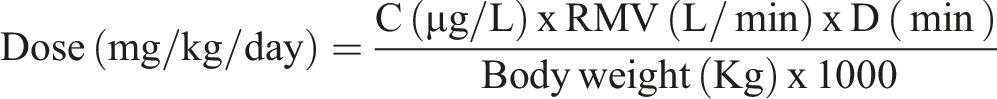Abstract
HFO-1234ze(E) is being developed as a next-generation propellant (excipient), with near zero global warming potential, for use in pressurized metered dose inhalers. In accordance with regulatory guidance, an assessment of the carcinogenic potential of HFO-1234ze(E) was required to gain regulatory approval as a new excipient. HFO-1234ze(E) was therefore evaluated in two-year carcinogenicity studies in mice and rats by the inhalation route of administration. Study assessments included in-life observations, organ weights, histopathology, and hematology. Group mean (sex combined) inhaled doses were 2132, 6218, and 21,193 mg/kg/day in mice and 379, 120,8 and 3918 mg/kg/day in rats; in both studies, control animals were exposed to air alone under the same conditions as HFO-1234ze(E)-exposed animals. HFO-1234ze(E) was well tolerated at all doses. There were no HFO-1234ze(E)-related in-life effects and no neoplastic or non-neoplastic findings or effects on hematology in either study. In summary, HFO-1234ze(E) was not carcinogenic in mice or rats. These data support the use of HFO-1234ze(E) as a medical propellant.
Keywords
Introduction
Pressurized metered dose inhalers (pMDIs) are an important option in the respiratory care of patients with chronic obstructive pulmonary disease (COPD) and asthma. pMDIs account for 78% of global inhaler use 1 and are listed as essential by the World Health Organization. 2
The propellant is a critical component of a pMDI and facilitates the generation of an aerosol cloud, containing one or more active ingredients, which can be inhaled by the patient. 3 Due to their ozone-depleting properties, the original chlorofluorocarbon (CFC) propellants were phased out and replaced by the hydrofluoroalkanes, HFA-134a or HFA-227ea. 4 More recently, environmental legislation, such as the European Union F-gas regulations, has however led to restrictions on the use of some HFAs given concerns relating to their global warming potential (GWP). 5 As a result, the pharmaceutical industry is actively advancing the development of pMDIs that use propellants with low GWP. One such alternative is the hydrofluoroolefin, HFO-1234ze(E) (trans-1,3,3,3-tetrafluoropropene), which has a GWP (100-year horizon) of 1.37, representing a significant improvement over both HFA-134a (GWP 1530) and HFA-227ea (GWP 3600). 6
To gain regulatory approval of a novel medical propellant for use in pMDIs, its safety must first be demonstrated in a series of non-clinical studies. As previously reported, the safety of HFO-1234ze(E) has been demonstrated following assessment in a battery of genetic toxicology, reproductive toxicology, repeat-dose toxicology, and safety pharmacology studies.7,8 Given that pMDIs are used frequently for long periods, it was necessary to characterize the carcinogenic potential of HFO-1234ze(E) in accordance with appropriate guidance.9-11 This paper describes the assessment of HFO-1234ze(E) in two-year carcinogenicity studies in mice and rats, by the inhaled route of administration in support of regulatory approval for use as a medical propellant in pMDIs.
Methods
Test Material
HFO-1234ze(E) (trans-1,3,3,3-tetrafluoropropene; CAS no.: 29118-24-9) was supplied in cylinders at a purity >99.99% by Honeywell (Buffalo, New York, USA). All batches were analyzed by using gas chromatography (GC)/mass spectroscopy (MS).
HFO-1234ze(E) Exposure and Atmosphere Analysis
Animals were exposed to air alone (control) or HFO-1234ze(E), once daily for 1 or 2 h (mice) or 4 h (rats) by nose-only inhalation using a flow-through exposure chamber; surviving animals received the last dose the day before scheduled necropsy. The inhalation exposure system comprised a pressurized gas cylinder containing the test item, a modular flow through, snout-only inhalation exposure chamber (aluminum alloy construction comprising a base unit, 7 animal exposure sections each containing 20 exposure ports, and top section incorporating a central inlet), restraining tubes, and a pre-chamber. A separate chamber was used for each group and housed in a separate extract cabinet to avoid possible cross-contamination. Doses of HFO-1234ze(E) were achieved by varying the HFO-1234ze(E) concentration in the exposure systems while keeping the duration of exposure constant; control animals were exposed to filtered air only. An airflow rate of 60 L/minute was applied for all groups. Separate chambers were used for each treatment group. Animals were rotated across chamber levels on a weekly basis. HFO-1234ze(E) atmosphere concentrations were determined daily; from each chamber, up to 3 samples (20 mL) were collected into syringes fitted with a Luer lock and analyzed by using gas chromatography using a ZB-5, 30 m × 0.32 mm ID, 0.25 mm film thickness analytical column and flame-ionization detection.
Calculation of Estimated Achieved Dose
Dose levels (mg/kg/day) were calculated as follows; a deposition factor was not applied.
C = HFO-1234ze(E) concentration in air (1 part per million (ppm) = 4.66 µg/L based on a molecular weight of 114 g/mol).
RMV = Respiratory minute volume = 0.608 x body weight (Kg).0.85212
D = Duration of exposure.
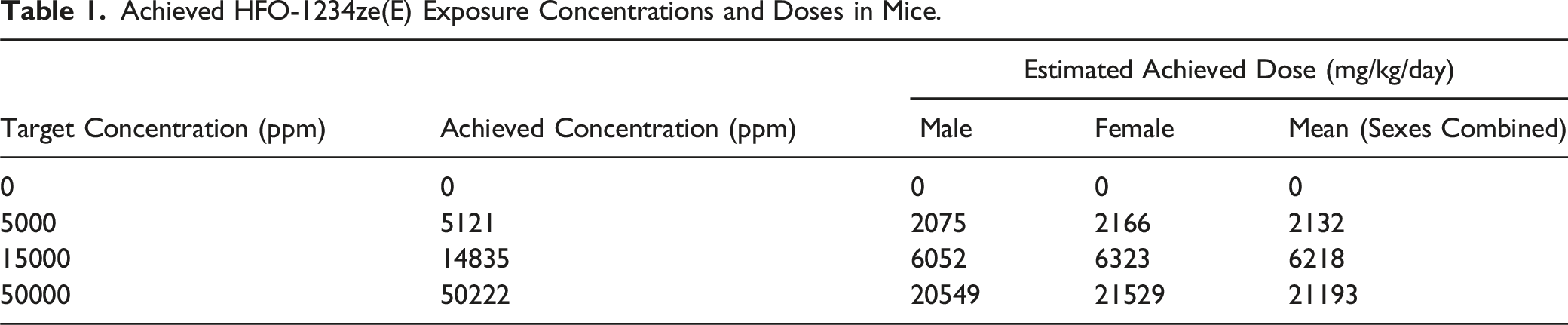
Achieved HFO-1234ze(E) Exposure Concentrations and Doses in Mice.
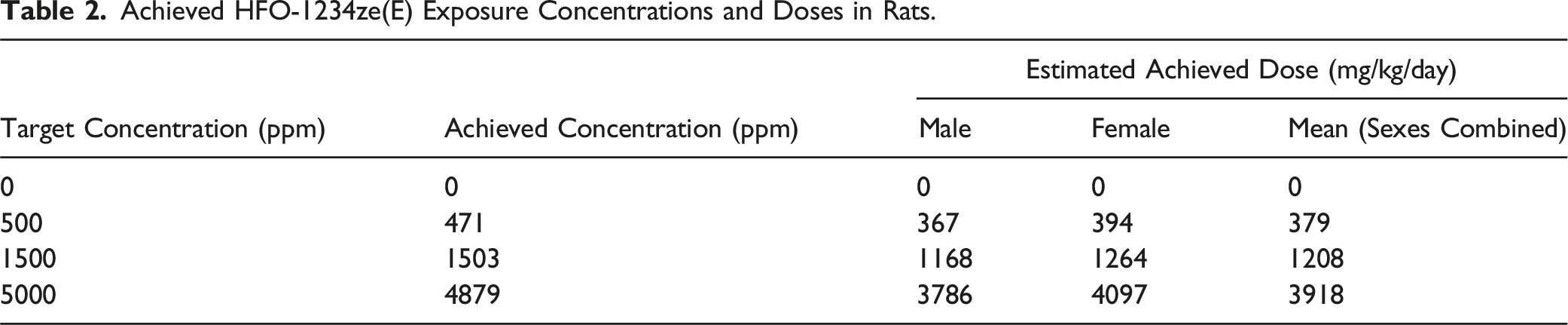
Achieved HFO-1234ze(E) Exposure Concentrations and Doses in Rats.
Animals and Animal Management
Crl:CD1(ICR) mice (5 to 6 weeks old at the start of dosing) and RccHanTM:WIST rats (6 to 7 weeks old at the start of dosing) were housed in groups of up to 3 (mice) or 5 (rats) of one sex in polycarbonate cages with a stainless steel mesh lids with wood shavings as bedding and provided ad-libitum access to food (Teklad 2014C diet) and water. Study rooms were maintained on a 12-hour light/dark cycle, at a controlled temperature (20 to 24°C) and relative humidity (40 to 70%). Environmental enrichment included plastic shelters, wooden balls/blocks, nestlets, and diamond paper twists (rats and mice) and swings (mice). Animals were acclimated to the exposure conditions once daily for at least 9 days prior to the first treatment day.
Study Design
Studies were conducted in AAALAC-accredited facilities (United Kingdom) in accordance with Good Laboratory Practice (GLP) standards, current guidelines for the non-clinical safety evaluation of pharmaceuticals, UK Home Office legislation (Animals [Scientific Procedures] Act 1986), and AstraZeneca’s Bioethics Standard. The number of animals used for both studies (69/sex/group for mice and 56/sex/group for rats) were the minimum considered necessary to ensure scientific integrity and regulatory acceptability; throughout, consideration was given to the welfare of individual animals with respect to the number and extent of procedures conducted. Study designs (including amendments) were agreed with the United States (US) Food and Drug Administration (FDA) Carcinogenicity Advisory Committee (CAC).
Dose Selection
Target HFO-1234ze(E) exposure concentrations were 5000, 15,000, or 50,000 ppm for mice (2 h/day) and 500, 1500, or 5000 ppm for rats (4 h/day); all animals were dosed 7 days per week. Concentrations and exposure durations were based on the outcome of 13- and 26-week toxicology studies in mice and rats, respectively, as described previously. 8 The respective high doses were selected to provide a sufficient exposure margin (on a mg/kg and mg/m2 basis) to the highest daily dose of HFO-1234ze(E) expected with any inhaled product. Lower doses provided approximately 3-fold separation across the respective studies.
During the mouse study, the incidence of non-HFO-1234ze(E)-related clinical observations in all groups (including air controls) was higher than expected and considered to be related to the duration of exposure (2 h). To ensure welfare, the study was refined to reduce the daily inhalation period from 2 h (weeks 1 to 49, inclusive) to 1 h (from Week 50). The target concentrations of HFO-1234ze(E) were not changed. Target and achieved concentrations and achieved doses are shown in Table 1 (mice) and Table 2 (rat).
Study Durations
The intended minimum dosing period for both studies was 104 weeks. Study-specific termination criteria were applied.
For mice, criteria included (sexes were considered separately) the following: • If, at any point, the control group fell to 20 animals, then all animals of that sex were to be euthanized (across all groups). • Up to Week 99, if survival in any HFO-1234ze(E)-treated group fell to 15 animals, then the remaining animals of that sex were to be euthanized (for the affected group). • From Week 100, if survival in any HFO-1234ze(E)-treated group fell to 15 animals, all remaining animals of that sex were to be euthanized (across all groups).
For rats, criteria included (sexes were considered separately) the following: • Up to Week 100, if survival in the control group fell to 15 animals, all remaining animals of that sex were to be euthanized (across all groups). • Up to Week 100, if survival in any HFO-1234ze(E)-treated group fell to 15 animals, then the remaining animals of that sex were to be euthanized (for the affected group). • From Week 101, if survival in any group fell to 15 animals, all remaining animals of that sex were to be euthanized (across all groups).
In-Life Assessments
All animals were assessed regularly for clinical observations, including palpation (at least twice daily), body weights (up to twice weekly), and food intake (up to weekly).
Necropsy
All animals terminated at scheduled or unscheduled intervals were euthanized by an overdose of intraperitoneal pentobarbitone sodium followed by exsanguination. For mice, surviving females were necropsied in Week 100 (target 50,000 ppm) or Week 101 (target 0, 5000, or 15,000 ppm) and surviving males in Week 105 (all doses). For rats, surviving animals were necropsied during weeks 105 and 106.
Tissue Preservation and Examination.

Histopathology and Microscopic Examination
Both carcinogenicity studies were evaluated and peer reviewed by veterinary pathologists experienced in the assessment of toxicologic pathology. Industry standard criteria and guidance were followed for the diagnosis of neoplastic findings. 13
Tissues for histology were dehydrated, embedded in paraffin wax, sectioned (four-to-five-micron thickness), and stained with hematoxylin and eosin prior to examination by using light microscopy. The pathology slides and draft pathology phase report were subject to a peer review by AstraZeneca. Factors contributing to death were recorded for any animals necropsied at unscheduled intervals or which died intercurrently.
Hematology
Blood samples for hematology were collected from surviving animals into tubes containing K2EDTA anticoagulant. In mice, terminal samples (0.3 mL via cardiac puncture) were collected in Week 100 (females at target 50,000 ppm), Week 101 (females at target 0, 5000, or 15,000 ppm), and Week 105 (males at all doses). For the rat study, samples (0.5 mL via jugular venipuncture) were collected in Week 104. Samples were analyzed for hematocrit, hemoglobin concentration, erythrocyte count, absolute reticulocyte count, mean cell hemoglobin, mean cell hemoglobin concentration, mean cell volume, red cell distribution width, platelet count, and leukocyte count (total and differential) using an ADVIA® Analyzer (Siemens Healthcare, Germany). Blood films (Romanowsky stain) were prepared and examined by using light microscopy in the event of flags from the analyzer.
Statistical Analysis
Statistical analysis was performed for males and females separately using the individual animal as the basic experimental unit, except for food consumption which was analyzed on a cage basis. The following comparisons were performed for each study: Group 1 versus 2, 3, and 4. Tests to compare survival were performed using log-rank and Wilcoxon tests, with a two-sided risk. Tests to compare tumor incidence were performed, with a one-sided risk for increasing incidence with dose. Non-palpable tumors were analyzed by the International Agency for Research on Cancer (IARC) asymptotic fixed interval-based prevalence and death rate tests. 14 Observable or palpable (superficial as in mammary or skin) tumors were analyzed using the methods previously described for analyzing survival, using the time to death or time of detection of the tumor (in weeks) as a surrogate for the tumor-onset time. Indication of a possible treatment effect was assessed based on rare or common tumor type, in line with the current FDA guidelines. 15 Site or tumor combinations were also statistically analyzed as per current guidance, 16 using the same statistical methods as described for the individual tumors (above).
Results
Mortality
Mouse
Incidence of Mortality and Statistical Analysis of Survival Data in Mice.
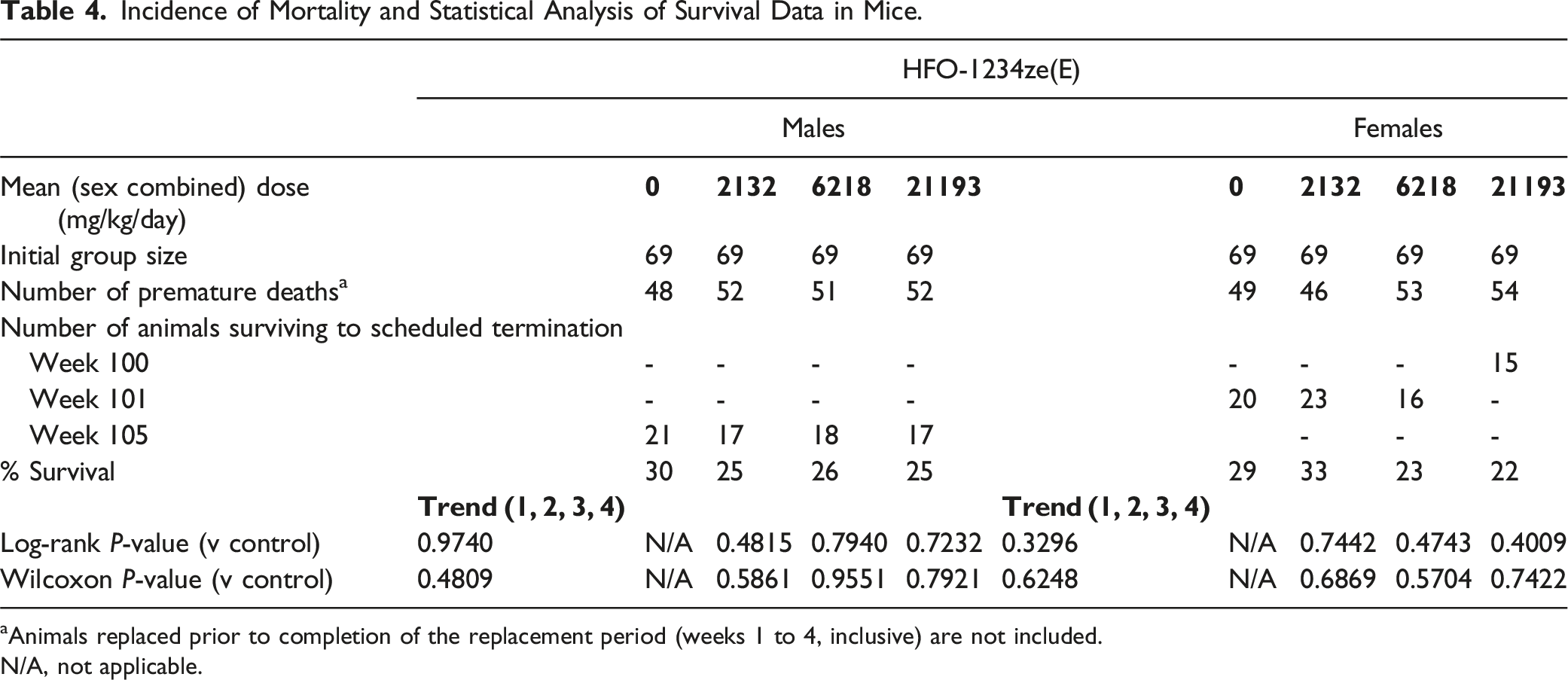
aAnimals replaced prior to completion of the replacement period (weeks 1 to 4, inclusive) are not included.
N/A, not applicable.
Rat
Incidence of Mortality and Statistical Analysis of Survival Data in Rats.
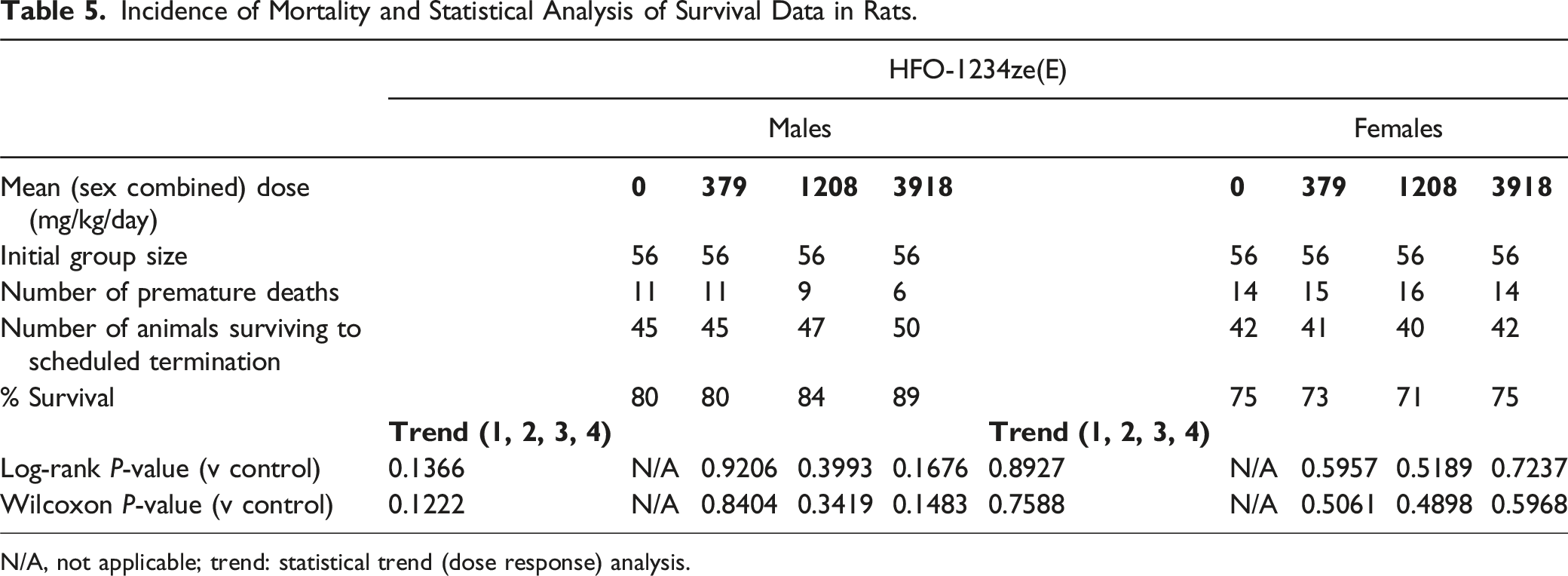
N/A, not applicable; trend: statistical trend (dose response) analysis.
Anatomic Pathology
Mouse
Factors contributing to death were typical of those commonly seen in Crl:CD1(ICR) mouse carcinogenicity studies and had a similar incidence across control and treated groups. No HFO-1234ze(E)-related macroscopic findings were observed for animals found dead or euthanized prematurely during the treatment period or at scheduled termination. There were no HFO-1234ze(E)-related non-neoplastic or neoplastic microscopic findings after 100-, 101-, or 104 weeks of exposure.
Mice: Group Distribution of Neoplastic (With Statistical Analysis) and Selected Non-neoplastic Lesions.
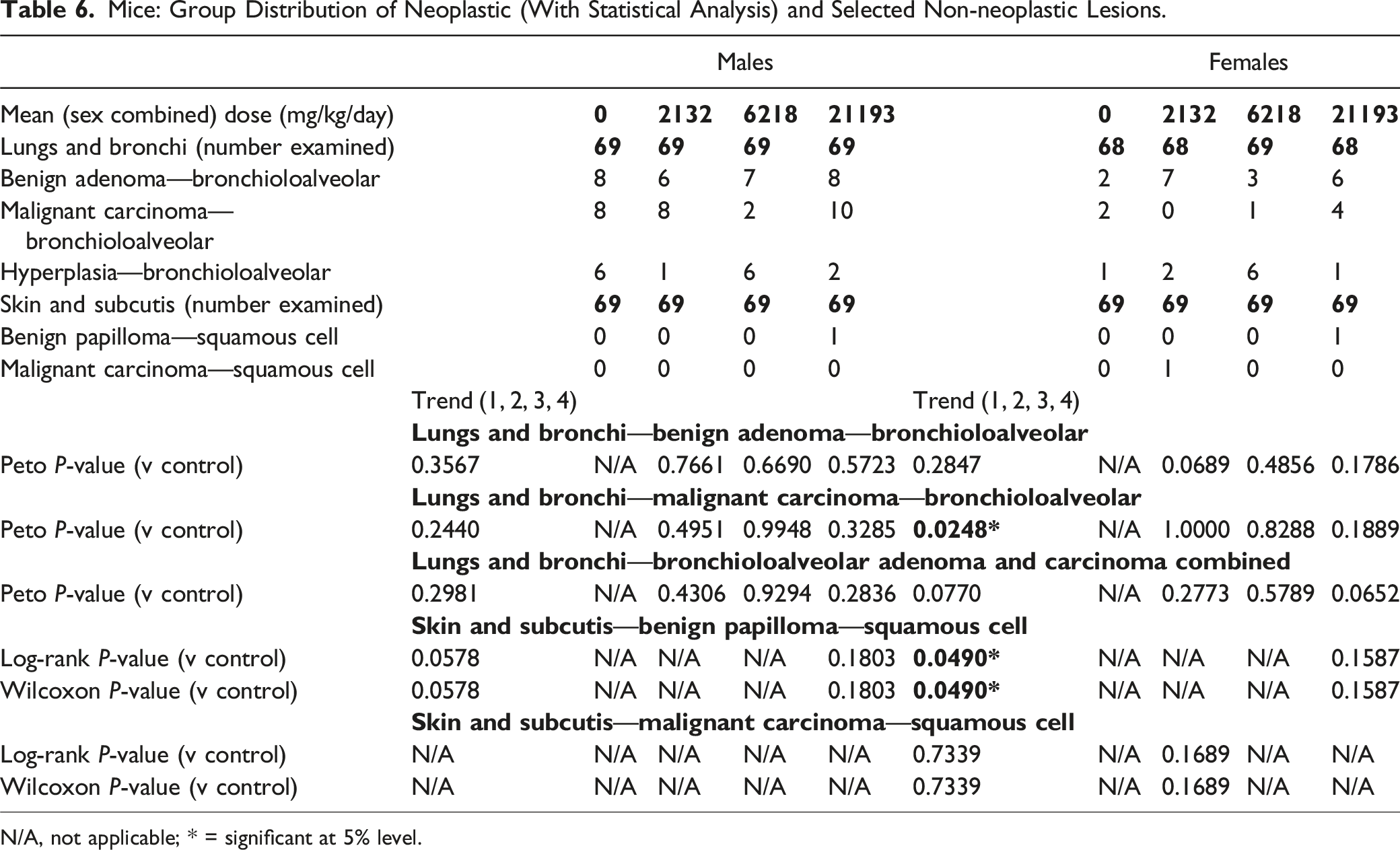
N/A, not applicable; * = significant at 5% level.
Test Facility Historical Control Histopathology Data From 104-Week Studies in Female Crl:CD1(ICR) Mice.
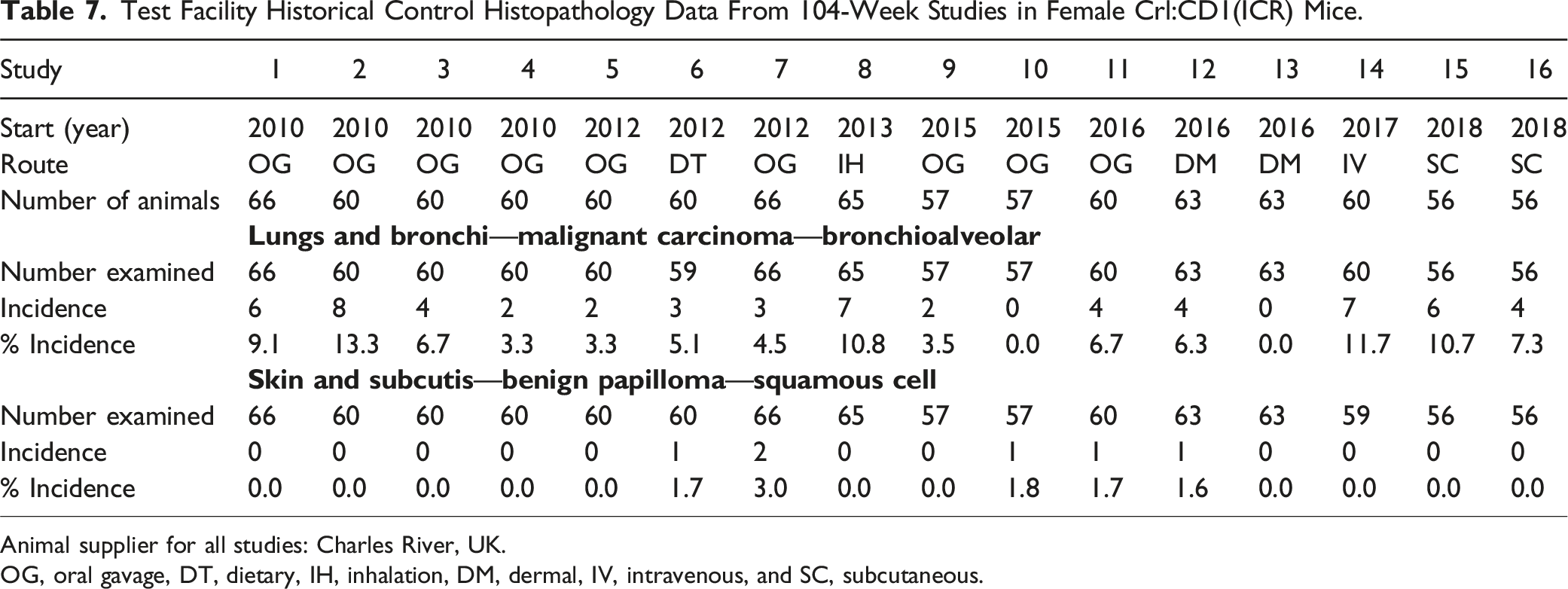
Animal supplier for all studies: Charles River, UK.
OG, oral gavage, DT, dietary, IH, inhalation, DM, dermal, IV, intravenous, and SC, subcutaneous.
Squamous cell papilloma was observed in the skin in one male (1/69; 1.4%) and one female (1/69; 1.4%) exposed to HFO-1234ze(E) at 21,193 mg/kg/day. These single incidences were within the range of test facility HCD for this tumor type in males (mean 0.4%; range 0-3%) and females (mean 0.6%; range 0-3%). As the background incidence was <1%, squamous cell papilloma was classified as “rare” as per FDA guidance. 15 When assessed for trend, with all terminal and decedent animals included in the analysis, this finding was statistically significant at the 5% level in females only (P = 0.0490 in both the log-rank test and the Wilcoxon test) but did not meet the FDA criteria for significance for rare tumors (P ≤ 0.025) in either sex. None of the pairwise comparisons were statistically significant in males or females. Based on the single incidence and a lack of statistical significance at the 2.5% level for rare tumors, these occurrences of squamous cell papilloma were considered incidental and not related to exposure to HFO-1234ze(E).
Rat
Factors contributing to death were typical of those commonly seen in RccHan™:WIST rat carcinogenicity studies and had a similar low incidence across control and treated groups.
There were no HFO-1234ze(E)-related macroscopic findings.
Rats: Group Distribution of Neoplastic (With Statistical Analysis) and Selected Non-neoplastic Lesions.
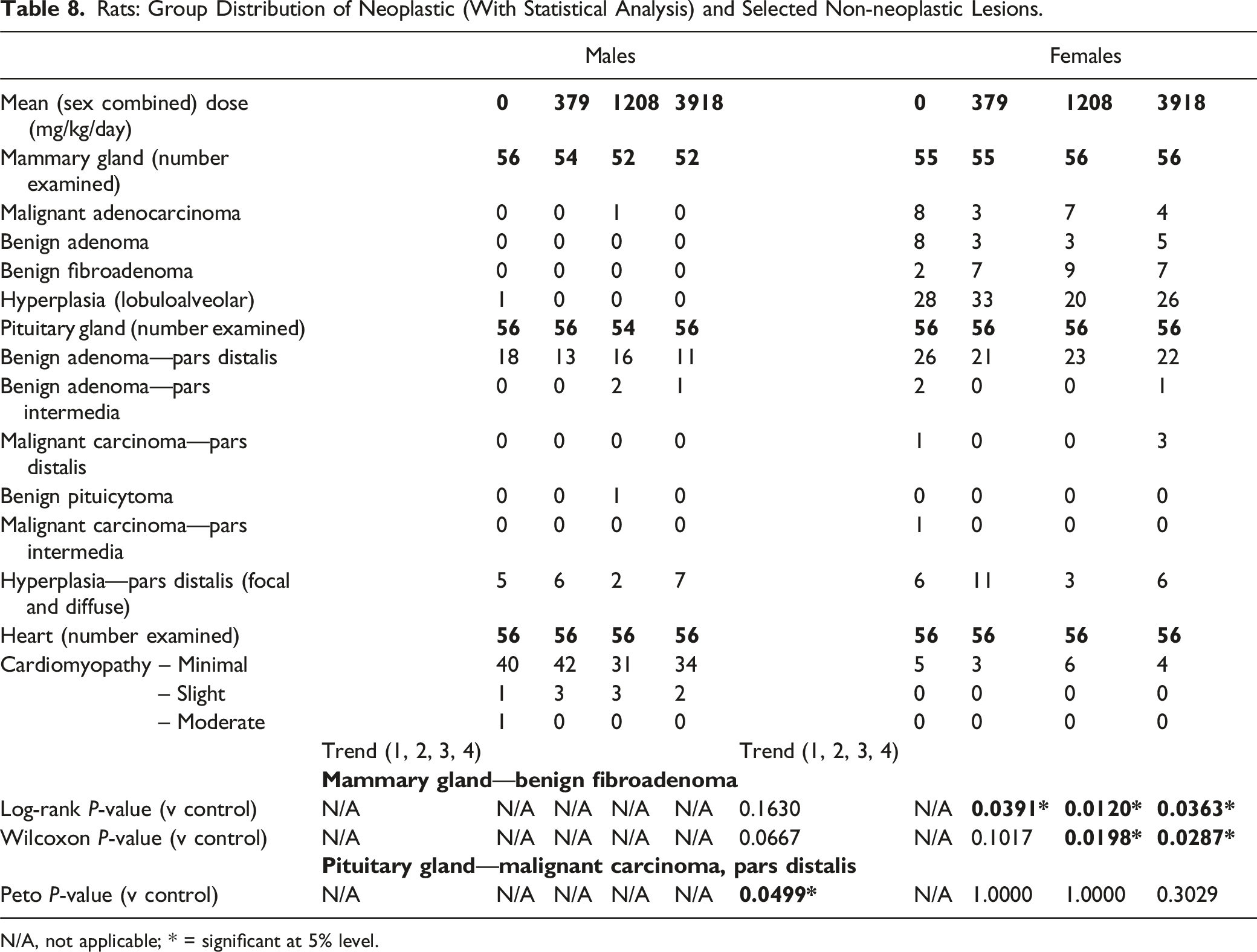
N/A, not applicable; * = significant at 5% level.
Test Facility Historical Control Histopathology Data From 104-Week Studies in Female Wistar Han Rats.
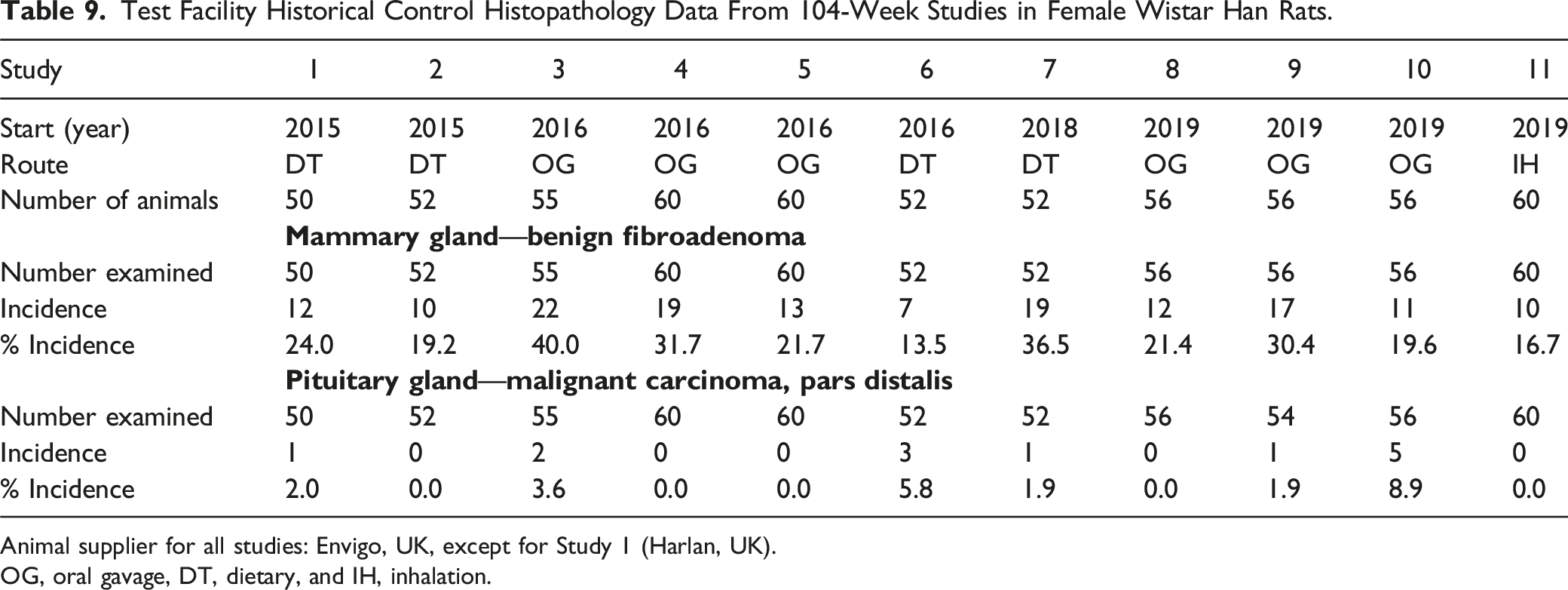
Animal supplier for all studies: Envigo, UK, except for Study 1 (Harlan, UK).
OG, oral gavage, DT, dietary, and IH, inhalation.
In females, the incidence of mammary gland fibroadenoma was marginally increased in all HFO-1234ze(E)-groups, but with no evidence of a dose response. For all HFO-1234ze(E)-treated groups, the increased incidence was statistically significant at a P-value of ≤0.05 using a log-rank test, while only animals exposed to an achieved concentration of 1208 and 3918 mg/kg/day demonstrated a statistically significant increase at a P-value of ≤0.05 using a Wilcoxon test. However, the increased incidence noted in all dose groups did not reach a level considered to be statistically significant using FDA guidance (P-value of 0.01). Fibroadenoma is a common tumor in aged Wistar rats including the RccHan™:WIST strain17,18 and test facility HCD for the RccHan™:WIST strain confirmed this, with a range of 13.5-40%. The incidence of fibroadenoma in the control group (3.6%) suggests that the apparent increase in incidence in treated animals is likely to have resulted from an abnormally low incidence in concurrent controls. Published ranges for mammary fibroadenoma in RccHan™:WIST18,19 confirm an overall incidence of 28.3%, further supporting the low incidence of fibroadenomas in the concurrent control group. The incidence of fibroadenoma in HFO-1234ze(E)-treated groups (13%, 16%, and 12.5%) all fell within the range of test facility HCD data (13.5-40%), with incidences in HFO-1234ze(E)-treated animals either below or at the low end of the test facility range.
The incidences of mammary gland adenoma and mammary gland adenocarcinoma were reduced in females in all HFO-1234ze(E) groups. In addition, the incidence of all benign mammary tumors or the combination of all benign and malignant mammary tumors was not increased in animals administered HFO-1234ze(E). The incidence of mammary hyperplasia also showed no increase in animals dosed with HFO-1234ze(E). The weight of evidence therefore supports that the marginally apparent increase in the incidence of mammary fibroadenomas in females was incidental and not HFO-1234ze(E)-related.
The incidence of pituitary carcinoma of the pars distalis was marginally increased in females dosed with HFO-1234ze(E) at an achieved concentration of 4879 ppm. Although this was statistically significant using a Peto trend test at a P-value of ≤0.05, this did not reach a level considered to be statistically significant using FDA guidance (P-value of 0.005). The incidence of pituitary carcinoma of the pars distalis in females exposed to an achieved concentration of 3918 mg/kg/day (5.4%) fell within the range of test facility HCD (0-8.9%). Carcinoma of the pars distalis of the pituitary gland in rats arises from adenoma of the pars distalis and cannot be readily distinguished from adenomas based on the nature of the cells or the architectural pattern of the neoplasia. Indeed, the distinction between adenoma and carcinoma of the pituitary gland is usually based on histological evidence of local infiltration into adjacent structures such as the brain.20,21 However, in accordance with standard practice, the pituitary gland is removed and separated from adjacent tissues at necropsy, which can make the distinction of carcinoma from adenoma more difficult. The development of proliferative lesions within the pars distalis of the pituitary shows a progression from hyperplasia to adenoma and carcinoma,21,22 and a true test article related effect should be reflected in differences in the incidence of each of these findings in the pars distalis, and not just the presence of carcinoma alone. The incidence of pituitary adenoma of the pars distalis in both sexes was slightly reduced in HFO-1234ze(E)-treated groups; there was no evidence of an HFO-1234ze(E)-related effect in the incidence of focal or diffuse foci of hyperplasia within the pars distalis of either sex. The combined incidence of carcinoma and adenoma of the pars distalis was reduced in all female treated groups. Overall, the weight of evidence supports that the marginal increase in the incidence of pituitary carcinoma of the pars distalis in females was incidental and not HFO-1234ze(E)-related.
There were no HFO-1234ze(E)-related non-neoplastic findings. In subchronic and chronic studies in rats, exposure to very high concentrations of HFO-1234ze(E) was associated with an exacerbation of rodent chronic progressive cardiomyopathy. 8 The incidence and severity of chronic cardiomyopathy in HFO-1234ze(E)-treated rats in the two-year carcinogenicity study were similar to concurrent controls (Table 8), confirming a lack of progression between the chronic and carcinogenicity studies.
Discussion
HFO-1234ze(E), a next-generation propellant with near zero global warming potential for use in inhaled pMDI drug products, was assessed in two-year carcinogenicity studies in mice and rats. In both studies, there were no HFO-1234ze(E)-related in-life findings and survival at the end of the studies was sufficient to enable an assessment of its carcinogenic potential.
Aged rodents develop a range of spontaneous neoplasms with tumor profiles that are often influenced by strain, breeder, and date of the in-life phase of the study.18,23-25 It is therefore important to consider strain and test facility specific HCD when evaluating the results of carcinogenicity studies.26,27 Further, test facility HCD is also used to establish whether a tumor is designated as common (spontaneous incidence >1%) or rare (spontaneous incidence ≤1%) with respect to statistical analysis.15,28 When analyzing carcinogenicity studies, the statistical methods recommended in the FDA draft guidance for industry 15 are closely followed by statisticians within the FDA Center for Drug Evaluation and Research as well as in pharmaceutical companies and Contract Research Organizations. The FDA guidance was originally developed for submissions that include 2 standard rodent carcinogenicity studies (i.e., the approach used to assess carcinogenicity risk with HFO-1234ze(E)) and recommends that a positive dose-response (or trend) in incidence rates of an individual tumor type be tested at 0.005 or 0.025 significance level and that a pairwise comparison of an increase in incidence rates in a treated group over the control group of the tumor type be tested at 0.01 or 0.05 significance level for a common or a rare tumor type, respectively. Use of these decision rules for individual tumor types results in an overall false-positive rate of approximately 10% in a standard submission containing 2 chronic studies in both sexes of rats and mice.29,30 A 10% overall false-positive rate has been determined appropriate when analyzing the results of carcinogenicity studies. None of the tumor types in HFO-1234ze(E)-treated mice or rats were statistically significant as per FDA guidance and differences in tumor incidence fell within test facility HCD, confirming they were not HFO-1234ze(E)-related.
An exacerbation of rodent progressive cardiomyopathy (RPCM), a commonly reported background finding in rats,31,32 was reported in male rats in toxicology studies with HFO-1234ze(E); in a 26-week repeat dose study, a No Observed Effect Level (NOAEL) of 4280 mg/kg/day (5000 ppm) was identified based on the presence of RPCM at the highest dose tested (12,000 mg/kg/day). 8 The incidence and severity of RPCM in the 104-week rat carcinogenicity study were comparable in control and high-dose (3918 mg/kg/day, 5000 ppm) animals, demonstrating no further exacerbation between 26 weeks and 2 years of treatment at this dose level.
HFO-1234ze(E): Summary of Safety Margins From Two-Year Carcinogenicity Studies.
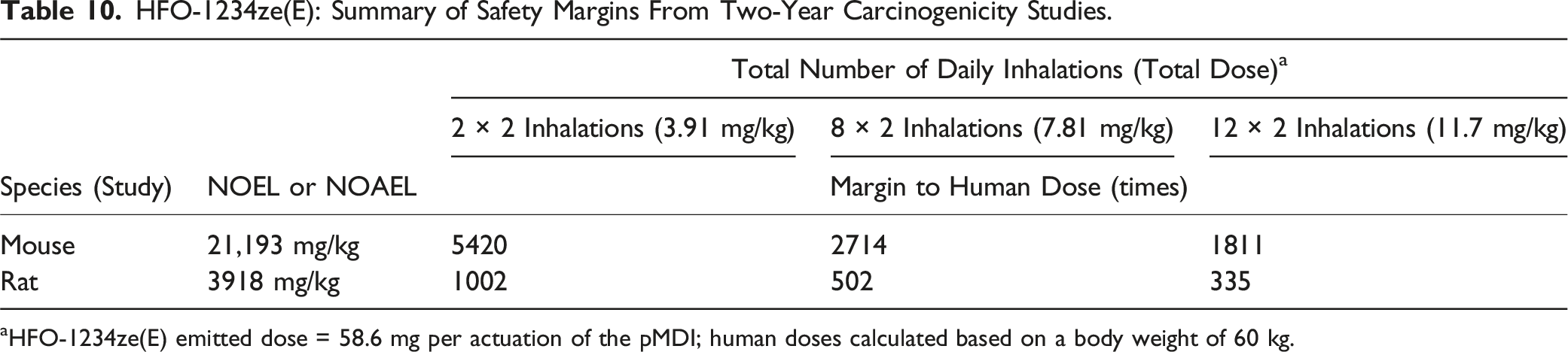
aHFO-1234ze(E) emitted dose = 58.6 mg per actuation of the pMDI; human doses calculated based on a body weight of 60 kg.
Conclusion
HFO-1234ze(E) was not carcinogenic in two-year inhalation carcinogenicity studies in mice or rats at doses which significantly exceed those expected in patients. These data support the use of HFO-1234ze(E) as a medical propellant for use in pMDIs.
Footnotes
Acknowledgments
The authors would like to acknowledge colleagues at Honeywell International Inc. and the department of Clinical Pharmacology and Safety Sciences at AstraZeneca for their support in the review of data and discussions throughout the lifecycle of this project.
Author Contributions
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Regulatory Science Associates (PC) was paid by AstraZeneca for inhalation toxicology consulting services. Labcorp (JS) were paid by AstraZeneca for the conduct of the mouse and rat carcinogenicity studies. PG, MJ, JC, RH, NW, LW, PA, and SP are employees of AstraZeneca and may hold stock.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.