Abstract
In silico modeling offers an opportunity to supplement and accelerate cardiac safety testing. With in silico modeling, computational simulation methods are used to predict electrophysiological interactions and pharmacological effects of novel drugs on critical physiological processes. The O’Hara-Rudy’s model was developed to predict the response to different ion channel inhibition levels on cardiac action potential duration (APD) which is known to directly correlate with the QT interval. APD data at 30% 60% and 90% inhibition were derived from the model to delineate possible ventricular arrhythmia scenarios and the marginal contribution of each ion channel to the model. Action potential values were calculated for epicardial, myocardial, and endocardial cells, with action potential curve modeling. This study assessed cardiac ion channel inhibition data combinations to consider when undertaking in silico modeling of proarrhythmic effects as stipulated in the Comprehensive in Vitro Proarrhythmia Assay (CiPA). As expected, our data highlight the importance of the delayed rectifier potassium channel (IKr) as the most impactful channel for APD prolongation. The impact of the transient outward potassium channel (Ito) inhibition on APD was minimal while the inward rectifier (IK1) and slow component of the delayed rectifier potassium channel (IKs) also had limited APD effects. In contrast, the contribution of fast sodium channel (INa) and/or L-type calcium channel (ICa) inhibition resulted in substantial APD alterations supporting the pharmacological relevance of in silico modeling using input from a limited number of cardiac ion channels including IKr, INa, and ICa, at least at an early stage of drug development.
Introduction
Cardiac ion channel evaluation, using both in vitro and in silico methods, is an important aspect of safety assessment during drug development to determine the potential risk of arrhythmia development such as ventricular tachycardia, torsades de pointes, or polymorphic ventricular tachycardia (TdP).1,2 In vitro evaluation of the potential of a drug to block the human ether-à-go-go related gene (hERG) channel (also called, rapid delayed rectifier potassium current Ikr), and other major channels such as the voltage-gated calcium (Cav) and sodium (Nav) channels, Cav 1.2 and Nav 1.5, respectively, are important during proarrhythmia risk assessment. 2 Kramer et al showed that drugs with Ikr block combined with late sodium and/or calcium current block had lower TdP risk than drugs that only blocked Ikr.2–4
Drug development does not systematically include all ion channels that are included in the O’Hara-Rudy model. Time and resources are the main reasons for conducting patch clamp analyses only on a subset of cardiac ion channels. Given the very high number of drug candidates in development, it would often not be feasible to evaluate inhibition for all ion channels, for all drug candidates. One important advantage of in silico modeling is the rapid generation of relevant safety data. In silico modeling can be used at very early stages of drug development when the work is initiated, and it can be repeated when additional ion channel data is generated for compounds that were selected from the preliminary data. Ion channels that have the greatest impact on cardiac repolarization (i.e., hERG, Nav1.5, and Cav1.2) are typically the first ones tested. Compounds that have favorable profiles for these channels are then advanced to later stages of development. 2 In silico modeling is used to predict proarrhythmic risk associated with new drugs by leveraging patch clamp data from individual ion channel and complementing this data using the O’Hara Rudy dynamic (ORd) model of human ventricular cells. This approach can alleviate some of the limitations associated with multi-ion channel in vitro methods, including sourcing human ventricular myocytes, and assays using non-cardiac or non-human cells. 5 The latter may not express subunit and anchoring proteins important for ionic channel kinetics necessary to assess arrhythmia risk.6,7 Other in silico models have been considered such as the TdP risk metric (qNet). This metric involves analyzing drug-induced changes in net charge during the action potential (AP).8–11 The O’Hara Rudy in silico model was developed to better understand human ventricular electrophysiology by dissecting the electrophysiology contribution of potassium [IKr (rapid delayed rectifier K+ current), IKs (slow delayed rectifier K+ current), IK1 (inward rectifier K+ current), Ito (transient outward K+ current)], calcium (L-type Ca2+), and sodium [INa (fast Na+ current)] channels. 5 In silico modeling methods derived from human data can improve translation potential for TdP risk, and have demonstrated higher or comparable sensitivity, specificity, and accuracy than those obtained in animal studies.10,12,13 For example, in silico dynamic modeling was demonstrated to improve the prediction of TdP risk from the interaction between the hERG channel (IKr) and a multi-ion channel drug. 14 As a constituent of the regulatory landscape, the Comprehensive In vitro Proarrhythmia Assay (CiPA) was developed to improve the prediction of arrhythmia by evaluating selected ion channels: IKr (Kv11.1/hERG), ICa (L-type; Cav1.2), INa (Nav1.5 peak and late current); Ito (Kv4.3); IKs (Kv7.1), and IK1 (Kir2.1). 1 However, currently, as per the International Council of Harmonization (ICH) in the S7B and E14 guidelines, only IKr (hERG), ICa, and INa are formally listed as components of the proarrhythmic risk assessment.15-18 This study aims to evaluate the impact of different ion channel combinations on proarrhythmic risk assessment when using an in silico model. More specifically, outcomes obtained with data from all channels included in the CiPA were compared to AP Duration (APD) outcome obtained from a partial CiPA panel with ion channels commonly included (IKr, ICa, and INa) in safety assessments. Additionally, the study applied in silico modeling to inhibition data recording using manual patch clamp with multiple drugs [cisapride (1 μM), terfenadine (1 μM), amiodarone (1 μM), and verapamil (10 μM)].
Since the Sodium (Nav1.5 peak and late current) channels are being considered as one, only 6 channels were evaluated in this paper, instead of 7. First, a single ion channel was tested with the O’Hara Rudy model with an inhibition percentage ranging from 0%–100%, followed by testing for a combination of multiple ion channels. Secondly, the APD at 30, 60, and 90% repolarization were evaluated with inhibition ranging from 0 to 100%. The analysis of the APD90 (action potential duration at 90% repolarization) of multiple combinations of ion channels with different inhibition percentages were also evaluated. Finally, triangulation, the repolarization time from APD30 to APD90, was assessed.
Materials and Methods
HEK 293 cells with stable ion channel expression were used in manual whole-cell patch clamp electrophysiological configuration (34 ± 1°C) with protocols as described previously. 19 For the recordings, tyrode solution (external solution) contained (mM): NaCl (137), KCl (4), CaCl2 (1.8), MgCl2 (1), Glucose (10), HEPES (10). pH was adjusted to 7.4 with NaOH. The pipette solution (internal solution) contained (mM): K-aspartate (130), MgCl2 (5), EGTA (5), Na2ATP (4), and HEPES (10) dissolved in Sterile Water for Injection USP. pH was adjusted to 7.2 with KOH. Junction potentials between bath and pipette solutions averaged 10.5 mV and were corrected for RMP measurements only. Borosilicate glass electrodes filled with pipette solution were connected to a patch clamp amplifier (Ax-opatch 200B; Axon). Electrodes had tip resistances of 2 to 5 MΩ. The O’Hara-Rudy model was applied to manual patch clamp inhibition profiles and the duration of the cardiac action potentials at 90% repolarization (APD90) was calculated at a stable pacing rate for single cells at 60 bpm (i.e., basic cycle length of 1000 ms). 5
The O’Hara Rudy in silico model was used to simulate different percentages of ion channel inhibition for 6 different ionic channels (IKr: Kv11.1/hERG; IK1: Kir 2.1; ICa: Cav1.2; INa: Nav1.5; Ito: Kv4.3; IKs: Kv7.1). 5 The in silico model was based on C++ code provided by the authors that was compiled using the open-source compiler Code::Blocks. Scripts for running simulations were coded in Python (Anaconda distribution, Anaconda Inc., Texas) in the Jupyter notebook environment (Jupyter, California).
The simulations included different scenarios with a range of inhibition percentages progressing from 0 to 100% for the six individual channels (IKr, ICa, INa, IK1, IKs, and Ito). The different sets of parameters tested included the percentage of inhibition of the six individual ion channels and a combination of multiple ion channels (six channels together, and 2 combinations of three channels: IKr, ICa, INa and IK1, Ito, IKs). The simulations also included baseline models from the different layers of the ventricles (endocardium (endo), myocardium (mid), and epicardium (epi)). Most graphs in this article display data from the mid (or M) layer which was considered representative of the overall effects of ion channel block, effects on the APD and the most proarrhythmic in terms of EAD (early after depolarization) notably.20,21
The APD30, APD60, and APD90 were calculated for every action potential simulated with the different percentages of inhibition. Triangulation was defined as (APD90 – APD30) for the same layer of the heart (i.e., endo, mid, and epi). 22 The total number of simulations was 194 for all the different ion channel combinations that were tested. Inhibition values for all the six CiPA ion channels (IKr, ICa, INa, IK1, IKs, Ito) and for a subset (IKr only, IKr, ICa, INa only) were tested with cisapride (1 μM), terfenadine (1 μM), amiodarone (1 μM), and verapamil (10 μM).
Results
Progressive Inhibition of Single Ion Channels (IKr, ICa, INa, IK1, IKs, Ito)
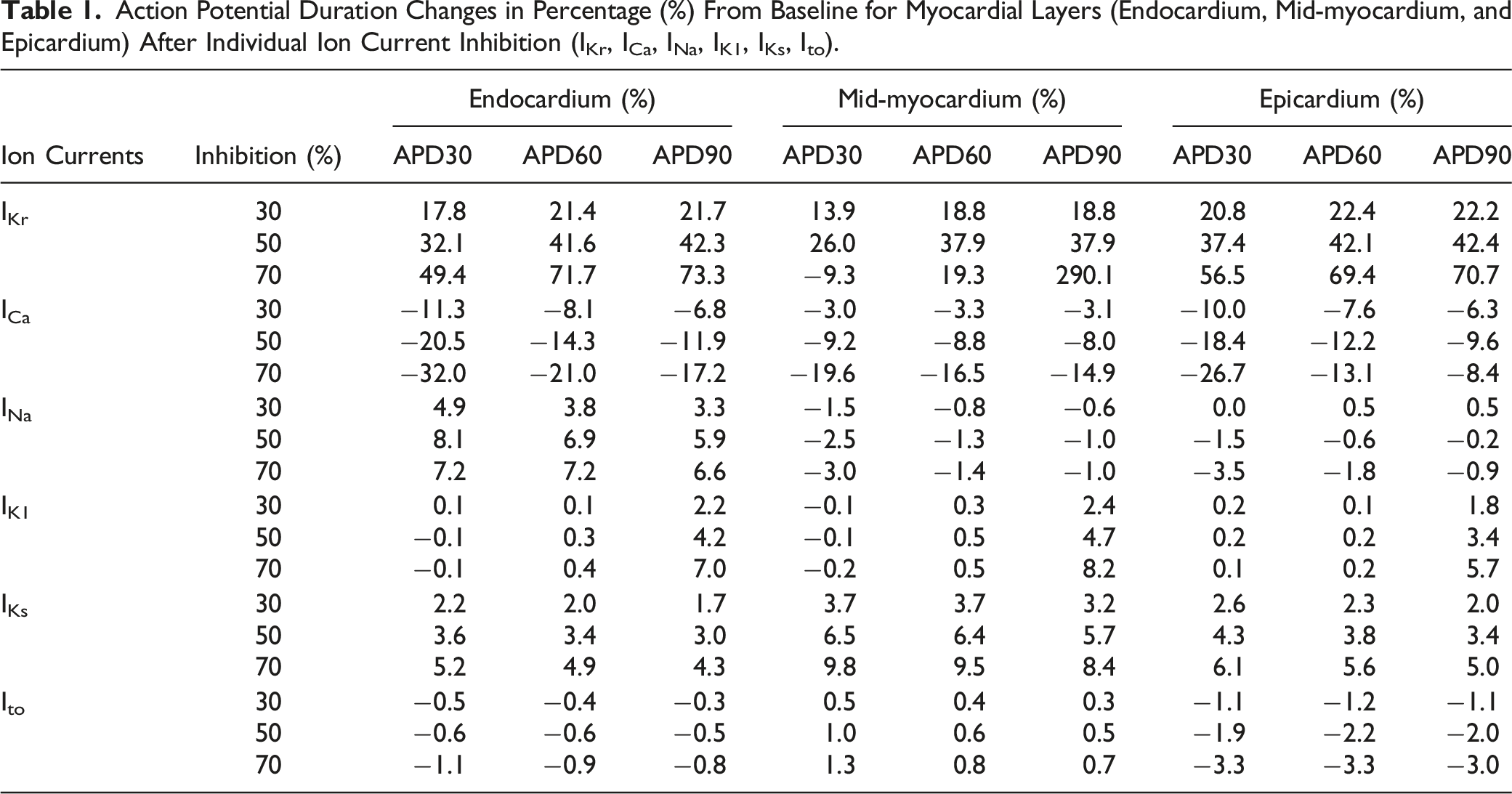
Simulations of single-channel inhibition are shown in Figure 1. The action potential for the myocardium with a progressive inhibition from 0 to 100% is represented for each individual channel. For IKr (Figure 1A), the repolarization duration increased with the progressive inhibition toward 100%. At 0% inhibition the end of repolarization was at 457 ms and for 60% inhibition the end of repolarization was at 645 ms. Repolarization duration values with inhibition greater than 60% (or ∼645 ms) triggered the development of an arrhythmia (EAD) and these are shown in Figure 1 with an APD simulation curve that did not return to the resting potential of −90 mV. For ICa (Figure 1B), at 0% inhibition, APD was 457 ms and APD decreased with increasing inhibition. At 90% inhibition, APD was 401 ms and for 100 % inhibition, the APD increased to 500 ms. The predicted inhibition effect on APD varied across different cardiac tissues layers. The magnitude of effect in absolute values for percent changes from baseline differed between myocardial layers and also between inhibition levels as illustrated for IKr (Table 1). For IKr, the effects on myocardial layers were greater in the following order (magnitude of effect) Mid > Endo > Epi (Table 1). For INa (Figure 1C), from 0 to 80 % inhibition, the APD from the peak to the resting potential was stable. Aberrant action potential waves appeared at 90 and 100% inhibition. For IK1 (Figure 1D), at 0% inhibition, APD was 457 ms and a progressive increase in APD was observed reaching 844 ms at 90 % inhibition. At 100% inhibition, return to the resting potential was very slow. For IKs (Figure 1E), APD increased from 457 ms at 0% inhibition to reach 500 ms at 100 % inhibition. Inhibition of Ito alone (Figure 1F) did not have any effect on the predicted APD. Complete action potential cycles with different inhibition percentages (0 to 100%), for cardiac ion channel currents derived from the mid myocardial layer. ( Action Potential Duration Changes in Percentage (%) From Baseline for Myocardial Layers (Endocardium, Mid-myocardium, and Epicardium) After Individual Ion Current Inhibition (IKr, ICa, INa, IK1, IKs, Ito).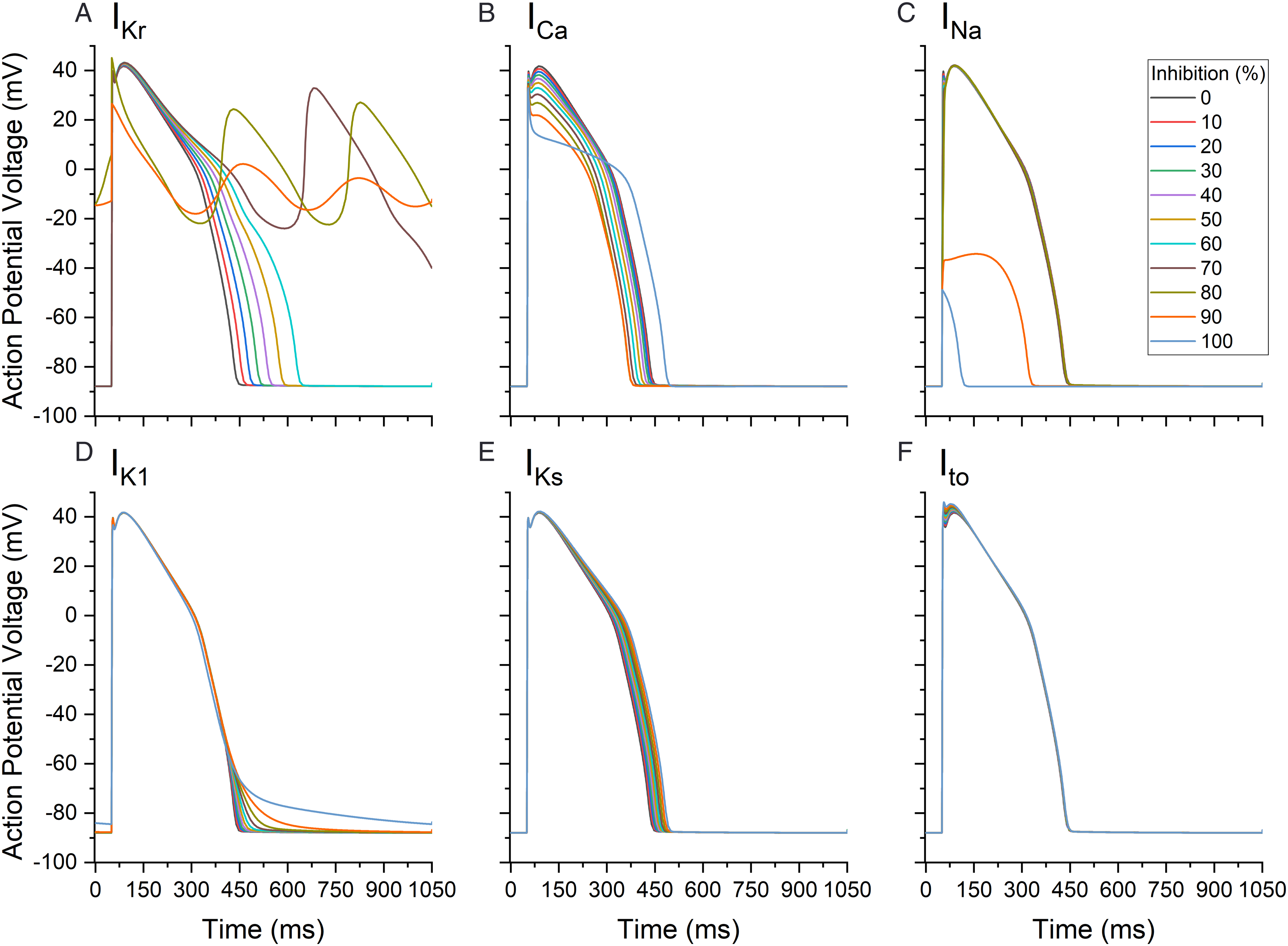
Progressive Inhibition of Multiple Ion Channels
Multiple channel inhibition simulations were performed as seen in Figure 2. At 0% inhibition for all 6 channels or all combinations (IKr, ICa, INa, IK1, IKs, Ito), the APD was 457 ms (Figure 2A). With 80% inhibition for all channels, APD was prolonged to 1034 ms and from 90 to 100% inhibition, the action potentials were aberrant in nature. The action potential for Figure 2A at inhibition of 90 to 100% had a low amplitude that decrease with higher inhibition, apart from the aberrant action potential no arrhythmia was detected. When the 3 channels that are most frequently tested during drug development were modulated (i.e., IKr, ICa, INa in Figure 2B), at 80% inhibition, APD was increased to 680 ms. Above 80% inhibition for these three currents, the action potential was aberrant, similar to the 6 channels mentioned above. With 80% inhibition for currents IKr, ICa, INa, IKs, the duration was 819 ms, (Figure 2C); however, when inhibition was above 80% no action potential was detected. For a combination of currents less often included in drug development, namely, IKr, IKs, IK1, and Ito, the APD at 50% inhibition increased to 711 ms and an aberrant action potential was generated at 60% inhibition and above (Figure 2D). When only IKr and INa were included at 60% inhibition, APD was increased to 676 ms but an aberrant action potential was only generated at 70% inhibition and above (Figure 2E). When IKr and ICa inhibition was simulated, APD reached 928 ms at 100% inhibition (Figure 2F). Action potential cycles with different inhibition percentages (0 to 100%) for selected cardiac current combinations examined at the mid myocardial layer. (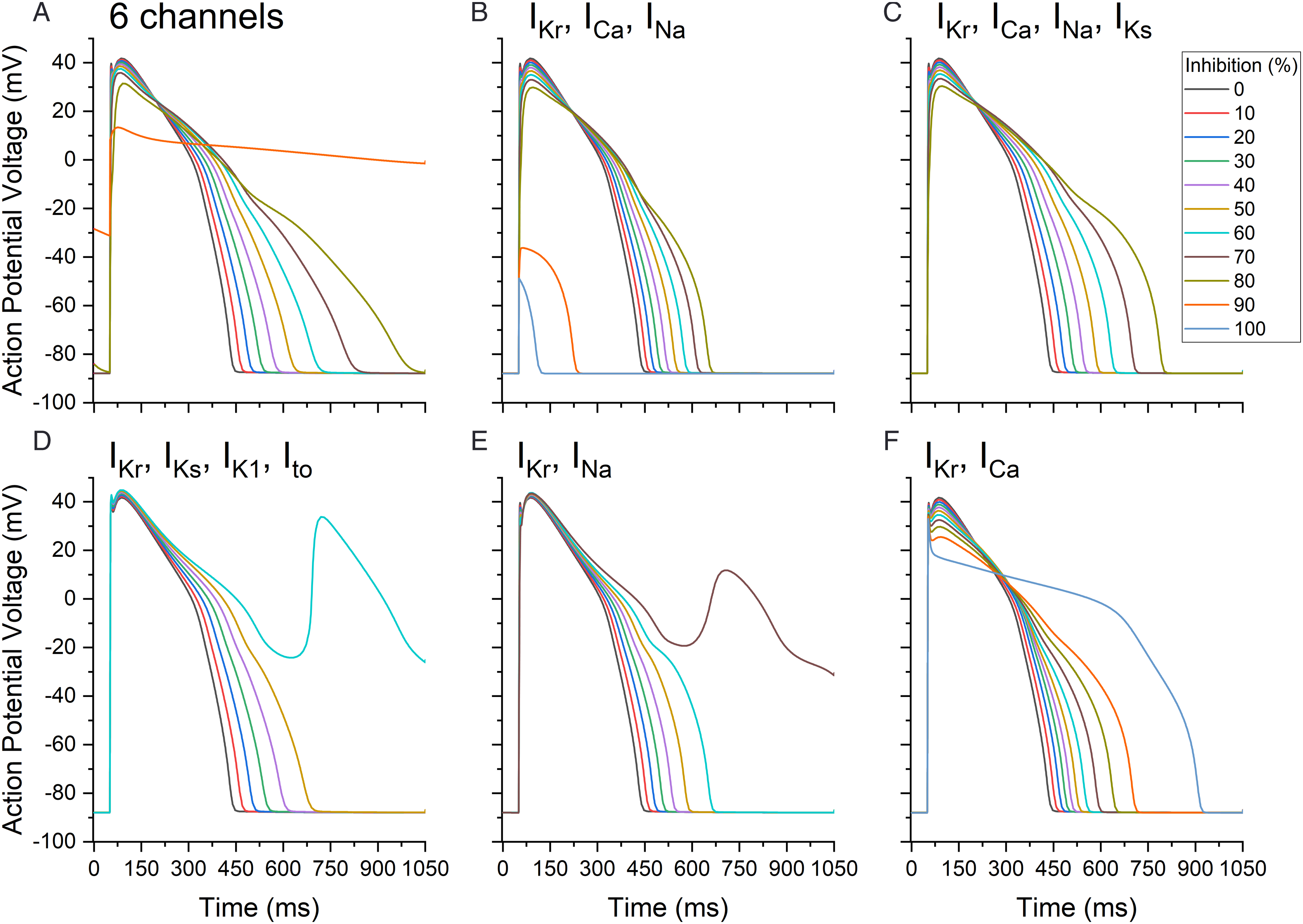
Triangulation and Action Potential Duration with the Progressive Inhibition of Multiple Ion Channel
The APDs at 30, 60, and 90% inhibition are represented in Figure 3A-3C for the different combinations of channels and in Figure 3D for the IKr current alone. The APD90 for the IKr inhibition was 575.3 ms, and when inhibition reached 70%, a predicted arrhythmia developed. The APD60 and APD90 values at 60% ion channel inhibition for IKr, ICa, and INa (Figure 3B), IKr, ICa, INa, and IKs (Figure 3C), and IKr (Figure 3D) were shorter in duration than the 6-channel combination (IKr, ICa, INa, IK1, IKs, Ito). At 80 % inhibition, APD for the 6-channel combination was increased (899.3 ms) and was longer than the other combinations: 594.8 ms for IKr, ICa, and INa (Figure 3B), and 734.3 ms for IKr, ICa, INa, and IKs (Figure 3C). Ion channel inhibition combinations and single-channel inhibition at different percentages (0 to 100%). (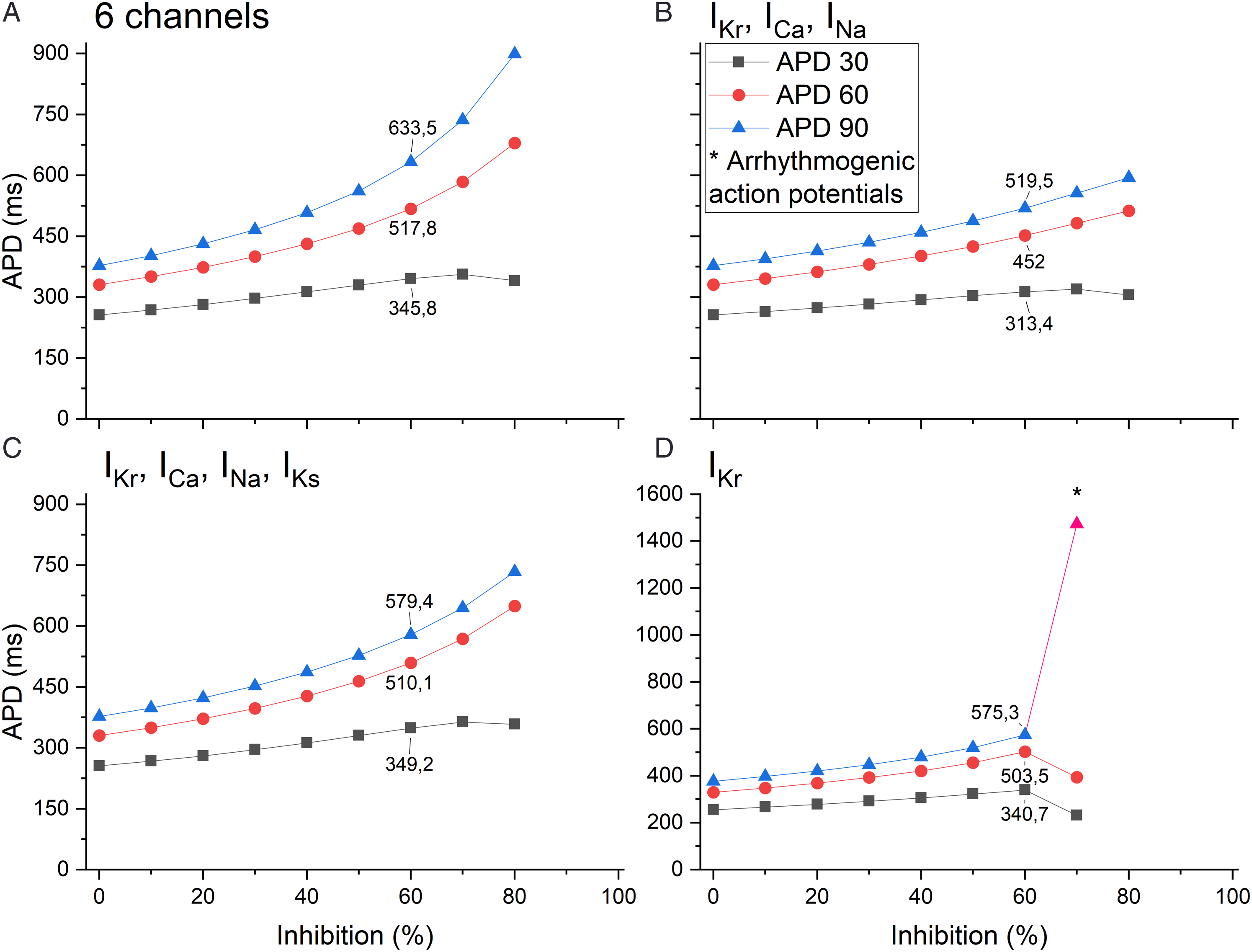
The APD90 at different inhibition percentages as well as new combinations of channels (IKr, ICa, INa, IK1; IKr, ICa, INa, Ito; IK1, IKs, Ito; and IKr, IK1, IKs, Ito) are presented in Figure 4. All combinations of channels had an increased APD as the inhibition percentage increased. The IKr alone and the IKr, IK1, IKs, Ito combinations had an arrhythmogenic action potential from 50% and 70% inhibition, respectively. As expected, the combination of IKr, IK1, IKs, and Ito potassium channels induced an increase in APD at a faster rate than other ion channel combinations or when compared to the 6-channel combination. At 40% inhibition of potassium channels, APD was at 614.2 ms compared to 508.2 ms for the 6-channel combination. Ion channel current inhibition (0 to 100%) for different combinations or single current using the action potential duration at 90%. The 6 channels include the following currents IKr, ICa, INa, IK1, IKs, Ito. The asterisk (*) represents an arrhythmia.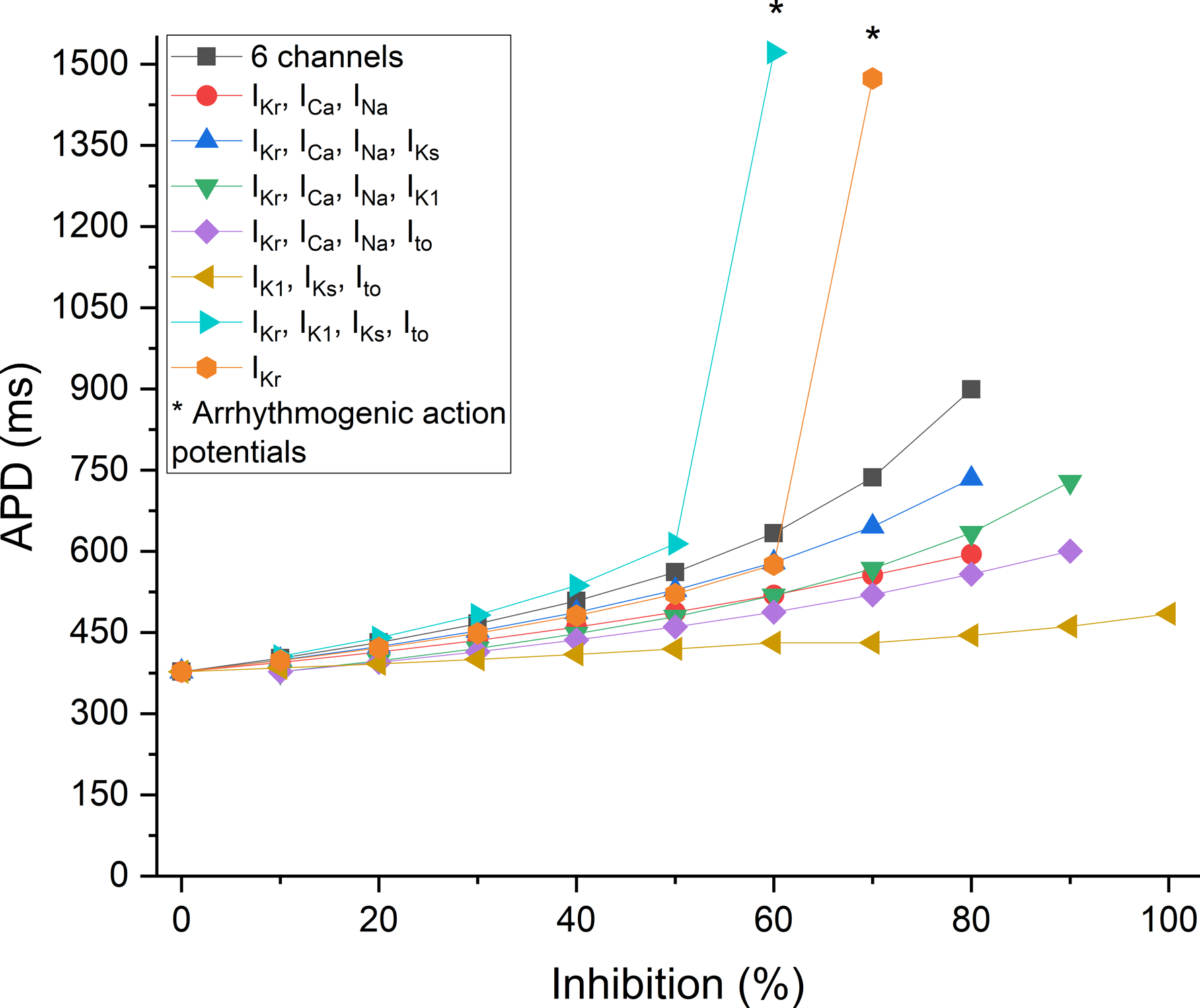
Finally, action potential triangulation (Figure 5) was calculated. Triangulation when considering the potassium channels only was qualitatively comparable to APD behavior as a function of inhibition percentage (Figure 4). Triangulation values for the combination of ion channel currents IKr, ICa, INa, IK1 increased at a lower percentage of inhibition than for IKr alone. The inhibition of IK1, IKs, Ito had the least impact on triangulation (121.2 ms at 0% inhibition and 188.3 at 100% inhibition). Ion channel inhibition (0 to 100%) combinations using triangulation (APD90 – APD30). The 6-channel combination is represented by the following currents: IKr, ICa, INa, IK1, IKs, Ito. The asterisk (*) represents an arrhythmia; those values are abnormal.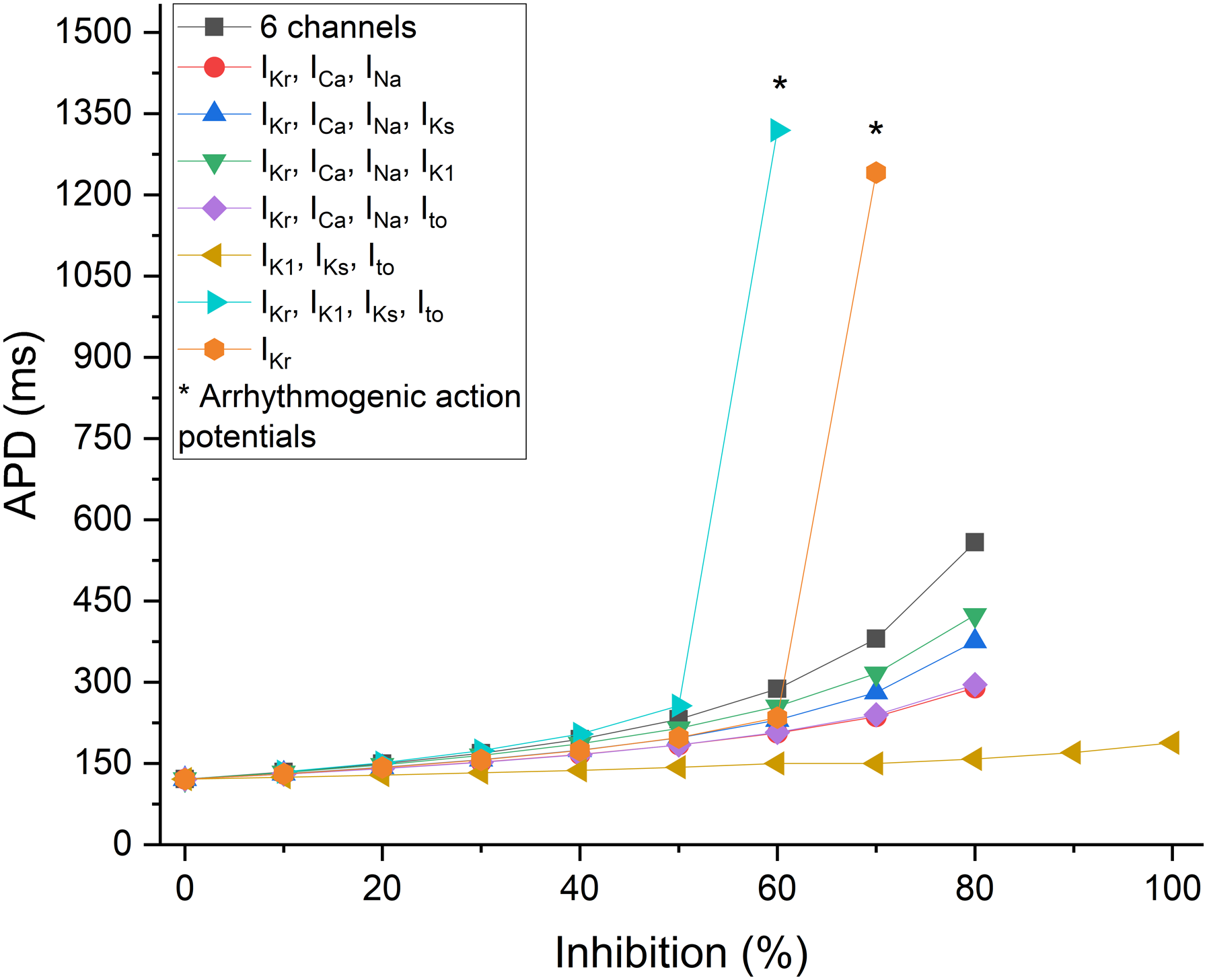
Evaluating Drug Effects and Action Potential Duration at 90% Repolarization Using the O’Hara-Rudy Model
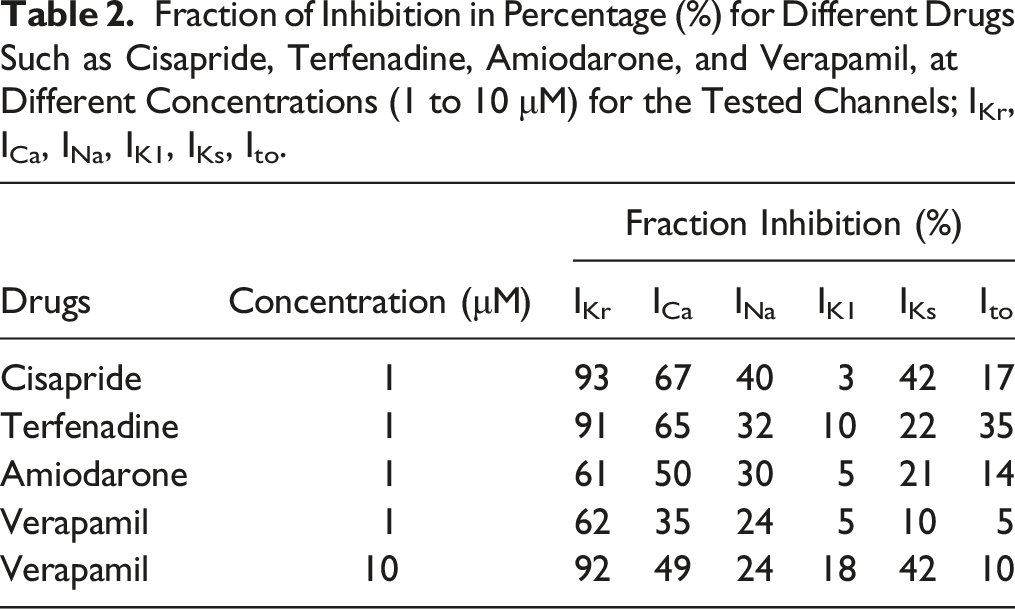
Fraction of Inhibition in Percentage (%) for Different Drugs Such as Cisapride, Terfenadine, Amiodarone, and Verapamil, at Different Concentrations (1 to 10 μM) for the Tested Channels; IKr, ICa, INa, IK1, IKs, Ito.
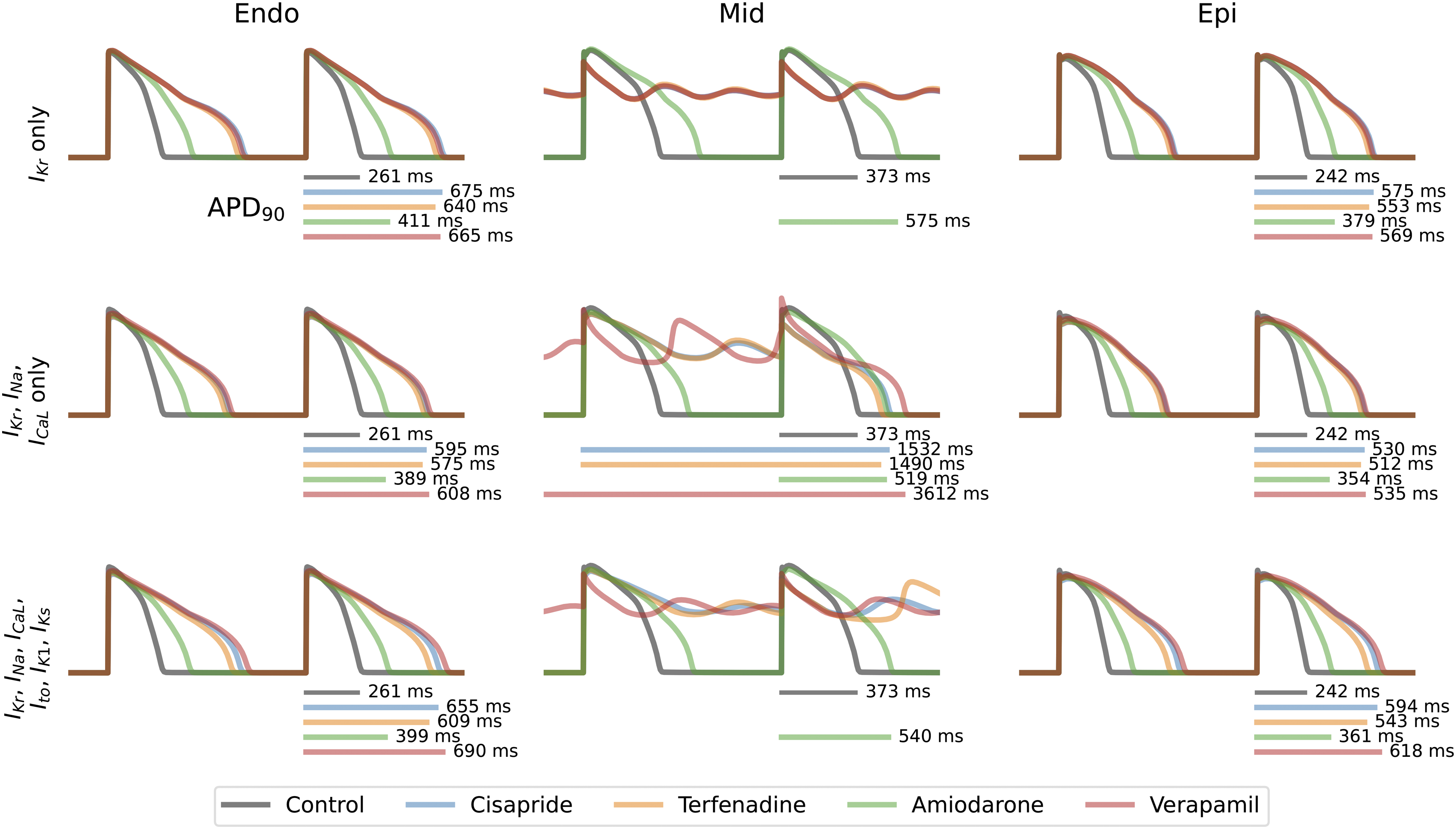
Ion channel inhibition combinations including IKr, ICa, INa, and IKr, ICa, INa, IK1, IKs, Ito, as well as the single-channel current IKr, in different layers of the heart (endo, mid, and epi) for the measurement of APD90. The control condition is depicted in gray, while cisapride (1 μM) is represented in blue, terfenadine (1 μM) in orange, amiodarone (1 μM) in green, and verapamil (10 μM) in red. The absence of a line for a drug indicates the presence of early afterdepolarization, which can lead to the development of ventricular arrhythmias.
Investigating the effects of different combinations of ion channel currents (IKr, ICa, INa and IKr, ICa, INa, IK1, IKs, Ito) on the endo and epi layers, exhibited similar increases in APD with multiple drugs. For the first combination, verapamil induced APD prolongation reaching 608 ms and 535 ms in the endo and epi layers, respectively. In the second combination, the increase was even more pronounced with values of 690 ms and 618 ms. Cisapride showed comparable increases in both the first combination (595 ms and 530 ms) and the second combination (655 ms and 594 ms). Terfenadine yielded similar effects as in the first combination (575 ms and 512 ms) and for the second combination (609 ms and 543 ms). Finally, amiodarone had the lowest impact of change in the APD, resulting in an increase of 389 ms and 354 ms for the first combination, and 399 ms and 361 ms for the second combination.
The mid layers for IKr, ICa, and INa ion channel combination exhibited severe APD90 prolongation in all the drugs tested, with verapamil showing the greatest increase of all drugs. When tested, verapamil prolonged the APD90 to a duration of 3612 ms. Cisapride and terfenadine showed similar APD90 increases (1532 ms and 1490 ms), respectively. In contrast, amiodarone showed a limited increase compared to other drugs with a value of 519 ms. As for the IKr, ICa, INa, IK1, IKs, Ito ion channel current combination only amiodarone resulted in a prolonged APD90 of 540 ms. However, the other drugs tested induced EADs and resulted in development of ventricular arrhythmias.
When using inhibition data from IKr, ICa, and INa, a higher increase in APD90 was noted for the endo and epi layers compared to in silico modeling using input only from the IKr channel. APD90 modeled from data obtained from IKr, ICa, INa, IK1, IKs, and Ito resulted in a larger increase in APD90 for the same layers compared to the aforementioned combination.
In contrast, for the mid myocardial layer, using input from a combination that includes IKr, ICa, INa showed a much more pronounced prolongation of APD90 compared to the 6-ion channel combination. These observations are consistent with the predicted outcome depicted in Figure 6, particularly in the middle panel representing the mid myocardial layers.
Discussion
Early in vitro screening for IKr (hERG) blocking effects is commonly conducted for small molecules to help identify any potential prolongation of the QT interval which can be associated with the development of TdP.23,24 When ICH S7B was first published in 2005, IKr was the only in vitro ion channel assessment required 15 ; however, subsequent guidance documents that have been released include considerations for the testing of the L-type calcium (ICa) and late sodium channels (INa).17,25 In silico modeling offers an opportunity to integrate in vitro data from individual ion channels into a single outcome to assess proarrhythmic risks 26 but concerns related to the value of in silico results obtained from a subset of ion channels have historically limited the use of this analysis in drug development. In this manuscript, we illustrated the use and relative value of in silico modeling using inhibition data from a limited number of ion channels.
The CiPA initiative was established to develop an innovative paradigm that could be used to assess proarrhythmic risk. As a regulatory based initiative, CiPA was developed to standardize and expand the safety testing strategies related to drug-induced delayed ventricular depolarization and eventually TdP. CiPA was innovative as it utilized new technologies and an expanded understanding of TdP mechanisms that extended beyond hERG (IKr) block alone. The four components of CiPA are (1) an in vitro assessment of a drug’s effects on multiple ion currents, (2) in silico modeling to assess the proarrhythmic risk, (3) in vitro effect on human stem cell derived ventricular cardiomyocytes, and finally (4) in vivo electrocardiogram biomarkers in phase I clinical trials.4,27 While components (1), (3), and (4) have been broadly discussed in scientific publications and within industry forums, there is limited information on the implementation of in silico testing strategies in drug development. This situation may be related to the role that hERG has played as a predominant mechanism for the development of drug-induced ventricular arrhythmias over in the last three decades. Indeed, hERG is the cardiac channel that is most often blocked by small chemical pharmaceutical agents, followed by Nav 1.5-late and Cav 1.2.14,28,29 Furthermore, drugs that block Cav 1.2 or Nav 1.5-late calcium, sodium channels at equal or greater concentrations required to block hERG will reduce the risk for the development of TdP arrhythmias.26,28,30 The drug development industry has deployed considerable efforts to avoid the clinical development of compounds that block any cardiac ion channel, but specifically to avoid any risk for the blockade of hERG (IKr) and resulting TdP liability. The in silico data presented herein highlights that only the combination of all the potassium channels tested (IKr, IK1, IKs, and Ito) results in the development of ventricular arrhythmia at an ion channel inhibition level that is lower than the IKr alone. The progressive inhibition for the 6-channel combination was the most resilient to arrhythmia development even though it had the longest APDs determined in this study. Combined blockade of these depolarizing ion channels in the heart reduces the potential for EAD generation26,30,31 and proarrhythmic risk (TdP) in the presence of a prolonged APD.
Triangulation is the repolarization time from APD30 to APD90 and it can be used as a biomarker of proarrhythmia. 32 This repolarization period creates a window during which EADs can occur. Blocking multiple channels in cardiomyocytes such as sodium, calcium and potassium channels can prevent the initiation of arrythmia that would be observed when selectively blocking the hERG channel. 33 Inhibition of IKr increases triangulation and the risk for arrhythmia. It has been previously shown that the inhibition of IKr and IKs was more arrhythmogenic than IKr channel inhibition alone 34 but the evaluation of drug effects on IKs is not generally included during early drug safety profiling. The fundamental rationale from quantifying QTc on ECG is related to its correlation with Phase 3 of the action potential when EADs can occur. When multi-channel inhibition is present or even when selective hERG block is identified, in silico modeling can be used to assess triangulation as an early indication of the potential for a new chemical compound to exhibit proarrhythmic risk.
The differences in APD90 prolongation threshold prior to predicting proarrhythmic effects between selective IKr and multi-ion channel blocking effects highlight the limitations of using in silico APD90 as a primary biomarker for quantifying the proarrhythmic risk of a compound. 7 The magnitude of APD90 prolongation alone does not fully inform on the proarrhythmic risk predicted by the in silico model. While most pharmacologists aim to develop safe compounds that will not inhibit cardiac ion channels, there are still several compounds under development that exhibit various profiles, ranging from selective to multi-ion channel inhibitory effects.
The early electrophysiological screening of pharmaceutical compounds often includes a limited cardiac ion channel panel usually comprised of IKr, ICa, and INa only.15-17 Selection of partial patch clamp panels is primarily driven by considerations related to probability and affordability. The hERG potassium channel is widely acknowledged for its notable pharmacological promiscuity, making it a pharmacological target that is frequently encountered by safety pharmacologists. 35 The hERG channel’s ability to engage with a diverse array of medications can be attributed to its distinctive ligand-binding site configuration, its hydrophobic nature, and the large channel vestibule.36,37 It had been estimated that up to 60% of drugs in development exhibit some level of hERG block.38,39 Although sodium and calcium channel blocks are less common, they still arise with sufficient frequency. In contrast, compounds that block the additional ion channels that are commonly involved with in silico modeling (i.e., IK1, IKs, and Ito) are relatively scarce. The above probabilities often direct the ion channel selection for early stage in vitro testing.
When the inhibition results justify further assessments and evaluation, in silico modeling of drug effects using these three ionic currents can provide a simple, yet relevant, analysis method that allows for the use of individual ion channel data in an integrated risk evaluation, as the marginal contribution to ADP90 values from the other three currents (i.e., Ito, IKs, and IK1) remains minor when taken individually. The presented results underscore the cumulative advantage of incorporating all six ion currents included in CiPA into in silico modeling compared to a partial panel. In silico models represent a theoretical aggregated response to compound effects on the APD and can describe the potential for development of proarrhythmic effects. Proceeding with in silico modeling using only the three primary cardiac ion currents commonly tested in early development only reveals part of the value provided by in silico modeling but can still contribute to an integrated risk assessment prior to the conduct of more complicated in vitro (cardiac left ventricular wedge preparation) or in vivo (telemetry dog study) cardiovascular assays. However, as has been shown, in silico modeling with IKr, ICa, and INa may overestimate compound safety, since ICa and INa counterbalance the APD prolonging effects of IKr. Additional potentially exacerbating cardiac currents (i.e., Ito, IKs, and IK1) are not usually included. As we know, IKr still provides the most critical contribution to ventricular repolarization and proarrhythmic risk and should remain the cornerstone of drug-induced TdP testing.
Novel in silico models have been developed to address some of the limitations of the O’Hara-Rudy model including the balance of ion channels. 40 In silico models have been reported to show a degree of prolongation and arrhythmia occurrence in excess of what is observed in in vitro tissues and QTc. 41 The current manuscript used 10% inhibition increments which represents a potential limitation of the analysis presented. More granular simulations with 1% inhibition increments may be valuable in the future when considering scenarios with drugs that are close to balanced blockers. In addition, some compounds have direct or indirect effect heart rate and in silico modeling at different cycle length may represent another opportunity to develop future testing paradigms early in drug development as in vitro assays may already inform on potential chronotropic effects of tested compounds.
The context of use for the in silico model is its application in the scenario of drug development. One of the principal components is to evaluate the TdP risk.
TdP, an infrequent form of ventricular tachycardia, manifests as gradual alterations in QRS complexes on an electrocardiogram, accompanied by QTc prolongation. This interval prolongation of the QTc is associated with the potential for sudden cardiac death. The lengthening of repolarization induced by drugs elevates the risk of critical arrhythmias like TdP. No specific limit has been established for QTc prolongation to cause TdP, having a QTc exceeding 500 milliseconds amplifies the risk by two to three times, with a 5% to 7% escalation for each 10-ms increase. Numerous drugs have the capacity to induce QT prolongation and/or TdP, either independently or in situations involving drug interactions. 42
In drug development, the potency for hERG block is typically used for early screening of candidate drugs in development to mitigate the risk of QT prolongation. The IC50 values for hERG block are compared to predict or measure maximal plasma drug concentrations (Cmax). This can allow for the calculation of a predictive value (IC50/Cmax), referred to as the hERG safety margin. 43
TQT studies are recognized for their good specificity (low false-positive rate) but relatively poor sensitivity (higher false-negative rate), indicating their capability to accurately identify drugs that do not have a significant effect on the QTc interval (low false-positive rate).44,45
The exclusion of a positive compound is often associated with its role as a blocker of the hERG channel. There are exceptions, such as hERG inhibitors that do not induce Torsades de Pointes (TdP), and drugs that may cause TdP without being potent hERG inhibitors. On the other hand, a multi-ion channel blocker has the ability to inhibit the hERG channel. Because of its simultaneous impact on sodium and calcium channels, it may lead to a less pronounced increase in the QTc interval. In vitro studies often encounter challenges in accurately evaluating the impact of drugs acting as multi-ion channel blockers. 46
Conclusions
Our results confirm that manual patch clamp inhibition profiles can provide high quality input for use in in silico modeling and quantitatively predicts cardiomyocyte APD changes and consequently QT prolongation. The O’Hara-Rudy model, using the six cardiac ion currents proposed for CiPA, predicted changes in cardiac APD and consequently, ECG effects. The use of a partial ion channel panel (i.e., IKr only or IKr, ICa, INa only) illustrates the primary contribution of IKr in APD estimation with this model.
These findings emphasize the importance of considering the specific combination of ion channels that should be evaluated and the effects they have on the different layers of the heart when evaluating the effects of drugs on APD. The interactions between various channels and cardiomyocyte layers play a crucial role in determining the overall electrophysiological behavior of the heart and highlight the complexity of ventricular arrhythmia mechanisms with various contributions from relevant ion currents as shown by the data herein.
Manual patch clamp data obtained during drug development is readily available for use with in silico modeling methods and can be considered to generate pharmacologically relevant APD data even when only a subset of the cardiac ion channels are tested. While the O’Hara-Rudy in silico model, using the 6 most important cardiac ion currents (i.e., IKr, ICa, INa, IK1, IKs, Ito), yielded the expected results relative to expected drug effects, in silico modeling with data from a combination of channels or even a single channel (i.e., IKr) provides valuable results that can be used to estimate the risk of drug-induced ventricular proarrhythmia.
Footnotes
Author Contributions
Emmanuel Boulay contributed to conception and design, contributed to analysis and interpretation, and drafted manuscript; Eric Troncy contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Vincent Jacquemet contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Hai Huang contributed to design, contributed to acquisition and interpretation, and critically revised manuscript; Michael Pugsley contributed to design, contributed to interpretation, and critically revised manuscript; Anne-Marie Downey contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Rafael Venegas Baca contributed to acquisition and analysis and critically revised manuscript; Simon Authier contributed to conception and design, contributed to analysis and interpretation, and drafted manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
