Abstract
The subchronic toxicity and toxicokinetics of a combination of rabeprazole sodium and sodium bicarbonate were investigated in dogs by daily oral administration for 13 consecutive weeks with a 4-week recovery period. The dose groups consisted of control (vehicles), (5 + 200), (10 + 400), and (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate, 20 mg/kg of rabeprazole sodium only, and 800 mg/kg of sodium bicarbonate only. Esophageal ulceration accompanied by inflammation was observed in only one animal in the male (20 + 800) mg/kg rabeprazole sodium + sodium bicarbonate group. However, the severity of the ulceration was moderate, and the site of occurrence was focally extensive; thus, it was assumed to be a treatment-related effect of rabeprazole sodium + sodium bicarbonate. In the toxicokinetics component of this study, systemic exposure to rabeprazole sodium (AUClast and Cmax at Day 91) was greater in males than females, suggesting sex differences. AUClast and Cmax at Day 91 were increased compared to those on Day 1 in a dose-dependent manner. A delayed Tmax and no drug accumulation were observed after repeated dosage. In conclusion, we suggest under the conditions of this study that the no-observed-adverse-effect level (NOAEL) of the combination of rabeprazole sodium + sodium bicarbonate in male and female dogs is (10 + 400) and (20 + 800) mg/kg, respectively.
Keywords
Introduction
Inhibition of proton pump enzymes is an effective way to increase gastric luminal pH. 1 Proton pump inhibitors (PPIs) are considered most effective in regulating the suppression of acid secretion for treating acid-related diseases, such as duodenal ulcers, gastric ulcers, and gastroesophageal reflux diseases. PPIs are benzimidazole derivatives that convert to an active sulfenamide form, forming a covalent disulfide bond with the surface-exposed cysteines of the active parietal cell H+/K+-ATPase to inhibit the enzyme’s activity.2,3 As PPIs are unstable and degrade in the stomach’s acidic environment, enteric-coated formulations have been developed to protect the drugs during the transit through the stomach after oral administration of PPIs. 4
Rabeprazole, omeprazole, pantoprazole, lansoprazole, and esomeprazole are the most frequently prescribed PPIs on the market.5-8 Among them, rabeprazole is the most acid-labile, followed by lansoprazole, omeprazole, and lastly pantoprazole as the least acid-labile. 9 Despite rabeprazole’s highly acid-labile nature, it is manufactured, supplied, and administered as an enteric-coated formulation and delayed-release capsules to successfully treat many acid-related diseases. However, the disadvantage of enteric-coated formulations and delayed-release capsules is that both dosage forms are dissolved and absorbed in the intestine but not immediately in the stomach. Consequently, the drug onset time becomes delayed and fails to exert an immediate therapeutic effect. 10
As part of an effort to address the need to develop a PPI with a faster absorption capacity and an excellent safety profile, one promising approach is to utilize sodium bicarbonate. Commonly used as an antacid, sodium bicarbonate is also effective in temporarily relieving acid reflux symptoms, albeit by direct neutralization of stomach acid. While rabeprazole and sodium bicarbonate have different mechanisms of action, both are effective in alleviating symptoms of gastric diseases caused by decreased pH levels. Furthermore, the latter also protects rabeprazole from gastric acid. Considering the possible synergistic effects, a combined formulation incorporating sodium bicarbonate with rabeprazole was developed to achieve optimal pharmacological effects, stability, and safety.
This study aimed to evaluate the safety of the aforementioned combination of rabeprazole sodium and sodium bicarbonate for the investigational new drug application to the Korean Ministry of Food and Drug Safety. To assess the safety, the subchronic toxicity and toxicokinetics of rabeprazole sodium and sodium bicarbonate were investigated in beagle dogs by daily oral administration for 13 consecutive weeks with a 4-week recovery period. This study was conducted in accordance with the Korean Ministry of Food and Drug Safety test guideline 11 under the latest Good Laboratory Practice Regulation.
Materials and Methods
Materials and Preparation
Rabeprazole sodium and sodium bicarbonate were supplied by Korea United Pharm Inc. Acid-resistant capsules for filling rabeprazole sodium were procured from Capsule Supplies LLC (PureCaps USA, Stuyvesant, NY, USA). Dai Han Pharm Co., Ltd. (Seoul, Republic of Korea) provided the water for injection as the vehicle for suspension of sodium bicarbonate.
Without any preparation of the dosing formulation, the required amounts of the rabeprazole sodium were weighed on electronic balances (BCE224i-1SKR, Sartorius, Germany) and filled in acid-resistant capsules.
Animals and Animal Husbandry
Male and female beagle dogs were obtained from Kitayama Labes Co., Ltd. Hongo Beagles Farm (Ina City, Japan). They were approximately 6 months old and weighed between 6.54 and 9.10 kg at study initiation.
They were individually housed in stainless steel cages (850W × 900D × 850H). The animal rooms were under a 12-hour light-dark cycle (150-300 Lux), with mean temperatures of 20 ± 3°C and relative humidity of 30 to 70%. The dogs were fed 250 g of Canine Certified Diet (Cargill Agri Purina, Inc. Dangjin, Republic of Korea) once daily. Water was provided ad libitum via an automatic watering system. This study was approved by the Institutional Animal Care and Use Committee of Croen Inc. (Approval No.: 21D001).
Dose Range Finding Study
A 4-week repeated oral dose range finding study (Croen Inc. Study No.: C20DR-030N) was performed using (7.5 + 300), (15 + 600), and (30 + 1200) mg/kg/day of rabeprazole sodium + sodium bicarbonate doses. While no mortality was observed, diarrhea and vomiting were observed in all dosage groups in a dose-dependent manner. Based on these findings, (20 + 800) mg/kg/day of rabeprazole sodium + sodium bicarbonate was selected as the high dose level for the 13-week study, and (10 + 400) and (5 + 200) mg/kg/day doses were selected as the middle and low doses, respectively.
Study Design and Dosing
Study Design and Group Assignment.
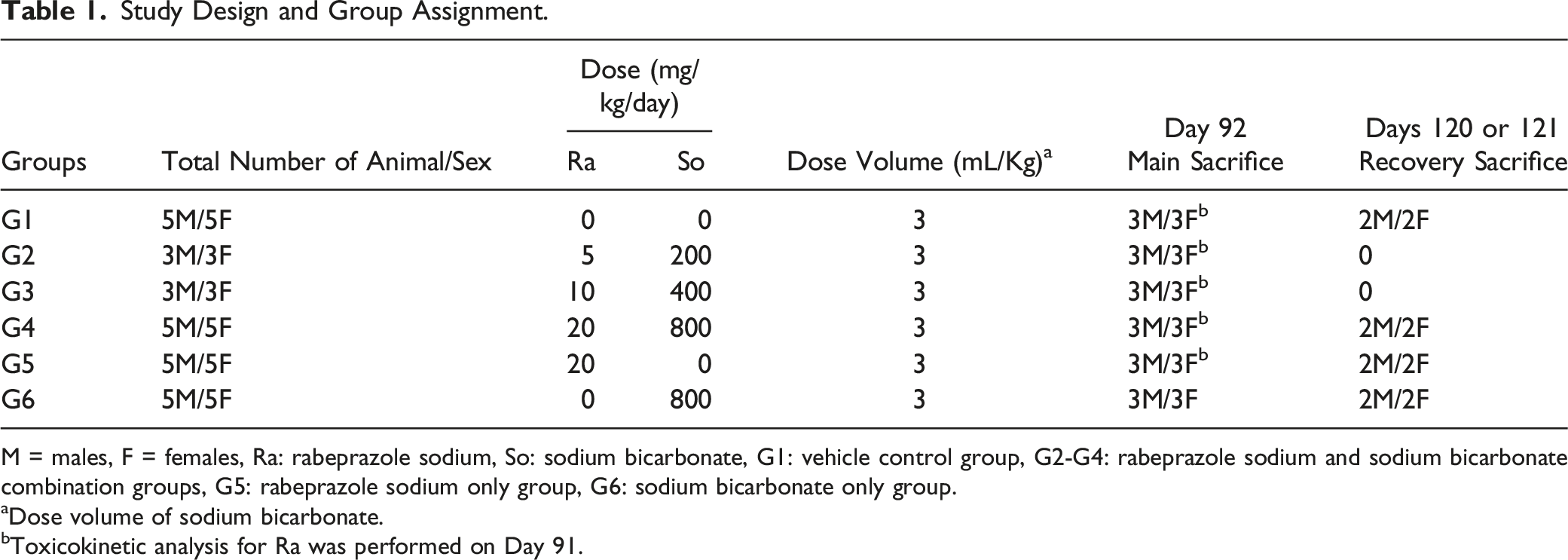
M = males, F = females, Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
aDose volume of sodium bicarbonate.
bToxicokinetic analysis for Ra was performed on Day 91.
To administer individual doses of rabeprazole sodium, which were calculated based on the most recently recorded body weights, the test animal’s mouth was held, the capsule was placed at the base of the tongue, and the mouth was closed. Subsequently, about 20 mL of water was fed, and the animal’s larynx was gently stroked to induce swallowing. After about 2 hours of administration of rabeprazole sodium dosing, sodium bicarbonate was administered at a dose volume of 3 mL/kg into the stomach using a disposable syringe (10 to 30 mL) with a catheter. The combination therapy for human is intended as a single oral pill, but this study administered the agents in sequence. Since it was difficult to produce a single oral pill for each dose in non-clinical study conducted before clinical trials, it was decided to administer using capsules. However, there was no capsule size for beagle dogs that could fill two test articles at the same time, so they were administered separately.
Dogs were dosed for approximately 91 consecutive days, with Day 1 as the first day of dosing. Animals (three/sex/group) from selected dose groups were necropsied on Day 92, with the remaining animals (two/sex/group) retained for a 4-week recovery period and then necropsied on approximately Day 121.
Clinical Observations, Body Weight, Food Consumption, and Ophthalmologic Examination
The animals were observed twice daily for clinical observations. The body weights were recorded before the first dose, weekly during the dosage period, and before the necropsy. Fasted body weights were recorded on the necropsy day to calculate organ/body weight ratios. Food consumption was recorded prior to the first dose and weekly during the dosage period. An ophthalmologic examination was conducted before the first dose, at week 13 of the dosing period and week 4 of the recovery period. The Mydriacyl Eye Drops 1% (Alcon, Lot No.: 19B12UA) was instilled into the eyes of all animals, and macroscopic examinations were conducted. The anterior segment of the eyes, transparent media and ocular fundus were evaluated using an ophthalmoscope (DM6C, Zumax, China).
Electrocardiogram
Electrocardiographic (ECG) examinations were conducted before the first dose, at week 13 of the dosing period and week 4 of the recovery period. The Standard Limb Leads and Amplification Unipolar Limb Leads (I, II, and III; aVR, aVL, and aVF) were recorded for the wave type forms using an electrocardiograph (CardiMax FX to 8222, Fukuda Denshi Co., Ltd., Japan). For the electrical axis and II Leads, the intervals including heart rate (HR, bpm), R to R interval (R to R, sec), P to R interval (P to R, sec), QRS duration (QRS, sec), Q to T interval (Q to T, sec), corrected QT (QTc), and mean electrical axis (AXIS, deg) were recorded. QTc was calculated using Bazett’ s formula (QTc = QT/
Urinalysis
Urinalysis was performed using approximately 2 mL of freshly collected urine samples from urine collection pans, and the total volume of urine collected for 24 hours was measured. Fresh urine samples were collected within approximately 3 hours after voiding before the first dose, at weeks 4 and 13 of the dosing period, and at week 4 of the recovery period. Urine was analyzed with a urine chemistry analyzer (UC-1000, Sysmex, Japan) and microscope (E200LED, Nikon, Japan). The following parameters were examined: urine color, pH, protein, glucose, ketone bodies, bilirubin, occult blood, urine sediments, and specific gravity.
Hematology and Serum Biochemistry
The blood samples were collected from fasted animals through the cephalic vein before the first dose, at weeks 4 and 13 of the dosing period, and week 4 of the recovery period. Hematology, coagulation indices, and serum biochemistry parameters were determined.
Approximately 1 mL of collected blood was placed into a tube containing anticoagulant dipotassium ethylenediaminetetraacetic acid (K2-EDTA). Hematology analysis was performed using a hematological autoanalyzer (XN-10, Sysmex, Japan). The analysis included the following: total erythrocyte count (RBC), hemoglobin concentration (HGB), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular hemoglobin (MCH), platelet count (PLT), reticulocytes (Retic), total leukocyte count (WBC), and WBC differential counting (neutrophil (NEU), lymphocyte (LYM), monocyte (MONO), eosinophil (EOS), and basophil (BASO)).
The blood samples were placed into a tube containing 3.2% sodium citrate and centrifuged at 3,000 rpm and 4°C for 10 minutes. The obtained plasma was used to measure the blood coagulation time (activated partial thromboplastin time and prothrombin time) using a coagulation time analyzer (CA 660, Sysmex, Japan).
Additional blood samples (approximately 2 mL) were centrifuged at 3,000 rpm and 4°C for 10 minutes. The obtained serum was analyzed using a serum biochemistry analyzer (7180, Hitachi, Japan) and an electrolyte analyzer (I-Smart 30, i-SENS, Inc., Korea). Measured parameters include the following: aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), γ-glutamyl transpeptidase (GGT), glucose (GLU), blood urea nitrogen (BUN), creatinine (CRE), total cholesterol (T-CHO), triglyceride (TG), total protein, albumin (ALB), total bilirubin (T-BIL), albumin/globulin ratio (A/G), calcium ion (Ca), inorganic phosphorus, sodium (Na), potassium (K), and chloride (Cl).
Necropsy and Organ Weight
At the scheduled termination, all surviving animals were euthanized by exsanguination under anesthesia using an intravenous injection of thiopental sodium (Choongwae Pharma. Corp., Korea). The organs and tissues in euthanized dogs' cranial, thoracic, and abdominal cavities underwent gross examination for visible defects. Absolute weights and relative organ-to-body weights (terminal body weights) ratios were determined. Tissues included the brain, spleen, epididymis, pituitary gland, adrenal gland, prostate gland, lung, kidneys, ovaries, heart, liver with gallbladder, uterus, thymus, testes, stomach, and thyroids with parathyroids.
Histopathology
Following excision, each organ was fixed in 10% neutral buffered formalin, except for the eyes which were fixed with Davidson’s fixative. The fixed organs were trimmed, processed, embedded in paraffin, and sections of 4–5 µm thickness were prepared. Hematoxylin and eosin-stained slides were prepared, and the resulting specimens were microscopically examined with an optical microscope.
Toxicokinetics (TK) Analysis
On Day 1 and week 13, approximately 2 mL of blood samples were collected from the cephalic or jugular vein using a 3 mL disposable syringe without anesthesia. The blood samples were placed into a tube containing K2-EDTA and centrifuged at 3,000 rpm and 4°C for 10 minutes to separate the plasma. A 1% diethylamine solution was incorporated as a stabilizer at a ratio of 10 µL per 100 µL of plasma, followed by storage in a deep freezer maintained at or below −60°C until analysis.
The blood collection time for the TK study was 1 hour after administration for the control group, and pre-dose, 30 min, 1, 2, 3, 4, 5, and 8 hours after administration for (5 + 200), (10 + 400), and (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate; 20 mg/kg of rabeprazole sodium only; and 800 mg/kg of sodium bicarbonate only groups.
Quantification of rabeprazole sodium in dog plasma was performed according to a previously validated method provided by Croen Inc. (“Analytical method validation of rabeprazole in beagle dog plasma using LC-MS/MS, Study No.: C20ZV-034G). Rabeprazole and the internal standard (amlodipine) were separated on a Poroshell 12 EC-120 column (4.6 ⅹ 150 mm, 4 μm, Agilent, Santa Clara, CA, USA). Samples were separated under isocratic elution (30:70, v/v) with .1% (v/v) formic acid in 10 mM ammonium acetate as the mobile phase A and .1% (v/v) formic acid in acetonitrile as the mobile phase B at a flow rate of .3 mL/min. An Agilent triple-quadrupole mass spectrometer 6470 (Santa Clara, CA, USA) was interfaced via an electrospray ionization (ESI) probe with an Agilent 1290 Infinity II liquid chromatograph. Agilent MassHunter software was used to acquire and process data. The mass spectrometer was operated in the positive ESI mode ([M + H]+) with selected multiple-reaction monitoring for all the analytes. The precursor-to-product ion transitions were monitored at m/z 360.2 → 242.1 for rabeprazole sodium and m/z 409.1 → 238.0 for the internal standard.
Toxicokinetic parameters such as the area under the plasma concentration-time curve (AUC0-last), maximum observed plasma concentration (Cmax), and time to Cmax (Tmax) were determined by PhoenixTM WinNonlin® (ver. 8.3.3.33, Pharsight) using the non-compartment model.
Statistical Analyses
The statistical variables examined in included body weight, food consumption, urine volume, hematology, clinical chemistry, and absolute and relative organ weights. Results from parametric multiple comparison procedures were subjected to a one-way analysis of variance (ANOVA) test. Significance was established at a probability threshold of P < .05. If the ANOVA results indicated statistical significance, Levene’s test was utilized to assess the equality of variances. If homoscedasticity was shown, Scheffe multiple range tests were employed for further analysis. In the case of a substantial deviation from homogeneity of variance, Dunnett’s T3-test was applied as a post-hoc analysis. All statistical analyses were performed with SPSS software (IBM® SPSS Statistics ver. 24).
Results
Clinical Observations
Clinical Signs in Male/Female Dogs.
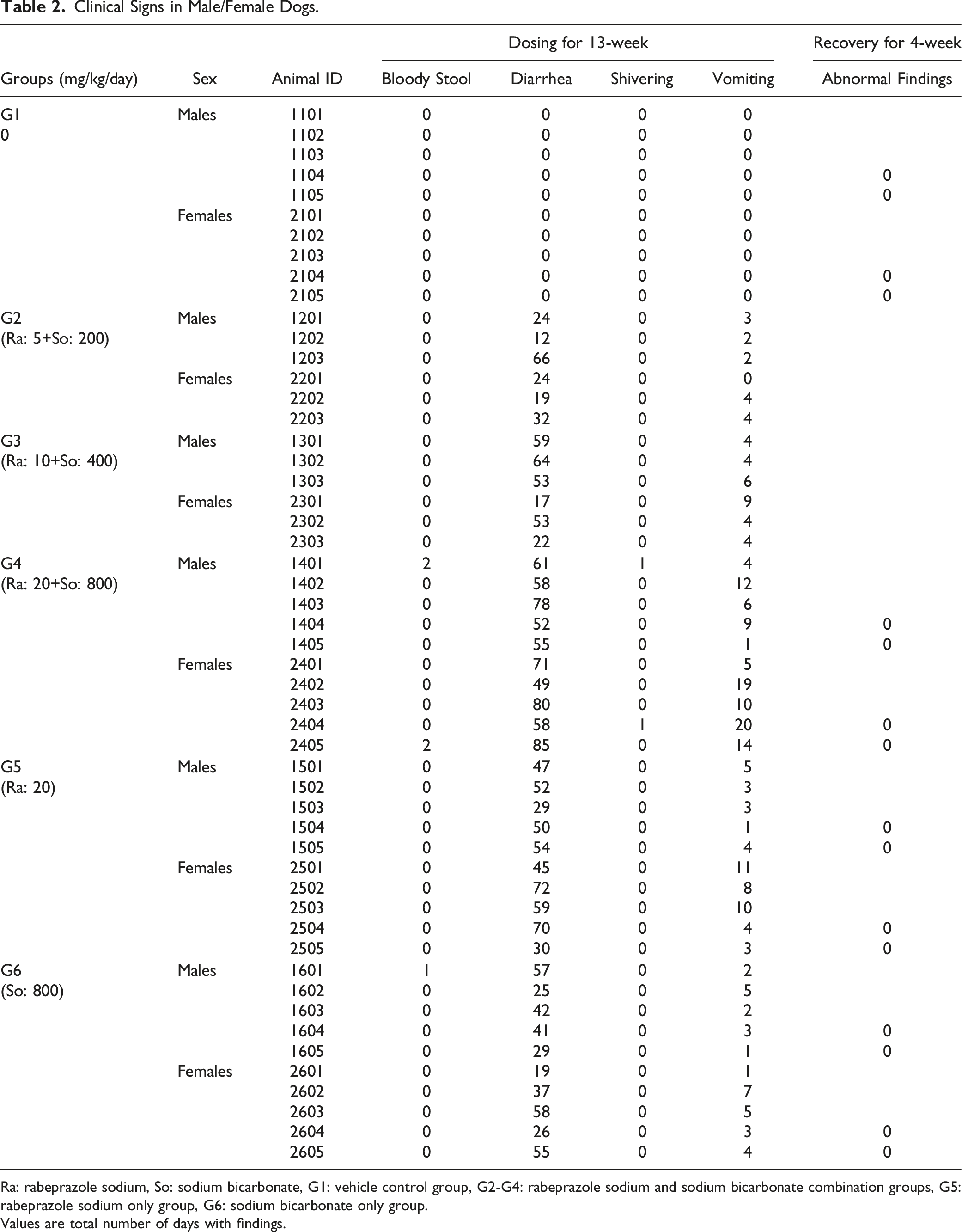
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
Values are total number of days with findings.
Body Weight, Food Consumption, Ophthalmologic Examination and Electrocardiogram
No abnormal findings or toxicologically significant differences were observed in all treated groups on body weight, food consumption, ophthalmologic examination, and electrocardiogram during the dosing and recovery periods (data not shown).
Urinalysis
At weeks 4 and 13 of the dosing period, the urinary pH of all groups administered with the rabeprazole sodium + sodium bicarbonate, 20 mg/kg of rabeprazole sodium and 800 mg/kg of sodium bicarbonate was higher than that of the control group. Changes in color, urinary protein, and occult blood were detected in some individuals, and epithelial cells were observed sporadically in the urine sediment.
Urinalysis Results at 4-Week of Dosing.
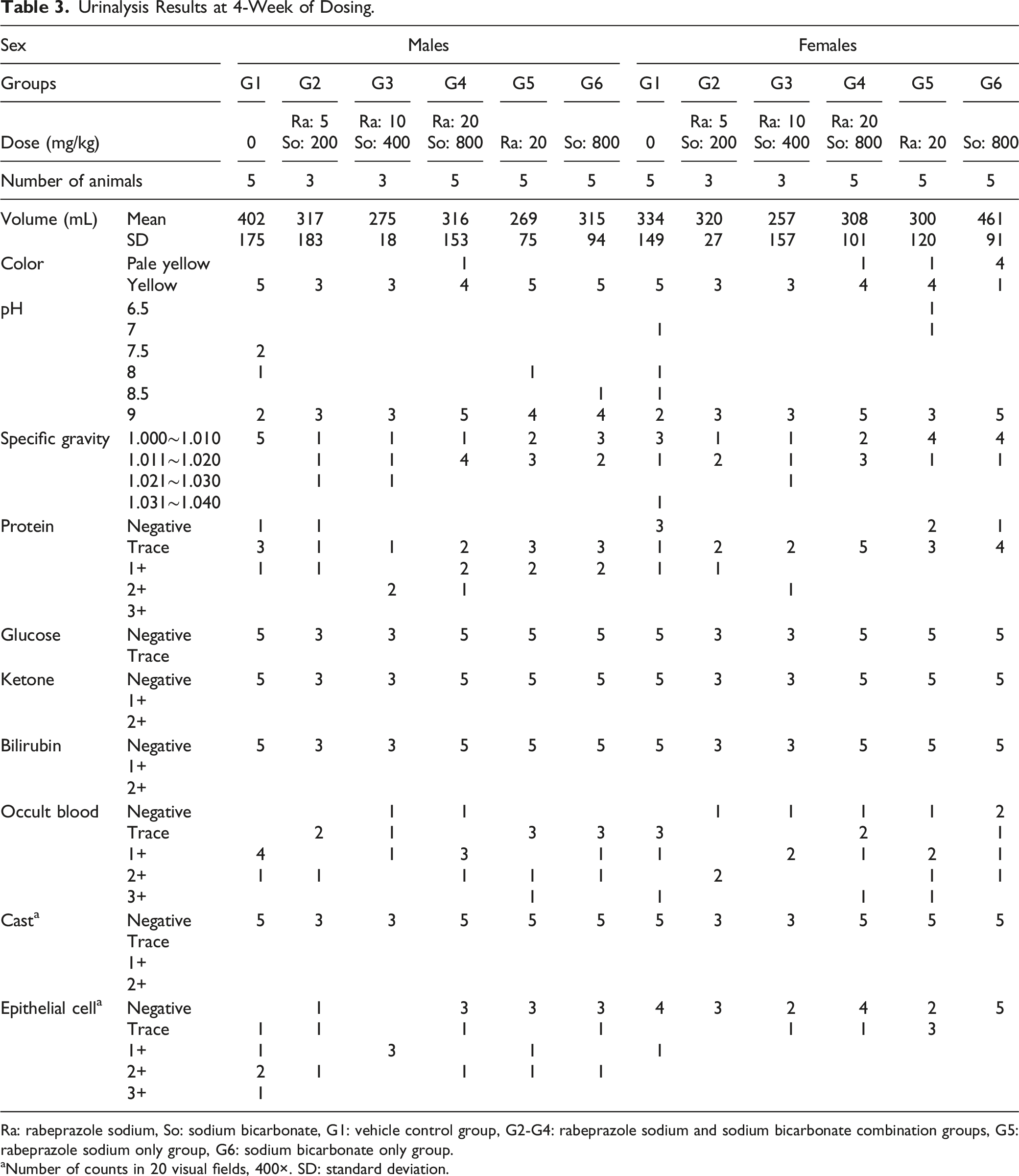
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
aNumber of counts in 20 visual fields, 400×. SD: standard deviation.
Urinalysis Results after 13-Week Treatment.
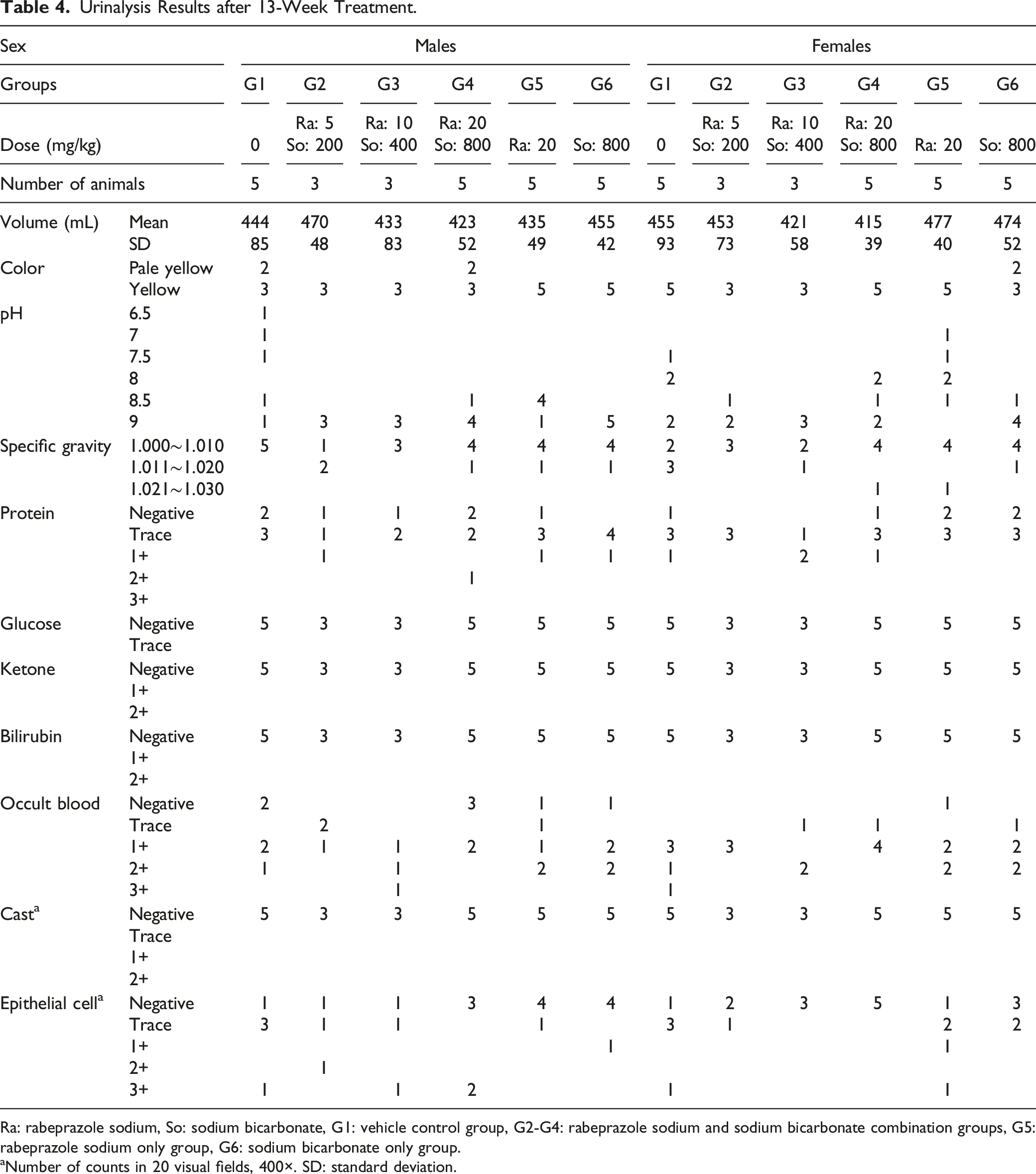
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
aNumber of counts in 20 visual fields, 400×. SD: standard deviation.
Urinalysis Results at 4-Week Recovery Groups After 13-Week Treatment.
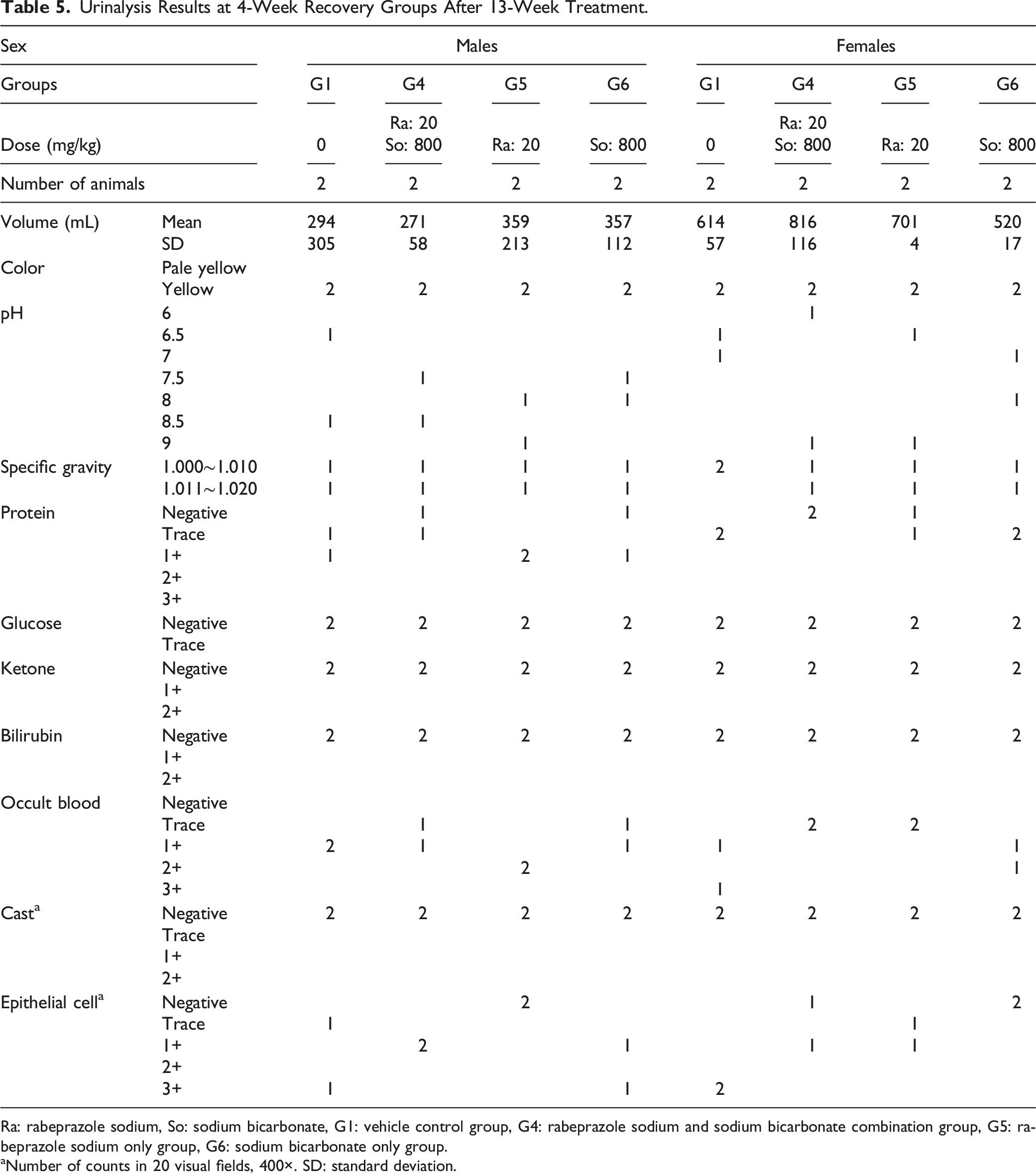
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G4: rabeprazole sodium and sodium bicarbonate combination group, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
aNumber of counts in 20 visual fields, 400×. SD: standard deviation.
Hematology
Hematological Findings at 4-Week of Dosing.
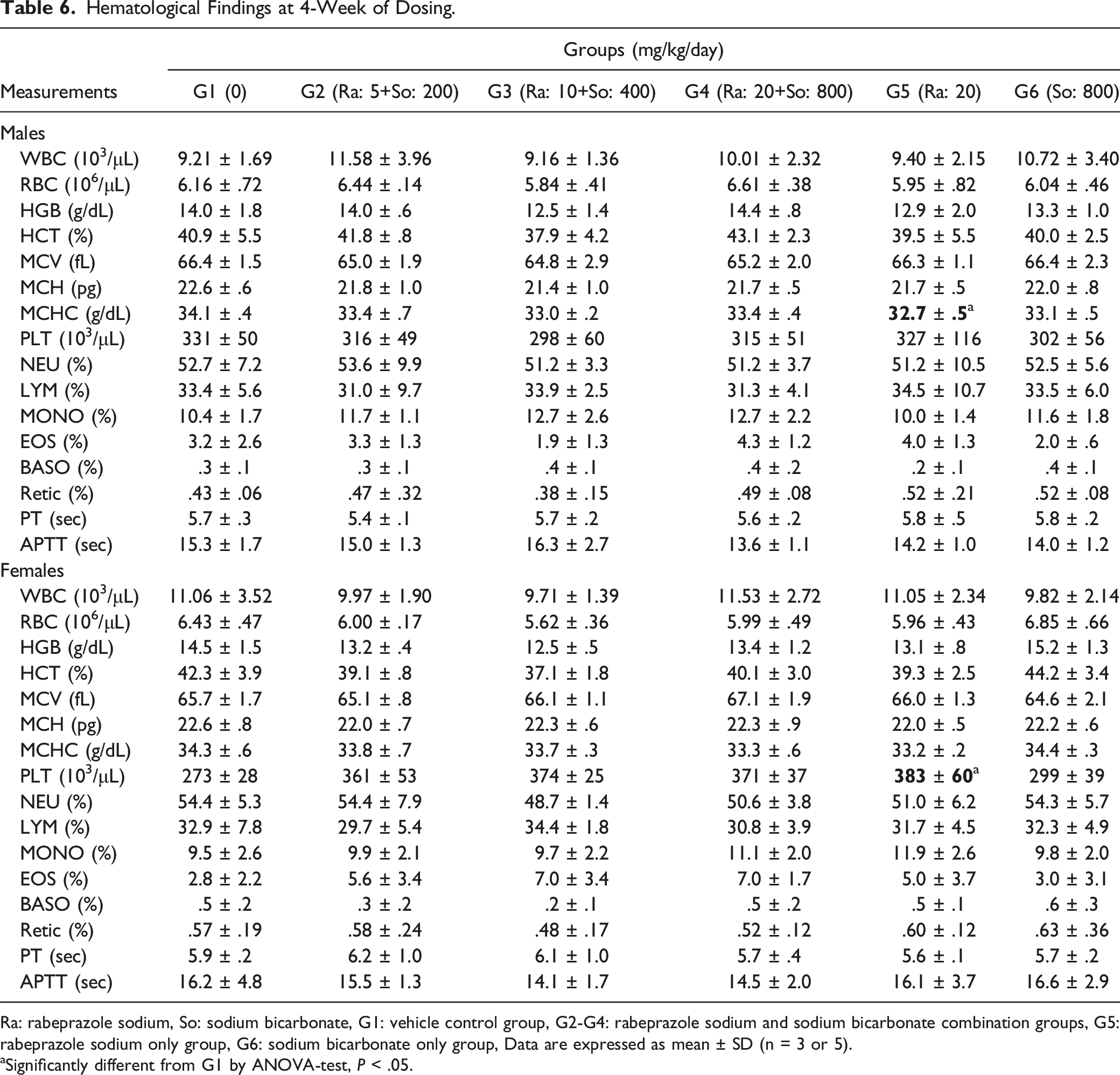
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group, Data are expressed as mean ± SD (n = 3 or 5).
aSignificantly different from G1 by ANOVA-test, P < .05.
Hematological Findings After 13-Week Treatment.
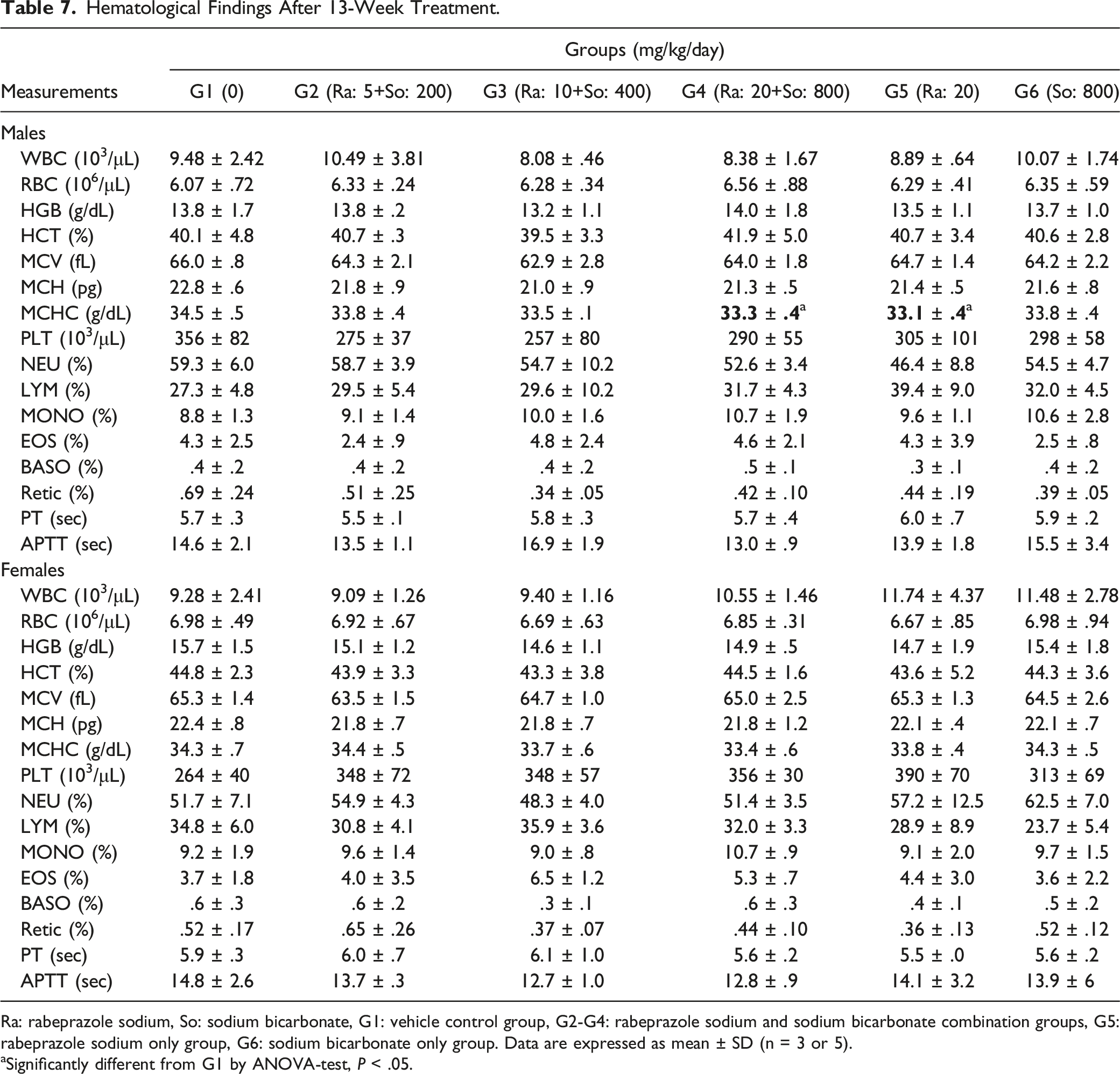
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group. Data are expressed as mean ± SD (n = 3 or 5).
aSignificantly different from G1 by ANOVA-test, P < .05.
No abnormal findings were observed on hematology pre-dosing and during the recovery periods (data not shown).
Blood Biochemistry
No abnormal findings or toxicologically significant differences were observed in all groups on blood biochemistry during the dosing (at weeks 4 and 13) and recovery periods (data not shown).
Organ Weights
Absolute (g) and Relative Organ (%) Weights in Male Dogs After 13-Week Treatment.
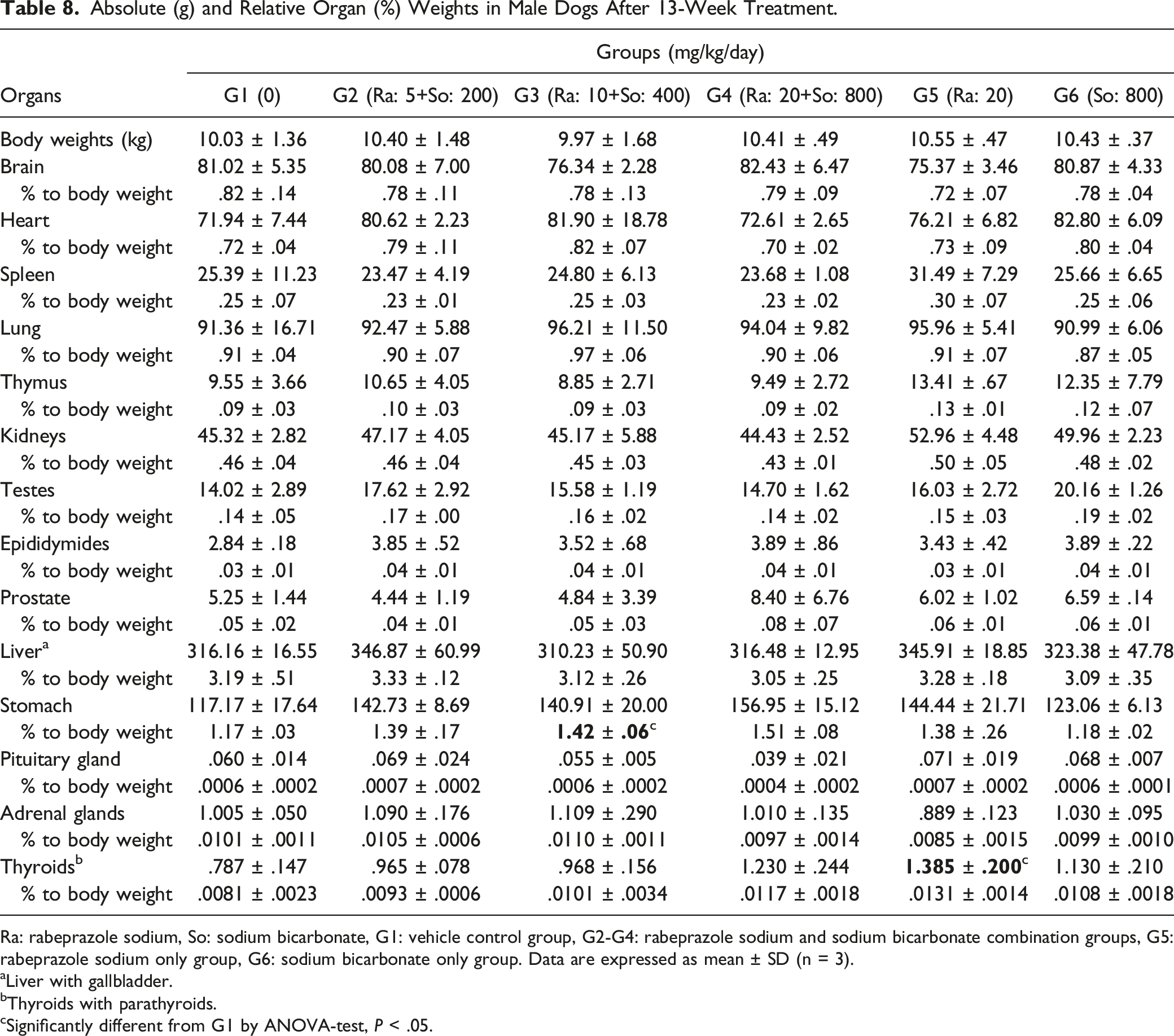
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group. Data are expressed as mean ± SD (n = 3).
aLiver with gallbladder.
bThyroids with parathyroids.
cSignificantly different from G1 by ANOVA-test, P < .05.
Absolute (g) and Relative Organ (%) Weights in Female Dogs After 13-Week Treatment.
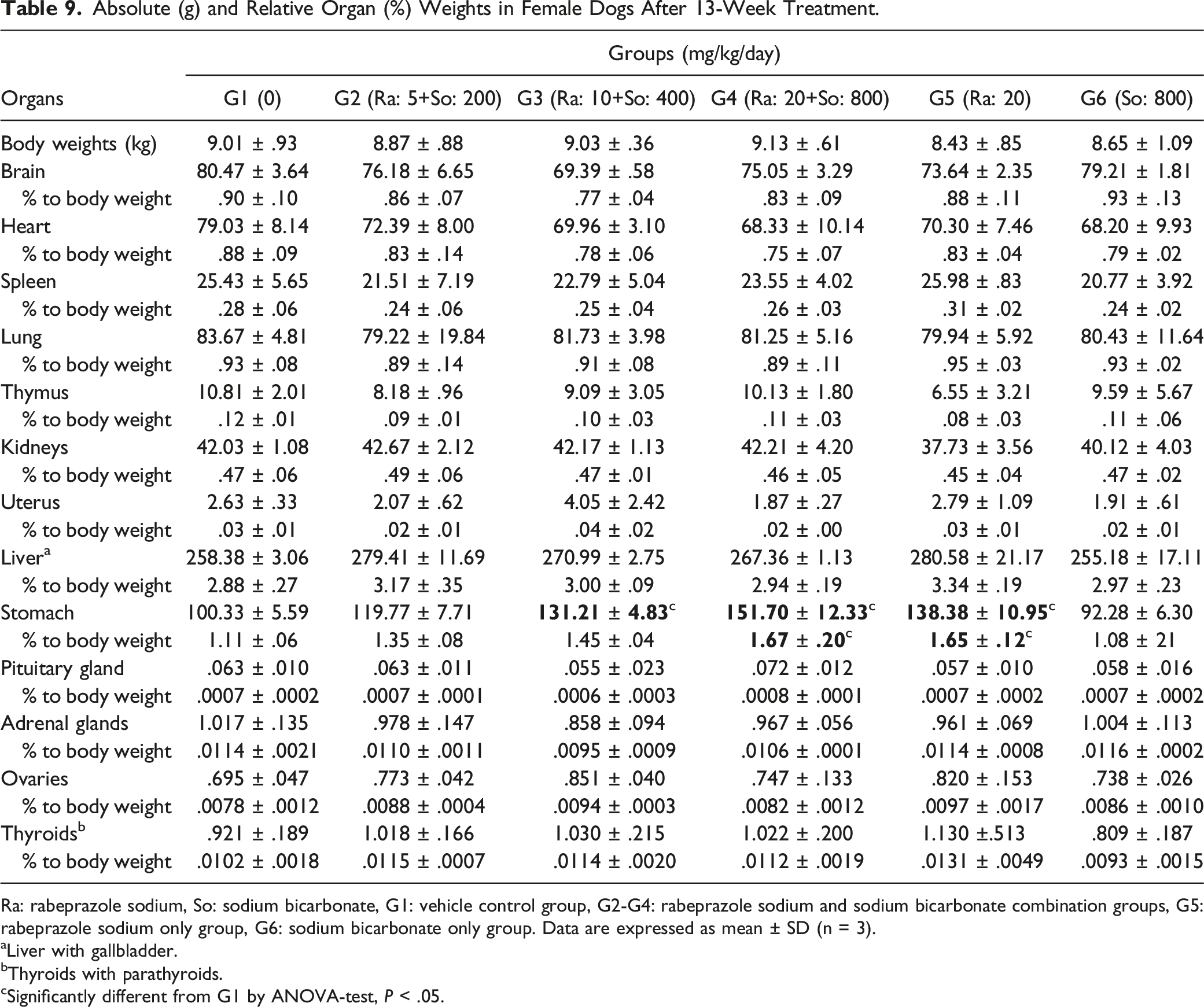
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group. Data are expressed as mean ± SD (n = 3).
aLiver with gallbladder.
bThyroids with parathyroids.
cSignificantly different from G1 by ANOVA-test, P < .05.
Absolute (g) and Relative Organ (%) Weights in Male Dogs at 4-Week Recovery Groups After 13-Week Treatment.
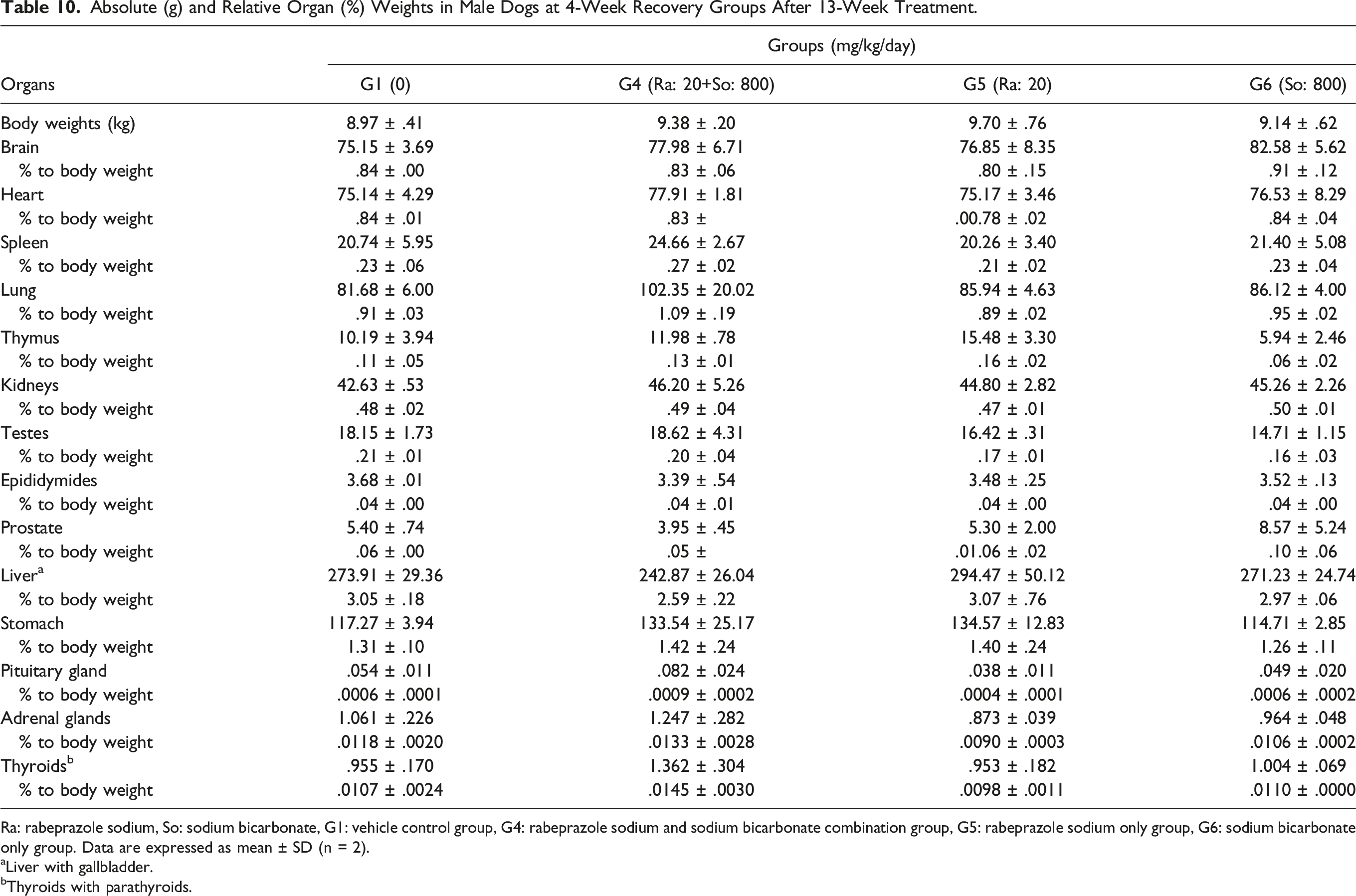
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G4: rabeprazole sodium and sodium bicarbonate combination group, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group. Data are expressed as mean ± SD (n = 2).
aLiver with gallbladder.
bThyroids with parathyroids.
Absolute (g) and Relative Organ (%) Weights in Female Dogs at 4-Week Recovery Groups After 13-Week Treatment.
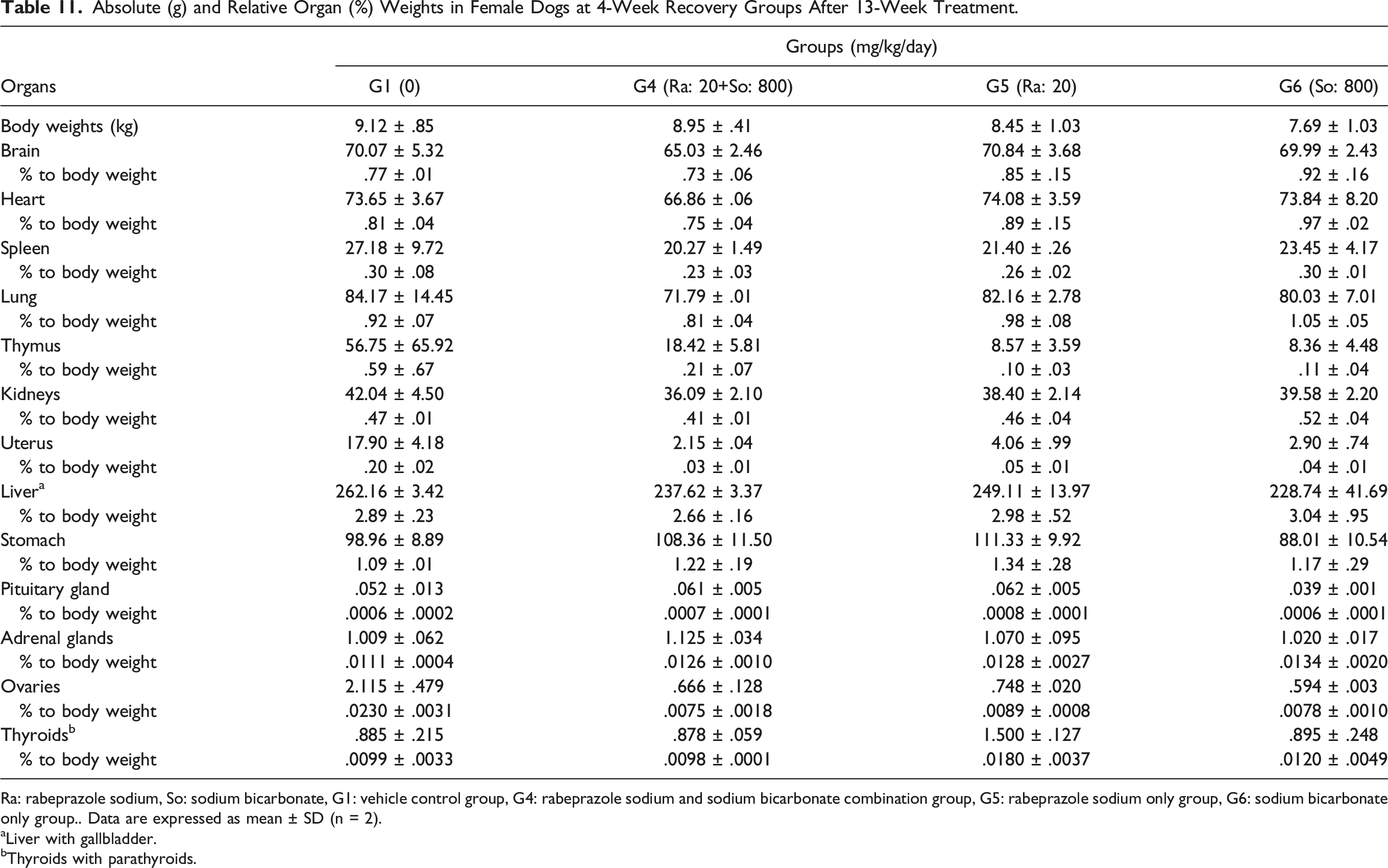
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G4: rabeprazole sodium and sodium bicarbonate combination group, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.. Data are expressed as mean ± SD (n = 2).
aLiver with gallbladder.
bThyroids with parathyroids.
In the 20 mg/kg of rabeprazole sodium group, the absolute (but not the relative) weight of thyroids with parathyroids of males significantly increased (P < .05) during the dosing period.
In all dose groups receiving rabeprazole sodium + sodium bicarbonate and 20 mg/kg of rabeprazole sodium only, a general trend or a dose-dependent significant increase (P < .05) in absolute and relative weights of the stomach was observed in both sexes during the dosing period.
Similar trends of increasing absolute and relative stomach weights were also observed in the (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate and 20 mg/kg of rabeprazole sodium only groups for both sexes during the recovery period. However, the increased stomach weights were not different from the control levels following the recovery period.
Necropsy Findings
No abnormal finding was observed in all groups on necropsy findings following the dosing and recovery periods (data not shown).
Histopathological Examinations
Histopathologic Findings After 13-Week Treatment.
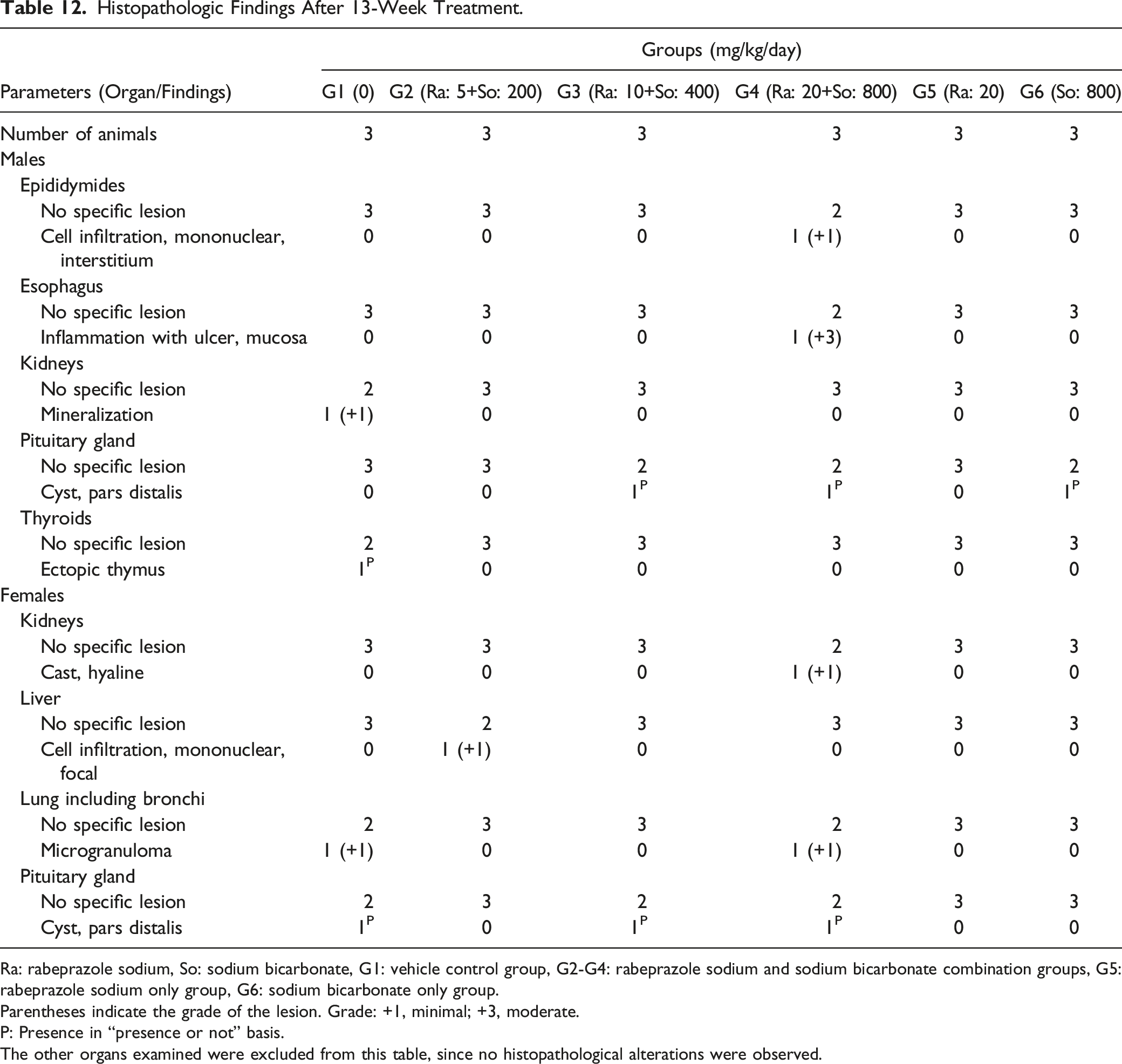
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
Parentheses indicate the grade of the lesion. Grade: +1, minimal; +3, moderate.
P: Presence in “presence or not” basis.
The other organs examined were excluded from this table, since no histopathological alterations were observed.
Histopathologic Findings at 4-Week Recovery Groups After 13-Week Treatment.
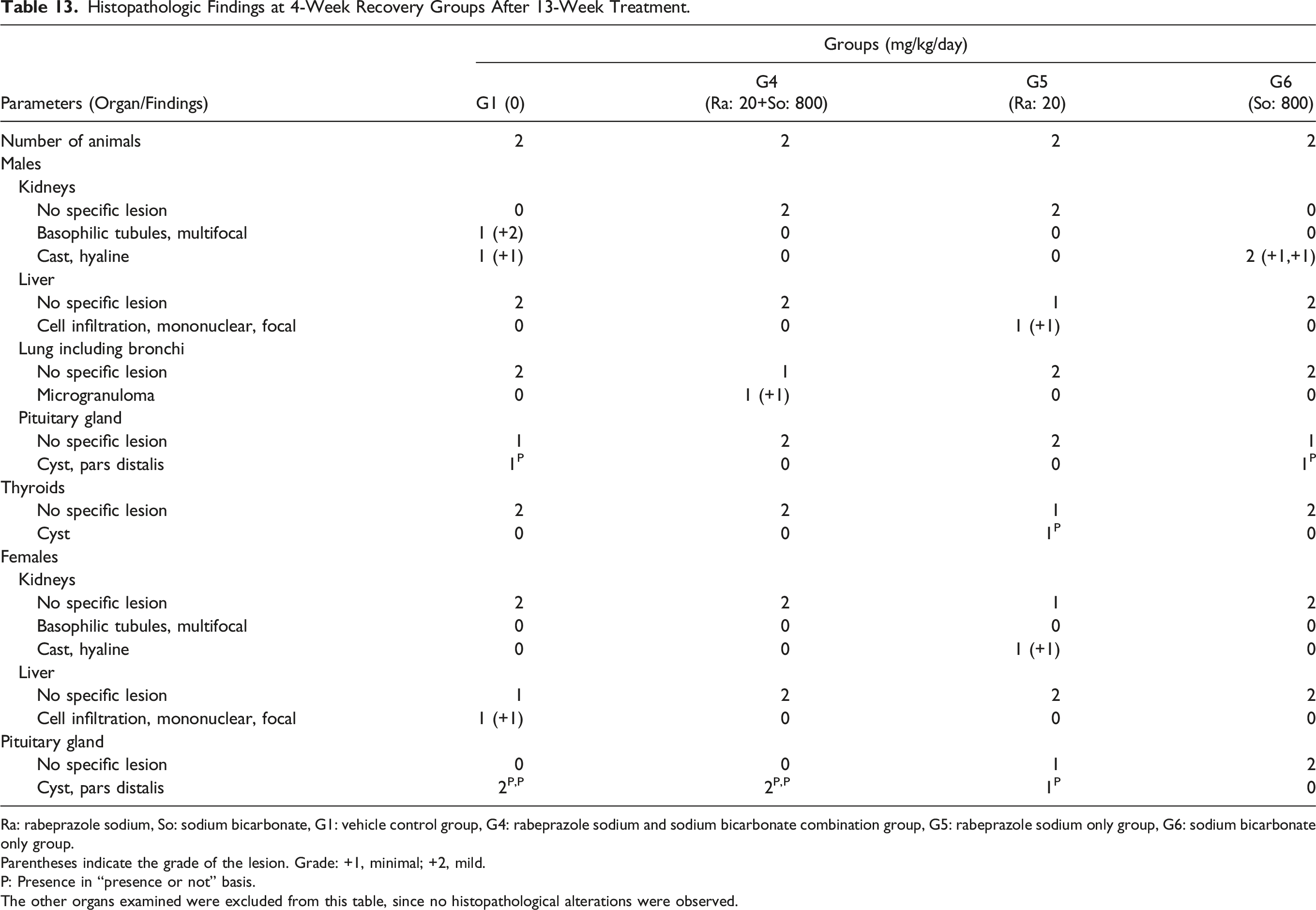
Ra: rabeprazole sodium, So: sodium bicarbonate, G1: vehicle control group, G4: rabeprazole sodium and sodium bicarbonate combination group, G5: rabeprazole sodium only group, G6: sodium bicarbonate only group.
Parentheses indicate the grade of the lesion. Grade: +1, minimal; +2, mild.
P: Presence in “presence or not” basis.
The other organs examined were excluded from this table, since no histopathological alterations were observed.
During the recovery period, a single case of multifocal basophilic tubules, hyaline cast in the kidneys, and cysts in the pars distalis of the pituitary gland were observed in the male control group. In the male (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate group, microgranuloma in the lung was observed in one case. In the male 20 mg/kg of rabeprazole sodium group, focal mononuclear cell infiltration in the liver and cyst in the thyroids were observed in one case. In the male 800 mg/kg of sodium bicarbonate group, hyaline cast in the kidneys was observed in both dogs, and cyst in the pituitary gland’s pars distalis was observed in one case. In the female control group, focal mononuclear cell infiltration in the liver was observed in one case, and a cyst in the pituitary gland’s pars distalis was observed in both dogs. In the female (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate group, a cyst in the pars distalis of the pituitary gland was observed in two cases. In the female 20 mg/kg of rabeprazole sodium group, a hyaline cast in the kidneys and a cyst in the pars distalis of the pituitary gland were observed in one case.
Toxicokinetics Study
Toxicokinetics in Male and Female Dogs After Day 1 and Day 91 of Dosing.
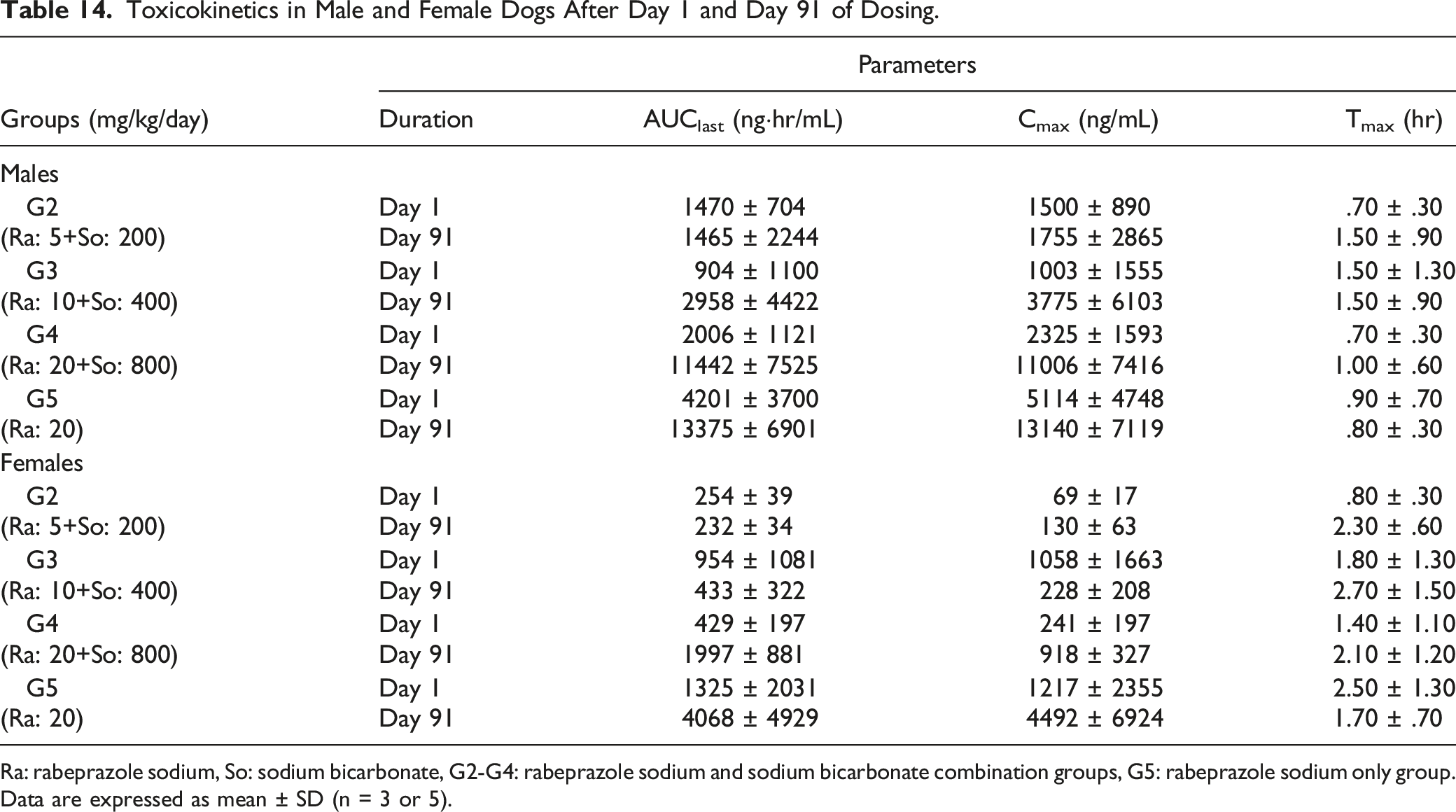
Ra: rabeprazole sodium, So: sodium bicarbonate, G2-G4: rabeprazole sodium and sodium bicarbonate combination groups, G5: rabeprazole sodium only group. Data are expressed as mean ± SD (n = 3 or 5).
Discussion
As part of an effort to enhance the stability of rabeprazole tablets in acidic environments, various approaches such as enteric-coated and delayed-release dosage forms have been developed. While the enhanced forms are more stable in the acidic gastric environment, the pharmacokinetic onset of action with such forms is slower than conventional PPIs. Thus, there is an emerging need to develop treatment options that retain the stability but also display a fast absorption capacity with an excellent safety profile. A combined formulation of sodium bicarbonate with rabeprazole sodium, aimed to achieve optimal pharmacological effects, stability, and safety, was conceived by utilizing sodium bicarbonate’s antacid properties to protect rabeprazole sodium from the eventual exposure to gastric acids after oral administration. To investigate the potential toxicity and target organs of the combination of rabeprazole sodium and sodium bicarbonate, a subchronic toxicity and toxicokinetics study of oral rabeprazole sodium and sodium bicarbonate was conducted in beagle dogs by administering daily doses for 13 consecutive weeks, followed by a 4-week recovery period.
The daily administration of (5 + 200), (10 + 400), and (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate, 20 mg/kg of rabeprazole sodium only, and 800 mg/kg of sodium bicarbonate only to beagle dogs for 13 weeks were well tolerated with no mortality observed.
Clinical signs observed in this study included diarrhea and vomiting in both male and female groups receiving rabeprazole sodium + sodium bicarbonate, 20 mg/kg of rabeprazole sodium only, and 800 mg/kg of sodium bicarbonate only. Based on the literature data, the main toxicities and side effects of PPIs reported in animals and humans include vomiting, stomach lesions,13,14 and diarrhea. 15 Common side effects of antacids include alkalosis, belching, nausea, abdominal distension, flatulence, diarrhea, and constipation. 16 Based on these documented side effects, the clinical signs observed in this study were considered to be treatment-related but non-adverse because no other changes were noted in body weight, food consumption, and hematology parameters. In addition, bloody stools, shivering, and salivation was incidentally observed, although infrequently, during the administration period in some animals from the (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate group and the 800 mg/kg of sodium bicarbonate group. However, no abnormal findings were observed at necropsy and following histopathological examination. Therefore, these observations were not considered toxicologically significant.
High urinary pH values were observed on weeks 4 and 13 of the dosing phase in both male and female of all groups receiving rabeprazole sodium + sodium bicarbonate, 20 mg/kg of rabeprazole sodium, and 800 mg/kg of sodium bicarbonate. The elevated pH values were assumed to be caused by sodium bicarbonate; most pH values were comparable to controls at the end of the 4-week recovery period. These changes were of no toxicological significance as sodium bicarbonate is known to alkalize urine. 16 In addition, changes in urine color, urinary protein, occult blood, and epithelial cells in the urine sediment were observed in most animals on weeks 4 and 13 of the dosing and recovery periods. These changes were minor and attributed to normal animal variability, and thus, they were considered non-adverse.
Decreased MCHC in males and increased PLT in females receiving 20 mg/kg of rabeprazole sodium noted on week 4 of the dosing period and decreased MCHC in males from the (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate and 20 mg/kg of rabeprazole sodium groups noted on week 13 of the dosing period were considered minor fluctuations with no toxicological significance.
Increased absolute and/or relative stomach weights in both sexes at all doses of rabeprazole sodium + sodium bicarbonate and 20 mg/kg of rabeprazole sodium groups were caused by the administration of rabeprazole sodium. These changes gradually trended towards control values during the recovery period, suggesting that the effects of rabeprazole sodium were reversible. Increased stomach weights are frequently reported in animal studies involving drugs similar to rabeprazole, such as omeprazole and pantoprazole, which are also PPIs that block gastric acid secretion by inhibiting an enzyme H+/K+-ATPase.17,18 Based on previous literature on drugs with similar therapeutic mechanism, the occurrence of increased stomach weights was not considered an unexpected toxicological finding in dogs receiving rabeprazole sodium + sodium bicarbonate. Additional histopathological examination of the stomach revealed that the combined administration of rabeprazole sodium and sodium bicarbonate did not exhibit any significant toxicological effects.
No treatment-related effects were found on necropsy findings.
Inflammation with ulceration of the esophageal mucosa was observed in one male receiving (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate. Although the inflammation in the esophagus was observed in only one animal, the severity of the lesion was moderate with the presence of ulceration, and the site of occurrence was focally extensive, so it was judged to be treatment-related since these findings are neither spontaneous nor common in beagle dogs. Furthermore, the lesions are unlikely to be caused by physical stimulation or vomiting during oral dosage. A few other findings were reported, such as hyaline cast in the kidneys, focal mononuclear cell infiltration in the liver, microgranuloma in the lung, a cyst in the pars distalis of the pituitary gland, a cyst in the thyroids, and mononuclear cell infiltration in the interstitium of epididymides. These changes were also observed in the control group, are within the range of background lesions commonly observed in this breed of dogs of the same age, or were distributed sporadically among control and treated groups. Therefore, they were not considered toxicologically significant.
The bioavailability of rabeprazole when dosed orally at a standard dose of 20 mg was 52%. After repeated ingestion of rabeprazole, no significant accumulation occurs because the elimination half-life is one hour after single dose and one and a half hours after repeated administrations.19–21
In the toxicokinetics part of this study, AUClast and Cmax values for females were lower than those of males from the (20 + 800) mg/kg rabeprazole sodium + sodium bicarbonate group on Day 91 (AUClast and Cmax, 17% and 8% of males, respectively). The observed differences were determined to be statistically significant, suggesting sex differences in the toxicokinetics of rabeprazole sodium + sodium bicarbonate.
The AUClast and Cmax at Day 91 were increased compared to Day 1 in a dose-dependent manner. These increases were because vomiting occurred in most animals of the high-dose group on Day 1; however, their vomiting symptom was sparsely observed or not observed on Day 91. Tmax was delayed, and repeated doses showed no sign of drug accumulation. As mentioned above, an ulceration of the esophagus was seen in one male receiving (20 + 800) mg/kg of the rabeprazole sodium + sodium bicarbonate, but not in the female group. Therefore, increased AUClast and Cmax at Day 91 after repeated dosage may have influenced the histopathological results of the esophagus in the male dog, and the observed clinical differences between male and female groups were taken into account when determining the no-observed-adverse-effect level (NOAEL) of the combination of rabeprazole sodium + sodium bicarbonate.
In females on Day 1 of the rabeprazole sodium + sodium bicarbonate group, increased AUClast and Cmax were more significant in the middle dose group than in the high dose group. This was confirmed to be due to vomiting in all animals of the high dose group; however, their vomiting symptom was sparsely observed or not observed on Day 91.
Considering the inflammation with a moderate ulceration of esophagus in male (20 + 800) mg/kg of rabeprazole sodium + sodium bicarbonate group and sex difference in toxicokinetics result, we suggest that under the conditions of this study, the NOAEL of the combination of rabeprazole sodium and sodium bicarbonate is (10 + 400) mg/kg and (20 + 800) mg/kg for male and female dogs, respectively.
Footnotes
Acknowledgments
We thank Daniel Lee of Croen Inc. for proofreading of this manuscript for better English presentation.
Author Contributions
Roh, JS., contributed to conception and design, drafted manuscript, and critically revised manuscript; Nam, KY., contributed to conception and design and critically revised manuscript; Jung, WT., contributed to conception and design and critically revised manuscript; Kim, YM., contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Hwang, EK., contributed to design, contributed to analysis and interpretation, and drafted manuscript; and Jeon, TW., contributed to conception, contributed to interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
