Abstract
Organophosphate pesticides are widely used; however, their use is limited due to neurotoxicity and, to a lesser extent, cardiotoxicity in humans. Given the high energy demands of cardiac muscle, which is characterized by a dense population of mitochondria, any damage to these organelles can exacerbate cardiotoxicity. This study aims to elucidate whether the cardiotoxic effects of organophosphate pesticides originate from mitochondrial dysfunction. To investigate this, in silico toxicogenomic analyses were performed using various tools, such as the Comparative Toxicogenomic Database, GeneMANIA, STRING, and Cytoscape. Results revealed that 11 out of the 13 WHO-recommended Class Ia organophosphate pesticides target genes associated with cardiotoxicity. Notably, three of these genes were mitochondrial, with catalase (CAT) being the common differentially expressed gene among parathion, methyl parathion, and phorate. Furthermore, protein-protein interaction analysis indicated a strong association between CAT and superoxide dismutase 2, mitochondrial (SOD2). Subsequently, isolated heart mitochondria were utilized to assess CAT and superoxide dismutase (SOD) activities in vitro. The findings demonstrated that at a concentration of 7.5 ng/µL, both methyl parathion and phorate significantly decreased CAT activity by approximately 35%. Moreover, phorate reduced total SOD and SOD2 activities by 17% and 19%, respectively, at the same concentration. In contrast, none of the three organophosphate pesticides induced the opening of the mitochondrial permeability transition pore. These results suggest that the reduction in CAT and SOD2 activities, critical antioxidant enzymes, leads to the accumulation of reactive oxygen species within mitochondria, ultimately resulting in mitochondrial damage. This mechanism likely underlies the observed cardiotoxicity induced by these organophosphate pesticides.
Keywords
Introduction
Organophosphates are a class of chemical compounds produced when phosphoric acid reacts with alcohol to form esters. They are active components in many pesticides and are commonly used in the production of insecticides and herbicides (e.g., tribufos) in agriculture, households, gardening, and veterinary practices. 1 Exposure to organophosphate pesticides (OPPs) can lead to neurotoxicity in both humans and insects through inhalation, ingestion, ocular contact, and dermal exposure. Neurotoxicity is caused by the inhibition of acetylcholinesterase in the nervous system, leading to an excess of the neurotransmitter acetylcholine and resulting in cholinergic toxidrome symptoms.2,3 In addition to neurotoxicity, OPPs can also cause cardiovascular complications. These cardiovascular effects are acutely life-threatening and include noncardiogenic pulmonary edema and arrhythmia, as well as both hypertension and hypotension.4-9
According to the World Health Organization’s (WHO) Recommended Classification of Pesticides by Hazard, Class Ia (extremely hazardous) includes 29 active ingredients in pesticides, and 13 of them belong to organophosphates. 10 Cardiac complications related to acute exposure to Class Ia OPPs, especially parathion (PT) and methyl parathion (MPT), have been reported in humans. These complications include noncardiogenic pulmonary edema and electrocardiographic abnormalities.4,11-13 Additionally, cardiotoxicity has been observed after PT and MPT exposure in in vitro studies using human iPSC cardiomyocytes and H9c2 cells (embryonic rat cardiomyocytes), as well as in vivo studies using bullfrog tadpoles, rats, and zebrafish embryos.14-18 Given the significant role of mitochondria in heart tissue, understanding whether these pesticides cause damage through mitochondrial pathways is essential to fully comprehending the cardiotoxic effects observed.
Mitochondria are particularly abundant in heart tissue, which requires a significant amount of energy. Any damage that may occur to mitochondria can lead to serious toxicity in the heart. It is unknown whether the cardiotoxic effects induced by these Class Ia OPPs are mediated through mitochondria, and the specific targets of OPPs within mitochondria are also unknown. This study aimed to analyze toxicogenomic data to identify common genes between Class Ia OPPs and cardiotoxicity and to determine whether this toxicity is mediated by mitochondria. We identified genes associated with mitochondria and explored the interactions among these genes. Additionally, we conducted in vitro studies using bovine heart mitochondria exposed to selected Class Ia OPPs identified from the toxicogenomic data.
Materials and Methods
Identifying the Genes Related to Cardiotoxicity and the Organophosphate Pesticides
To investigate the potential relationship between cardiotoxicity and Class Ia OPPs, data from the Comparative Toxicogenomics Database (CTD) (https://ctdbase.org) 19 were retrieved on December 9, 2023. Cardiotoxicity data were sourced from the “Direct Evidence” section of CTD, where “M” denotes “marker/mechanism” and “T” denotes “therapeutic.” Here, “marker/mechanism” refers to a gene that may be a biomarker of a disease (e.g., increased expression of gene X correlates with cardiovascular diseases) or play a role in the etiology of a disease (e.g., mutations in gene X cause cardiovascular complications). “Therapeutic” denotes a gene that is or may be a therapeutic target in the treatment of a disease (e.g., targeted reduction of gene X expression reduces susceptibility to cardiotoxicity). Utilizing the MyVenn CTD tool (https://ctdbase.org/tools/myVenn.go), the common genes associated with the OPPs and cardiotoxicity were identified. 19 Additionally, the proteins encoded by these genes, specifically their involvement in the mitochondrial proteome, were determined for further studies. OPPs that interact with these mitochondrial proteins were also identified.
Exploring Gene-Gene and Protein-Protein Interactions
GeneMANIA (http://genemania.org) was used to study the network of gene-gene interactions of the selected gene. 20 Homo sapiens was chosen as the target organism in the analysis. For protein-protein interactions (PPI) associated with the selected proteins, the String v.12 database was used (https://string-db.org/cgi). 21 The protein was further selected through the use of the Multiple Proteins by the Names/Identifiers Tool, specifying the species as “Homo sapiens” and ensuring a highest confidence score of .9. The final PPI network was constructed using Cytoscape 3.10.1. 22
In Vitro Analyses
Chemicals
Parathion (98.92%, CAS: 56-38-2), methyl parathion (98.44 %, CAS: 298-00-0), and phorate (95.24%, CAS: 298-02-2) were purchased from Dr. Ehrenstorfer GmbH and were soluble in cyclohexane. Therefore, in addition to dimethyl sulfoxide (DMSO), cyclohexane was used as a control in all in vitro studies. Pyrogallol, an autoxidizing compound that serves both as a source and a scavenger of oxygen-free radicals, was obtained from Carl Roth GmbH. All additional reagents employed in this study were of analytical grade.
Preparation of Mitochondria from the Bovine Heart
Mitochondrial isolation was carried out following the protocol described in a previous study. 23 To begin with, the heart of a freshly slaughtered bovine was washed immediately with ice-cold phosphate-buffered saline (100 mM PBS with pH 7.4, containing .9% NaCl). The tissues were then placed in Petri dishes containing ice-cold sucrose-mannitol buffer (pH 7.5) consisting of 20 mM HEPES, 70 mM sucrose, 220 mM mannitol, 2 mM EDTA, and .5 mg/mL of bovine serum albumin and minced using a scalpel. Next, 1.5 g of tissue sample was homogenized in 10 volumes of sucrose-mannitol buffer. The resulting homogenate was centrifuged at 600 g for 10 min and then at 650 g for another 10 min. The supernatant obtained was further centrifuged at 8,000 g for 15 min to isolate crude mitochondria. The crude mitochondrial pellet was gently washed six times with PBS and centrifuged at 9,000 g for 5 min to obtain pure mitochondria. In experiments where a mitochondrial matrix was required, the mitochondria were sonicated three times at 30 kHz for 10 s each. Subsequently, the lysate was centrifuged at 147,000 g for 1 h at 4°C to separate the mitochondrial matrix and membrane.
Measurement of Catalase Activity
The mitochondrial matrix (20 µL at 1 mg/mL) and 2.5, 5, and 7.5 ng/µL of PT, MPT, and phorate at final concentration were separately added to 50 mM PBS (pH: 7.00). The dose levels of pesticides were obtained from the literature.18,24 Subsequently, 1 mL of 30 mM hydrogen peroxide (H2O2) was added, and the cuvettes were mixed for 30 s. The enzymatic degradation of H2O2 was then monitored for 2 min at 240 nm. 25
Measurement of Superoxide Dismutase Activities
The total activity of superoxide dismutase (SOD) was measured using spectrophotometric assessment in a 50-mM Tris-HCl buffer with a pH of 8.2 and 1 mM EDTA. Pyrogallol was used as the substrate, following the method outlined by Marklund and Marklund. 26 The mitochondrial matrix (1 mg/mL) was incubated in the buffer for 20 s, after which pyrogallol and the pesticide (2.5, 5, and 7.5 ng/µL at final concentration) were added, and the autooxidation of the substrate was observed for 2 min. This was done by monitoring the reduction in absorbance at 420 nm. The difference in autooxidation rates with and without the mitochondrial matrix was used to calculate the total SOD activity. To measure SOD2 (MnSOD) activity specifically, SOD1 (Cu-Zn SOD) was inhibited with KCN, following the same approach used for total SOD measurement. SOD1 activity was calculated by subtracting SOD2 activity from total SOD activity.
Evaluation of the Mitochondrial Permeability Transition
Mitochondrial swelling, which is an indicator for mitochondrial permeability transition pore (mPTP) opening, was measured using the method described by Masubuchi et al. 27 Bovine heart mitochondria at a concentration of 1 mg/mL were exposed to PT, MPT, and phorate at three different doses (2.5, 5, and 7.5 ng/µL at the final concentration)18,24 in a reaction medium consisting of 125 mM sucrose, 50 mM KCl, 5 mM HEPES, 2 mM KH2PO4, 1 mM MgCl2, and pH 7.2 for 10 min. The incubation with pesticides was performed with and without the addition of CaCl2 (250 µM final concentration) as a positive control for inducing mPTP opening. This is labeled as “+Ca” in the result graph. Absorbance was measured at 1-min intervals over a 10-min period. The decrease in absorbance indicated an increase in mitochondrial swelling.
Statistical Analysis
The data were presented as the mean ± standard error of the mean (SEM) from three or more independent experiments, each of which was conducted in triplicate. Statistical analysis was conducted using GraphPad Prism Software version 8 (San Diego, CA, USA) for Windows. Unpaired and two-tailed Student’s t-tests were utilized for statistical comparisons, with a confidence level of 95%. Significance was accepted when P ≤ .05.
Results
Common Genes Associated with Organophosphate Pesticides and Cardiotoxicity, and Interactions
Genes proven by in vitro and in vivo studies to be associated with cardiotoxicity.

Overlapped genes between the OPPs and cardiotoxicity.
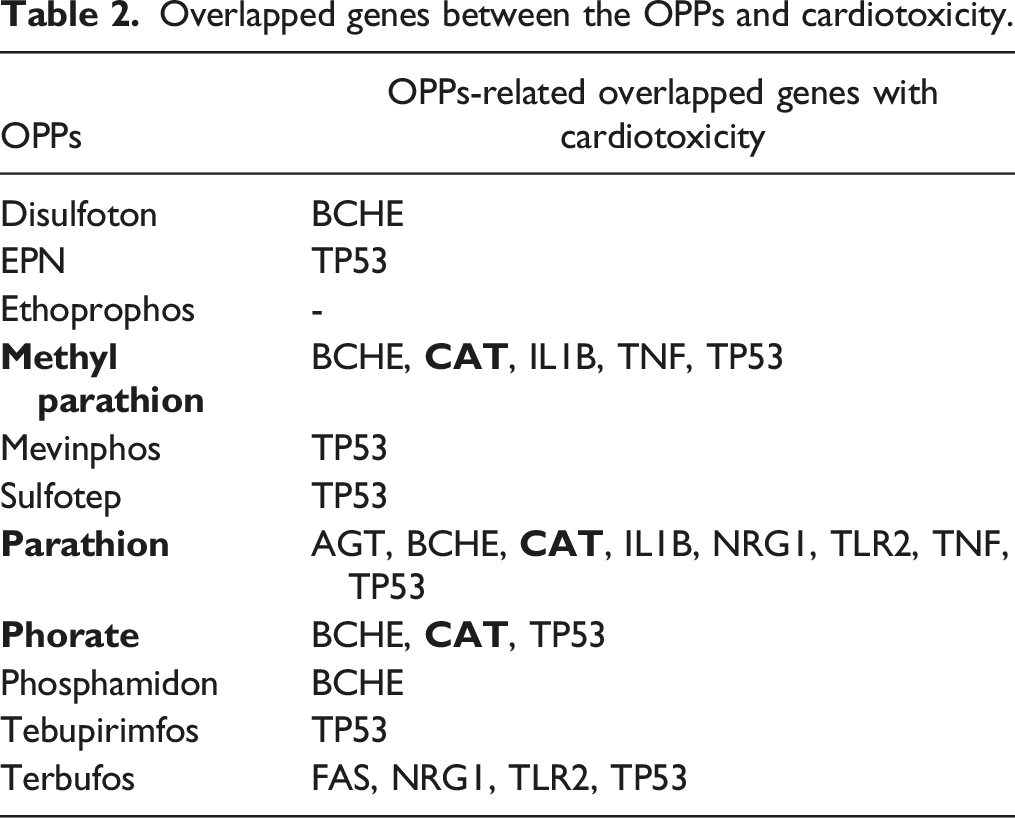
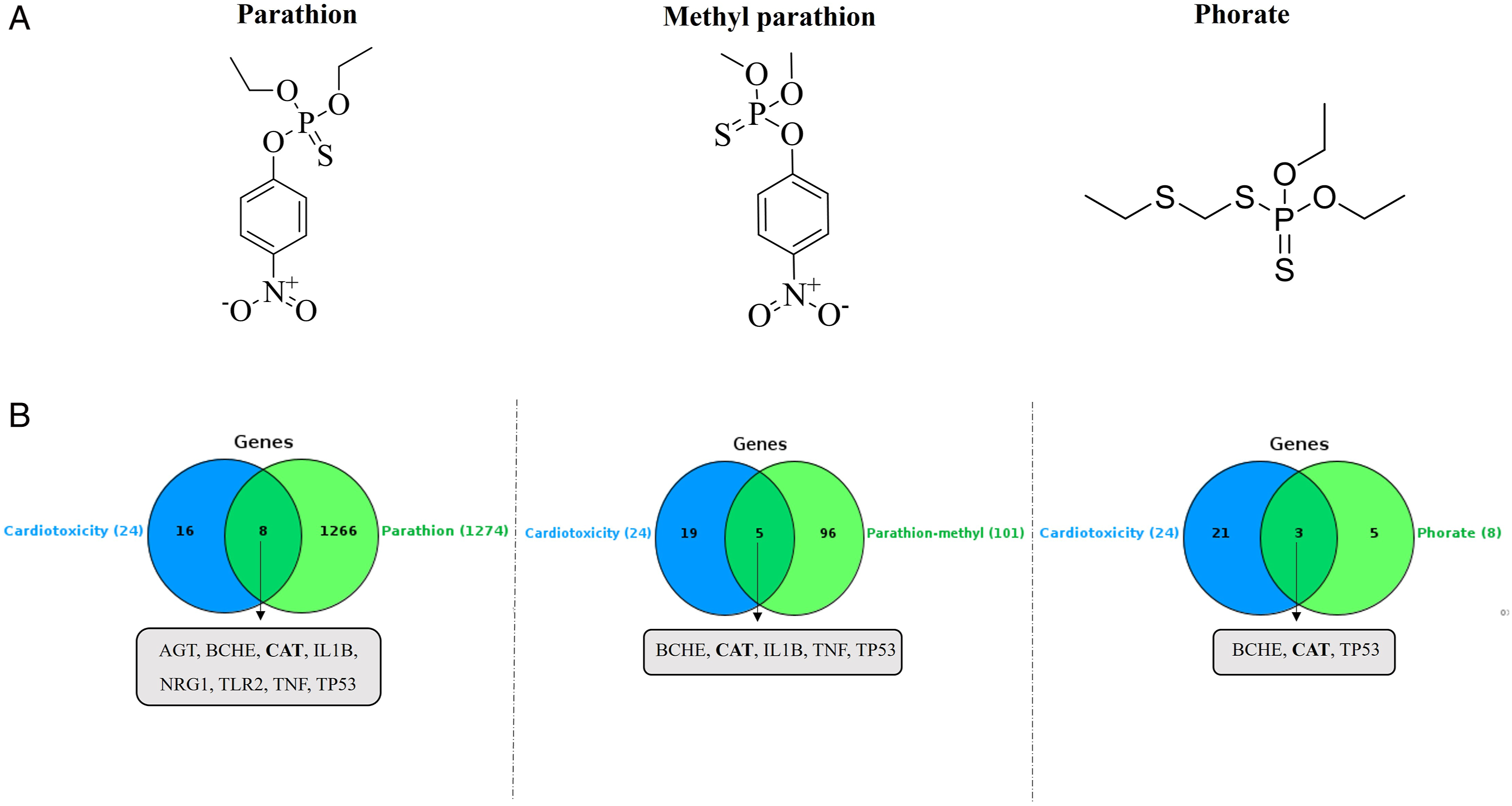
(A) Molecular structures of parathion, parathion-methyl, and phorate. (B) Venn diagrams depicting the overlap of genes between an OPP and cardiotoxicity.
Gene-Gene and Protein-Protein Interactions of Catalase
After identifying CAT as a mitochondrial protein in the relationship between OPPs and cardiotoxicity, the official gene symbol “CAT” was input into the GeneMANIA online plugin to generate a network. The analysis revealed that physical interactions (77.64%) were the predominant factors between CAT and other interacting genes (Figure 2(A)). Nodes in the outer ring depict genes associated with CAT, according to GeneMANIA. (A) Gene network of CAT analyzed using GeneMANIA. (B) Protein-protein interactions of orange nodes analyzed through STRING and Cytoscape.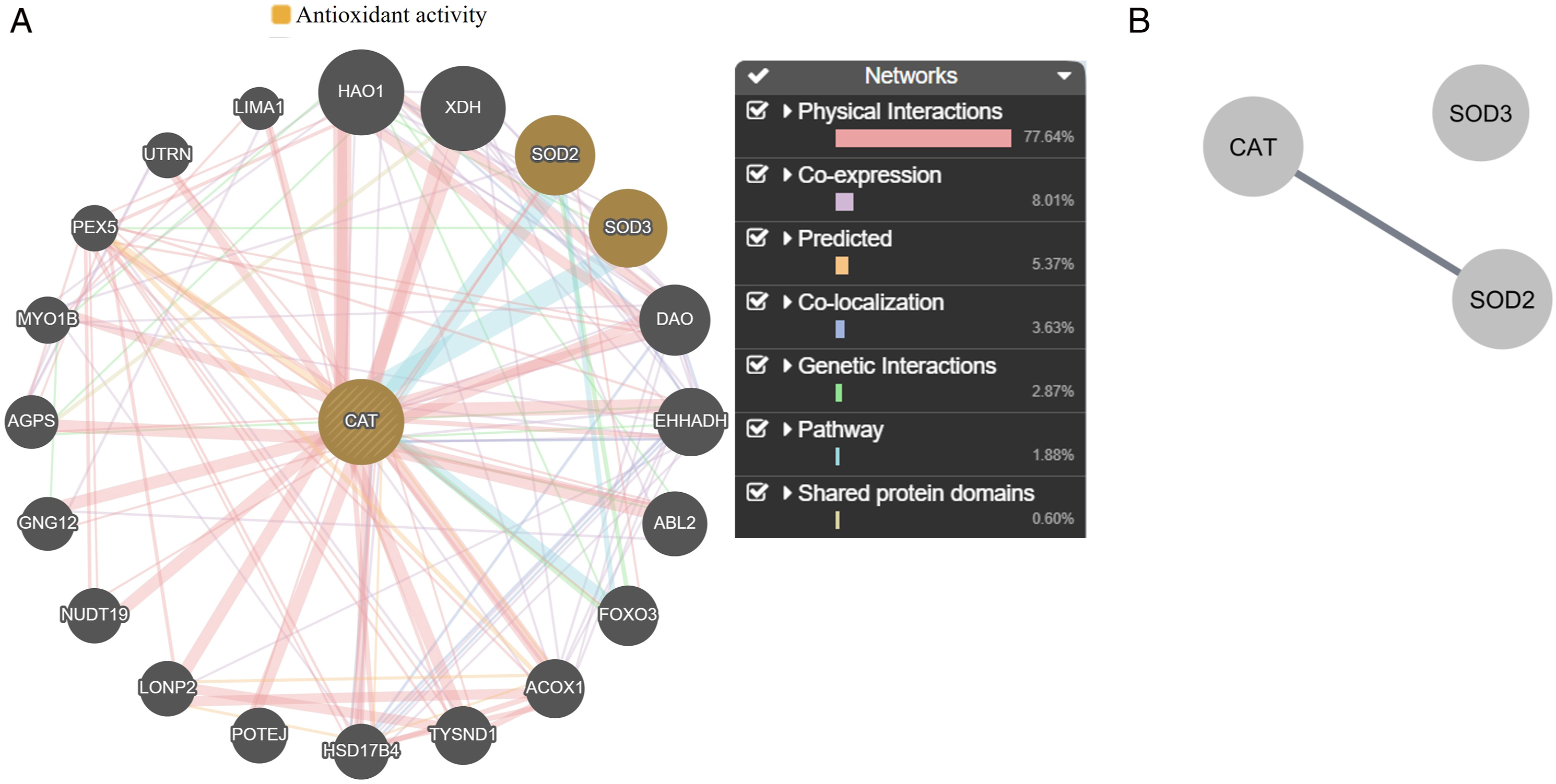
In the function section of GeneMANIA, the antioxidant activity was selected because of CAT. Two orange nodes were also related to the antioxidant activity apart from CAT, with a false discovery rate of 3.56 ×10−2 analyzed using GeneMANIA (Figure 2(A)). Regarding the PPI network (Figure 2(B)), it revealed three nodes and one edge (PPI enrichment P-value: .000991). CAT only interacted with the mitochondrial protein SOD2 with the highest confidence interaction score (.9) in the STRING database.
MPT and Phorate Reduced CAT Activity in the Mitochondria
After isolating the mitochondria from the bovine heart, they were sonicated to obtain the mitochondrial matrix. This matrix was then incubated with PT, MPT, and phorate to test the activity of CAT. PT did not affect CAT activity at any of the three doses tested. However, 7.5 ng/µL of MPT or phorate reduced CAT activity by approximately 35% compared to DMSO and cyclohexane. The results are presented in Figure 3. Effect of PT, MPT, and phorate on CAT activity in isolated heart mitochondria. Groups were compared to both DMSO and cyclohexane (n = 3; *P ≤ .05; ***P ≤ .001).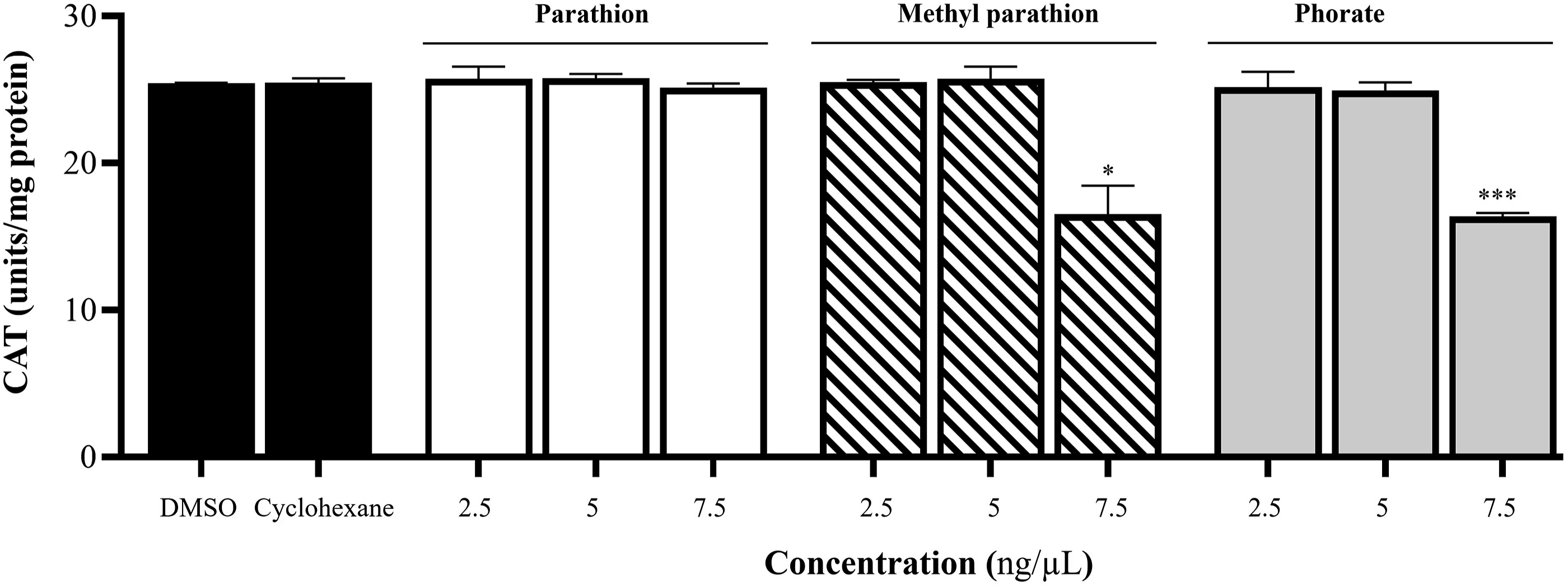
Phorate Reduced SOD2 Activity in the Mitochondria
The mitochondrial matrix was incubated with PT, MPT, and phorate at the indicated concentrations to measure total SOD activity. SOD1 was calculated by subtracting SOD2 activity from total SOD activity. While PT and MPT did not affect total SOD activity, 7.5 ng/µL of phorate reduced total SOD activity by approximately 17% (Figure 4) compared to DMSO and cyclohexane. On the other hand, none of the three pesticides affected SOD1 activity at the tested concentrations (Figure S3). Effect of PT, MPT, and phorate on total SOD activity in isolated heart mitochondria. Groups were compared to both DMSO and cyclohexane (n = 3; ***P ≤ .001).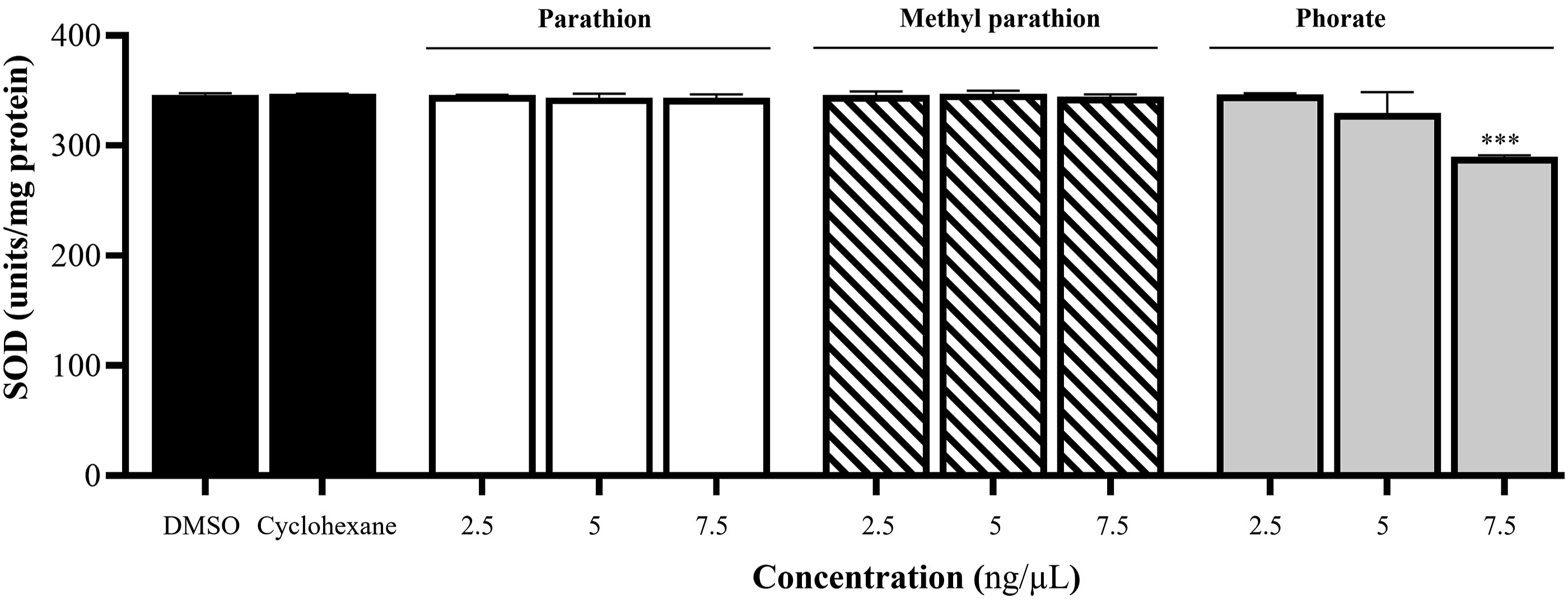
For SOD2 (MnSOD) activity, SOD1 was inhibited with KCN, and the mitochondrial matrix was incubated with PT, MPT, and phorate. PT and MPT did not affect SOD2 activity at any of the three doses tested compared to DMSO and cyclohexane. However, 7.5 ng/µL of phorate reduced SOD2 activity by 19% compared to DMSO and cyclohexane. The results are presented in Figure 5. Effect of PT, MPT, and phorate on SOD2 activity in isolated heart mitochondria. Groups were compared to both DMSO and cyclohexane (n = 3; ****P ≤ .0001).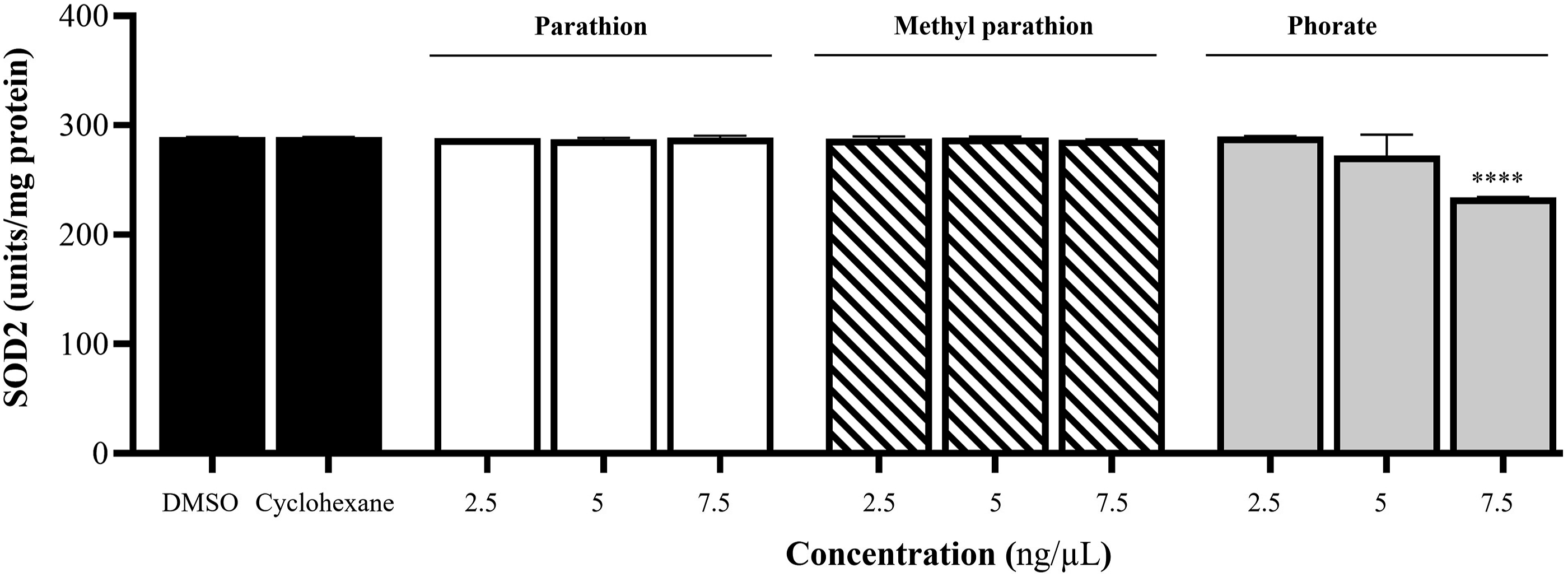
PT, MPT, and Phorate Did Not Induce mPTP Opening in the Mitochondria
To determine whether mPTP opening, which forms on the inner mitochondrial membrane in pathological conditions, is induced by PT, MPT, and phorate, freshly isolated mitochondria (1 mg/mL) were incubated with these OPPs at doses reported in the literature18,24 and also incubated with 250 µM Ca+2 as a positive control. The results showed that PT, MPT, and phorate, at all three doses tested, did not induce mPTP opening in the presence of calcium (+Ca, as shown in Figure 6) and without +Ca (data not shown in Figure 6) compared to +Ca. Only +Ca induced the mPTP opening compared to -Ca (the only mitochondria present) because +Ca was used as the positive control. Effect of PT, MPT, and phorate on mPTP opening in isolated heart mitochondria (n = 3; ****P ≤ .0001; compared to -Ca). Cyclo. = cyclohexane.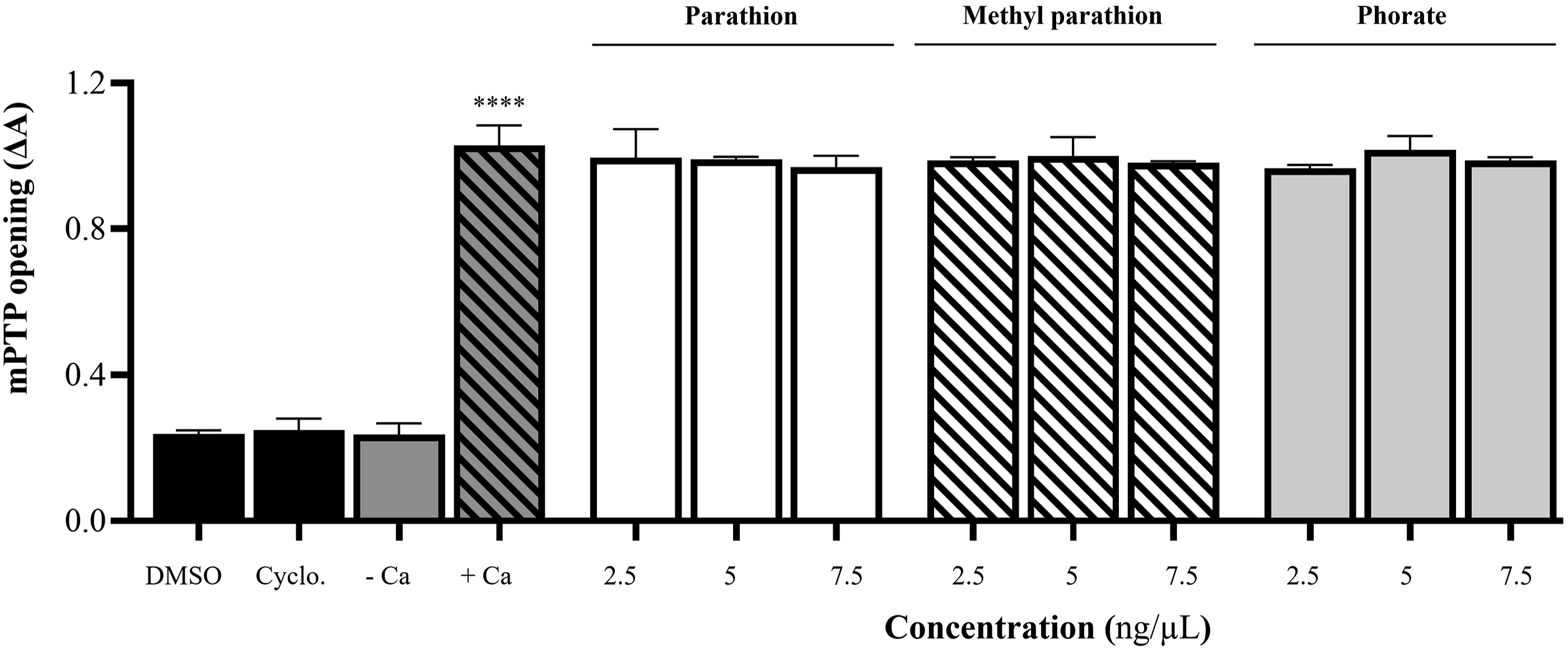
Discussion
Organophosphate pesticides (OPPs) have been restricted due to their neurotoxicity.2,3 They have also been linked to cardiotoxicity in humans.4-9,11-13 Karki et al. 4 reported that cardiac complications such as electrocardiographic abnormalities and noncardiogenic pulmonary edema developed in 23 (poisoning with methyl parathion) of 37 poisoning cases. Kidiyoor et al. 12 and Joshi et al. 13 reported a rare myocardial infarction due to parathion consumption. Muthu et al. 29 observed abnormal ECG with sinus tachycardia and nonspecific ST-T wave changes in a 19-year-old woman as a result of accidental exposure to phorate.
This study was aimed at investigating whether the cardiotoxicity caused by OPPs is related to mitochondria. In this study, first, the relationship between the OPPs, which are in the extremely hazardous class (Class Ia recommended by WHO), 10 and cardiotoxicity was screened using in silico methods. The results showed that 24 genes were associated with cardiotoxicity, and 11 of the Class Ia OPPs were related to this cardiotoxicity (Table 1). Out of these 24 genes, three (AGT, FAS, and CAT) were mitochondrial, and only catalase (CAT) was common to PT, MPT, and the phorate of 11 OPPs (Table 2).
CAT is an antioxidant enzyme found in peroxisomes and the mitochondrial matrix in humans and other mammals, as indicated by Human MitoCarta3.0. 28 It plays a vital role in breaking down hydrogen peroxide into water and oxygen, which is essential for maintaining the oxidant-antioxidant balance. In silico analysis (Figure 2(B)) strongly associates CAT with mitochondrial SOD (SOD2; MnSOD). Therefore, total SOD, SOD2 activities, and also SOD1 activities are considered, as Human MitoCarta3.0 29 reports the presence of SOD1 in human mitochondria. If CAT and SOD activities are reduced, hydrogen peroxide and superoxide radicals, respectively, can accumulate, potentially exacerbating cardiotoxicity.
In vitro studies showed that PT did not affect CAT and SOD activities, while MPT and phorate decreased CAT activity (Figure 3). Additionally, only phorate reduced total SOD and SOD2 activities (Figures 4 and 5). On the other hand, PT, MPT, and phorate did not induce mPTP opening. While there are no in vitro and in vivo studies related to phorate-cardiotoxicity in the literature, there are a limited number of in vitro and in vivo studies regarding PT and MPT cardiotoxicity. For example, Wen et al. 15 reported that when they exposed H9c2 embryonic rat cardiomyocytes to MPT, cell viability decreased and apoptosis increased. In another in vitro study, Sirenko et al. 14 reported that they created moderate cardiophysiological and mitochondrial effects when they exposed human iPSC-derived cardiomyocytes to PT for 30 min. In in vivo studies, Costa et al. 16 observed a reduction in tadpoles' cardiac ventricular mass when they exposed bullfrog tadpoles to MPT. Wen et al. 17 reported that SOD activity in the heart tissues of rats exposed to MPT decreased by approximately 10% compared to the control. Chen et al. 18 reported that when they applied 10 mg/L of MPT to zebrafish embryos, total SOD activity decreased significantly and CAT activity increased.
Since mitochondria are the organelle where large amounts of reactive oxygen species are produced, the activity of antioxidant enzymes here is crucial for mitochondrial functioning. Reducing CAT and SOD2 activities results in the accumulation of H2O2 and superoxide radicals and causes mitochondrial damage.
Conclusions
The present study highlights that MPT and phorate may induce the cardiotoxicity via mitochondria by decreasing the antioxidant enzymes activities. The mitochondrial CAT plays an important role in MPT and phorate-related cardiotoxicity. MPT and phorate at the highest tested concentration reduced CAT and SOD activities. The results show that CAT and SOD are important targets in MPT and phorate-related cardiotoxicity. On the other hand, it remains unknown whether the decreases in the activity of these enzymes result from reduced mRNA expression or protein synthesis of the relevant enzymes. Therefore, assessing CAT and SOD expressions along with protein amounts can offer more detailed insights into the literature on this subject.
Supplemental Material
Supplemental Material - Mitochondrial Impact of Organophosphate Pesticide-Induced Cardiotoxicity: An In Silico and In Vitro Study
Supplemental Material for Mitochondrial Impact of Organophosphate Pesticide-Induced Cardiotoxicity: An In Silico and In Vitro Study by Fuat Karakuş, Ege Arzuk, and Ali Ergüç in International Journal of Toxicology
Footnotes
Author Contributions
Karakuş, F contributed to conception and design, acquisition, drafted manuscript and critically revised manuscript. Arzuk, E contributed to analysis and interpretation and critically revised the manuscript. Ergüç, A contributed to analysis and critically revised the manuscript. All authors gave final approval and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
