Abstract
Nanoplastics (NPx) can enter living organisms, including humans, through ecosystems, inhalation, and dermal contact and can be found from the intestine to the brain. However, it is unclear whether NPx accumulates and affects the dopamine system. In this study, we investigated the effects of NPx on the dopamine system in cultured murine cerebral cortex neurons. Cultured cerebrocortical neurons were treated with 100 nm NPx at the following concentrations for 24 h: 1.896 × 105, 3.791 × 106, 7.583 × 107, 1.571 × 109, 3.033 × 1010, and 3.033 × 1011 particles/mL. Dopamine-associated proteins were analyzed using immunofluorescence staining. NPx treatment induced its accumulation in neurons in a dose-dependent manner and increased the levels of dopamine receptors D1 and D2 and their co-expression. However, NPx treatment did not affect the levels of other dopamine receptors, dopamine transporters, tyrosine hydroxylase, and microtubule-associated protein 2, or synaptophysin in neuronal structures. This study demonstrated that NPx is a potential modulator of the dopamine system via its receptors rather than its synthesis and reuptake in neurons and may be associated with dopamine-based psychiatric disorders.
Introduction
Industrial development contributes to the convenience of human life but causes the overproduction of environmental pollutants. Among many pollutants, nanoplastics (NPx) are degraded microplastics and are released from sources such as electronics and commercial products. 1 NPx are defined as plastic particles less than 1 μm. In contrast, microplastics refer to plastic particles between 1 μm and 1000 μm. 2 Notably, since the size of NPx is similar to that of a virus, they can easily penetrate biological membranes. Therefore, living organisms are more susceptible to these than to microplastics.2,3
NPx can enter living organisms, including humans, through ecosystems, inhalation, and dermal contact.
4
In the case of humans, NPx are found in the intestine, liver, lungs, and kidney.
1
In
DA is a major neurotransmitter that originates from the ventral tegmental area and the substantia nigra, which is compact and innervates the prefrontal cortex, striatum, and amygdala.9,10 Tyrosine hydroxylase synthesizes DA from tyrosine; calcium influx subsequently activates the vesicular monoamine transporter (VMAT)-2 to release DA into the extracellular space. Extracellular DA can then either undergo reuptake by the DA transporter (DAT) on presynaptic nerve terminals or bind to 5 types of DA receptors, characterized as D1R, D2R, D3R, D4R, and D5R.10,11 DA receptor binding specifically affects synaptic plasticity via various signal cascades, and all these processes are considered part of the DA system. Alterations in the DA system have been implicated in several psychiatric disorders, including schizophrenia, attention deficit hyperactivity disorder, and Parkinson’s disease.12-14 The accumulation of pollutants in living organisms affects brain functions associated with the DA system. The influx of perfluoroalkyl substances as pollutants decreased the DA content and tyrosine hydroxylase expression in the brain,
15
whereas lead exposure increased tyrosine hydroxylase and DAT levels in the brain.
16
Particulate matter exposure accelerated dopaminergic neuron death in a Parkinson’s disease cellular model.
17
However, the effects of NPx on the DA system have rarely been reported. In zebrafish, neither bisphenol A nor 50 nm NPx caused any changes in DA levels, although their combination induced an increase in DA levels in the head of zebrafish.
18
Using SH-SY5Y cells as representative dopaminergic neurons, 50 nm NPx led to cell death by activating mitophagy through the adenosine monophosphate-activated protein kinase (AMPK)/Unc-51 like autophagy activating kinase 1 (ULK1) pathway.
19
This indicates that NPx, similar to other pollutants, can diffuse into the brain and affect the DA system. However, the exact mechanism through which NPx affect the DA system is unclear; therefore, in this study, we investigated the effects of NPx on the DA system
Materials and Methods
Primary Cell Culture
Animal experiments were conducted in accordance with the Institutional Animal Care and Use Committee of Daegu Catholic University (IACUC-2023-030). Institute of Cancer Research (ICR) mice, an outbred strain, were chosen for primary cell culture because they have a similar genetic background to humans, which can simulate the genetic diversity found in humans. 20 Cerebrocortical neurons from ICR mice were cultured on embryonic day 14 (Youngnam-Bio, Inc, Daegu, Korea). After pregnant ICR mice were anesthetized, embryos were separated from the dams, their brains were removed from the skull, and cerebrocortical neurons were dissected. Eight-well chamber slides were coated with 5.0 μg/mL poly-D-lysine (catalog #P6407, Sigma-Aldrich, St. Louis, MD, USA) for 24 h prior to cell harvesting. Cerebrocortical neurons were cultured at 1.5 × 105 cells/well on these coated slides in a neurobasal medium (catalog # 21103049, Gibco, Rockville, MD, USA) supplemented with 12% horse serum (catalog # 26050070, Gibco), 0.6% glucose, and 2 mM L-glutamine, in an incubator controlled at 37°C with 5% CO2. One day after incubation, the medium was replaced with a fresh neurobasal medium supplemented with 2% B-27 (catalog # 17504044, Gibco). After 4 days of cultivation, the cells were treated with 100 nm NPx (Catalog # 64010-15, Polysciences, Warrington, PA, USA) at the following concentrations: 1.896 × 105, 3.791 × 106, 7.583 × 107, 1.571 × 109, 3.033 × 1010, and 3.033 × 1011 particles/mL. The same doses were used for 100 nm fluorescent-NPx (Catalog # PS100-FC-1, Nanocs, New York City, NY, USA). As a control, the vehicle (VEH) was treated with distilled water at the same volume as the NPx solution. After 24 h, all cells were treated with 4% paraformaldehyde for 2 h, and immunostaining was performed.
Fluorescence Immunostaining
The mouse monoclonal anti-tyrosine hydroxylase (TH; catalog # ab129991, Abcam, Cambridge, UK), rabbit polyclonal anti-DA transporter (DAT; catalog # sc-14002, Santa Cruz, Dallas, TX, USA), rabbit polyclonal anti-DA (catalog # ab6427, Abcam), rabbit polyclonal anti-D1R (catalog # sc-14001, Santa Cruz), mouse monoclonal anti-D2R (catalog # sc-5303, Santa Cruz), mouse monoclonal anti-D3R (catalog # sc-136170, Santa Cruz), rabbit polyclonal anti-D4R (catalog # ab20424, Abcam), rabbit polyclonal anti-D5R (catalog # MBS2516950, Mybiosource, San Diego, CA, USA), mouse monoclonal anti-synaptophysin (SYP, catalog #s5768, Abcam), rabbit polyclonal anti-SYP (catalog # PA1-1043, Invitrogen, Carlsbad, CA, USA), mouse monoclonal anti-microtubule-associated protein 2 (MAP2; sc-32791, Santa Cruz), and rabbit polyclonal anti-MAP2 (AB5622, Merck Millipore, Burlington, MA, USA) were used as primary antibody. Twenty-four hours after NPx treatments, the cells were fixed with 4% paraformaldehyde for 2 h and washed three times with phosphate buffer saline (PBS). The cells were subsequently incubated with primary antibodies for 24 h at 4°C. After treatment, secondary Alexa Fluor 488 goat anti-mouse IgG antibody (catalog # A-11001, Life Technologies, Waltham, MA, US) and Alexa Fluor 594 goat anti-rabbit antibody (catalog # A-11012, Life Technologies) were added for a 2 h incubation at room temperature. For nuclei visualization, 4′,6-diamidino-2-phenylindole (DAPI; catalog # 10236276001, Roche, Basel, Switzerland) was added for a 10 min incubation. After washing thrice with PBS, the immunostained cells were mounted with an aqua-poly mount (catalog # 18606, Polysciences, Warrington, PA, USA). Images of stained cells were captured using a fluorescence microscope (Leica DM2500, Leica Microsystems, Wetzlar, Germany). Detailed information on immunostaining for the “no primary antibody” controls is provided in Supplemental Materials and Methods.
Image Analysis
Images were captured at random culture sites for each treatment condition using a fluorescence microscope (Leica DM 2500, Leica Microsystems). Quantification of fluorescent signals in the images was conducted using ImageJ software (version 0.19.0, JASP Team, 2024). Fluorescence intensity was calculated as the number of cells with fluorescent signals divided by the total number of cells with DAPI staining in each image; this was then expressed as a percentage of the VEH ratio.
Statistical Analysis
All results are expressed as mean ± standard error. These were analyzed using OriginPro (version 2024; OriginLab Corporation, Northampton, MA, USA). All data were analyzed using a one-way analysis of variance (ANOVA) with post-hoc Tukey’s test. Statistical significance was set at
List of Abbreviations.
Results
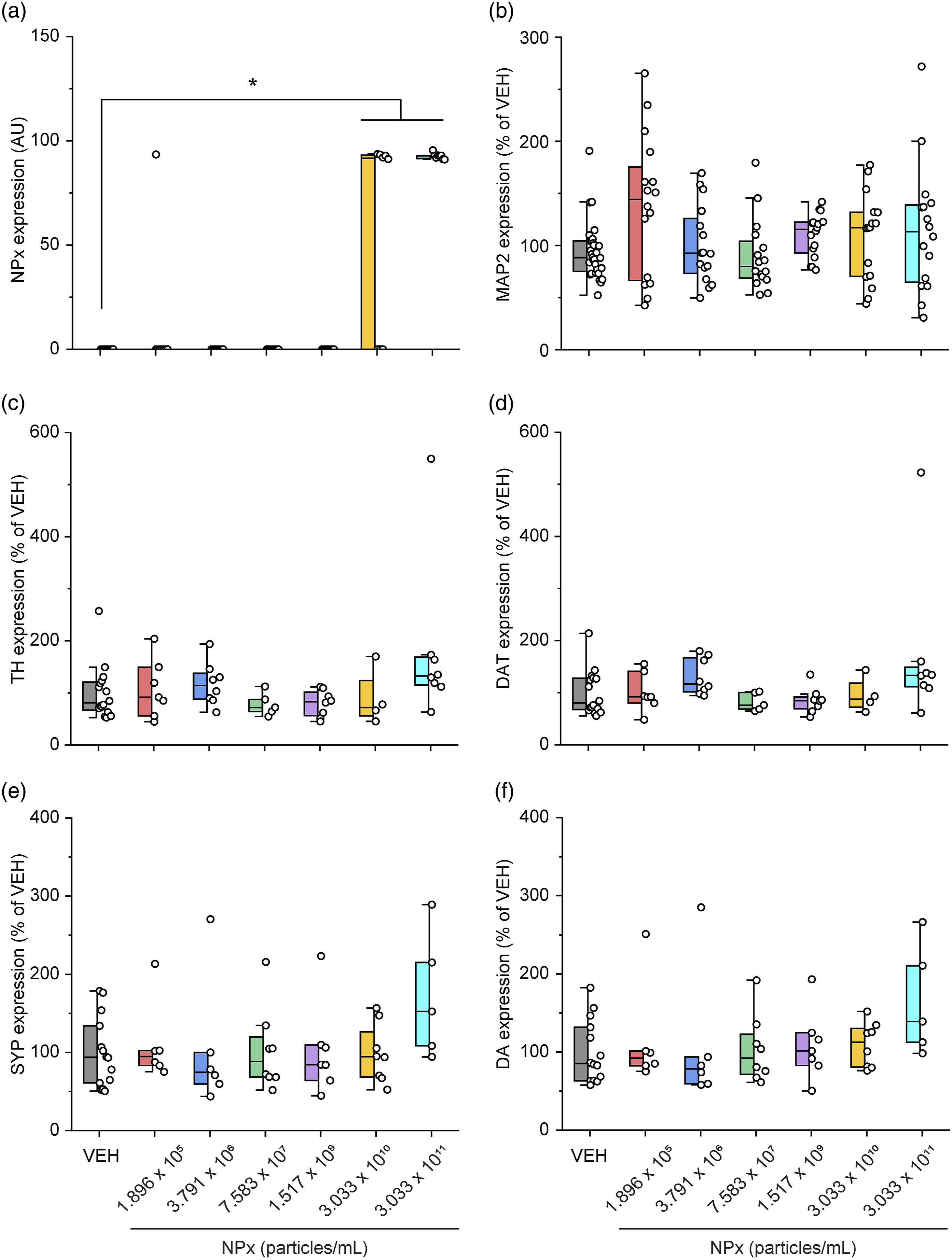
NPx treatment induced the accumulation of NPx at 3.033 × 1010 and 3.033 × 1011 particles/mL (F (6.57) = 28.107, NPx accumulation visualized with fluorescent-NPx and the effect of NPx on the DA system in cerebrocortical neurons. Fluorescent images of cerebrocortical neurons treated with NPx showing DAPI (blue color) and (a) NPx (green color) and MAP2 (red color); (b) TH (green) and the DAT (red); and (c) SYP (green) and DA (red). DA, dopamine; DAPI, 4′,6-diamidino-2-phenylindole; MAP2, microtubule-associated protein 2; NPx, nanoplastics; SYP, synaptophysin; TH, tyrosine hydroxylase; VEH, vehicle. The intensity of NPx accumulation and protein expression modulated by NPx treatment. The intensity of fluorescence for (a) NPx, (b) MAP2, (c) TH, (d) DAT, (e) SYP, and (f) DA. A one-way ANOVA with a post-hoc Tukey’s test was used to analyze the fluorescence intensities for significance, with 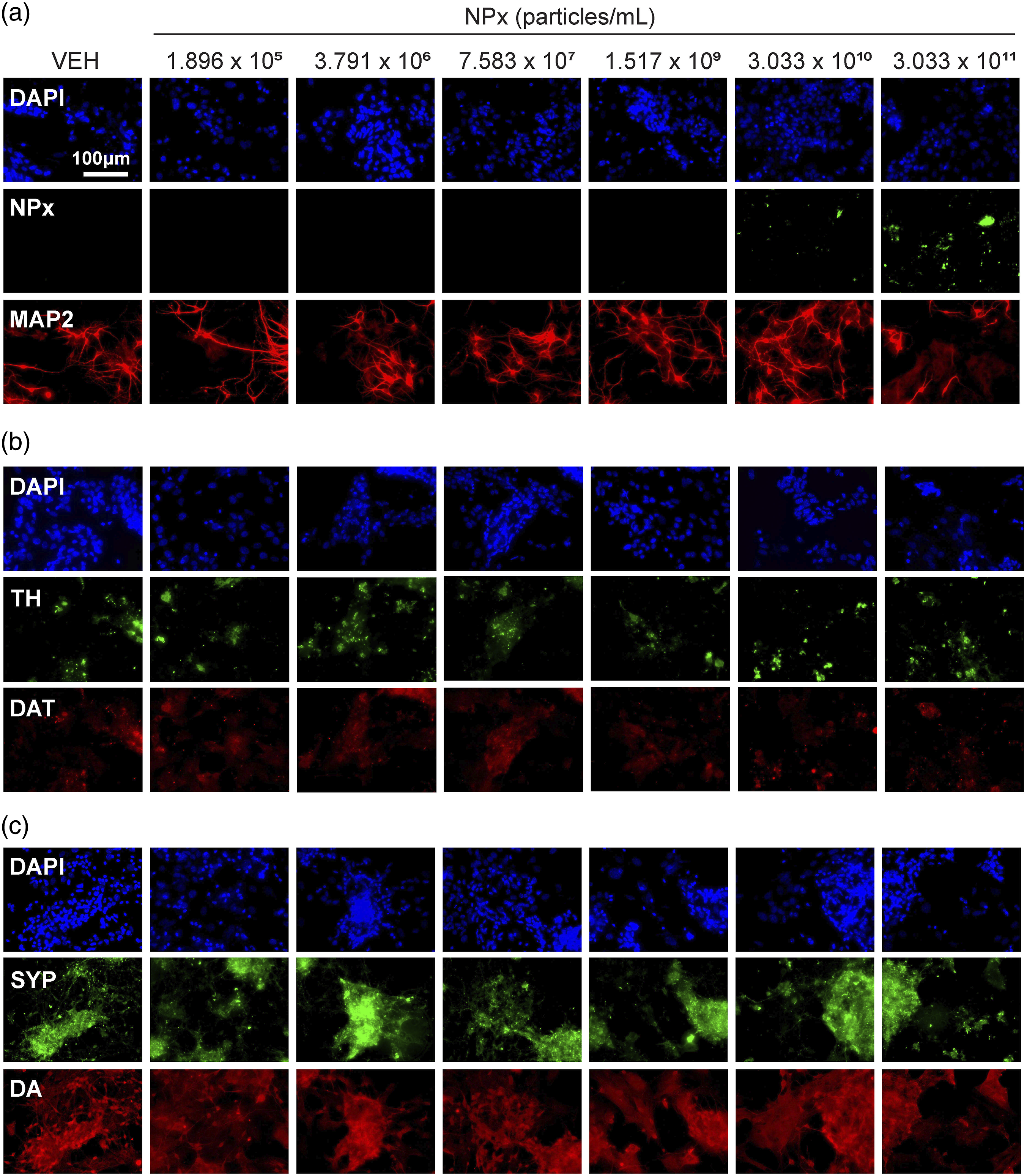
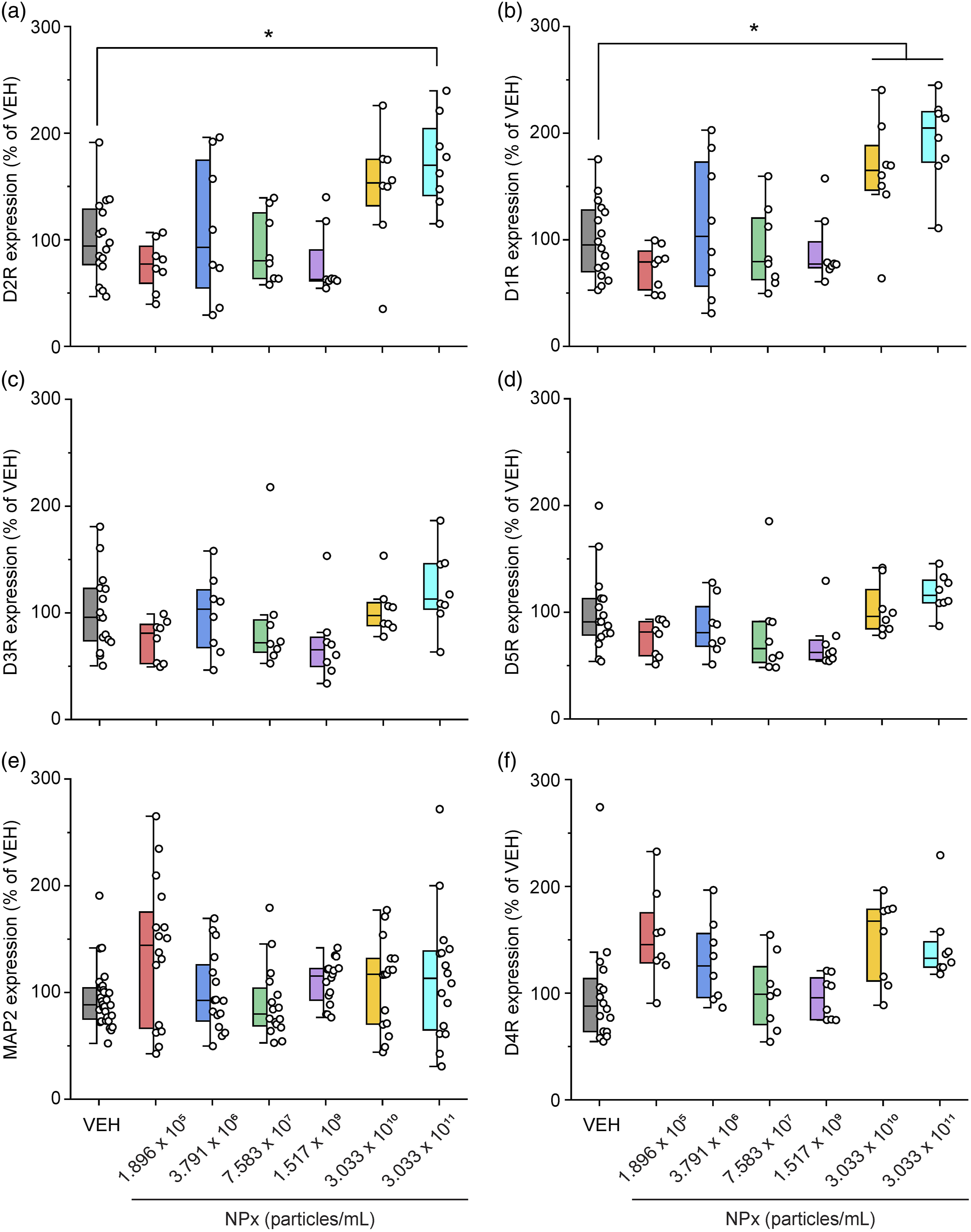
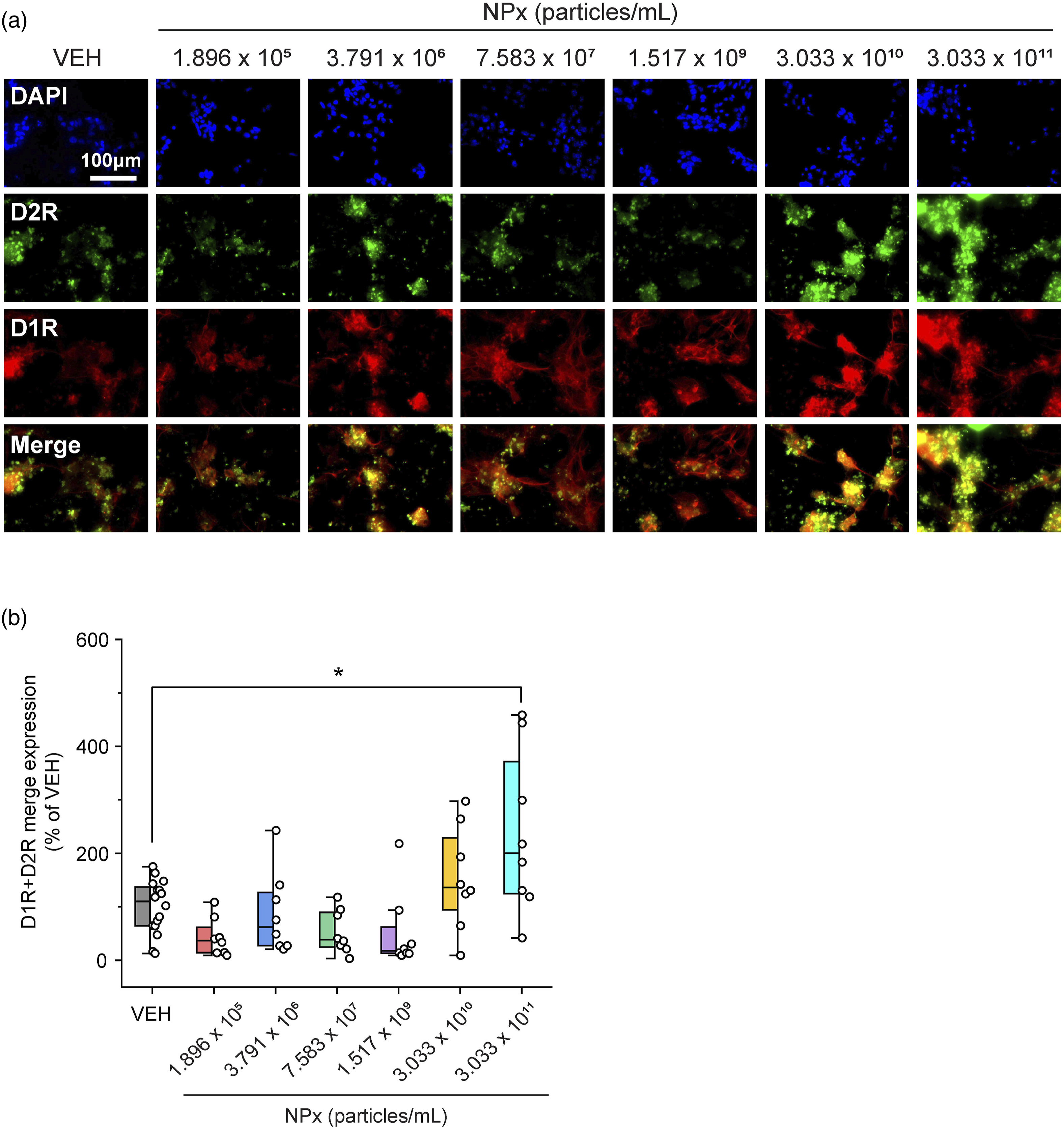
Of the five types of DA receptors, NPx treatment increased the levels of D1R and D2R. D1R levels increased with the two highest doses of NPx compared with VEH treatment (F (6.57) = 9.026, The effects of NPx on DA receptor expression in cerebrocortical neurons. Fluorescent images of cerebrocortical neurons treated with NPx showing DAPI (blue color) and (a) D2R (green color) and D1R (red color), (b) D3R (green) and D5R (red), and (c) MAP2 (green) and D4R (red). D1R, dopamine D1 receptor; D2R, dopamine D2 receptor; D3R, dopamine D3 receptor; D4R, dopamine D4 receptor; D5R, dopamine D5 receptor; DA, dopamine; DAPI, 4′,6-diamidino-2-phenylindole; MAP2, microtubule-associated protein 2; NPx, nanoplastics; VEH, vehicle. The intensity of DA receptor and MAP2 expression modulated by NPx treatment. The intensity of fluorescence for (a) D2R, (b) D1R, (c) D3R, (d) D5R, (e) MAP2, and (f) D4R. A one-way ANOVA with a post-hoc Tukey’s test was used to analyze the fluorescence intensities for significance, with The intensity of D1R and D2R co-expression modulated by NPx treatment. (a) Fluorescent images of cerebrocortical neurons treated with NPx showing DAPI (blue color), D2R (green color), D1R (red color), and its merge (yellow color). (b) The intensity of fluorescence for the merge of D1R and D2R. A one-way ANOVA with a post-hoc Tukey’s test was used to analyze the fluorescence intensities for significance, with 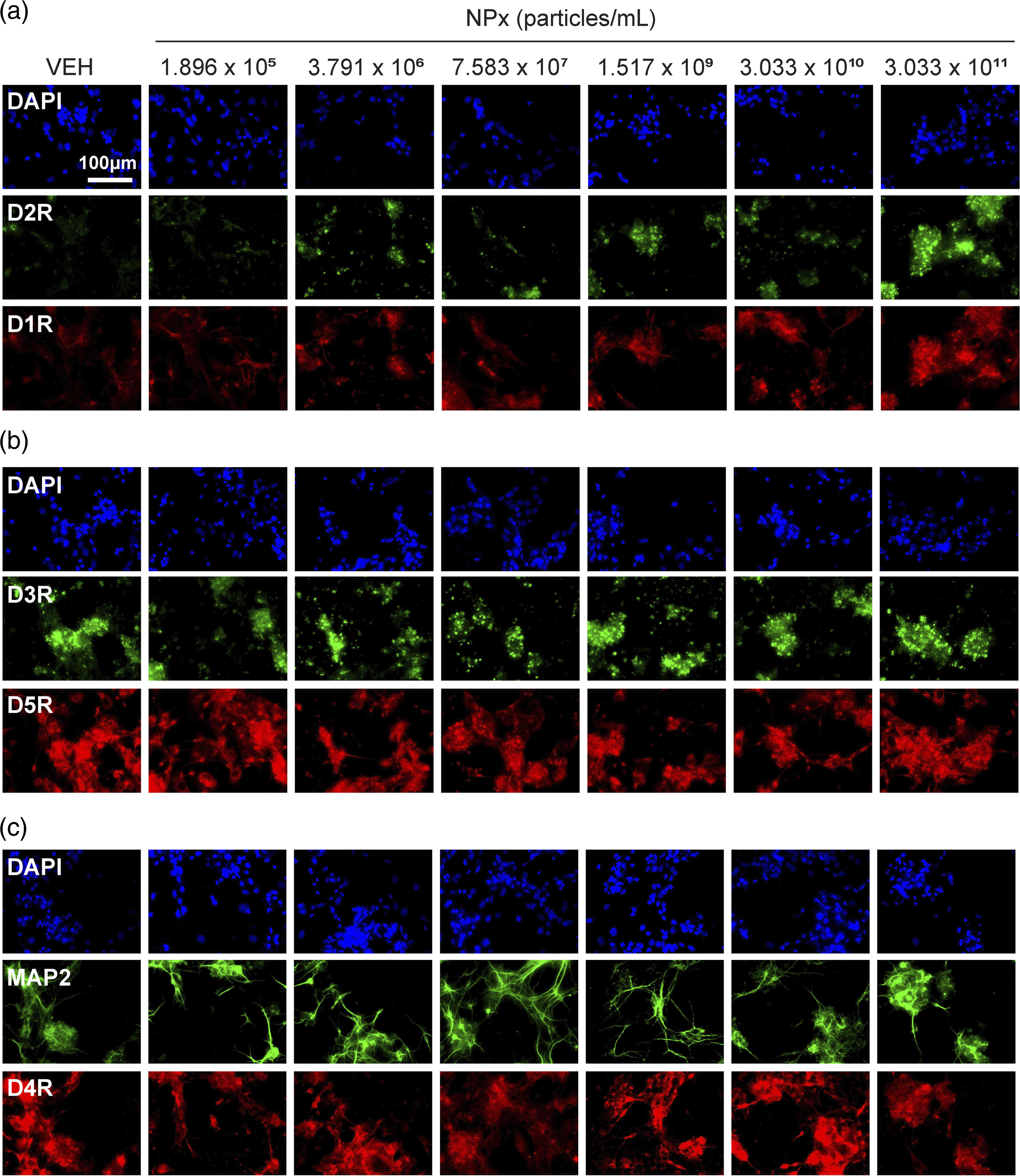
Discussion
We investigated the effects of NPx on the DA system of cultured cerebrocortical neurons. We found that NPx accumulated in the neurons and increased the levels of D1R, D2R, and their co-expression. These findings indicate that NPx affects the DA systems in neurons.
NPx has been reported to accumulate both
Alterations in DA receptor levels were induced only in the high-dose NPx treatment group. In our study, 100 nm NPx were treated at concentrations between 1.896 × 105 particles/mL and 3.033 × 1011 particles/mL, which equates to a range of 0.156 ng/mL to 250 μg/mL. A previous study has reported that 50–200 μg/mL of 100 nm NPx in the mixed neurons induced neuronal apoptosis with intracellular accumulation of NPx.
25
Therefore, the NPx doses used in our study are typical doses for studying NPx toxicity. However, these concentrations of NPx may not be appropriate in human or
In mice, administering 50 and 500 nm NPx for 24 h induced biodistribution over a wide range, from the intestine to the brain. Furthermore, their repeated administration over 28 days resulted in increased accumulation and reactive oxygen species generation in the intestine. 28 Moreover, 50 nm NPx administered in mice for 32 weeks increased levels of reactive oxygen species and malondialdehyde and decreased the activity of superoxide dismutase and glutathione peroxide in the intestine. 29 In Caco-2 cells, 8 weeks of 50 nm NPx treatment was reported to cause increased oxidative DNA damage with increasing accumulation. 30 Recently, 2 weeks of 100 nm NPx administration in mice was reported to induce increased locomotion under L-DOPA treatment. 31 These results suggest that NPx can accumulate in living organisms and affect neuronal function via the DA system, although the size and dose of NPx, as well as the duration and tissue type treated with NPx, may also affect their accumulation and effect.
NPx treatment specifically increased D1R and D2R levels but not DA synthesis or reuptake. There are five types of DA receptors: D1R, D2R, D3R, D4R, and D5R. D1R induces increased intracellular levels of cAMP by activating adenylate cyclase, whereas D2R causes decreased intracellular levels of cAMP by inhibiting adenylate cyclase. 11 In contrast, D1R–D2R heterodimers exhibit different functions from those of the D1R or D2R homodimers. For example, the heterodimers induce intracellular calcium release by activating phospholipase C, 32 and this signal affects synaptic plasticity and causes psychiatric disorders. Furthermore, D1R–D2R heterodimers induce calmodulin-dependent protein kinase II (CamKII) activation and modulate brain-derived neurotrophic factor (BDNF) and glutamate receptor subunit epsilon-2 (GluN2B). 32 Activated D1R–D2R heterodimers also affect DA- and cAMP-regulated phosphoprotein of molecular weight 32 kDa (DARPP-32)/extracellular regulated kinase (ERK), which are associated with the D1R homodimer and protein kinase B (PKB or Akt)/glycogen synthase kinase-3 (GSK3), which are associated with the D2R homodimer.33,34 These interactions suggest that D1R–D2R heterodimers affect neural plasticity both individually and in relation to the signaling cascade of D1R or D2R homodimers, thereby causing depression, addiction, anxiety, and schizophrenia.32-34 In our study, the levels of D1R and D2R, as well as their co-expression, were highest at the highest concentration of NPx. This suggests that the effect of NPx in cerebrocortical neurons is associated with D1R–D2R heterodimers. However, the effects of NPx on the signaling cascades of D1R, D2R, and D1R–D2R heterodimers need further investigation.
Previous studies have reported that NPx administration induces impairment of brain function. In mice, 531.2 ± 97.83 nm NPx decreased motility and spatial memory, 24 and 23.03 ± .266 nm NPx increased anxiety and decreased defensive responses, with no changes in motility. 35 Moreover, prenatal treatment with 193 nm NPx led to offspring that exhibited depressive and abnormal social behaviors without any alterations in spatial or object memory. 36 Notably, DA receptor-associated signaling cascades have been found to be associated with various behaviors, including spatial memory, anxiety, depression, and social ability.11,32 Therefore, disruptions in the DA system can cause various psychiatric disorders, including Parkinson’s disease, major depression disorder, and schizophrenia.12,37-39 These results suggest that an altered DA system induced by NPx treatment may be associated with the neuronal dysfunction that causes these psychiatric disorders, although the mechanism through which NPx treatment affects neuronal function by modulating the DA system requires further investigation.
Unlike DA receptor alterations, DA levels showed no changes with NPx treatment. In
This study has several limitations. First, although NPx did not induce alterations in DA synthesis or reuptake, DA degradation may have been affected. Exposure to mercury as a pollutant showed a negative correlation with monoamine oxidase in the cerebral cortex of wild river otters.
42
Further research is required to elucidate the effects of NPx on DA degradation. Second, our study used only 100 nm NPx; however, NPx consist of complexes of various sizes in nature. Exposure to complexes of 50 nm and 500 nm showed greater accumulation than exposures to complexes of 50 nm or 500 nm alone in the organs of mice.
28
Therefore, further research is needed to clarify the effect of exposure to complexes of NPx of various sizes. Third, the duration of NPx treatment in this study was only 24 h; however, increasing the NPx treatment duration has been shown to induce an increase in oxidative DNA damage.
30
Recently, changes in D1R and DAT levels were reported after only 24 h of
In this study, NPx accumulated in the cerebrocortical neurons, resulting in increased levels of D1R, D2R, and their co-expression. These findings indicate that NPx is a potential modulator of the DA system in cerebrocortical neurons and may be associated with DA system-based psychiatric disorders.
Supplemental Material
Supplemental Material - The Effects of Nanoplastics on the Dopamine System of Cerebrocortical Neurons
Supplemental Material for The Effects of Nanoplastics on the Dopamine System of Cerebrocortical Neurons by Na-Hyun Kim and Young-A Lee in International Journal of Toxicology.
Footnotes
Author Contributions
Kim, Na-Hyun contributed to acquisition and analysis and drafted manuscript; Lee, Young-A contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. Both authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (Ministry of Science and ICT, MSIT) (NRF-2020R1F1A1048370).
Ethical Statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
