Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Basic Red 76, which is reported to function in cosmetics as a hair colorant and hair-conditioning agent. The Panel reviewed the available data to determine the safety of this ingredient. The Panel concluded that Basic Red 76 is safe for use as a hair dye ingredient in the present practices of use and concentration described in the safety assessment.
Keywords
Introduction
This is a safety assessment of Basic Red 76 as used as a hair dye ingredient. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), Basic Red 76 is a monoazo color that functions as a hair colorant and hair-conditioning agent. 1 However, according to the US Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP) database, this ingredient is reported to also be used in nail products. 2 Although Basic Red 76 is exempt for use as hair dye, use in nail products is not an approved use for this cosmetic ingredient, and therefore, the Panel is not reviewing the safety for such use.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Much of the data included in this safety assessment were gathered from the opinions of European scientific committees, specifically, the Scientific Committee on Consumer Safety (SCCS) 3 and Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers (SCCNFP). 4
Chemistry
Definition and Structure
As given in the Dictionary, Basic Red 76 (CAS No. 68391-30-0) is the azo dye that conforms to the following structure (Figure 1)
1
: The azo dye, Basic Red 76.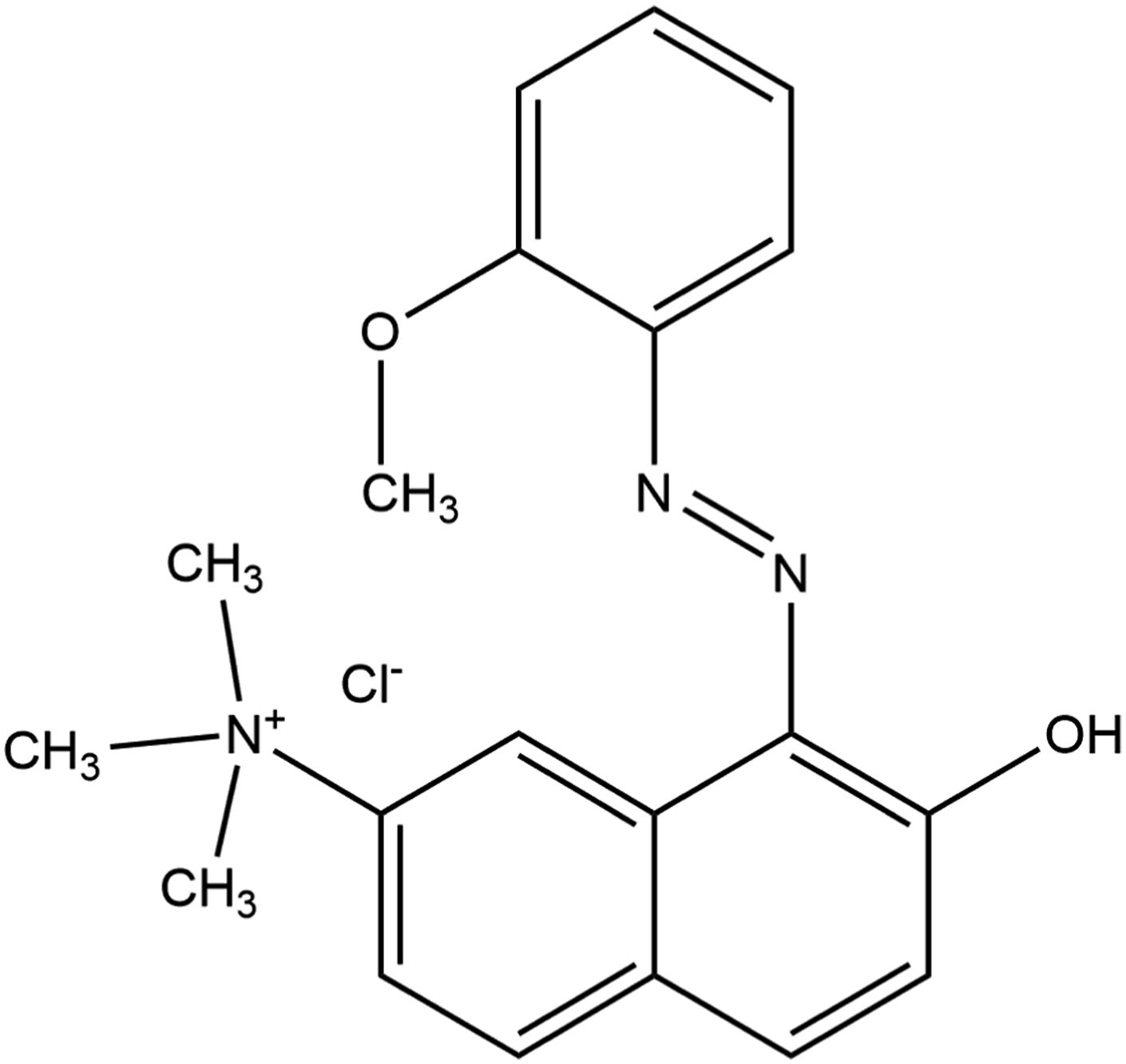
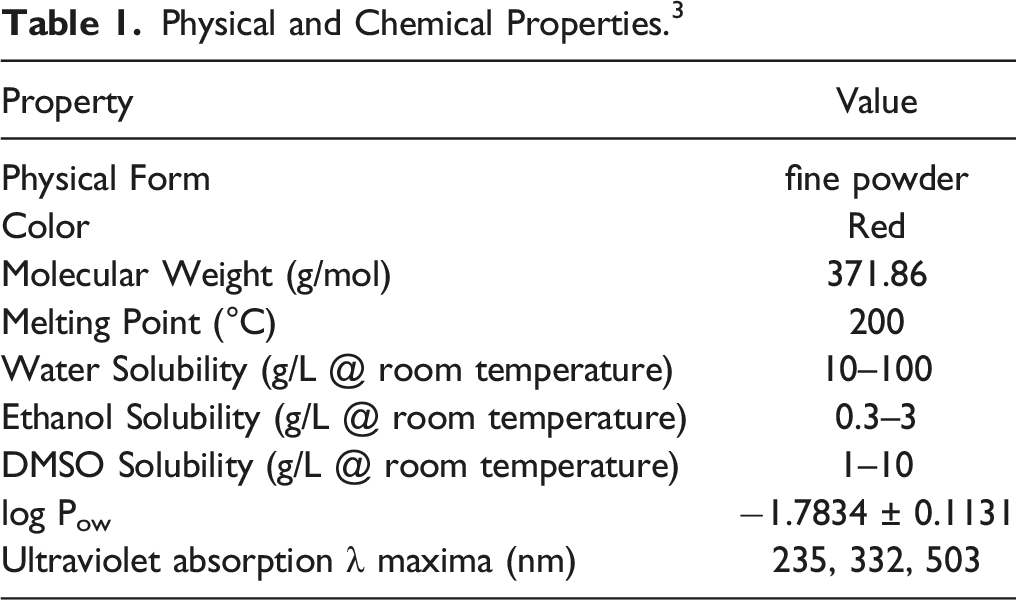
Physical and Chemical Properties
Physical and Chemical Properties. 3
Method of Manufacture
While no methods were found in the publicly available literature specific to the preparation of Basic Red 76, most azo dyes are synthesized in the same manner. 5 The first of two steps in the classic synthesis of dyes like Basic Red 76 involves the diazotization of a primary aromatic amine (e.g., 2-methoxyaniline), in a cold aqueous, acidic solution, with sodium nitrite. The resulting diazonium salt is highly reactive, and an arylazo-dehydrogenation reaction with an aromatic alcohol (e.g., 7-hydroxy-N,N,N-trimethylnaphthalen-2-aminium chloride) quickly results in an azo dye.
Impurities/Components
Impurities/Components.
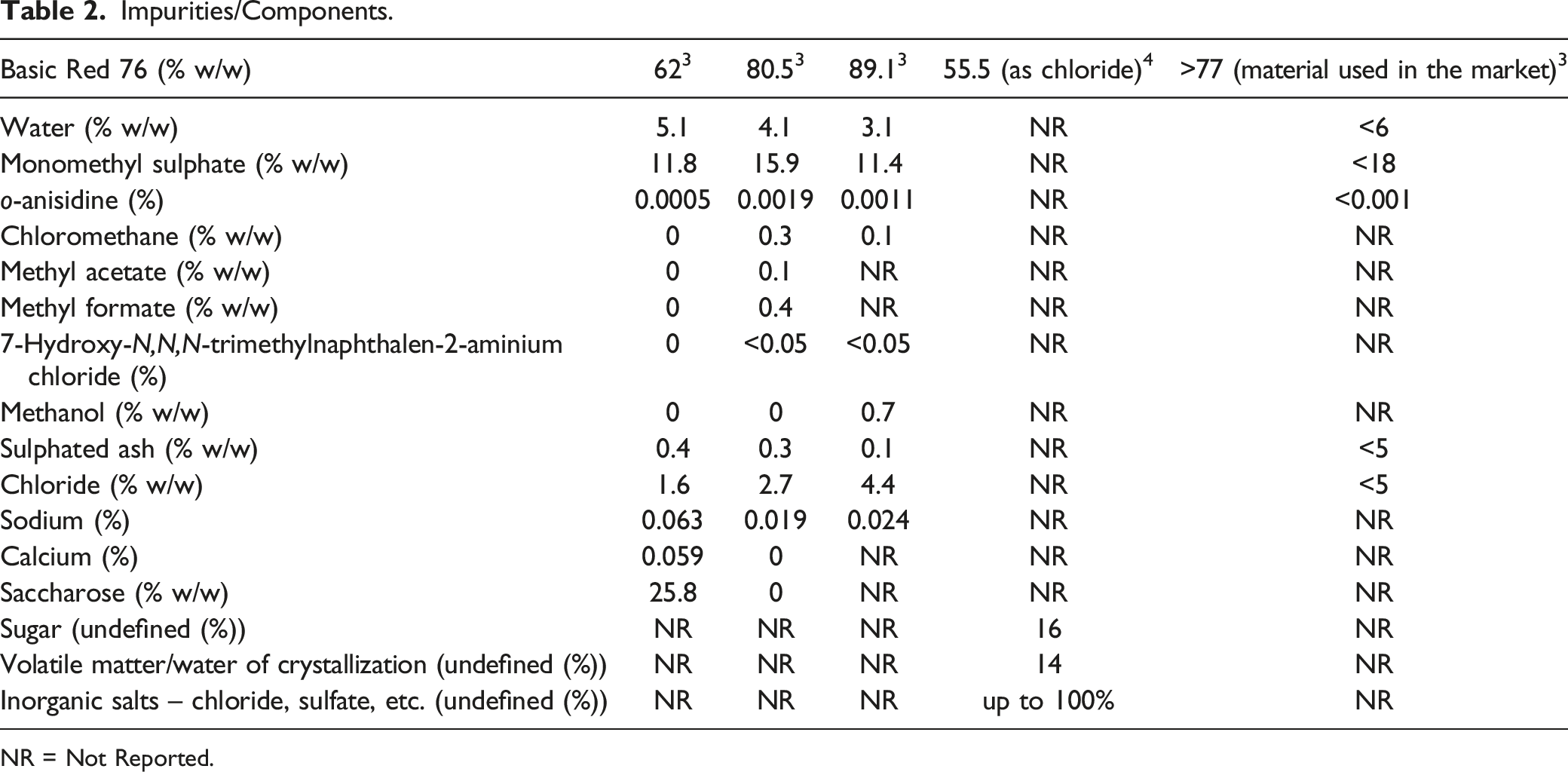
NR = Not Reported.
Use
Cosmetic
The safety of the cosmetic ingredient addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Frequency (2019) and Concentration of Use (2017) of Basic Red 76.

NR = no reported use.
*Because an ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
Basic Red 76 is considered a coal tar hair dye for which regulations require caution statements and instructions regarding patch tests in order to be exempt from certain adulteration and color additive provisions of the US Federal Food, Drug, and Cosmetic Act. In order to be exempt, the following caution statement must be displayed on all coal tar hair dye products: Caution - this product contains ingredients which may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dyeing the eyelashes or eyebrows; to do so may cause blindness.
Product labels shall also bear a caution statement and patch test instructions for determining whether the product causes skin irritation. The Panel recommends that an open patch test be applied and evaluated by the beautician and/or consumer for sensitization 48 hours after application of the test material and prior to the use of a hair dye formulation.
In 2012, a report was published regarding such self-testing for contact sensitization to hair dyes. 8 These authors concluded that, in its present form, the hair dye self-test has severe limitations. An accompanying editorial performed on behalf of the European Society of Contact Dermatitis (ESCD) asserted that industry is focusing on predicting the risks from exposure to hair dyes by having millions of European consumers perform a self-test prior to each hair dying, stating that it is the opinion of the ESCD that attention must be given to reducing the risks of serious allergic reactions by improving the safety of the products themselves. 9
Additionally, according to a report published in 2018, a different method of patch-testing was suggested utilizing more relevant protocols regarding exposure time and test preparation that reflect the actual use conditions with hair dying. 10 According to this study, a self-test protocol for an allergy alert test (AAT) was developed that elicits a self-noticeable alert signal in p-phenylenediamine (PPD)-allergic consumers after a 45 minute exposure to the hair dye mixed with developer.
Basic Red 76 is listed in the EU Cosmetics Regulation 1197/2013 Annex III, and is allowed in non-oxidative hair dye products at a maximum concentration of 2%. 11 According to the SCCS, Basic Red 76 containing up to18% methyl sulfate does not pose a risk to the health of the consumer when used as a non-oxidative hair dye with a maximum head-on concentration of 2.0%. 3
Toxicokinetic Studies
Azo bond cleavage and reduction is mediated by enzymes found in the liver, skin, and intestines. 12 Responsible cofactors and enzymes include nicotinamide adenine dinucleotide (NADH), cytochrome P450 reductase, and NAD(P)H quinone oxidoreductase. Both skin and intestinal microflora have been shown to reduce azo linkage, forming aromatic amines (e.g., o-anisidine). The produced aromatic amines can potentially have a greater expected absorption rate than the dye from which they are derived from.
Dermal Penetration
Animal
A two-part dermal/percutaneous absorption study was performed according to Organization for Economic Cooperation and Development (OECD) test guideline (TG) 428.3,13 Four replicates from each animal (one male, one female) of dermatomed pig skin, 0.75 mm thick, were used per experiment. In experiment A, 2% test material (80.5% Basic Red 76) in direct dye was applied to skin samples. Experiment B involved 2% test material (80.5% Basic Red 76) in water. In both experiments, applications of approximately 20 mg/cm2 were applied to the skin. Skin discs of 1.0 cm2 were exposed to the test substance for 30 minutes, and then rinsed. The receptor fluid used was a phosphate buffered saline. Within 48 hours after application, the percent of applied dose of Basic Red 76 recovered in the stratum corneum, epidermis/dermis, and receptor fluid were 1.44, 0.046, and 0.0049%, respectively. Samples treated with the aqueous solution displayed penetration amounts of 3.87% in the stratum corneum, 1.77% in the epidermis/dermis, and 0.012% in the receptor fluid. The amount of the test substance that was considered bioavailable from the direct dye cream and the aqueous solution was 1.96 ± 0.83 μg/cm2 and 6.52 ± 3.58 μg/cm2, respectively.
Human
Ten male subjects had 20 μL of a 1 mM test material (55.5% Basic Red 76), in 40% aqueous isopropanol, applied to five separate areas (5.3 cm2) of the inner forearm. 4 The dye stains were removed by ten repeated strippings with tape after 10 minutes, 24, 48, and 72 hours, and the amount of dye that potentially penetrated was estimated. The dye was not suspected to have been diffused into the horny layer, and the researchers concluded that the dye was not absorbed by the skin. No other information regarding this study was provided.
Absorption, Distribution, Metabolism, and Excretion (ADME)
Animal
Dermal
An application of 200 μL of a hair setting lotion containing 0.1% test material (55.5% [14C]-labelled Basic Red 76, labeling regiochemistry not stated) was applied to the skin of 3 Sprague-Dawley rats over an area of 1.5” × 1.5”, corresponding to an exposure of 31.3 mg/cm2 skin (and 31.3 μg Basic Red 76/cm2 skin). 4 The rats were anesthetized for 1 hour after application, then they were fitted with a collar to prevent licking. Radioactivity recovered from feces and urine was less than 0.2 and 0.3% of the applied dose, respectively. A maximum total absorption was calculated to be 0.5%, corresponding to a maximum of 0.15 μg/cm2 of skin. Excretion of radioactivity in urine and feces was measured for 24 hours after application. The amount of radioactivity recovered in the carcass or organs was not determined.
Other studies were performed using a setting lotion and shampoo formulation containing 0.2 and 0.5% test material (55.5% [14C]-labelled Basic Red 76, labeling regiochemistry not stated), respectively.
4
Application was performed on the clipped (but not shaven) skin of Wistar rats. After an application of 100 μL to 5 Wistar rats/sex, treatment sites were covered with a non-occlusive glass capsule containing small holes. Exposure occurred for 24 hours. In rats dosed with the setting lotion formulation containing 0.2% of the test material (25 μg of test material/cm2 skin), more than 80% of the applied radioactivity was recovered on the hair, and about 10% was recovered on the skin. The radioactivity recovered in the urine and feces was 0.07 and 0.16%, respectively. No radioactivity was detected in the carcasses. In a different study where rats were exposed for 24 hours to 70 and 140 μL of a shampoo containing 0.5% test material (55.5% [14C]-labelled Basic Red 76, labeling regiochemistry not stated),
Parenteral
Three male Wistar rats were given a single intravenous (i.v.) dose of 2.5 mg/kg bw of the test material (55.5% [14C]-labelled Basic Red 76, labeling regiochemistry not stated) in physiological saline. 4 Approximately 63 and 15% of the administered test substance was recovered in the feces and urine, respectively, over a duration of 24 hours. The level of radioactivity detected in the carcass 24 hours after the administrated dose was approximately 9%. In another study, mice were given a single subcutaneous dose of 5 mg/kg of the same test substance. Two minutes after administration, 31% of the radioactivity was present in the liver and kidneys, 9% in the small intestine, and 1.3% in the lungs. After 24 hours, the total radioactivity in the liver, kidneys and lungs decreased to 33.7% of the given dose. Specific radioactivity was highest in the cecum and large intestine by the end of the study.
Toxicological Studies
Acute Toxicity Studies
Oral
In an acute toxicity study, 3 CF1 mice were treated with a single oral dose of 1, 2.51, or 5.01 g/kg bw test material (55.5% Basic Red 76) in a volume of 20 to 40 mL/kg, and 10 male mice received the top dose of 10 mg/kg bw (method of oral dosing was not provided). 4 The animals were observed for 7 days after treatment. Lethargy and breathing disorders were observed in mice given 10 g/kg bw of the test substance. The LD50 value was reported to be >10 g/kg bw.
Wistar rats (3/sex) were given a single oral dose of 2 g/kg bw test material (62% Basic Red 76) in propylene glycol via gavage.3,14 Three male and one female rat displayed hunched posture on the first day of treatment. Red staining of the back and/or snout and/or head was observed in one female and two male mice. Red and/or yellow feces and/or urine were seen in all animals. The established oral LD50 value was >2 g/kg bw.
CFY rats (2/sex/group) were given a single oral dose (0, 0.1, 1, 4, 8, or 16 g/kg bw) of the test material (55.5% Basic Red 76) in 1% aqueous methylcellulose (method of oral dosing was not provided). 4 Animals were observed for 14 days after treatment. All animals survived treatment. Lethargy, piloerection, decreased respiratory rate, and hunched posture were observed. At the 8 and 16 g/kg bw dose levels, red staining of the urine and feces was noted. The acute lethal dose of the test substance was reported to be >16 g/kg bw.
Short-Term Toxicity Studies
Oral
Wistar MuRa Han 67 SPF rats (20/sex/group) were given 0 or 200 mg/kg bw of the test material (55.5% Basic Red 76) in a volume of 10 mL/kg water via gavage 5 days per week for 12 weeks in a screening study. 4 All animals survived the duration of the experiment. Aggressive behavior was apparent in all dosed animals. In males, body weight gain was similar to the control group, however, in females, slight but significantly lower mean body weights were recorded (95–96% of control) on the 5th, 7th, 9th, and 12th week, and at the end of the study. Colored urine was observed in all dosed animals. Increases in the mean cell volume and hematocrit values were noted in male rats and some female rats. In male rats, a slight increase in cerebral weights, as compared to the control groups, was observed. In females, kidney, heart, and liver weights were lower than those of control animals. The no-observable-adverse-effect-level (NOAEL) was reported to be <200 mg/kg bw/d.
Subchronic Toxicity Studies
Oral
Groups of 10 female and 10 male Sprague-Dawley CD rats were dosed with 0 and 20 mg/kg bw of the test material (55.5% Basic Red 76), in a volume of 10 mL/kg aqueous solution. 4 The test article was administered 5 days/week for 13 weeks by gavage. No mortalities were reported. Body weight gain was similar in the control and treated groups. No other effects were noted. The dose of 20 mg/kg bw/d was determined to be a “no effect level.”
SPF-bred Wistar rats (12/sex/group) were given a single daily dose of the test material (80.5% Basic Red 76) in distilled water via gavage for 90 days.3,15 Rats received doses of 0, 60, 250, or 1000 mg/kg bw/d. One female dosed with 60 mg/kg bw/d was found dead on day 60; however, a gavage error was considered to be the cause of death. Staining of body parts and discoloration of the feces/urine was observed. Infrequent and intermittent clonic spasms were observed in some test animals in all dose groups. No relevant body weight or food intake level changes were noted. Destruction of red blood cells, increased tissue iron in the spleen and liver, and increased serum bilirubin levels were noted in animals dosed with 250 and 1000 mg/kg bw/d. Thyroid follicular cell hypertrophy and adenohypophyseal cell hypertrophy was observed in rats given 1000 mg/kg bw/d; however, this effect is not considered relevant to humans as rats have a significantly higher sensitivity to this effect. At 60 mg/kg bw/day, decreased red blood count cells, hemoglobin levels, hematocrit levels, and mean corpuscular hemoglobin concentrations were seen. The NOAEL was reported to be 60 mg/kg/d. This study also included recovery groups of 5 rats/sex/dose group. The animals in these groups were examined after treatment, for four weeks. The hematological effects observed in the treated animals were widely resolved during the 28-day recovery period.
Developmental and Reproductive Toxicity Studies
Doses of 0, 60, 250, and 1000 mg/kg bw/d of the test substance (89.1% Basic Red 76) were given via gavage to Wistar rats (24/group) on days 6 to 20 of gestation.3,16 All females were killed and examined 21 days after mating. In rats treated with 60 mg/kg bw/d, no signs of developmental toxicity were observed. At the 250 mg/kg bw/d dose level, decreased body weight, weight gain, and food consumption was observed in maternal rats; a decrease in fetal body weight was also reported. Similar findings were seen in rats treated with 1000 mg/kg bw/d, however, signs of toxicity were more pronounced in the group treated with 250 mg/kg bw/d. Slight increases in the thinning of the central tendon region of the diaphragm and left-sided umbilical artery was observed in offspring. The maternal and developmental NOAEL was determined to be 60 mg/kg bw/d. Another study was performed using Sprague-Dawley CD rats. 4 Rats were given the test substance (55.5% Basic Red 76) in distilled water in doses of either 0 (20 rats) or 50 mg/kg (25 rats) bw/d on days 6–15 of gestation via gavage. On day 20 of gestation, the dams were killed. No adverse effects were reported in dams or fetuses treated with 50 mg/kg bw/d.
Genotoxicity
Genotoxicity Studies.
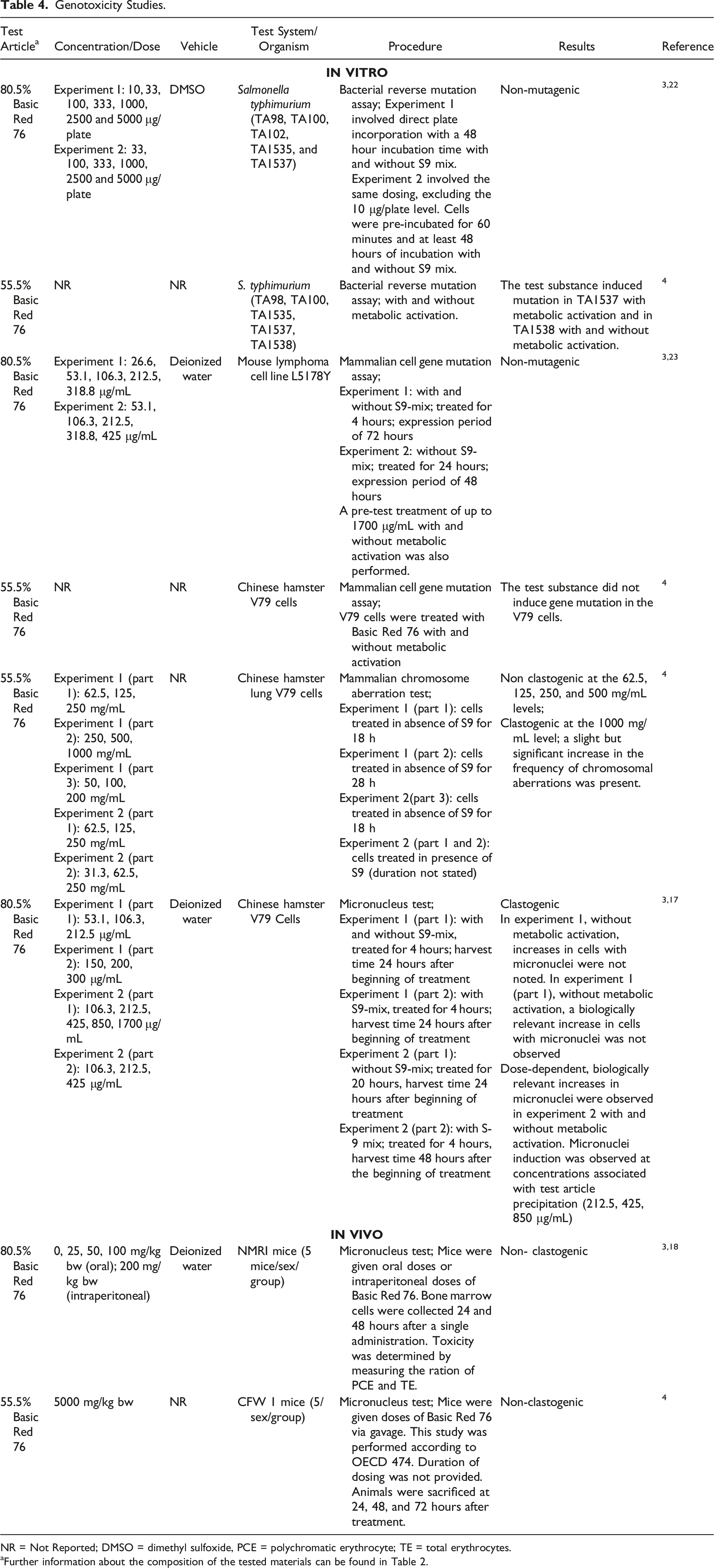
NR = Not Reported; DMSO = dimethyl sulfoxide, PCE = polychromatic erythrocyte; TE = total erythrocytes.
aFurther information about the composition of the tested materials can be found in Table 2.
Basic Red 76 was generally not genotoxic. Negative results were observed in Ames tests at concentrations of up to 5000 μg/plate, with or without metabolic activation (Salmonella typhimurium TA98, TA100, TA102, TA1535, and TA1537
Carcinogenicity
Information regarding the carcinogenicity of Basic Red 76 was not found; however, the potential metabolites of Basic Red 76, such as o-anisidine, have induced a number of multi-organ tumors according to third party summaries of animal studies cited by National Industrial Chemicals Notification and Assessment Scheme (NICNAS). 12 Both malignant and benign tumors in the bladder, spleen, subcutaneous tissues, kidneys, adrenal gland, liver, mammary glands, skin, blood, blood vessels, thyroid, lungs, gallbladder and renal pelvis, have been associated with the exposure of the metabolized aromatic amines. NICNAS, however, did not provide any specific evidence stating that Basic Red 76 is metabolized to o-anisidine.
Dermal Irritation and Sensitization
Irritation
Animal
A skin irritation test was performed according to OECD TG 404.3,19 A semi-occlusive patch containing 500 mg of the test substance (62% Basic Red 76) was applied to approximately 150 cm2 of shaved skin of 3 New Zealand White rabbits. Patches were removed after 4 hours. Skin was evaluated 1, 24, 48, and 72 hours, as well as 7, 10, and 14 days after removal of dressing. No visible signs of irritation were observed. Minimal red staining was noted in all animals.
In another study, a 24-h occlusive patch of 0.5 g undiluted test material (62% Basic Red 76) was applied to 1 in2 of intact or scarified skin of the back of 3 New Zealand White rabbits. 4 No reactions were reported. A similar study was performed with the same test substance that was dampened for adhesion. A dose of 0.5 g of the test material (62% Basic Red 76) was dampened with 0.5 mL distilled water and applied to a 1 in2 area of intact or scarified skin of the back of 3 New Zealand White rabbits. No reactions were recorded.
Sensitization
Animal
Twenty-five µL of the test material (62% Basic Red 76) was applied in concentrations of 2.5, 5, and 10% in a 7:3 v/v ethanol:water mixture.3,20 Ten percent was the highest technically applicable concentration in the vehicle. Applications were made on the earlobes of mice (4 females/group) in a local lymph node assay (LLNA). The test substance was applied once daily for 3 days. Five days after the first treatment, mice were given an intravenous injection of radiolabeled thymidine. Mice were killed 5 hours after thymidine administration. The draining lymph nodes were excised, pooled, placed in scintillation vials, and tested for proliferative capacity. The stimulation index values were 0.9, 1.1, and 1.3, for the 2.5, 5, and 10% dose levels, respectively. The test material was considered to be non-sensitizing.
In a Magnusson-Kligman test performed according to OECD TG 406, the sensitization potential of the test material (55.5% Basic Red 76) was evaluated using 10 female Dunkin-Hartley guinea pigs. 4 Intradermal induction consisted of injections of the material solution (0.1% test material in water), Freund’s Complete Adjuvant (FCA) diluted with an equal volume of water, and a 1:1 mixture of the material solution and FCA. One week after the administration of injections, a solution of 75% w/v of the test substance in distilled water was applied to the skin. A challenge patch was applied 2 weeks later at a concentration of 25% w/v of the test material. Irritation was noted after administration of the intradermal injection in all animals, which was still present at the time of topical induction. Half of the test animals displayed erythema after the challenge phase that was resolved by 48 hours. The sensitization potential was considered to be equivocal.
Ocular Irritation Studies
Animal
An ocular irritation test was performed according to OECD TG 405.3,21 The test material (62% Basic Red 76; 0.1 g) was instilled into the conjunctival sac of 3 New Zealand White rabbits. Treated eyes were rinsed following a 24-hour exposure period. Scoring occurred 1, 24, 48, and 72 hours, and 7 days, after instillation. Redness of the conjunctivae and sclerae, discharge, and chemosis were apparent at the beginning of treatment, but were no longer present after 72 hours. Minimal staining of the eyes was observed after 1 and 24 hours in all subjects. Staining was present in two animals at the 48-hour mark, and in one animal at the 72-hour mark. No abnormalities or corrosion was reported in the cornea or iris of test animals.
In a similar study using 3 New Zealand White rabbits, the test material (55.5% Basic Red 76) in physiological saline was instilled into the conjunctival sac (0.1 mL) of one eye of each rabbit. 4 The concentration of the test substance used was 0.5%. Eye reactions were recorded after 30 and 60 minutes, and 24 and 48 hours. No effects on the cornea or iris were reported; however, discoloration was noted.
Hair Dye Epidemiology
Hair dyes may be broadly grouped into oxidative (permanent) and direct (semi-permanent) hair dyes. The oxidative dyes consist of precursors mixed with developers to produce color, while direct hair dyes are a preformed color. Basic Red 76 is a direct hair dye ingredient. While the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair dye use and disease, such studies do provide broad information. The Panel determined that the available hair dye epidemiology data do not provide sufficient evidence for a causal relationship between personal hair dye use and cancer. A detailed summary of the available hair dye epidemiology data is available at https://www.cir-safety.org/cir-findings.
Summary
Basic Red 76 is a monoazo color that is reported to function as a hair colorant and hair-conditioning agent. Synthesis of the dye includes diazotization of a primary aromatic amine in a cold, aqueous, acidic solution with sodium nitrite. The resulting diazonium salt is highly reactive and an arylazo-dehydrogenation reaction with an aromatic alcohol quickly results in an azo dye.
According to 2019 VCRP survey data, Basic Red 76 is reported to be used in 48 hair-coloring formulations and 2 nail polish and enamel formulations. The results of the concentration of use survey conducted by the Council indicate that the highest concentration of use reported for Basic Red 76 was 0.35% in hair dyes and colors. Basic Red 76 is listed in the EU Cosmetics Regulation 1223/2009 Annex III, and is allowed in non-oxidative hair dye products at a maximum concentration of 2.0%.
In a dermal absorption study involving pig skin, 2% test material (80.5% Basic Red 76) was applied to samples in either a direct dye cream or aqueous formulation. Applications were performed in amounts of approximately 20 mg/cm2. The amount of the test material that was considered to be bioavailable by the direct dye cream and the aqueous solution was 1.96 ± 0.83 μg/cm2 and 6.52 ± 3.58 μg/cm2, respectively. In a human study, 20 μL of 1 mM of the test substance (55.5% Basic Red 76) in aqueous isopropanol was applied to the forearm. The dye stains were removed by ten repeated strippings with tape after 10 minutes, 24, 48, and 72 hours. The dye was not suspected to have diffused into the horny layer.
When an application of 200 μL of a hair setting lotion containing 0.1% test material (55.5% [14C]-labelled Basic Red 76) was applied to the skin of rats over an area of 1.5” × 1.5,” the amount of radioactivity recovered from feces and urine (measured for 24 hours after application) was less than 0.2 and 0.3% of the applied dose, respectively. A maximum total absorption was calculated to be 0.5%, corresponding to a maximum of 0.15 μg/cm2 of skin. Other studies were performed in which a setting lotion and shampoo formulation containing 0.2 and 0.5% test material (55.5% [14C]-labelled Basic Red 76), respectively, were applied to the skin of Wistar rats for 24 hours.
In rats dosed with 0.2% test material (55.5% [14C]-labelled Basic Red 76), over 80% of the applied radioactivity was recovered on the hair. The radioactivity recovered in the urine and feces was 0.07 and 0.16%, respectively. In rats treated with the formulation containing 0.5% test material, 93-102% of the applied radioactivity was recovered in hair rinsings. Approximately 2.1 and 1.7% radioactivity was recovered on the skin treated with 70 and 140 μL of the shampoo, respectively. The radioactivity recovered in the urine was <0.007% in males and 0.002% in females. Less than 10% of the applied radioactivity was observed in the feces of treated animals. Three male Wistar rats were given a single i.v. dose of 2.5 mg/kg bw of test material (55.5% [14C]-labelled Basic Red 76) in physiological saline. Approximately 63 and 15% of the administered dose was recovered in the feces and urine, respectively. In a similar study, mice were given a single subcutaneous dose of 5 mg/kg of the same test substance. Two minutes after administration, 31% radioactivity was present in the liver and kidneys, 9% in the small intestine, and 1.4% in the lungs. After 24 hours, the total radioactivity in the liver, kidneys and lungs decreased to 33.7% of the given dose.
The oral LD50 of a test substance containing Basic Red 76 was >10 g/kg bw (55.5% Basic Red 76) in CF1 mice, >2 g/kg bw (62% Basic Red 76) in Wistar rats and >16 g/kg (55.5% Basic Red 76) bw in CFY rats. These values were the highest doses tested in each study. Some signs of toxicity were observed.
Decreases in organ weights and increases in the mean cell volume and hematocrit values were noted when Wistar MuRa Han 67 SPF rats were dosed via gavage 5 days a week for 12 weeks (test material, 55.5% Basic Red 76; 200 mg/kg bw). The dose of 20 mg/kg bw/d was determined to be a no effect level in Sprague-Dawley CD rats dosed by gavage for 5 days/week for 13 weeks (test material, 55.5% Basic Red 76). Toxic effects included lowered body and organ weights. The established NOAEL in a 90-day study involving Wistar rats dosed via gavage was 60 mg/kg/d (test material, 80.5% Basic Red 76).
No signs of developmental toxicity were observed in Wistar rats given 60 mg/kg bw/d on days 6–20 of gestation via gavage (test material, 89.1% Basic Red 76); however, toxic effects were noted at the 250 mg/kg bw/d dose level and higher when the same test substance was used. The maternal and developmental NOAEL was determined to be 60 mg/kg bw/d. In a different study, no adverse effects were reported in Sprague-Dawley CD rats given up to 50 mg/kg bw/d via gavage on gestation days 6–15 (test substance, 55.5% Basic Red 76).
Mixed results were seen in Ames tests and mammalian gene mutation assays using Chinese hamster V79 cells with and without metabolic activation. Negative results were seen in a mammalian cell gene mutation assay using mouse lymphoma L5178Y cells. A chromosomal aberration assay using Chinese hamster V79 cells yielded negative results at up to 500 mg/mL; however, at the 1000 mg/mL concentration level, without metabolic activation, Basic Red 76 was clastogenic. An in vitro micronucleus test in V79 cells yielded positive results at concentrations as low as 212.5 μg/mL, with and without metabolic activation; however, in part one of the same experiment, negative results were seen at concentrations as high as 300 μg/mL with metabolic activation. Negative results were observed in in vivo micronucleus assays in mice at up to 5000 mg/kg.
No information regarding the carcinogenicity of Basic Red 76 was found. However, possible metabolites of Basic Red 76 (e.g., o-anisidine) have induced a number of multi-organ tumors according to several animal studies cited by NICNAS. (NICNAS, however, did not provide any specific evidence stating that Basic Red 76 is metabolized to o-anisidine.)
No irritation was reported when New Zealand White rabbits were dermally dosed with 500 mg or 0.5 g/in2 of the test substance (62% Basic Red 76) under an occlusive patch. In an LLNA, Basic Red 76 was considered a non-sensitizer when 25 μL of the test substance (62% Basic Red 76) was applied to mouse earlobes at a concentration of up to 10%. In a Magnusson-Kligman test, the sensitization potential of the test material (55.5% Basic Red 76) was evaluated using Dunkin-Hartley guinea pigs. Intradermal induction consisted of injections of the material solution (0.1% test material in water), Freund’s Complete Adjuvant (FCA) diluted with an equal volume of water, and a 1:1 mixture of the material solution and FCA. One week after the administration of injections, a solution of 75% w/v of the test substance in distilled water was applied to the skin. A challenge patch was applied 2 weeks later at a concentration of 25% w/v of the test material. Half of the test animals displayed erythema after the challenge phase that was resolved by 48 hours.
Ocular irritation was observed in New Zealand White rabbits following instillation of 0.1 g of the test material (62% Basic Red 76) into the conjunctival sac; this effect was resolved within 72 hours. In a different study, no irritation was reported when the test substance (55.5% Basic Red 76), at a concentration of 0.5%, in physiological saline, was placed in the conjunctival sac of New Zealand White rabbits.
The most recent comprehensive review of available epidemiology studies concluded that there is insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers. A summary of the data is available in the CIR hair dye epidemiology resource document and can be found on the CIR website (https://www.cir-safety.org/cir-findings).
Discussion
Basic Red 76 is a direct dye that is reported to function in cosmetics as a hair-colorant and hair-conditioning agent. However, according to US FDA VCRP data, this ingredient is reported as also being used in nail products. Use in nail products is not an approved use for this cosmetic ingredient, and therefore, the Panel is not reviewing the safety for such use.
The Panel found that the systemic toxicity, developmental/reproductive toxicity, genotoxicity, and irritation data in this report were sufficient. The Panel recognized the positive results of some genotoxicity studies, but considered them to be potentially misleading. Positive results were only obtained in in vitro studies, and at concentrations much higher than what would be used in cosmetics. All in vivo genotoxicity assays performed using Basic Red 76 yielded negative results, suggesting that the positive results seen in in vitro studies were not of concern. In addition, the Panel noted the carcinogenic potential of the aromatic amines formed by the metabolism of Basic Red 76. The concern regarding these metabolites was mitigated considering Basic Red 76 is poorly absorbed, is used in rinse-off products, and is used at very low concentrations. In addition, because use of this ingredient results in minimal dermal absorption, the actual exposure to these metabolized aromatic amines would be insignificant in light of cosmetic use conditions.
The Panel also considered the current recommendations for patch-testing, and noted that concerns over patch testing are on-going. New methods for patch-testing using relevant protocols regarding application time and actual use conditions are continuously being developed. The Panel suggests that the patch-test instructions provided by manufacturers should be followed before hair dye use. In addition, hair dyes containing Basic Red 76, as coal tar hair dye products, are exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act, when the label bears a caution statement and patch test instructions for determining whether the product causes skin irritation. The Expert Panel expects that following this procedure will identify prospective individuals who would have an irritation/sensitization reaction and allow them to avoid significant exposures.
In considering hair dye epidemiology data, the Panel concluded that the available epidemiology studies are insufficient to conclude there is a causal relationship between hair dye use and cancer and other endpoints. Details of these studies can be found in the CIR hair dye epidemiology resource document.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Basic Red 76 is safe for use as a hair dye ingredient in the present practices of use and concentration described in the safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
