources, which might not entirely reflect practical enforcement subtleties or the most recent informal regulatory developments. Second, the review is concerned with significant jurisdictions, and though it mentions emerging markets, it is not an exhaustive treatment of all regional blocs or countries. Third, the dynamic state of regulatory reform implies that some recent or future reforms are not represented fully at the time of writing. Lastly, the research focuses on regulatory design and purpose, rather than empirical results like actual product safety in the real world, innovation levels, or consumer feedback, which would necessitate specialized field research and data gathering.
Future Recommendations
To fill the gaps of concern and enable the further development of effective, science-driven, and internationally harmonized regulatory frameworks, a number of recommendations are made: • Facilitate Global Harmonization and Definitive Definitions: Global cooperation should work towards the establishment of a globally recognized definition and classification for cosmeceuticals, along with harmonized ingredient safety requirements, claims substantiation, and labeling rules. • Improve Post-Market Surveillance and Enforcement: Investment in effective adverse event reporting systems, digital surveillance technologies, and coordinated enforcement efforts particularly in growing markets will prove essential for timely risk management and consumer protection. • Align Ethical and Environmental Imperatives: Regulatory systems should further encourage cruelty-free testing, eliminate environmentally toxic substances, and encourage sustainable packaging and sourcing, building on collaborations with scientific and non-governmental bodies. • Adjust to Digital Commerce and Cross-Border Trade: Regulators need to enhance digital surveillance, establish standards for online labeling and claims, and encourage international coordination to avoid the spread of counterfeit or harmful products in e-commerce platforms. • Support SMEs and Promote Inclusive Compliance: Small- and medium-sized enterprises must be given targeted guidance, capacity-building, and transitional support by policymakers and regulators to ensure that regulatory modernization doesn’t strangle innovation or market variety. • Encourage Evidence-Based Policy and Continued Research: There exists an urgent need for empirical investigation into the actual effects of regulatory adjustments on product safety, innovation, consumer confidence, and access to markets. Open data sharing and cooperation with academic and public health organizations should be encouraged.
In conclusion, despite all the advances that have been made, the future of regulatory cosmeceutical and cosmetic governance is all about the creation of responsive, harmonized, and science-informed frameworks that safeguard consumers, encourage responsible innovation, and meet the ethical and environmental challenges of a fast-changing industry. Resolution of the limitations elucidated herein by continued research and stakeholder interaction will be critical to bringing about these objectives and guaranteeing the sustainable development of the industry globally.
Footnotes
Author Contributions
Munish thakur : Drafting and Manuscript writing
Rajni Bala : Concept, Idea and Editing
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
