Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 8 palm tree (Euterpe edulis (juçara) and Euterpe oleracea (açaí))-derived ingredients as used in cosmetic products; these ingredients are reported to function mostly as skin conditioning agents. The Panel reviewed relevant data relating to the safety of these ingredients in cosmetic formulations. Industry should continue to use good manufacturing practices to limit impurities. The Panel concluded that palm tree (açaí and juçara)-derived ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction

Definitions and Functions of the Ingredients in This Safety Assessment. 1

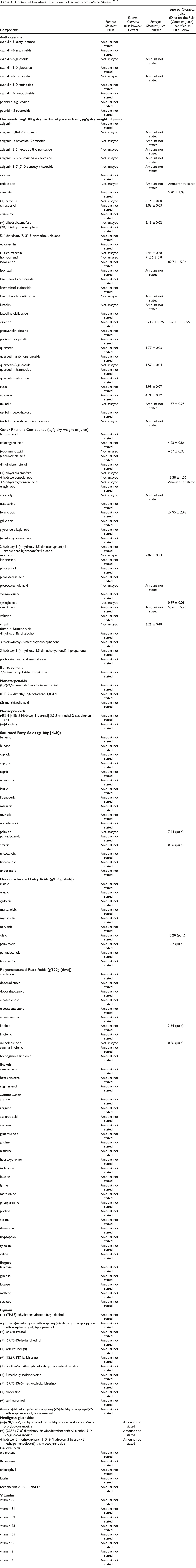
The ingredient group that is being reviewed in this safety assessment (Euterpe oleracea (açaí)- and Euterpe edulis (juçara)-derived ingredients) was formed based on the supposition that ingredients from a given genus and species, and on a closely related species (i.e., edulis and oleracea), would have constituents in common. For example, both species have the following constituents in common: catechin, chlorogenic acid, cyanidin-3-glucoside, cyanidin-3-rutinoside, ellagic acid, ferulic acid, gallic acid, pelargonidin-3-glucoside, and peonidin-3-rutinoside.2-14
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/ supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Botanicals, such as Euterpe edulis- or Euterpe oleracea-derived ingredients, may contain hundreds of constituents. In this assessment, the Panel reviewed the potential toxicity of each of the botanical ingredients as a whole, complex mixture. The Panel did not review the potential toxicity of the individual constituents.
Because the safety of Euterpe oleracea-derived ingredients is being reviewed in this safety assessment, it should be noted that the Panel published a safety assessment on Euterpe Oleracea Fruit Oil (and other plant-derived fatty acid oils) in 2017. 15 Based on the available data, the Panel concluded that the ingredients included in that report are safe in the present practices of use and concentration described in the safety assessment. The full report on these ingredients can be accessed on CIR website (https://www.cir-safety.org/ingredients).
Given some similarities in composition (based on the available data) between different plant parts of Euterpe oleracea, data on plant parts that are not identified in the names of the cosmetic ingredients that are being reviewed in this safety assessment are included. Additionally, although Euterpe edulis fruit oil was reviewed previously (and therefore is not among cosmetic ingredients being reviewed), data on the oil are also included to supplement the data in this report.
It is often not known how the substance being tested in a study compares to the ingredient that is being used in cosmetics. In the report text, if it is known that the material being tested is a cosmetic ingredient, the Dictionary naming convention will be used (i.e., the names of cosmetic ingredients are capitalized, without italics (e.g., Euterpe Edulis Fruit Extract)). If it is not known that the test substance is the same as the cosmetic ingredient, then the taxonomic naming conventions will be used (i.e., with genus and species name, italicized (e.g., a Euterpe edulis fruit extract)).
Chemistry
Definition and General Characterization
The definitions and reported functions in cosmetics of these ingredients, according to the Dictionary, are presented in Table 1. 1 The Euterpe oleracea-derived ingredients are associated with two generic CAS numbers, i.e., 879496-95-4 and 906351-38-0; CAS numbers are not identified for the Euterpe edulis-derived ingredients.
Generic Plant Part Definitions as They Apply to Palm Tree-Derived Ingredients. 1

Method of Manufacture
Euterpe Oleracea Fruit Extract
The method of manufacture for a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) provided by a supplier is as follows: 20 Euterpe oleracea fruit is processed (mechanical grinding/milling). This process is followed by aqueous extraction (at specific pH and temperature) for a specified duration. The aqueous fruit extract is then subjected to tangential flow filtration to isolate the desired components. Addition of Lactobacillus ferment is the next step, and batch adjustments are made if needed (refiltration). A sample is then subjected to quality control, after which the material is packed and sampled for microbiological analysis prior to shipment.
Euterpe Oleracea Juice
According to one manufacturer of a Euterpe oleracea juice, for use in foods, this juice is obtained by cold pressing the thin pulp of the ovoidal fruit (berry) of Euterpe oleracea Mart. 21
The method of manufacture for Euterpe Oleracea Juice (undiluted, freeze dried), provided by a supplier, is as follows: 22 Euterpe oleracea is cold-pressed for juice. This process is followed by filtration to remove unnecessary plant matter. The filtrate is then freeze dried, and batch adjustments are made, if necessary. A sample is then subjected to quality control, after which the material is packed. The packed material is then sampled for microbiological analysis prior to shipment, and it is reconstituted with water for use.
Euterpe Oleracea Pulp Powder
In one production method, the fruit pulp obtained from Euterpe oleracea fruit harvested in Brazil was frozen. 23 Samples of spray-dried pulp were obtained using an industrial scale spray dryer system and anionic maltodextrin DE10 was used as a carrier agent.
Composition/Impurities
Euterpe Edulis Fruit Extract
Composition Data on Euterpe Edulis Fruit Extract (Various Extractants). 24

*The chemical constituents of the extracts were identified by comparing their retention indices and making computer matches with the National Institute of Standards and Technology library provided by the computer controlling the gas chromatography-mass spectrometry system.
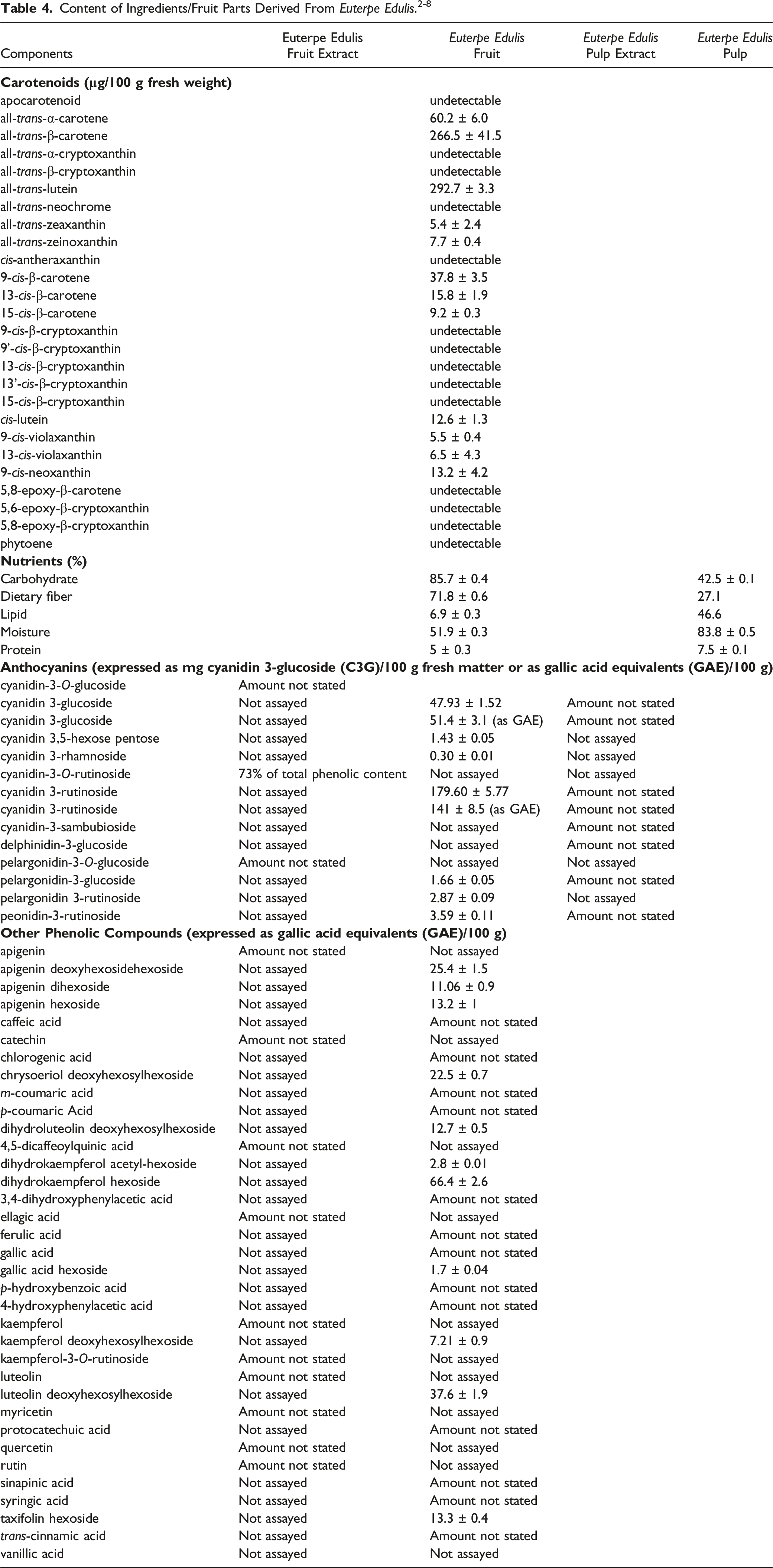
According to research investigating the major anthocyanins (type of flavonoid) and non-anthocyanin phenolic compounds in a Euterpe edulis fruit extract, high amounts of anthocyanins, approximately 26 mg/g dry weight basis (dwb), of a total of 31 mg/g dwb of phenolic compounds, were detected. 2 Cyanidin-3-O-rutinoside was the most abundant anthocyanin (73% of the total phenolic content). It should be noted that an analysis of Euterpe edulis fruit for phenolics yielded a value of 4087 mg/100 g dwb for soluble phenolics in pulp from fruits collected in southeastern Brazil. 3 However, a lower value of 1695 mg/100 g dwb for soluble phenolics in this fruit (from Minas Gerais State, a state in the north of Southeastern Brazil) has also been reported. 4 Furthermore, Euterpe edulis fruit is rich in oleic and palmitic fatty acids. 16
Euterpe Edulis Fruit Extract and Euterpe Edulis Juice Extract
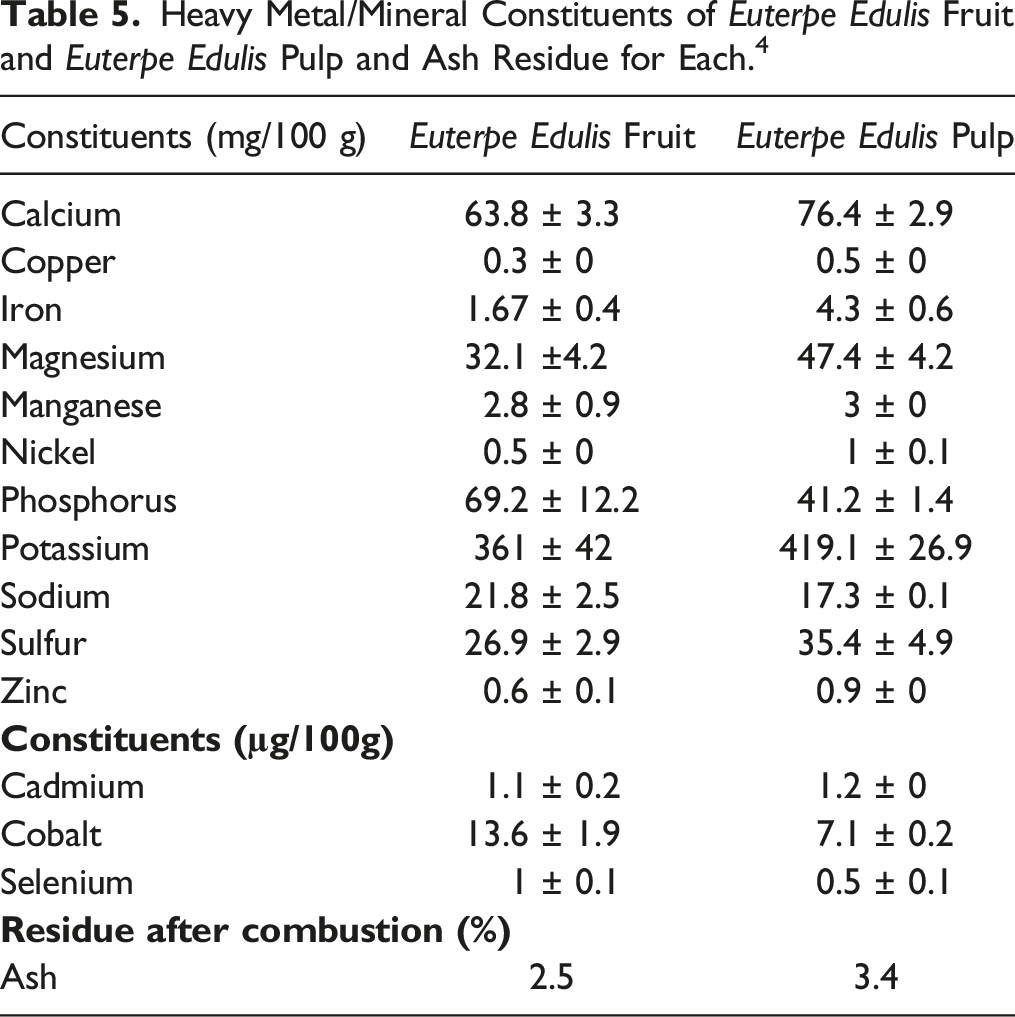
Heavy Metal/Mineral Constituents of Euterpe Edulis Fruit and Euterpe Edulis Pulp and Ash Residue for Each. 4
Euterpe Oleracea Fruit Extract
The heavy metals content of Euterpe Oleracea Fruit Extract (powder) has been described as follows: arsenic (<0.1 ppm), cadmium (<0.01 ppm), mercury (<0.005 ppm), lead (<0.05 ppm), and copper (0.3 ppm). 25 A supplier reported that a Euterpe Oleracea Fruit Extract trade name mixture contains 98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment. 14 This supplier has certified that this product does not contain the 26 allergenic chemical substances that are restricted by the European Union, nor does it contain pesticides exceeding the limitations established by the US Environmental Protection Agency.14,26 Heavy metals, lead, arsenic, microbial content, yeast and mold, and gram negative bacteria are below detection limits.
Euterpe Oleracea Fruit Extract (test material Euterpe oleracea fruit)
Euterpe oleracea Martius, as a native fruit of the Amazon rainforest, has been described as highly contaminated in microbiological terms. 17 The fruit is said to be subject to natural microbiological contamination and one of the main sources of this contamination is water, considering that more than 50% of the municipalities located in the Brazilian Amazon do not use chlorinated water. Euterpe oleracea fruit from Brazil and the United States (US) were analyzed for 174 different pesticides, using liquid chromatography-tandem mass spectrometry (LC-MS/MS) and gas chromatography-tandem mass spectrometry (GC-MS/MS). 27 Euterpe oleracea fruit that was harvested and lyophilized in Brazil had no detectable pesticides. There also were no detectable pesticides in 7 out of 12 samples of Euterpe oleracea fruit in the US. However, the following pesticides were detected in the 5 other samples of Euterpe oleracea fruit in the US: methoxyfenozide (0.2 ng/g), metalaxyl (0.2 ng/g), boscalid (2.6–3 ng/g), imidacloprid (0.9 ng/g), bifenazate (1.6–2.5 ng/g), carbendazim (0.9 ng/g), hexythiazox (0.6 ng/g), and pyraclostrobin (0.1 ng/g).
The following heavy metals have been detected in Euterpe oleracea fruit: lead, cadmium, mercury, and arsenic. 10 Additionally, the following elements have been detected in Euterpe oleracea fruit: potassium, magnesium, phosphorus, calcium, sodium, zinc, iron, and copper. Ash residue in the amount of 1.68 ± 0 g/100 g (dwb) remained after the combustion of Euterpe oleracea fruit. 28
Euterpe Oleracea Fruit Extract and Euterpe Oleracea Juice
*dwb = dry weight basis.
According to a supplier’s specification for a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment), the ferulic acid content ranges from 4% to 5%. This material is a clear to slightly hazy liquid. 14
Euterpe Oleracea Seed Powder
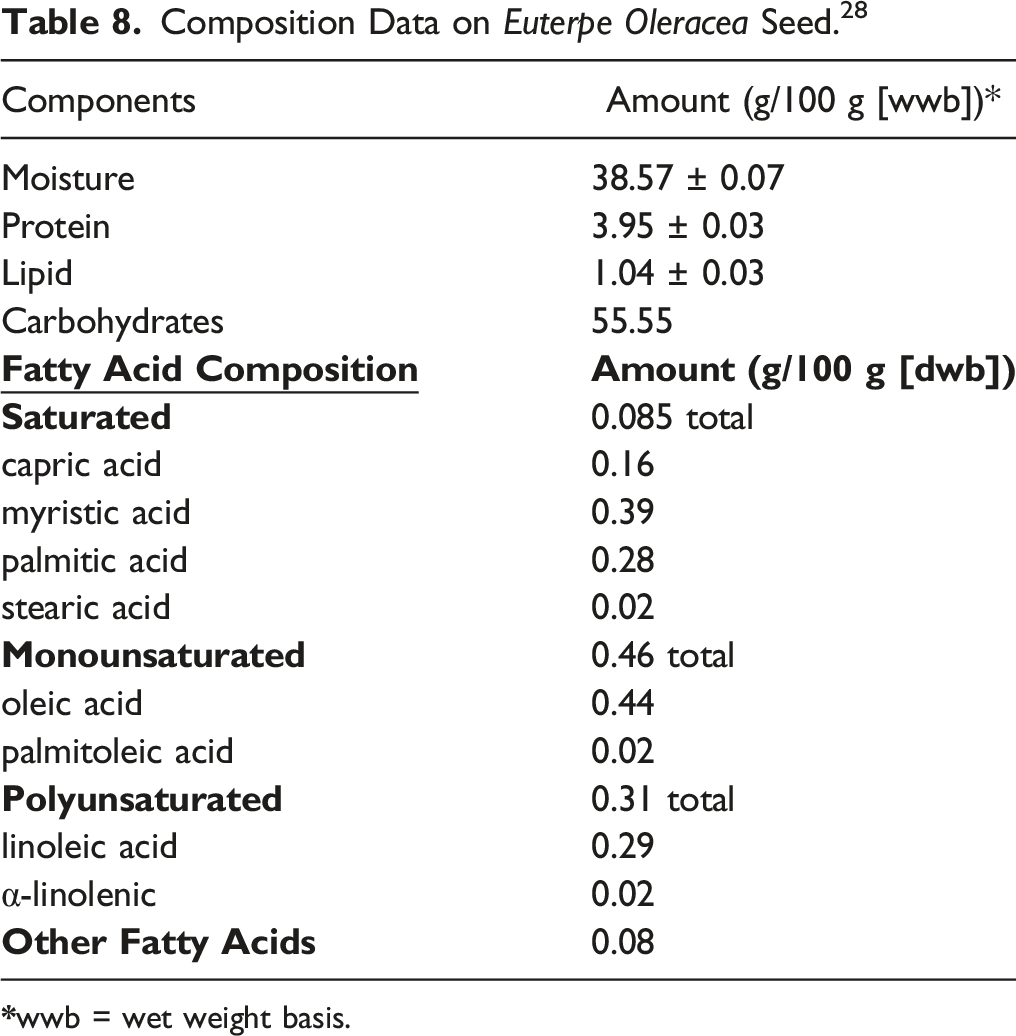
Composition Data on Euterpe Oleracea Seed. 28
Euterpe Oleracea Pulp Powder (Euterpe oleracea pulp)
Ash residue in the amount of 3.78 ± 0.06 g/100 g (dwb) remained after the combustion of Euterpe oleracea pulp. 28
Euterpe Oleracea Seed Powder (Euterpe oleracea seed)
Ash residue in the amount of 1.44 ± 0.01 g/100 g (dwb) remained after the combustion of Euterpe oleracea seed. 28
Use
Cosmetic
The safety of palm tree-derived ingredients is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 31 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 32
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
NR = Not Reported.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
cNot specified that these products are sprays or powders, but it is possible the use can be as a spray or powder, therefore the information is captured in both categories.
According to VCRP and Council survey data, the following 3 ingredients are not being used in cosmetic products: Euterpe Edulis Fruit Extract, Euterpe Edulis Juice Extract, and Euterpe Oleracea Seed Powder.
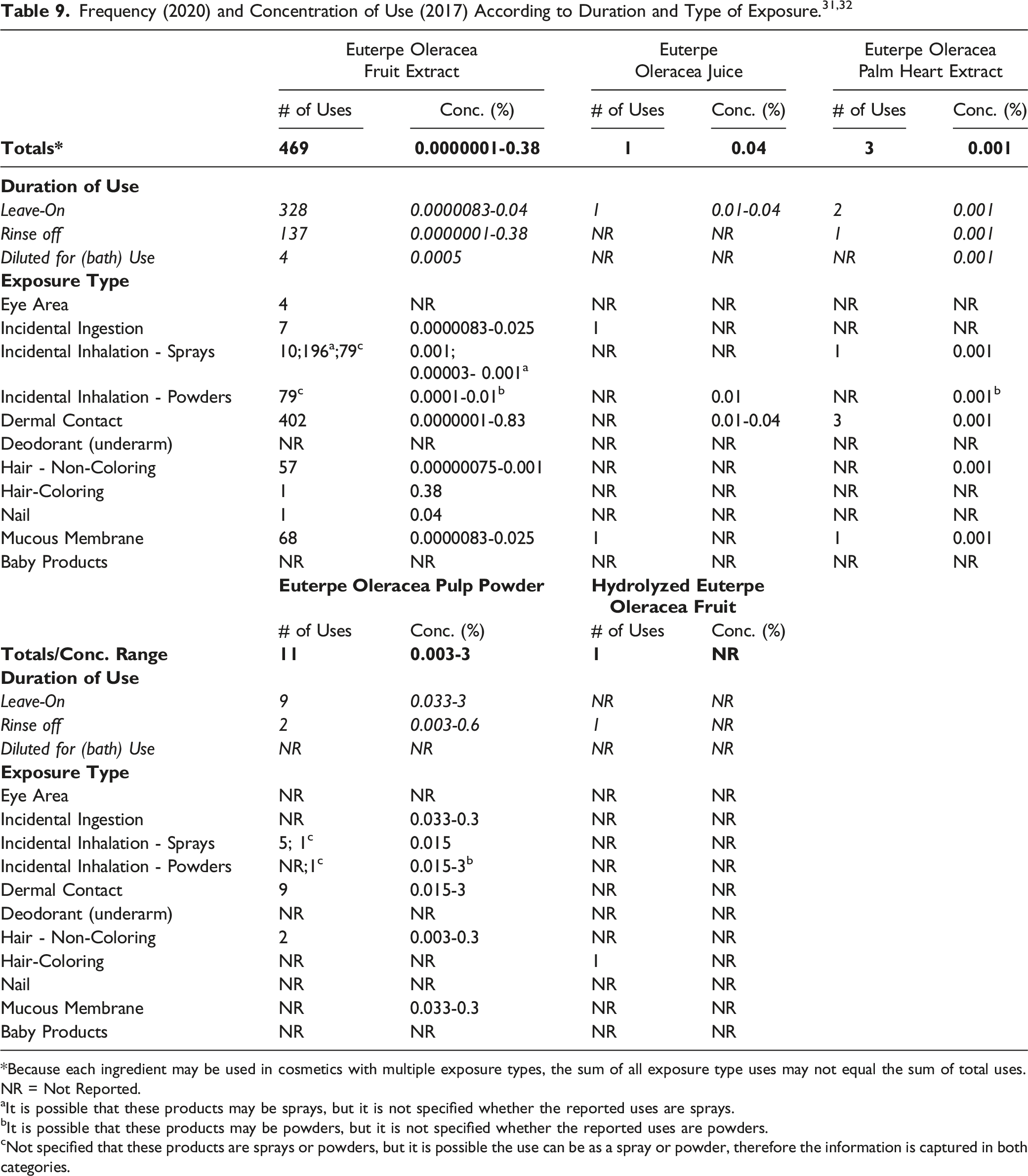
Cosmetic products containing palm tree-derived ingredients may be applied to the skin or, incidentally, may come in contact with the eyes (e.g., Euterpe Oleracea Fruit Extract). Euterpe Oleracea Fruit Extract, Euterpe Oleracea Juice, Euterpe Oleracea Palm Heart Extract, and Euterpe Oleracea Pulp Powder are ingredients that are used in products that come in contact with mucous membranes during product use (ingredient use concentrations: 0.0000083–0.3%). Additionally, Euterpe Oleracea Fruit Extract and Euterpe Oleracea Pulp Powder could be incidentally ingested (at maximum use concentrations up to 0.025% [lipstick] and 0.3% [lipstick], respectively). Products containing palm tree-derived ingredients may be applied as frequently as several times per day and may come in contact with the skin for variable periods following application. Daily or occasional use may extend over many years.
The following palm tree-derived ingredients are being used in products that are sprayed: Euterpe Oleracea Fruit Extract (0.001% in pump hair spray), Euterpe Oleracea Palm Heart Extract (0.001% in colognes and toilet waters), and Euterpe Oleracea Pulp Powder (0.015% in colognes and toilet waters). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.33-36 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.33,34 The only use of palm tree-derived ingredients in powders is being reported for Euterpe Oleracea Juice, which is being used at concentrations up to 0.01% in face powders. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.37-39
The palm tree-derived ingredients reviewed in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 40
Non-Cosmetic Use
Euterpe oleracea extract is not the name of any of the ingredients that are being reviewed in this safety assessment, but has the same CAS number (879496-95-4) as the following ingredients that are being reviewed: Euterpe Oleracea Fruit, Euterpe Oleracea Palm Heart Extract, Euterpe Oleracea Pulp Powder, Euterpe Oleracea Pulp Powder, and Euterpe Oleracea Seed Powder Extract. However, it should be noted that Euterpe oleracea extract (also known as acai berry extract) is a food flavoring agent or adjuvant. 41 According to another source, Euterpe oleracea extract is a hydroalcoholic (ethanol and water) extract of Euterpe oleracea berry stones (pits). 42 Because the safety of Euterpe Oleracea Palm Heart Extract is being reviewed in this report, it is also important to note that heart of palm is the edible part of the apical meristem of palms (Euterpe oleracea and Euterpe edulis) and is considered a gourmet vegetable. 43 Furthermore, it should be noted that both Euterpe oleracea and Euterpe edulis fruits are typically consumed in the Amazon region of Brazil. 44
Toxicokinetic Studies
Dermal Penetration
Data on the dermal penetration of the palm tree-derived ingredients reviewed in this safety assessment were neither found in the published literature, nor were these data submitted. Dermal penetration data were not expected to be found because each botanical ingredient is a mixture of hundreds of constituents.
Absorption, Distribution, Metabolism, and Excretion
Human
Oral
Euterpe Oleracea Juice and Euterpe Oleracea Pulp Powder (test material Euterpe oleracea pulp)
An acute 4-way crossover clinical trial that involved oral dosing with the following was performed using 12 subjects: Euterpe Oleracea Juice, Euterpe oleracea pulp, applesauce (control), and a non-antioxidant beverage (control). 45 An oral dose of Euterpe Oleracea Juice or Euterpe oleracea pulp (7 mL/kg) was administered after a washout phase and overnight fast, and plasma was repeatedly sampled over 12 h. Urine was sampled over a 24-h period after dosing. Plasma anthocyanin (antioxidant) concentrations were determined over a period of 0–12 h. Noncompartmental pharmacokinetic analysis of total anthocyanins, quantified as cyanidin-3-O-glucoside, indicated maximum plasma concentration (Cmax) values of 2321 and 1138 ng/L at maximum concentration times (tmax) of 2.2 and 2.0 h, and area under the concentration-time curve (AUClast; last refers to AUC up to the last measurable concentration) values of 8568 and 3314 ng h/L for Euterpe oleracea pulp and Euterpe Oleracea Juice, respectively. Nonlinear mixed effect modeling identified dose volume as a significant predictor of relative oral bioavailability in a negative nonlinear relationship for Euterpe oleracea pulp and Euterpe Oleracea Juice. Additionally, after consumption of Euterpe oleracea pulp, applesauce, and Euterpe Oleracea Juice, plasma antioxidant capacity was statistically significantly increased (P < 0.01) when compared to the non-antioxidant control beverage. Individual increases in plasma antioxidant capacity of up to 2.3- and 3-fold for Euterpe Oleracea Juice and Euterpe oleracea pulp, respectively, were observed. Both applesauce and Euterpe oleracea pulp induced statistically significantly higher plasma antioxidant activities than Euterpe Oleracea Juice (P < 0.05). The non-oxidant control beverage also caused an increase in the antioxidant capacity of the plasma when compared to the baseline, which may have resulted from its fructose content. The antioxidant capacity in the urine, generation of reactive oxygen species, and uric acid concentrations in plasma were not significantly altered by the treatments. The results of this study indicate that anthocyanins from Euterpe oleracea are bioavailable in human subjects after consumption of Euterpe Oleracea Juice and Euterpe oleracea pulp in moderate amounts.
Toxicological Studies
Acute Toxicity Studies
Oral
Euterpe Oleracea Juice (test material Euterpe oleracea pulp-enriched fruit and berry juice)
The acute toxicity of a Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was evaluated in accordance with Organization for Economic Co-operation and Development (OECD) test guideline (TG) 423. 46 The juice is described as a functional beverage containing 19 fruits and berries. The concentration of Euterpe oleracea pulp (predominant ingredient in the juice) in the juice was not stated. Two groups of Wistar rats (Crl:(WI) BR strain; 5 males and 5 females per group) received single oral doses by gavage of 5 g/kg and 20 g/kg, respectively. Dosing was followed by a 14-day observation period and gross necropsy was performed on day 15. None of the animals died and there were no treatment-related clinical or behavioral signs. For female rats, the mean body weight gain (on days 1 and 2 and during the last week) in the 20 g/kg dose group was statistically significantly lower when compared to the 5 g/kg group. However, the total body weight gain of females in the 20 g/kg dose group was not statistically significantly different when compared to the 5 g/kg dose group. At necropsy (both dose groups) on day 15, there was no evidence of gross lesions in any organ, and all organs were free of gross pathological changes. It was concluded that the acute oral LD50 for the test substance was >20 g/kg.
Short-Term Toxicity Studies
Oral
Euterpe oleracea fruit oil
The short-term oral toxicity of Euterpe oleracea fruit oil was evaluated using groups of 6 Wistar rats. 47 Euterpe oleracea fruit oil (doses of 30 mg/kg, 100 mg/kg, or 300 mg/kg) in 1% Tween 80 was administered by gavage daily (at 24-h intervals) for 14 consecutive days. At the dose of 300 mg/kg, but not at lower doses, some animals began to display signs of toxicity, such as diarrhea and bristling of the hair. Information on mortalities or microscopic changes was not reported.
Subchronic Toxicity Studies
Oral
Euterpe Oleracea Juice (test material Euterpe oleracea pulp-enriched fruit and berry juice)
The subchronic oral toxicity of Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was evaluated using groups of 40 Wistar rats (SPF Hsd.Brl.Han strain; 20 males and 20 females per group). 46 The juice is described as a functional beverage containing 19 fruits and berries. The concentration of Euterpe oleracea pulp (predominant ingredient in the juice) in the juice was not stated. The test substance was administered daily by gavage for 90 days to 3 groups at doses of 10, 20, and 40 g/kg, respectively. Necropsy was performed on day 91. The vehicle control group was dosed with saline, and there was also an untreated control group. When compared to the control groups, there were no treatment-related, statistically significant changes in the following in surviving animals of all 3 dose groups: body weight, food and water consumption, ophthalmology, organ weights, urinalysis, hematological and clinical chemistry, or gross pathology. Three animals died during the study (1 female at 10 g/kg; 1 male at 20 g/kg; and 1 male at 40 g/kg). The animals that died did not have clinical symptoms prior to death. With the exception of signs of suffocation/aspiration congestion (due to problems with the gavage administration of the test substance; not considered test substance-related), there was no evidence of histopathological lesions or injury to tissues or organs. The only statistically significant difference observed was in mean adrenal weight (values not stated) relative to the brain weight in the 20 mg/kg dose group when compared to untreated female controls. Whether or not the change in adrenal weight in treated animals was an increase or decrease when compared to controls was not stated. The no-observed-adverse-effect-level (NOAEL) was determined to be 40 g/kg/day for male and female rats.
Developmental and Reproductive Toxicity Studies
Data on the developmental and reproductive toxicity of palm tree-derived ingredients reviewed in this safety assessment were neither found in the published literature, nor were these data submitted.
Genotoxicity Studies
Genotoxicity Studies on Palm Tree-Derived Ingredients and Related Components of Euterpe edulis and Euterpe Oleracea.

In Vitro
Euterpe edulis fruit pulp (9% in water) was genotoxic (at 25 to 250 μg/plate, but not at higher doses), without (but not with) metabolic activation, in one Salmonella typhimurium strain (TA97) in the Ames test. 24 In the same test, the authors noted a clear trend for the genotoxicity of this test substance in strains TA98 and TA100 at doses ranging from 25 to 250 μg/plate without metabolic activation. Euterpe edulis fruit pulp (9% in water) was also genotoxic in the micronucleus assay (RAW264.7 mouse macrophage-like cells; genotoxic at the entire range of concentrations tested (0.27 to 10.8 mg/mL)). 24 Euterpe edulis fruit oil was non-genotoxic in the cytokinesis-block micronucleus assay (human peripheral blood lymphocytes and HepG2 human hepatoma cells; concentrations up to 1000 μg/ml) or in the comet assay (human peripheral blood lymphocytes and HepG2 human hepatoma cells; concentrations up to 1000 μg/ml). 48
An Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) was non-genotoxic, with and without metabolic activation, in the Ames test (S. typhimurium strains and an Escherichia coli strain; doses up to 5000 μg/plate). 49 A Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was non-genotoxic, with and without metabolic activation, in the Ames test (S. typhimurium strains; doses up to 5 μg/plate), and non-genotoxic, with and without metabolic activation, in the chromosomal aberration assay (Chinese hamster lung cells; concentrations up to 5000 μg/ml) and in the L5178Y/TK+/− mouse lymphoma assay (concentrations up to 500 μg/ml). 46 The juice is described as a functional beverage containing 19 fruits and berries. The concentration of Euterpe oleracea pulp (predominant ingredient in the juice) in the juice was not stated.
In Vivo
Euterpe edulis fruit pulp extract (9% in water) was genotoxic in a micronucleus assay using bone marrow erythrocytes from rats that were dosed with up to 180 mg/kg by gavage for 3 days. 24 However, in a second study using the same protocol and doses, Euterpe edulis fruit pulp extract (9% in water) was non-genotoxic. 24 Negative results were also obtained in the comet assay (single cell gel electrophoresis [SCGE] test) using this test article involving randomly selected cells in blood from rats receiving doses (by gavage) up to 180 mg/kg, and in another comet assay involving randomly selected cells in human blood that was drawn after oral ingestion of 300 mL/day for 5 days.
Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was non-genotoxic in the micronucleus assay (mouse bone marrow erythrocytes from mice receiving daily oral or intraperitoneal (i.p.) doses of 100 μg/150 μl saline). 46 The juice is described as a functional beverage containing 19 fruits and berries. The concentration of Euterpe oleracea pulp (predominant ingredient in the juice) was not stated. Euterpe oleracea fruit pulp was non-genotoxic in the micronucleus assay (mouse bone marrow erythrocytes and peripheral blood erythrocytes from mice receiving either single or 14 days of oral doses up to 16.67 g/kg), and was non-genotoxic in the comet assay involving mouse peripheral blood erythrocytes, liver cells, and kidney cells from mice orally receiving doses up to 16.67 mg/kg for 1 or 14 days. 50 In rats dosed (by gavage) with Euterpe oleracea fruit oil (doses up to 300 mg/kg), there was no significant induction of DNA strand breaks in the comet assay (peripheral blood, bone marrow, liver cells, and testicle cells), but there was minor DNA damage in a few nucleoids (after dosing with 300 mg/kg). 47 Euterpe oleracea fruit oil was non-genotoxic in the micronucleus assay (bone marrow erythrocytes from rats receiving doses up to 300 mg/kg by gavage for 14 days).
Carcinogenicity Studies
Data on the carcinogenicity of palm tree-derived ingredients reviewed in this safety assessment were neither found in the published literature, nor were these data submitted.
Anti-Carcinogenicity Studies
Euterpe Oleracea Fruit Extract
The anti-tumorigenicity of Euterpe Oleracea Fruit Extract (hydroalcoholic extract) was evaluated using 2 groups of 40 female Wistar rats. 42 Twenty rats were dosed orally (200 mg/kg, by gastric intubation) with a saline solution of the fruit extract for 16 consecutive weeks. The control group (20 rats) was dosed with saline according to the same procedure. One day after starting dosing with Euterpe Oleracea Fruit Extract, mammary carcinogenesis was induced in all animals by subcutaneous (s.c.) injection of 25 mg/kg of 7,12-dimethylbenz[a]anthracene (DMBA) in the mammary gland. The animals were palpated in the mammary gland once per week to detect the presence of breast tumors. At the end of the treatment period, the animals were killed and tumor tissues as well as heart, liver, and kidney samples were examined histologically. In the control group, the tumor incidence rate was 100%. However, in the group dosed with Euterpe Oleracea Fruit Extract, the tumor incidence rate was markedly reduced to 50%. In both groups, mammary tumors displayed adhesions and a cystic pattern near the site of tumor induction. However, there was no significant difference in tumor volume (control: 4.151 ± 0.8 mL; Euterpe Oleracea Fruit Extract: 3.971 ± 1.3 mL) and tumor weight (control: 3.012 ± 0.5 g; Euterpe Oleracea Fruit Extract: 2.52 ± 0.7 g).
Survival analysis indicated that Euterpe Oleracea Fruit Extract increased survival (P = 0.0002, long-rank test) and reduced the number of deaths (P = 0.0036, Chi-square test). Cumulative survival periods of 15.15 weeks and 12.75 weeks were reported for test and control animals, respectively. The mortality rate in the control group was 65% (13 deaths), and the mortality rate was 15% (3 deaths) after dosing with Euterpe Oleracea Fruit Extract. There was no evidence of toxicity of the extract, based on food consumption, body weight, and activity levels, when compared to results for the 20 control rats. Histopathological results for the liver and kidneys indicated a protective effect of Euterpe Oleracea Fruit Extract, because, in the control group, there was an increase in fibrosis, atypical cells, and hemorrhagic microenvironment. There were no morphological differences in heart tissue between test and control rats. The researchers concluded that Euterpe Oleracea Fruit Extract (hydroalcoholic extract) exhibited anti-tumorigenic activity in DMBA-induced breast cancer. 42
Euterpe Oleracea Pulp Powder
A study was performed to investigate the protective effect of Euterpe Oleracea Pulp Powder (spray-dried) intake on colon carcinogenesis induced by 1,2-dimethylhydrazine. 51 Four groups of 10 rats received 4 s.c. injections of 1,2-dimethylhydrazine (40 mg/kg) for 4 weeks (twice a week), for initiation of colon carcinogenesis. A fifth group (5 rats) received similar injections of ethylenediaminetetraacetic acid (EDTA; 1,2-dimethylhydrazine vehicle). The groups were then fed a diet containing 2.5% or 5.0% Euterpe Oleracea Pulp Powder, or a diet containing 0.2% N-acetylcysteine (antioxidant and anti-carcinogenic agent) for 10 weeks, using aberrant crypt foci (ACF) as the endpoint. Additionally, two groups were fed a standard diet or a diet containing 5.0% Euterpe Oleracea Pulp Powder for 20 weeks, using colon tumors as the endpoint. In the assay using ACF as the endpoint, a reduction in the number of aberrant crypts and ACF were observed in the groups fed 5.0% Euterpe Oleracea Pulp Powder for 10 weeks (37% aberrant crypts and 47% ACF inhibition, P = 0.036) and 0.2% N-acetylcysteine (39% aberrant crypts and 41% ACF inhibition, P = 0.042). In the assay using colon tumors as the endpoint, a reduction in the number of invasive tumors (P < .005; % reduction not stated) and tumor multiplicity (P = 0.001; % reduction not stated) was observed in the group fed with 5.0% Euterpe Oleracea Pulp Powder for 20 weeks. Also, a reduction in tumor Ki-67 (human protein strictly associated with cell proliferation) cell proliferation (P = 0.003) and net growth index (P = 0.001) was observed in the group fed 5.0% Euterpe Oleracea Pulp Powder. The researchers concluded that the results of this study indicate that Euterpe Oleracea Pulp Powder feeding may reduce the development of chemically-induced rat colon carcinogenesis.
Another study was performed to evaluate whether feeding with Euterpe Oleracea Pulp Powder attenuates the initiation step of chemically-induced mouse colon carcinogenesis. 23 Euterpe oleracea fruit pulp was frozen and samples of spray-dried pulp (powder) were obtained. (The production method for this powder is stated in the Method of Manufacture section of this report.) This study involved male Swiss mice (3 groups of 15 (Groups 1–3); 1 group of 5 (Group 4)). Group 1 was fed a low-fat diet and Groups 2 and 3 were fed a low-fat diet containing 2.5% and 5% Euterpe Oleracea Pulp Powder, respectively, during weeks 1 to 4. The positive control group (Group 4) was fed a low-fat diet containing 0.1% indole-3-carbinol during weeks 1 to 3. All groups received an i.p. injection of the colon carcinogen azoxymethane (AOM) at week 3. Some mice from groups 1 to 3 (n = 5 mice/group) and all 5 mice from group 4 were killed 24 h after the first injection of AOM at week 3, and liver samples were collected for immunohistochemical and glutathione analysis. The remaining mice (Groups 1-3; n = 10 mice/group) received a second i.p. injection of AOM at week 4 and were fed a high-fat diet to accelerate the development of preneoplastic ACF until week 14. At week 3, both dietary Euterpe Oleracea Pulp Powder doses (2.5% or 5.0%) reduced (P < 0.001; % reduction not stated) peripheral blood cell DNA damage induced by AOM. Also, 5.0% Euterpe Oleracea Pulp Powder increased (P = 0.002; % increase not stated) hepatic total glutathione. At week 14, 5.0% Euterpe Oleracea Pulp Powder reduced (P < 0.05; % reduction not stated) ACF multiplicity. The researchers concluded that these findings indicate that feeding with Euterpe Oleracea Pulp Powder attenuates chemically-induced mouse colon carcinogenesis by increasing total glutathione and attenuating DNA damage and preneoplastic lesion development.
Other Relevant Studies
Effect on Mast Cell Activation
Euterpe Oleracea Pulp Powder (test material Euterpe oleracea pulp)
The pretreatment of immunoglobulin E (IgE)-sensitized mouse primary cultured mast cells with Euterpe oleracea pulp caused dramatic suppression of antigen-induced degranulation in a dose-dependent manner (1 to 1000 ng/mL). 52 Furthermore, Euterpe oleracea pulp suppressed IgE-mediated degranulation and transcription of the cytokine genes from a cultured mast cell line of rat basophilic leukemia (RBL)-2H3 cells. The results also suggest that Euterpe oleracea pulp could selectively inhibit a high affinity IgE receptor (FcεRI) signaling pathways, and indicate that the FcεRI-mediated complementary signaling pathway was suppressed by Euterpe oleracea pulp. The authors noted that these results demonstrate that Euterpe oleracea Pulp is a potent inhibitor of IgE-mediated mast cell activation.
Cytotoxicity
Euterpe Oleracea Fruit Extract
A cellular viability assay was performed to assess the potential for a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) to increase cellular metabolic activity in human dermal fibroblasts cultured for 24 h with test article concentrations of 0.01, 0.1, and 1% (in Dulbecco’s modified eagle medium). 53 In this assay, resazurin (nonfluorescent dye) is converted to resorufin, a fluorescent dye, by respiring mitochondria. Healthy cells in a proliferative state will be able to easily convert resazurin to resorufin without harming the cells. A proliferative cellular state is indicated by an increase in the signal generated by resazurin conversion. Cellular metabolism was expressed as viability (% of control). When compared to the control (unnamed), all concentrations of the Euterpe Oleracea Fruit Extract trade name mixture increased cellular metabolism. At a concentration of 0.01%, viability was 117.31% of the control. No change was seen in 99.35% compared to control at a concentration of 0.10%. The increase in the fluorescent signal indicated an increase in cellular metabolism and viability after incubation with the trade name mixture.
The anti-carcinogenicity potential of Euterpe Oleracea Fruit Extract (hydroalcoholic extract) was evaluated in vitro in a study using cell viability as the toxicity endpoint. 54 The malignant cell lines derived from human mammary adenocarcinoma (MCF-7 and MDA-MB-468 cells) and human colon adenocarcinomas (Caco-2 and HT-29) were treated with 10, 20, and 40 μg/mL Euterpe Oleracea Fruit Extract for 24 h and 48 h. After treatment, cell viability was measured using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays, and cell morphological features were observed by light and transmission electron microscopy. The data were analyzed statistically. Of all the cell lines tested, MCF-7 was the only line that responded to Euterpe Oleracea Fruit Extract treatment (cytotoxic effect). Significant reduction (P < 0.01; % reduction not stated) in cell viability and altered cell morphological features (by inducing the appearance of autophagic vacuoles) was noted at all concentrations. It was concluded that Euterpe Oleracea Fruit Extract possesses anti-tumorigenic potential in the MCF-7 cell line.
Euterpe Oleracea Pulp Extract
The antiproliferative activity of a Euterpe oleracea pulp extract (polyphenolic extract, concentrations ranging from 0.04 to 12 μg of GAE)/mL was evaluated in a cell culture model using HT-29 colon carcinoma cell viability as the endpoint. 55 Cell numbers were determined after 48 h of incubation. Total cell numbers were indicative of the proliferative activity of HT-29 cells and the cytotoxic effect of Euterpe oleracea pulp extract. The extract caused significant decreases (P < 0.01; % decrease not stated) in total cell numbers in a concentration-dependent manner.
Dermal Irritation and Sensitization Studies
Irritation
In Vitro
Euterpe Oleracea Fruit Extract
The skin irritation potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) was evaluated using the EpiDermTM model (reconstructed human epidermis) assay. 56 The test substance was applied to tissue inserts, which were incubated for 60 minutes. Tissue viability was measured by dehydrogenase conversion of MTT, present in mitochondria, into blue formazan salt. Skin irritation potential of the test substance is dictated by the reduction in tissue viability of exposed tissues when compared to the negative control (sterile Dulbecco’s phosphate buffered saline). Sodium dodecyl sulfate (5%) served as the positive control. An irritant is predicted if the mean relative tissue viability of the 3 tissues exposed to the test substance is reduced by 50% of the mean viability of the negative controls, and a non-irritant’s viability is >50%. The trade name mixture was classified as a non-irritant in this assay.
Sensitization
In Vitro/In Chemico
Euterpe Oleracea Fruit Extract
The in vitro skin sensitization antioxidant/electrophile response element (ARE)-nuclear factor (erythroid-derived 2; Nrf2) luciferase test method was used to evaluate the sensitization potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment). 57 This test method (validated by independent peer review by the European Union Reference Laboratory for Alternatives to Animal Testing (EURL)-European Center for the Validation of Alternative Methods (ECVAM)) addresses the induction of genes that are regulated by AREs by skin sensitizers. The sensitization assay in this study utilizes the KeratinoSensTM method. Collectively, an immortalized adherent human keratinocyte cell line (HaCaT) was incubated for 48 h with 12 concentrations of the trade name mixture ranging from 0.98 μM to 2000 μM. Cinnamic aldehyde (4 μM to 64 μM) and 1% dimethyl sulfoxide served as positive and negative controls, respectively. There was no statistically significant increase in luciferase expression, and the Euterpe Oleracea Fruit Extract trade name mixture was not predicted to be a skin sensitizer.
The skin sensitization potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) was evaluated using the direct peptide reactivity assay (DPRA; an in chemico method). 58 This assay is designed to mimic the covalent binding of electrophilic chemicals to nucleophilic centers in skin proteins by quantifying the reactivity of chemicals towards the model synthetic peptides containing cysteine and lysine. The mean percent depletion of cysteine and lysine was 3.20%, interpreted as minimal reactivity in the assay and yielding a prediction of no sensitization.
Human
Euterpe Oleracea Pulp Powder
A human repeated insult patch test (HRIPT) involving a face and neck product containing 3% Euterpe Oleracea Pulp Powder was performed using 214 subjects. 59 Testing occurred over a 6-week period. During induction, a 2 cm × 2 cm occlusive patch containing the product (0.2 mL or 0.2 g) was applied for 24 h to the infrascapular area of the back (to the right or left of midline) or to the upper arm. This procedure was repeated for a total of 9 induction applications, and sites were evaluated at 48-h intervals. For 24-h patch applications on Fridays, sites were evaluated on the following Monday (i.e., 72 h after patch application). The evaluation of sites after the 9th patch application was followed by a 10- to 15-day non-treatment period, after which (at week 6) the challenge phase was initiated. A challenge patch was applied for 24 h to a new test site, and reactions were scored at 24 h, 48 h, and 72 h after patch application. Definite erythema and damage to the epidermis, but no edema, were observed (at 5th induction evaluation) in 1 subject. Thereafter, the product was applied to a new site and reactions were not observed for the remainder of the induction period or during the challenge phase. The authors concluded that there was no evidence of sensitization to the product tested in this study.
Ocular Irritation Studies
In Vitro
Euterpe Oleracea Fruit Extract
The EpiOcularTM model (human corneal epithelial model) assay was used to evaluate the irritation potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment). 56 The test substance was applied to tissue inserts and incubated for 30 min. Tissue viability was measured by dehydrogenase conversion of MTT, present in the cell mitochondria, into blue formazan salt. Ocular irritation potential of the test substance is dictated by the reduction in tissue viability of exposed tissues when compared to the negative control (sterile deionized water). Methyl acetate served as the positive control. An irritant is predicted if the mean relative tissue viability of the 2 tissues exposed to the test substance is reduced by 60% of the mean viability of the negative controls, and a non-irritant’s viability is >40%. The trade name mixture was classified as a non-irritant in this assay.
Summary
The safety of 8 palm tree (Euterpe edulis (juçara) and Euterpe oleracea (açaí))-derived ingredients as used in cosmetics is reviewed in this safety assessment. According to the Dictionary, these ingredients function mostly as skin conditioning agents in cosmetic products. Euterpe Oleracea Pulp Powder and Euterpe Oleracea Seed Powder also function as abrasives and exfoliants in cosmetics.
Information on the method of manufacture of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) from a supplier indicates that the process involves the aqueous extraction of Euterpe Oleracea Fruit. Additionally, this trade name mixture and Euterpe Oleracea Juice have been analyzed for the 26 fragrance allergens that are required to be listed on the product label in the European Union if they exceed a certain concentration. Both were found not to contain these allergenic flavors or fragrances, neither directly nor through cross contamination. The same supplier’s impurities specifications for a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) include the following: heavy metals (<20 ppm), lead (<10 ppm), arsenic (<2 ppm), microbial content (<100 cfu/g; no pathogens), yeast and mold (<100 cfu/g), and gram-negative bacteria (0 cfu/g). Data provided by the same supplier indicate that pesticides present in this trade name mixture do not exceed the EPA’s limits.
According to 2020 VCRP data, Euterpe Oleracea Fruit Extract is reported to be used in 469 cosmetic products (328 leave-on products, 137 rinse-off products, and 4 products that are diluted for (bath) use). Of the palm tree-derived ingredients that are being reviewed in this safety assessment, this is the greatest reported use frequency. The results of a concentration of use survey conducted by the Council in 2017 indicate that Euterpe Oleracea Pulp Powder is being used at maximum use concentrations up to 3% in leave-on products (face and neck products [not spray]) and maximum use concentrations up to 0.6% in rinse-off products (moisturizing products [not spray] and paste masks [mud packs]). These are the highest use concentrations in leave-on and rinse-off products that are being reported for the palm tree-derived ingredients that are being reviewed in this safety assessment. According to VCRP and Council survey data, the following 3 ingredients that are being reviewed are not being used in cosmetic products: Euterpe Edulis Fruit Extract, Euterpe Edulis Juice Extract, and Euterpe Oleracea Seed Powder.
The results from a clinical trial involving 12 subjects who consumed an oral dose (7 mL/kg) of Euterpe Oleracea Juice or Euterpe oleracea pulp indicated that anthocyanins from Euterpe oleracea are bioavailable in human subjects after consumption of Euterpe Oleracea Juice and Euterpe oleracea pulp in moderate amounts.
The acute toxicity of a Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was evaluated using 2 groups of 10 Wistar rats that received single oral doses of 5 g/kg and 20 g/kg, respectively. The acute oral LD50 was reported as > 20 g/kg.
In groups of 6 Wistar rats, Euterpe oleracea fruit oil (doses of 30 mg/kg, 100 mg/kg, or 300 mg/kg) in 1% Tween 80 was administered by gavage daily for 14 consecutive days. At the dose of 300 mg/kg, but not at lower doses, some of the animals had signs of toxicity such as diarrhea and bristling of the hair. In a 16-week study involving 20 Wistar rats dosed orally with Euterpe Oleracea Fruit Extract and s.c. with DMBA, there was no evidence of toxicity of the extract, based on food consumption, body weight, and activity levels. There were no morphological differences in heart tissue between test and control rats.
The subchronic oral toxicity of Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was evaluated using groups of 40 Wistar rats. The test substance was administered daily for 90 days to 3 groups at oral doses of 10, 20, and 40 g/kg, respectively. There were no treatment-related, statistically significant changes in the following in surviving animals of all 3 dose groups: body weight, food and water consumption, ophthalmology, organ weights, urinalysis, hematological and clinical chemistry, or gross pathology. The 3 animals that died during the study did not have clinical symptoms prior to death, and there was no evidence of histopathological lesions or injury to tissues or organs. An NOAEL of 40 g/kg/day was reported.
Components of Euterpe edulis and Euterpe oleracea were evaluated in in vitro genotoxicity tests. Euterpe edulis fruit pulp (9% in water) was genotoxic without metabolic activation in one S. typhimurium strain in the Ames test, and in the in vitro micronucleus assay. In the Ames test on Euterpe edulis fruit pulp (9% in water), there was also a clear trend for genotoxicity in strains TA98 and TA100 at concentrations ranging from 25 to 250 μg/plate without metabolic activation. Euterpe edulis fruit oil was non-genotoxic in the cytokinesis-block micronucleus assay and in the comet assay. A Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) was non-genotoxic, with and without metabolic activation, in the Ames test (S. typhimurium strains and an E. coli strain). Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine) was non-genotoxic in the Ames test, the chromosomal aberration assay, and in the L5178Y/TK+/− mouse lymphoma assay.
In vivo genotoxicity test results for components of Euterpe edulis and Euterpe oleracea have also been reported. Euterpe edulis fruit pulp (9% in water) was genotoxic in one micronucleus assay (dosing by gavage; doses up to 180 mg/kg), but was non-genotoxic in another micronucleus assay (doses up to 180 mg/kg) using the same procedure or in comet assays (involved blood from rats dosed orally with up to 180 mg/kg; blood from humans dosed orally with up to 300 mg/kg). Euterpe oleracea pulp-enriched fruit and berry juice (fortified with glucosamine, daily oral [100 μg/150 μl saline] or i.p. [100 μg/150 μl saline] doses) was non-genotoxic in the micronucleus assay. Euterpe oleracea fruit pulp (doses up to 16.67 g/kg) was non-genotoxic in the micronucleus assay and in the comet assay. Results for Euterpe oleracea fruit oil (doses up o 300 mg/kg) in the comet assay indicated no significant induction of DNA strand breaks, but there was minor DNA damage in a few nucleoids. Euterpe oleracea fruit oil (doses up to 300 mg/kg) was also non-genotoxic in the micronucleus assay.
The anti-tumorigenicity of Euterpe Oleracea Fruit Extract has been demonstrated both in vivo (rats, breast cancer study) and in vitro (human mammary adenocarcinoma cell line). In vivo anti-carcinogenic activity of Euterpe Oleracea Pulp Powder has been demonstrated in colon cancer studies involving rats. In another study, the antiproliferative activity of Euterpe oleracea pulp extract was evaluated in a cell culture model using colon carcinoma cells, and a significant decrease in total cell numbers was reported.
When compared to the control (details not provided), a Euterpe Oleracea Fruit Extract trade name mixture increased cellular metabolism and viability at all test concentrations (0.01%, 0.1%, and 1%) in human dermal fibroblasts in vitro. In an in vitro study in which IgE-sensitized mouse mast cells were treated with Euterpe oleracea pulp, the test material was found to be a potent inhibitor of IgE-mediated mast cell activation.
A Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment) was classified as a non-irritant when skin irritation was evaluated using the EpiDermTM model (reconstructed human epidermis) assay.
The in vitro skin sensitization ARE-Nrf2 luciferase test method was used to evaluate the sensitization potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment). This test method involved incubation of the HaCaT cell line with concentrations ranging from 0.98 μM to 2000 μM, and the trade name mixture was not predicted to be a skin sensitizer. The same trade name mixture was evaluated for sensitization potential using the DPRA and was predicted to be a non-sensitizer.
An HRIPT involving a face and neck product containing 3% Euterpe Oleracea Pulp Powder was performed using 214 subjects. The authors concluded that there was no evidence of sensitization to the product tested in this study.
The EpiOcularTM model (human corneal epithelial model) assay was used to evaluate the ocular irritation potential of a Euterpe Oleracea Fruit Extract trade name mixture (98% Euterpe Oleracea Fruit Extract and 2% Lactobacillus ferment). The trade name mixture was classified as a non-irritant in this assay.
Discussion
The ingredient group reviewed in this safety assessment (Euterpe edulis (juçara) and Euterpe oleracea (açaí)-derived ingredients) was formed based on the supposition that ingredients from a given genus and species, and on a closely related species (i.e., edulis and oleracea), would have constituents in common. For example, both species have the following constituents in common: catechin, chlorogenic acid, cyanidin-3-glucoside, cyanidin-3-rutinoside, ellagic acid, ferulic acid, gallic acid, pelargonidin-3-glucoside, and peonidin-3-rutinoside. Except for Euterpe Oleracea Palm Heart Extract, for which composition data are absent, the Panel agreed that the available data indicate that the composition of the fruit from the two species are similar.
The Panel discussed the issue of incidental inhalation exposure from powders and hair sprays. The Council survey results indicate that Euterpe Oleracea Fruit Extract is being used in pump hair sprays at concentrations up to 0.001%. Also, Euterpe Oleracea Juice is being used at concentrations up to 0.01% in face powders. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The skin sensitization potential of a face and neck product containing 3% Euterpe Oleracea Pulp Powder was evaluated in a study involving 214 subjects, and the results were classified as negative. However, definite erythema and damage to the epidermis (but no edema) were observed in 1 subject at the 5th induction evaluation. No other effects were observed when the induction site was moved, or after the challenge application. The test concentration evaluated in this study is the highest maximum use concentration in leave-on products that is reported in this safety assessment. Furthermore, the in vitro/in chemico sensitization data on Euterpe Oleracea Fruit Extract were classified as negative. After reviewing these data, the Panel also noted that the available composition data do not indicate the presence of any sensitizing constituents.
The Panel also expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Earlier in the review process, the Panel was of the opinion that the available data were insufficient to make a determination that Euterpe Oleracea Palm Heart Extract is safe under the intended conditions of use in cosmetic formulations. However, the Panel now agrees that the available data support the safe use of this ingredient in cosmetics. Prior to making this decision, the Panel noted that palm heart is the edible part of the palm tree, and that the published literature lacks any evidence of consumption-related adverse effects such as contact sensitization or colitis. Furthermore, they noted that the absence of these effects is supported by clinical experience, and have also taken into consideration the low use concentrations of Euterpe Oleracea Palm Heart Extract in rinse-off and leave-on products.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.