Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reassessed the safety of Capryloyl Salicylic Acid in cosmetic products; this ingredient is reported to function as a skin conditioning agent. The Panel reviewed relevant data relating to the safety of this ingredient in cosmetic formulations, and concluded that the available data are insufficient to make a determination that Capryloyl Salicylic Acid is safe under the intended conditions of use in cosmetic formulations.
Keywords
Introduction
In 2003, the Expert Panel for Cosmetic Ingredient Safety (Panel) published a safety assessment of Salicylic Acid and 16 salicylates. 1 That safety assessment included Capryloyl Salicylic Acid, which was included in the grouping because of structural similarity. However, as this ingredient is a ketone, it does not belong in that family of ingredients; therefore this separate report was prepared. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), Capryloyl Salicylic Acid is reported to function as a skin conditioning agent. 2
The published data in this document were identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and endpoints that the Panel evaluates, is available on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties. Much of the data included in this safety assessment was found at the European Chemicals Agency (ECHA) and National Industrial Chemicals Notification and Assessment Scheme (NICNAS) websites.3,4 Please note that these websites provide summaries of information from other studies, and it is those summary data that are reported in this safety assessment when ECHA or NICNAS is cited.
Chemistry
Definition and General Characterization
Capryloyl Salicylic Acid (CAS No. 78418-01-6) was previously, erroneously, defined as the ester of salicylic acid and caprylic acid. However, it has become apparent that Capryloyl Salicylic Acid is instead the 5-capryl ketone of salicylic acid. As such, this chemical is structurally distinct from the salicylate carboxyl esters. It is now defined as the organic compound that conforms to Figure 1.
2
Capryloyl salicylic acid.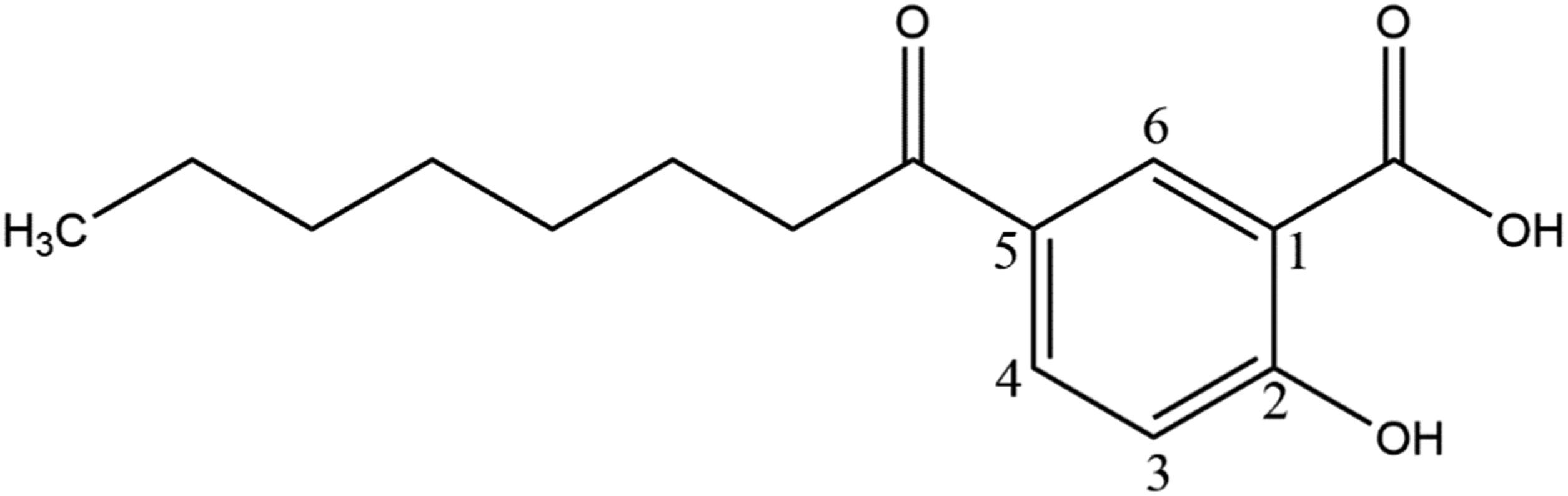
Chemical and Physical Properties
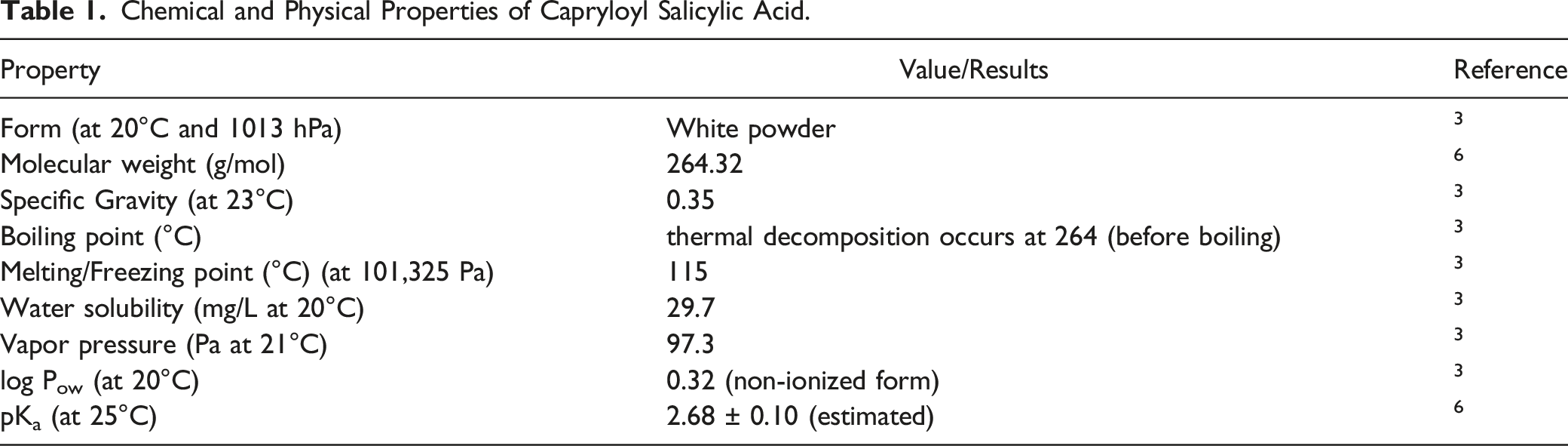
Chemical and Physical Properties of Capryloyl Salicylic Acid.
Method of Manufacture
The synthetic method of manufacture for Capryloyl Salicylic Acid is based on Friedel-Crafts acylation of methyl salicylate by capryloyl chloride and aluminum trichloride to yield the methyl ester of Capryloyl Salicylic Acid. 7 This ester is hydrolyzed with sodium hydroxide, after which acidification yields Capryloyl Salicylic Acid.
Impurities
It has been reported that the structural isomer, 3-capryloyl salicylic acid, is a highly plausible contaminant of Capryloyl Salicylic Acid. 7
Use
Cosmetic
The safety of this cosmetic ingredient is evaluated based, in part, on data received from the United States (US) Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. 8 Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category. 9
Frequency (2019) and Concentration (2018) of Use According to Duration and Exposure.
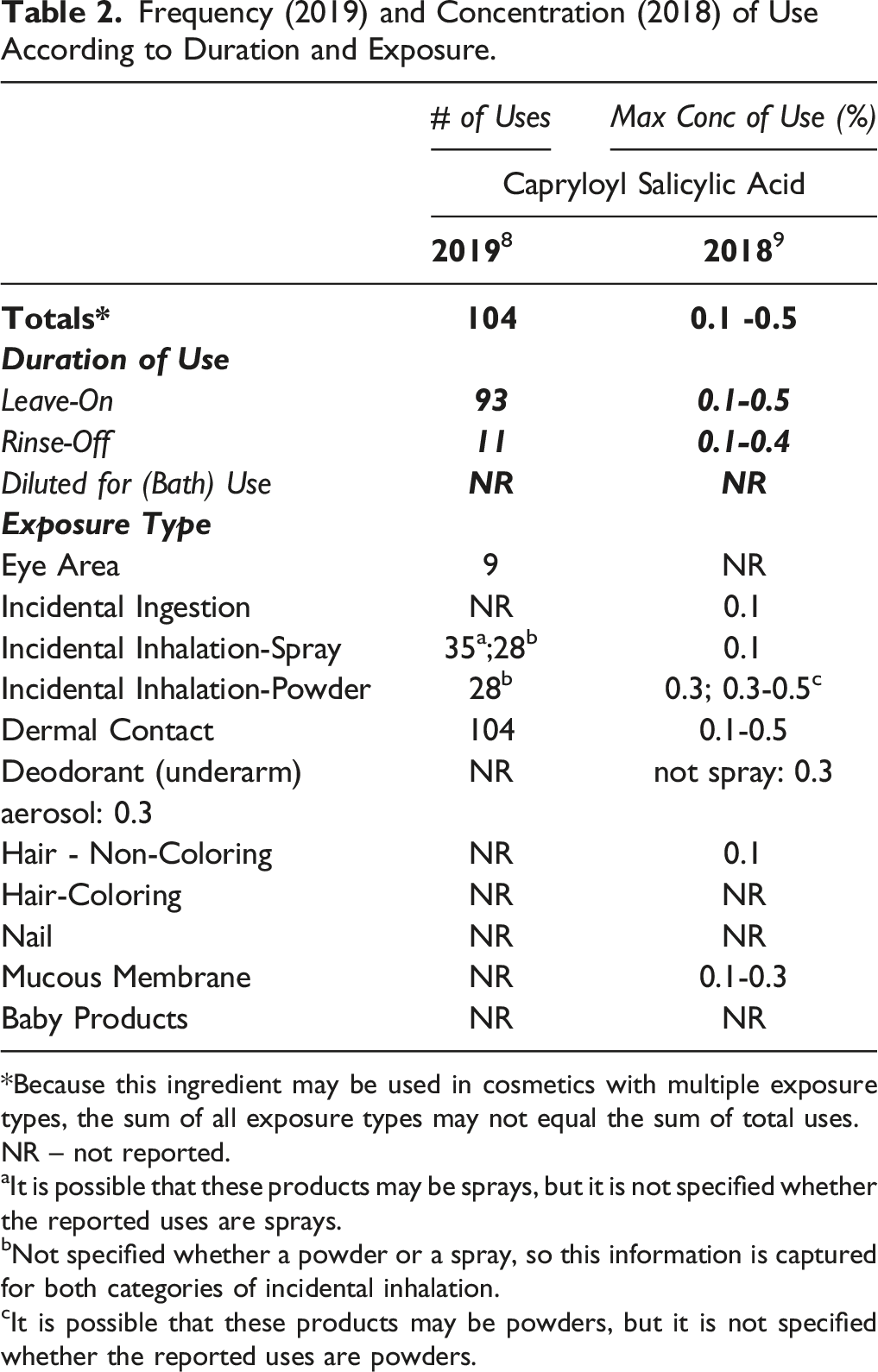
*Because this ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
NR – not reported.
aIt is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible that these products may be powders, but it is not specified whether the reported uses are powders.
Cosmetic products containing Capryloyl Salicylic Acid may be applied to the skin at concentrations up to 0.5% (moisturizing products, not spray) and may come in contact with the eyes during use of eye lotions and other eye makeup preparations (use concentrations were not reported by industry). Capryloyl Salicylic Acid also could be incidentally ingested during product use (e.g., use concentrations up to 0.1% in lipsticks). Products containing Capryloyl Salicylic Acid may be applied as frequently as several times per day and may come in contact with the skin for variable periods following application. Daily or occasional use may extend over many years.
Capryloyl Salicylic Acid is reported to be used in deodorant sprays (aerosolized) at concentrations up to 0.3%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.10-13 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.10,11 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 11 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Capryloyl Salicylic Acid is being used in face powders at concentrations up to 0.3%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.14-16
Toxicokinetic Studies
Dermal Penetration
In Vitro
The penetration of Capryloyl Salicylic Acid through human skin was assessed using a Franz diffusion chamber and skin samples. 5 The amount of Capryloyl Salicylic Acid that penetrated deeper than the stratum corneum after a 16-h contact time was significantly lower than its parent acid, salicylic acid, in the same vehicle. Thus, only approximately 6% of Capryloyl Salicylic Acid was found to penetrate deeper than the stratum corneum, after 16 h, versus 58% for salicylic acid.
Human
The skin penetration of Capryloyl Salicylic Acid was also assessed using a standard stripped skin method. 5 Capryloyl Salicylic Acid was applied to the skin (number of subjects involved not stated), followed by rinsing with water/alcohol 30 min later. There was no indication that a vehicle for the test substance was used. Strips of stratum corneum were then harvested, using adhesive tape, and analyzed for test material content. An estimation of the level of penetration through the skin over a 4-day period was determined using the stored concentration method. Based on this method, 17.1% of the applied test material was found in the stratum corneum, versus 9.7% of salicylic acid applied. Data relating to distribution of most of the Capryloyl Salicylic Acid applied to the skin was not included.
Absorption, Distribution, Metabolism, and Excretion
Data on the absorption, distribution, metabolism, and excretion of Capryloyl Salicylic Acid were neither found in the published literature, nor were these data submitted.
Toxicological Studies
Acute Toxicity Studies
Dermal
The acute dermal toxicity of Capryloyl Salicylic Acid was studied using Sprague-Dawley rats (5 males, 5 females). 3 The study was performed according to Organization for Economic Co-operation and Development (OECD) Test Guideline (TG) 402. A semi-occlusive patch (7 cm × 4 cm) containing the test substance (in peanut oil) was applied for 24 h to an area defined as 10% of the total body area. This amounted to a single dose exposure of 2000 mg/kg. Dosing was followed by a 14-day observation period, after which the animals were killed. Necropsy was performed, but tissues were not retained. None of the animals died during the observation period, but the following signs of toxicity were observed: hunched posture, lethargy, and piloerection during the first day. With the exception of 1 male rat, body weight gain was not affected by treatment. The following adverse dermal reactions were observed: edema and blanching and hard, light brown colored scabs at the application site. These findings were accompanied by loss of the upper layers of skin and fur, resulting in purple/pink areas. The distribution of toxicity signs/adverse dermal reactions in the dosed group was not reported. The LD50 was >2000 mg/kg.
Oral
The acute oral toxicity of Capryloyl Salicylic Acid (in peanut oil) was evaluated using groups of 10 Sprague-Dawley rats (5 males, 5 females/group). 3 The four dose groups received single oral doses (by gavage) of 2530, 3000, 3557, or 4217 mg/kg. The study was performed according to OECD TG 401. A control group was not included in the study. Dosing was followed by a 14-day observation period. All animals were subjected to gross necropsy. The following major signs of toxicity were observed in all dose groups: hunched posture, piloerection, lethargy, ptosis, decreased or gasping respiration, red/brown staining around the snout or mouth, and ataxia and/or increased salivation. Incidences of reduced body weight gain or body weight loss also occurred in all dose groups. Distended abdomen and/or emaciation were observed in animals of the 3 higher dose groups. Pallor of the extremities, with an isolated incidence of tiptoe gait, was observed in animals of the 3557 mg/kg dose group. Mortalities in all dose groups were reported as follows: 2530 mg/kg (1 female); 3000 mg/kg (4 males and 2 females); 3557 mg/kg (3 males, 3 females), and 4217 mg/kg (4 males, 4 females). The following abnormalities were observed at necropsy of animals that died during the study: hemorrhagic or abnormally red lungs; dark liver or patchy pallor of the liver; pale spleen; pale or dark kidneys; hemorrhage or sloughing of the glandular gastric epithelium; and hemorrhage of the small and/or large intestines. Abnormalities observed at necropsy of animals (2530 g/kg group) that were killed at the end of the study were described as occasional white foci (∼1 mm × 1 mm covering 75% of the non-glandular gastric epithelium). At necropsy, there were no abnormalities in animals that received a dose of 3000 mg/kg or higher and were killed at the end of the study. The combined oral LD50 (males + females) for the test material was 3354 mg/kg (95% confidence limit between 2834 and 3970 mg/kg).
Results relating to acute oral toxicity are included in a micronucleus test on Capryloyl Salicylic Acid (in 0.5% carboxymethylcellulose aqueous vehicle). 4 The micronucleus test (described later in this report), performed in accordance with OECD TG 474, used groups of 10 (5 males, 5 females per group) Swiss CD-1 mice. A single dose of the test substance (500, 1000, or 2000 mg/kg) was administered by gavage to 3 groups. Two female mice and 1 male mouse dosed with 2000 mg/kg were found dead 24 h after dosing. Piloerection was observed in all animals on the same day of dosing with 1000 mg/kg. Piloerection and swollen abdomen were observed in the 2000 mg/kg dose group.
Results relating to acute oral toxicity are also included in an unscheduled DNA synthesis test on Capryloyl Salicylic Acid (in 0.5% in carboxymethylcellulose aqueous vehicle). 4 The assay was performed in accordance with OECD TG 486 using groups of 4 Sprague-Dawley rats. In 2 tests, single doses of 500, 1000, or 2000 mg/kg were administered by gavage. The animals were killed at 14 h in one test and at 2 h in the other test. No mortalities or clinical signs were observed. (Results relating to genotoxicity are included in that section of this report.)
Short-Term Toxicity Studies
Dermal
A short-term dermal toxicity study (10-day study) on Capryloyl Salicylic Acid (in hexaethylene glycol (PEG-6)) was performed using groups of 5 female rats of the Crl:CD(SD)BR (VAF plus) strain. 3 The test substance was applied for 6 h directly to the back (not less than 10% of body area; constant volume of 2 mL/kg) once daily at concentrations of 2% and 5%. A third group was treated with vehicle only. The test protocol was similar to OECD TG 410. None of the animals died during the study. The animals were killed at the end of the 10-day dosing period and subjected to gross necropsy. Tissues were examined microscopically. There were no treatment-related changes in food consumption or body weight gain. The authors noted that occasional transient weight losses were observed which, because of the small group sizes, skewed the means. Other than the scabbing that was observed at necropsy, there were no other necropsy findings. The no-observed-effect-levels (NOEL) for local and systemic effects of Capryloyl Salicylic Acid were 2% and >5%, respectively. Additional results from this study are included in the Skin Irritation section of this report.
In a guinea pig maximization test of Capryloyl Salicylic Acid (in 0.5% aqueous methylcellulose), performed according to OECD TG 406, clinical observations were made. 4 Twenty Dunkin-Hartley guinea pigs (10 males, 10 females) were tested with this ingredient in this study. During topical induction, Capryloyl Salicylic Acid was applied at a concentration of 1%. One animal was found dead on day 18, but microscopic examination revealed no apparent abnormalities. The tissues from the dead animal examined microscopically were not stated. None of the other animals had clinical signs; body weight gains were comparable to the control animals. Results relating to skin sensitization potential are included in that section of this report.
Oral
A short-term oral toxicity study on Capryloyl Salicylic Acid (in PEG-6) was performed using groups of 10 or 20 rats of the CRL:CD(SD)BR strain. 3 The control group and highest dose group (initially, 300 mg/kg/day) each consisted of 20 rats (10 males, 10 females per group). Each of the remaining dose groups (10, 30, and 100 mg/kg/day) consisted of 10 rats (5 males, 5 females per group). The animals were dosed orally (by gavage) daily for 28 days in accordance with OECD TG 407. Two recovery groups (5 males, 5 females per group; for control and highest dose groups) were maintained un-dosed for an additional 14 days after the last day of dosing (day 28). The dose level in the highest dose group was reduced from 300 to 200 mg/kg/day on day 13 due to adverse clinical signs, including death. The animals were subjected to gross necropsy and microscopic examination of tissues at the end of the study. Five rats from the highest dose group died during the study, and the following clinical signs were observed: rough coat, piloerection, post-dose salivation. A slight reduction in body weight gain (13 to 14%) was also noted in males of the highest dose group during treatment and non-treatment periods, and in females of the 100 mg/kg dose group during treatment only. Hematology and blood chemistry evaluations did not reveal any adverse effects. At necropsy, a dose-related increase in the incidence of stomach abnormalities was observed (at the end of both the treatment and the treatment-free period) for animals dosed with 100 mg/kg/day and 300/200 mg/kg/day. Microscopic findings included hyperplasia of the non-glandular stomach in animals of the 300/200 mg/kg/day group. This finding was accompanied by chronic inflammation and ulceration. Hyperplasia was also observed in 1 male in the 100 mg/kg/day dose group. Similar, but less severe, hyperplastic lesions were also observed in recovery animals that were previously dosed with 300/200 mg/kg/day. Hyperplasia of the non-glandular mucosa of the stomach was less severe at the end of the treatment-free period, which indicates that some recovery had taken place. Additionally, reversibility was observed and the effects were limited to the non-glandular stomach, and, thus, were considered to be of doubtful relevance to humans. The NOEL for local effects was 30 mg/kg/day, and the NOEL for systemic effects was >100 mg/kg/day.
Subchronic Toxicity Studies
Data on the subchronic toxicity of Capryloyl Salicylic Acid were neither found in the published literature, nor were these data submitted.
Chronic Toxicity Studies
Data on the chronic toxicity of Capryloyl Salicylic Acid were neither found in the published literature, nor were these data submitted.
Developmental and Reproductive Toxicity Studies
Dermal
The developmental toxicity of Capryloyl Salicylic Acid (in PEG-6) was evaluated using groups of 24 pregnant female Sprague-Dawley rats. 3 The 2 test groups received doses of 40 mg/kg/day and 100 mg/kg/day, respectively, on gestation days 6 to 15. The vehicle control group was dosed with PEG-6. The test substance and vehicle control were each applied for 6 h directly to skin on the back (not less than 10% of the body area). The rats wore Elizabethan collars during the 6-h exposure period, and the application collars were rinsed after 6 h. On gestation day 20, all females were killed and subjected to necropsy. Numbers of corpora lutea and live and dead implantations were recorded. Live fetuses were examined for external and visceral abnormalities. One-half of the fetuses were subsequently examined for skeletal abnormalities. No premature deaths or treatment-related clinical signs were recorded during the study. There was no treatment-related effect on mean maternal food consumption and body weight. However, when compared to the control group, a statistically significant reduction in body weight gain values was reported for the 40 mg/kg/day dose group (gestation days 9–12) and for the 100 mg/kg/day dose group (gestation days 6–15). There were no treatment-related effects on pregnancy parameters, mean fetal weight, and incidences of major external, visceral, or skeletal abnormalities.
When compared to the background range, a higher (but not statistically significant) increase in the incidence of minor skeletal abnormalities and variants (including incomplete ossification of the sacral neural arch) was observed in the vehicle control group and both dose groups. The historical control range was from 0% to 9%, compared to a value of 18.3% for the incidence of sacral neural arch incomplete ossification in the vehicle control group. Therefore, the authors noted the likelihood that the incidence of the skeletal minor abnormalities observed in both dose groups was also overestimated. Furthermore, they noted that these statistically non-significant skeletal findings in both dose groups were likely related to the transient, but statistically significant, decrease in body weight gain observed in dams (gestation days 9–12). This decrease was attributed to the moderate or severe skin lesions that led to pain and stress in the animals. Therefore, it was determined that the increase in the incidence of skeletal variations reported for fetuses from both groups were likely secondary to maternal toxicity (induced by local effects leading to pain and stress) and not indicative of a teratogenic effect. (Results relating to maternal skin irritation potential are included in the Skin Irritation section of this report.) A no-observed-adverse-effect-level (NOAEL) was not reported in this study summary. However, the following conclusion is reported in a summary of this study from a different source, “The NOAEL for developmental toxicity was established as 40 mg/kg/day, based on an increase in the incidence of fetuses with incomplete ossification of the sacral neural arch at 100 mg/kg/day.” 4 The NOAEL was not established for maternal toxicity, as treatment-related effects (local reaction at the site of administration and reductions in body weight gain) were observed at both doses tested.
Oral
The reproductive and developmental toxicity of Capryloyl Salicylic Acid (in PEG-6) was evaluated using groups of 20 (10 males, 10 females per group) Wistar Hannover rats. 3 The animals were dosed orally (by gavage) with the test substance once daily in accordance with OECD TG 421. The 3 dose groups received 10 mg/kg/day, 30 mg/kg/day, and 100 mg/kg/day, respectively (dose volume of 4 mL/kg/day). Male rats were dosed 2 weeks prior to mating, during the mating period, and up to 5 weeks post-mating (50 days total). Female rats were dosed 2 weeks prior to mating, during the mating period (up to 14 days), during gestation, and at least 4 additional days during the lactation period (40 to 49 days total). The vehicle control group was dosed with PEG-6. At the end of the dosing period, all parental animals were killed and subjected to gross necropsy. Histopathological examination of tissues was performed. There were no mortalities or clinical signs that were attributed to treatment with the test substance, and there were no effects on body weight and food consumption. The following parameters in treated animals were similar to those of the vehicle control group: reproductive performance of males and females, mating, fertility, gestation, and live birth indices. There were no treatment-related effects on weights of testes, epididymides, ovaries, uterus, and cervix. Furthermore, there were no treatment-related findings at necropsy or microscopic examination. There also were no effects on the clinical condition of pups, body weight, or sex ratio. No macroscopic findings were noted in pups that were killed on day 4 post-partum. The NOEL of Capryloyl Salicylic Acid was considered to be 100 mg/kg/day for the following: parental toxicity, embryo-fetal developmental toxicity, and pup development until day 4 post-partum.
Genotoxicity Studies
Genotoxicity Studies on Capryloyl Salicylic Acid.

In Vitro
The genotoxicity of Capryloyl Salicylic Acid (in ethanol) was evaluated in the Ames test using the following bacterial strains, with and without metabolic activation: Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2 uvr A. 3 Doses up to 1000 μg/plate were tested, and the test material was classified as non-genotoxic. The mammalian chromosome aberration test involving Chinese hamster ovary cells (with and without metabolic activation) was used to evaluate the genotoxicity of Capryloyl Salicylic Acid (in ethanol) at concentrations of 50 μg/mL and 80 μg/mL. 4 The test material was classified as clastogenic with, but not without, metabolic activation.
In Vivo
The genotoxicity of Capryloyl Salicylic Acid (in 0.5% carboxymethylcellulose aqueous vehicle) was evaluated in the micronucleus test using groups of 10 (5 males, 5 females per group) mice of the CD-1 strain. 3 A single dose of the test substance (250, 500, or 1000 mg/kg) was administered by gavage to the 3 groups. The test material was classified as non-clastogenic. In a second micronucleus test, Capryloyl Salicylic Acid (in 0.5% aqueous carboxymethylcellulose) was administered to groups of 10 (5 males, 5 females per group) Swiss CD-1 mice. 4 A single dose of the test material (500, 1000, or 2000 mg/kg) was administered by gavage to the 3 groups. A slight increase in the polychromatic erythrocytes (PCEs)/normochromatic erythrocytes (NCEs) ratio was observed in males of the 2000 mg/kg dose group, only at the 24-h sampling time. This finding was indicative of an inhibitory effect on erythropoietic cell division. Results indicated that the test material was non-clastogenic in this assay. (Results relating to the acute oral toxicity of this test material are included in that section of this report.) The unscheduled DNA synthesis test was also used to evaluate the genotoxicity of Capryloyl Salicylic Acid (in 0.5% aqueous carboxymethylcellulose), using groups of 4 Sprague-Dawley rats. 4 In 2 tests, single doses of 500, 1000, or 2000 mg/kg were administered by gavage, and the test material was classified as non-clastogenic in both. (Results relating to acute oral toxicity are included in that section of this report.)
Carcinogenicity Studies
Data on the carcinogenicity of Capryloyl Salicylic Acid were neither found in the published literature, nor were unpublished data submitted.
Dermal Irritation and Sensitization Studies
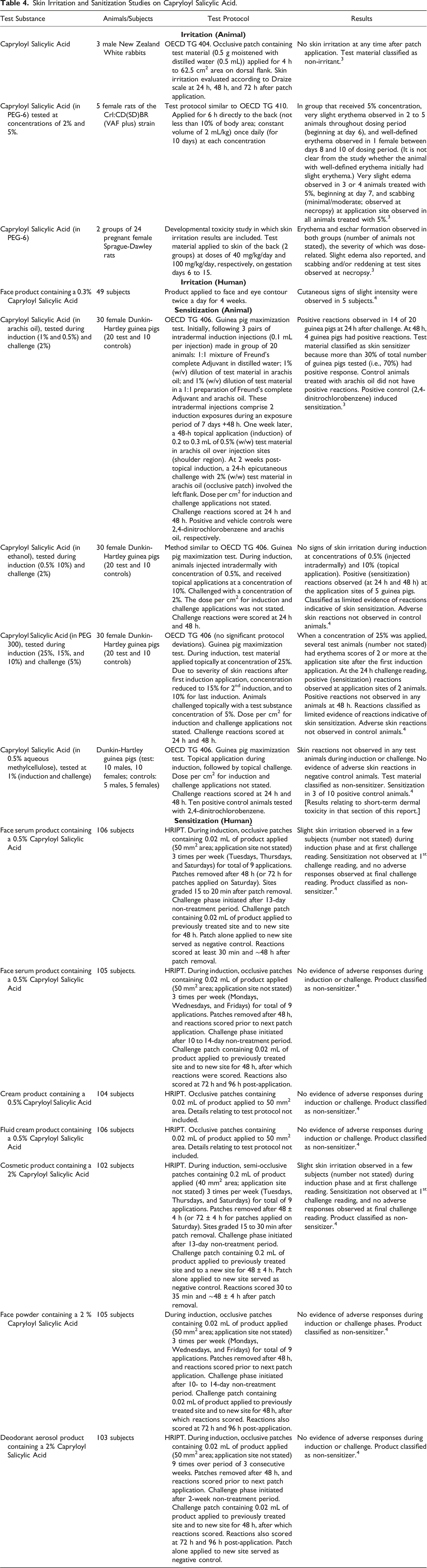
Skin Irritation and Sanitization Studies on Capryloyl Salicylic Acid.
The skin irritation potential of Capryloyl Salicylic Acid was evaluated in a 4-h occlusive patch test using 3 male New Zealand White rabbits. 3 The animals were patch tested with 0.5 g (62.5 mm2 patch) of the test material, and results were negative. In a study involving groups of 5 female rats of the Crl:CD(SD)BR (VAF plus) strain, Capryloyl Salicylic Acid (in PEG-6) was applied to the back (constant volume of 2 mL/kg) once daily for 10 days. The test material was applied at concentrations of 2% and 5%; only the 5% concentration caused very slight erythema (in 2 to 5 rats) and edema (3 or 4 rats). Minimal/moderate scabbing at the application site was also observed. (Additional results from this study are included in the Short-Term Dermal Toxicity section of this report.) Skin irritation data are reported in a developmental toxicity study (described previously) on Capryloyl Salicylic Acid (in PEG-6) involving 2 groups of 24 pregnant female Sprague-Dawley rats. 3 The test material was applied to skin of the back (2 groups) at doses of 40 mg/kg/day and 100 mg/kg/day, respectively, on gestation days 6 to 15. Erythema and eschar formation were observed in both groups (number of animals not stated), the severity of which was dose-related. Slight edema was also reported, and scabbing and/or reddening at test sites was observed at necropsy.
Reactions described as cutaneous signs of slight intensity were observed in 5 of 49 subjects after application of a face product containing 0.3% Capryloyl Salicylic Acid to the face and eye contour twice daily for 4 weeks. 4
The skin sensitization potential of Capryloyl Salicylic Acid (in arachis oil) was evaluated in the guinea pig maximization test, using 20 female Dunkin-Hartley guinea pigs. 3 Induction involved intradermal injection and topical application of 1% and 0.5% concentrations, respectively. This was followed by challenge with 2% (occlusive patch). The test material was classified as a skin sensitizer. In another maximization test, 20 guinea pigs were tested with Capryloyl Salicylic Acid (in ethanol). 4 Induction involved intradermal injection and topical application of 0.5% and 10% concentrations, respectively. There were no signs of skin irritation during induction. The animals were challenged with 2%. Sensitization reactions were observed (at 24 h and 48 h) at the application sites of 5 guinea pigs. However, these findings were classified as limited evidence of skin sensitization. The skin sensitization potential of Capryloyl Salicylic Acid (in PEG-300) was also evaluated in the maximization test, using 20 guinea pigs. 4 The induction phase involved concentrations of 25%, 15%, and 10%. When a concentration of 25% was applied, several test animals (number not stated) had erythema scores of 2 or more at the application site after the first induction application. The concentration was decreased during induction because of skin irritation, and induction was followed by topical challenge with 5%. At 24 h, but not 48 h, sensitization reactions were observed at the application sites of 2 animals. These findings were classified as limited evidence of skin sensitization. Capryloyl Salicylic Acid (in 0.5% aqueous methylcellulose) was evaluated for sensitization potential in the guinea pig maximization test, using 20 Dunkin-Hartley guinea pigs. 4 A test concentration of 1% was used for topical induction and challenge. Skin reactions were not observed in any of the test animals during induction or the challenge phase, and the test material was classified as a non-sensitizer. Results relating to short-term dermal toxicity reported in this maximization test are included in that section of this report.
In 2 human repeated insult patch tests (HRIPTs), involving 106 subjects in one study and 105 subjects in the other, a face serum product containing 0.5% Capryloyl Salicylic Acid was classified as a non-sensitizer. 4 In the study involving 106 subjects, slight skin irritation was observed in a few subjects (number not stated) during the induction phase and at the first challenge reading. A cream product and a fluid cream product (each containing 0.5% Capryloyl Salicylic Acid) were also classified as non-sensitizers in HRIPTs involving 104 subjects and 106 subjects, respectively. A cosmetic product containing 2% Capryloyl Salicylic Acid was classified as a non-sensitizer in an HRIPT involving 102 subjects. 4 Slight skin irritation was observed in a few subjects (number not stated) during the induction phase and at the first challenge reading. A face powder containing 2% Capryloyl Salicylic Acid and a deodorant aerosol product containing 2% Capryloyl Salicylic Acid were also classified as non-sensitizers in HRIPTs involving 105 subjects and 103 subjects, respectively. 4
Photosensitization/Phototoxicity
In Vitro
The phototoxicity of Capryloyl Salicylic Acid (100% pure, in dimethyl sulfoxide (DMSO)) was evaluated using the in vitro 3T3 neutral red uptake (NRU) phototoxicity test. 17 The study was performed in accordance with the OECD Guideline for Testing of Chemicals Draft Proposal for a New Guideline, issued in 2000. Balb/c 3T3 mouse fibroblasts (in microtiter plates) were treated with a range of test substance concentrations in 2 independent experiments. In the first experiment, Capryloyl Salicylic Acid was tested at concentrations ranging from 0.316 to 1000 μg/mL in the absence of ultraviolet (UV) light, and at concentrations ranging from 0.316 to 100 μg/mL in the presence of UV light. In the second experiment, Capryloyl Salicylic Acid was tested at concentrations ranging from 5 to 100 μg/mL in the absence of UV light, and at concentrations ranging from 1.25 to 30 μg/mL in the presence of UV light. Chlorpromazine (in phosphate-buffered saline (PBS)) and 1% v/v DMSO (in PBS) served as positive and negative controls, respectively. Both experiments involved the exposure of cells to the test substance, negative control, or positive control at a volume of 100 μL. For UV exposure of test plates, the long-wavelength ultraviolet (UVA) light source was described as a solar lamp fitted with a filter against mid-wavelength ultraviolet (UVB) emission. After a 1-h incubation period in the dark, test cultures were exposed to UVA for ∼40 min in order to achieve a UVA dose of 5 J/cm2.
At the end of the incubation period, the cells were examined microscopically for evidence of cytotoxicity. Optical densities (OD) of test plates were read at a wavelength of 540 nm (i.e., OD540), and mean ODs were calculated. Half maximal inhibitory concentration (IC50) values were calculated for the test substance and positive control using a curve fitting program. Photo-irritation factors (PIF = (IC50 in the absence of UVA) ÷ (IC50 in the presence of UVA)) for the test substance and positive control were also calculated. According to the evaluation criteria that were used, a test article was considered to be phototoxic in this assay if a marked decrease in cell viability (as measured by OD540 in the NRU) was observed in the presence of UVA (by comparison with the viability seen in the absence of UVA), such that PIF values of ≥5 were obtained. A test article was considered to be non-phototoxic in this assay if there was no marked decrease in cell viability when cells were exposed to the test article in the absence and presence of UVA, or if similar toxic profiles were observed in the absence and presence of UVA (PIF <5). The test yielded PIFs of 4 (experiment 1) and 2.6–1.7 (experiment 2). Based on these PIF values, the author concluded that, according to the proposed OECD guideline evaluation criteria, Capryloyl Salicylic Acid was not phototoxic in the in vitro 3T3 NRU phototoxicity test. The negative and positive controls yielded acceptable responses, and the study was considered valid. In light of the author’s conclusion, it should be noted that, according to the current OECD TG for the in vitro 3T3 NRU phototoxicity test (OECD TG 432), PIF values are to be interpreted based on the following criteria: a test substance with a PIF of <2 predicts “no phototoxicity,” a PIF of >2 and <5 predicts “probable phototoxicity,” and a PIF of >5 predicts “phototoxicity.” 18 Therefore, using these criteria, Capryloyl Salicylic Acid would have been classified as probably phototoxic.
Ocular Irritation Studies
Animal
The ocular irritation potential of Capryloyl Salicylic Acid was evaluated using 3 New Zealand White rabbits, according to OECD TG 405. 3 The test substance (∼65 mg) was instilled into the eye, and exposure was not followed by ocular rinsing. Reactions were scored at 24 h, 48 h, and 72 h after instillation, and the animals were observed for a total of 14 days after exposure. Diffuse corneal opacity, iridial inflammation, and moderate or severe conjunctivitis were observed in all treated eyes at 1 h post-instillation and in 2 treated eyes at 24 h, 48 h, and 72 h post-instillation. In the eye of 1 treated animal, corneal opacity increased and areas of opalescent corneal opacity with iridial inflammation and moderate to severe conjunctival irritation were observed at 48 h and 72 h post-instillation. Adverse effects observed on the nictitating and/or conjunctival membranes in the eye of this animal were described as pale appearance, small green-colored or white areas, and areas of hemorrhage. In another rabbit, diffuse corneal opacity, iridial inflammation, and minimal conjunctival irritation persisted in the treated eye on day 7; these effects were resolved by day 14. Circumcorneal vascularization and convoluted eyelids were also noted in this animal, and the nictitating membrane was also pale in appearance. In the third rabbit, corneal opacity increased and opalescent corneal opacity with pannus formation (indicative of irreversible ocular damage) had developed on day 14. It was not possible to assess iridial inflammation at this time, and minimal conjunctival irritation with convoluted eyelids were also observed. Capryloyl Salicylic Acid was classified as a severe irritant to the rabbit eye.
Human
A face product containing 0.3% Capryloyl Salicylic Acid was applied to the face and eye contour of 49 subjects twice a day for 4 weeks. 4 The group included subjects with sensitive skin, sensitive eyes, non-sensitive eyes, and contact lenses wearers. According to the authors, “observations were recorded before application and at least 10 min after the first application and the last application respectively.” Microscopic examinations of ocular and periocular structures revealed no appearance of ocular physical signs or palpebral signs. Colorimetric examinations of the cornea and the conjunctiva revealed a maximal corneal index of 0.50% and a maximal conjunctival index of 0%, indicating an absence of toxicity to the conjunctiva and a very slight toxicity to the cornea. Clinical examinations revealed an ocular irritation rate of 0.03% and an ocular comfort rate of 99.83%. Additionally, the product did not induce any pathology that was specific to contact lenses wearers.
An eye contour product containing 0.3% Capryloyl Salicylic Acid was applied to the face and eye contour of 50 subjects twice a day for 4 weeks. 4 The group included subjects with sensitive eyes, non-sensitive eyes, and contact lenses wearers. Observations were recorded before application and at least 10 min after the first application and the last application, respectively. The product induced moderate ocular burning in 1 subject with sensitive eyes, and slight ocular stinging in 1 contact lenses wearer. Biomicroscopic examinations of ocular and periocular structures revealed 2 bilateral occurrences of bulbar conjunctival redness in 2 subjects. Colorimetric examinations of the cornea and the conjunctiva revealed a maximal corneal index of 0% and a maximal conjunctival index of 0%, indicating an absence of toxicity to the conjunctiva and very slight toxicity to the cornea. Clinical examinations revealed an ocular irritation rate of 0.04% and an ocular comfort rate of 99.88%. Additionally, the product did not induce any pathology that was specific to contact lenses wearers.
Clinical Studies
Case Reports
A female patient who used day and night creams containing Capryloyl Salicylic Acid presented with dermatitis of the face, which was first observed 3 months earlier. 19 Positive patch test reactions (+) to both products and to Capryloyl Salicylic Acid (1% in alcohol) were reported. Another female patient who used the same night cream containing Capryloyl Salicylic Acid also presented with facial dermatitis and had a positive patch test reaction to this ingredient (1% in alcohol).
A female patient presented with a pruritic erythematous rash that arose on her face 10 days after application of a cream containing Capryloyl Salicylic Acid (concentration not stated). A positive allergic reaction (++) to 1% Capryloyl Salicylic Acid in alcohol was observed in the patient (at 48 h and 96 h), but not in 15 healthy control subjects.
In a letter to the editor on the preceding 2 case reports, the author stated that Capryloyl Salicylic Acid is unlikely to be significantly allergenic, and is therefore unlikely to be the cause of the contact allergy reported. 7 However, the structural isomer, 3-capryloyl salicylic acid, is a highly plausible contaminant of Capryloyl Salicylic Acid and is likely to be sufficiently allergenic to account for the observed contact allergy.
Other Clinical Reports
In a split-face study, 44 female volunteers with mild to moderate facial hyperpigmentation and fine lines/wrinkles were randomized, and a Capryloyl Salicylic Acid containing peel was applied to one side of the face. 20 Increasing peel concentrations were applied (5–10% Capryloyl Salicylic Acid) based on the tolerance level of the subjects and clinical observations of an expert dermatologist for 12 weeks at biweekly intervals. Results indicated that there were no significant changes in erythema for Capryloyl Salicylic Acid from baseline values when compared with pre-peel to pre-peel and post-peel to post-peel at different weeks.
Summary
Capryloyl Salicylic Acid was previously, erroneously, defined as the ester of salicylic acid and caprylic acid. However, is has become apparent that Capryloyl Salicylic Acid is, instead, the 5-capryl ketone of salicylic acid. Capryloyl Salicylic Acid can be manufactured via Friedel-Crafts acylation of methyl salicylate with capryloyl chloride and aluminum trichloride, to yield the methyl ester of Capryloyl Salicylic Acid, which is then hydrolyzed with sodium hydroxide, after which acidification yields this ketone, Capryloyl Salicylic Acid.
Capryloyl Salicylic Acid is reported to be used in 104 cosmetic products (93 leave-on and 11 rinse-off). The results of a concentration of use survey conducted in 2018 indicate that Capryloyl Salicylic Acid is used at concentrations up to 0.5% (in moisturizing products, not spray), which is the highest reported maximum use concentration for leave-on formulations. In rinse-off products, Capryloyl Salicylic Acid is used at concentrations up to 0.4% (in paste masks and mud packs), which is the highest reported maximum use concentration for rinse-off formulations.
In vitro skin penetration data (human skin samples) indicate that, after 16 h of contact, ∼6% of the applied Capryloyl Salicylic Acid was found to “penetrate deeper than the stratum corneum.” Using a standard tape stripping method for determining skin penetration (number of subjects not stated), it was determined that 17.1% of Capryloyl Salicylic Acid applied to the skin of human subjects was found in the stratum corneum over a 4-day period.
The acute dermal toxicity of Capryloyl Salicylic Acid was studied using Sprague-Dawley rats (5 males, 5 females). An LD50 of >2000 mg/kg was reported. The following adverse dermal reactions were observed after a single dermal dose of 2000 mg/kg: edema and blanching and hard, light brown colored scabs at the application site.
The acute oral toxicity of Capryloyl Salicylic Acid (in peanut oil) was evaluated using groups of 10 Sprague-Dawley rats (5 males, 5 females/group). The highest dose administered was 4217 mg/kg. The combined oral LD50 (males + females) was 3354 mg/kg (95% confidence limit between 2834 and 3970 mg/kg). Abnormalities observed at necropsy of animals (2530 g/kg group) that were killed at the end of the study were described as occasional white foci (∼1 mm × 1 mm covering 75% of the non-glandular gastric epithelium). At necropsy, there were no abnormalities in animals that received a dose of 3000 mg/kg or higher and were killed at the end of the study.
Results relating to the acute oral toxicity of Capryloyl Salicylic Acid (in 0.5% carboxymethylcellulose aqueous vehicle) are reported in a micronucleus test involving groups of 10 (5 males, 5 females per group) Swiss CD-1 mice. A single dose of the test substance (500, 1000, or 2000 mg/kg) was administered by gavage to the 3 groups. Two female mice and 1 male mouse dosed with 2000 mg/kg died. Results relating to acute oral toxicity of Capryloyl Salicylic Acid (in 0.5% carboxymethyl-cellulose aqueous vehicle) were also reported in an unscheduled DNA synthesis test using groups of 4 Sprague-Dawley rats. Single doses of 500, 1000, or 2000 mg/kg were administered by gavage, and no mortalities or clinical signs were observed.
A short-term (10-day) dermal toxicity study on Capryloyl Salicylic Acid (in PEG-6) was performed using groups of 5 female rats of the Crl:CD(SD)BR (VAF plus) strain. The test substance was applied for 6 h directly to the back once daily at concentrations of 2% and 5%. The NOEL for local and systemic effects was considered to be 2% and >5%, respectively. Results relating to short-term dermal toxicity of Capryloyl Salicylic Acid (in 0.5% aqueous methylcellulose) are reported in a guinea pig maximization test involving 20 Dunkin-Hartley guinea pigs. One animal was found dead on day 18, but microscopic examination revealed no apparent abnormalities.
A short-term (28 day) oral toxicity study on Capryloyl Salicylic Acid (PEG-6) was performed using groups of 10 or 20 rats of the CRL:CD(SD)BR strain. The highest dose group (initially 300 mg/kg/day) consisted of 20 rats (10 males, 10 females per group). Each of the remaining dose groups (10, 30, and 100 mg/kg/day) consisted of 10 rats (5 males, 5 females per group). The NOEL for local effects was 30 mg/kg/day, and the NOEL for systemic effects was >100 mg/kg/day.
The developmental toxicity of Capryloyl Salicylic Acid (in PEG-6) was evaluated using groups of 24 pregnant female Sprague-Dawley rats. The test material was applied for 6 h directly to skin on the back (not less than 10% of the body area). One group was dosed with 40 mg/kg/day and the other group was dosed with 100 mg/kg/day on gestation days 6 to 15. The NOAEL for developmental toxicity was established as 40 mg/kg/day, based on an increase in the incidence of fetuses with incomplete ossification of the sacral neural arch at 100 mg/kg/day. The NOAEL was not established for maternal toxicity, as treatment-related effects (local reaction at the site of administration and reductions in body weight gain) were observed at both doses tested. The reproductive and developmental toxicity of Capryloyl Salicylic Acid (in PEG-6) was evaluated using groups of 10 male and 10 female Wistar Hannover rats. The animals were dosed orally (by gavage) with the test substance once daily. The 3 dose groups received 10 mg/kg/day, 30 mg/kg/day, and 100 mg/kg/day, respectively (dose volume of 4 mL/kg/day). Males were dosed for 50 days total and females were dosed for 40 to 49 days total. The NOEL was considered to be 100 mg/kg/day for the following: parental toxicity, embryo-fetal developmental toxicity, and pup development until day 4 post-partum.
In the Ames test, Capryloyl Salicylic Acid (in ethanol) was non-genotoxic in S. typhimurium strains TA98, TA100, TA1535, and TA1537 and E coli strain WP2 uvr A when evaluated at doses up to 1000 μg/plate with and without metabolic activation. Capryloyl Salicylic Acid (in ethanol) was classified as clastogenic with, but not without, metabolic activation, in the mammalian chromosome aberration test involving Chinese hamster ovary cells. The test material was evaluated at concentrations of 50 μg/mL and 80 μg/mL in this assay.
Capryloyl Salicylic Acid (in 0.5% aqueous carboxymethylcellulose) was classified as non-clastogenic when evaluated in in vivo micronucleus tests using groups of 5 male and 5 female mice of the CD-1 strain. Single doses of the test substance up to 2000 mg/kg were administered by gavage. The in vivo unscheduled DNA synthesis test was also used to evaluate the genotoxicity of Capryloyl Salicylic Acid (in 0.5% aqueous carboxymethylcellulose) using groups of 4 Sprague-Dawley rats. Single doses of 500, 1000, or 2000 mg/kg were administered by gavage, and the test material was classified as non-clastogenic.
When 3 male New Zealand White rabbits were patch tested with 0.5 g of Capryloyl Salicylic Acid, skin irritation was not observed. However, skin irritation was reported in other types of toxicity tests. In a short-term dermal toxicity study, daily (for 10 days) applications of Capryloyl Salicylic Acid (in PEG-6) at a concentration of 5% caused very slight erythema and edema in a group of 5 female rats of the Crl:CD(SD)BR (VAF plus) strain. Scabbing was observed in all 5 rats at necropsy. Very slight erythema was observed in 2 animals and well-defined erythema in 1, and very slight edema was observed in 2 or 4 animals.
In a developmental toxicity study, Capryloyl Salicylic Acid (in PEG-6) was applied to the backs of pregnant female Sprague-Dawley rats (2 groups of 24) at doses of 40 mg/kg/day and 100 mg/kg/day, respectively, on gestation days 6 to 15. Erythema and eschar formation, dose-related in severity, were observed (number of animals not stated).
Reactions described as cutaneous signs of slight intensity were observed in 5 of 49 subjects after application of a face product containing 0.3% Capryloyl Salicylic Acid to the face and eye contour twice daily for 4 weeks.
Three maximization tests involved groups of 20 Dunkin-Hartley guinea pigs tested with Capryloyl Salicylic Acid (different vehicles used). A challenge concentration of 2% (after induction with 0.5% and 10%) was sensitizing to 5 of 20 guinea pigs, and a challenge concentration of 5% (after induction with 10%, 15%, and 25%) was sensitizing to 2 of 20 guinea pigs. The absence of skin sensitization was noted in a group of 20 guinea pigs challenged with a concentration of 1% (after induction with 1%). Additionally, findings relating to skin irritation (number of animals with reactions not stated) during induction were reported in the 3 maximization tests. A test concentration of 25% applied topically was irritating, whereas, 0.5% (injected intradermally) and 10% (topical application) were not. In a fourth guinea pig maximization test, the following results reported after challenge with 2% Capryloyl Salicylic Acid: Positive reactions were observed in 14 of 20 guinea pigs at 24 h after challenge. At 48 h, 4 guinea pigs had positive reactions. The test material was classified as skin sensitizer because more than 30% of total number of guinea pigs tested (i.e., 70%) had a positive response. Positive reactions were observed at the 24-h challenge reading, but not at the 48-h reading.
In 2 HRIPTs involving 106 subjects and 105 subjects, respectively, face serum products containing 0.5% Capryloyl Salicylic Acid was classified as a non-sensitizer. In the HRIPT involving 106 subjects, slight skin irritation was observed in a few subjects (number not stated) during the induction phase and at the first challenge reading. A cream product and a fluid cream product (each containing 0.5% Capryloyl Salicylic Acid) were also classified as non-sensitizers in HRIPTs involving 104 subjects and 106 subjects, respectively. A cosmetic product containing 2% Capryloyl Salicylic Acid was classified as a non-sensitizer in an HRIPT involving 102 subjects. Slight skin irritation was observed in a few subjects (number not stated) during the induction phase and at the first challenge reading. A face powder containing 2% Capryloyl Salicylic Acid and a deodorant aerosol product containing 2% Capryloyl Salicylic Acid were also classified as non-sensitizers in HRIPTs involving 105 subjects and 103 subjects, respectively.
The phototoxicity of Capryloyl Salicylic Acid (100%, in DMSO) was evaluated using the in vitro 3T3 NRU phototoxicity test. The study was performed in accordance with the OECD Guideline for Testing of Chemicals Draft Proposal for a New Guideline, issued in 2000. In the first experiment, Capryloyl Salicylic Acid was tested at concentrations ranging from 0.316 to 100 μg/mL in the presence of UVA light. The second experiment involved test concentrations ranging from 1.25 to 30 μg/mL in the presence of UVA light. The test yielded PIFs of 4 (experiment 1) and 2.6–1.7 (experiment 2), and Capryloyl Salicylic Acid was classified as non-phototoxic. However, retroactive application of the current OECD test guideline, would probably have classified Capryloyl Salicylic Acid as phototoxic.
Capryloyl Salicylic Acid was classified as being a severe ocular irritant in a test involving 3 New Zealand White rabbits. The test substance (∼65 mg) was instilled into the eye, and exposure was not followed by ocular rinsing.
A face product containing 0.3% Capryloyl Salicylic Acid was applied to the face and eye contour of 49 subjects twice a day for 4 weeks. Clinical examinations revealed an ocular irritation rate of 0.03%. In another test, an eye contour product containing 0.3% Capryloyl Salicylic Acid was applied to the face and eye contour of 50 subjects twice a day for 4 weeks, and clinical examinations revealed an ocular irritation rate of 0.04%
Positive patch test reactions to 1% Capryloyl Salicylic Acid have been reported in case reports, one of which reported no reactions in a control group of 15 subjects.
Discussion
The Panel discussed the issue of skin sensitization potential for this ingredient. Capryloyl Salicylic Acid induced skin sensitization in guinea pig maximization tests at challenge concentrations of 2% (after induction with up to 10%) and 5% (after induction with up to 25%), but not at 1% (after induction with 1%). However, in HRIPTs, cosmetic products containing 0.5% or 2% Capryloyl Salicylic Acid were classified as non-sensitizing. After reviewing the HRIPT results and considering that the highest reported maximum use concentration of Capryloyl Salicylic Acid is 0.5% in leave-on cosmetic products, the Panel was reassured that the sensitization potential of exposure to this ingredient via cosmetic use is not a risk. Furthermore, dermatologists on the Panel stated that, based on their clinical experience, Capryloyl Salicylic Acid is not a sensitizer.
The Panel noted the absence of carcinogenicity data from this safety assessment. However, it was agreed that due to the predominance of negative genotoxicity data on Capryloyl Salicylic Acid and the absence of structural alerts in the chemical structure, the carcinogenic potential of this ingredient is not a concern.
The Panel also discussed the issue of incidental inhalation exposure from powders and hair sprays. The Council’s survey results indicate that Capryloyl Salicylic Acid is being used in deodorant sprays (aerosolized) at concentrations up to 0.3%. Also, Capryloyl Salicylic Acid is being used in face powders at concentrations up to 0.3%. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
In response to the Panel’s request for phototoxicity data, the results of an in vitro 3T3 NRU phototoxicity test were provided by the Council. The study was performed in accordance with the OECD Guideline for Testing of Chemicals Draft Proposal for a New Guideline (2000). According to the evaluation criteria that were used, a test article was considered to be phototoxic in this assay if a marked decrease in cell viability (as measured by OD540 in the NRU) was observed in the presence of UVA (by comparison with the viability seen in the absence of UVA) such that PIF values of ≥5 were obtained. A test article was considered to be non-phototoxic in this assay if there was no marked decrease in cell viability when cells were exposed to the test article in the absence and presence of UVA, or if similar toxic profiles were observed in the absence and presence of UVA (PIF <5). PIFs of 4 and 2.6–1.7 were reported in separate experiments that were performed. Based on these PIF values, the author concluded that, according to the proposed OECD TG evaluation criteria at that time, Capryloyl Salicylic Acid was not phototoxic in the in vitro 3T3 NRU phototoxicity test. However, the Panel noted that, according to the current OECD TG for the in vitro 3T3 NRU phototoxicity test, the results of this test are to be interpreted based on the following criteria: a test substance with a PIF of <2 predicts “no phototoxicity,” a PIF of >2 and <5 predicts “probable phototoxicity,” and a PIF of >5 predicts “phototoxicity.” Thus, the Panel agreed that, according to the current criteria, Capryloyl Salicylic Acid (PIFs of 4 and 2.6–1.7) would be classified as probably phototoxic in the in vitro 3T3 NRU phototoxicity test. Furthermore, the Panel agreed that because this test is prone to false positives, additional data would be needed in order to evaluate the phototoxicity potential of Capryloyl Salicylic Acid.
Impurities data were also previously requested by the Panel. Data on impurities were not provided, and the request for these data remains. Thus, the Panel determined that the available data are insufficient to determine the safety of Capryloyl Salicylic Acid as used in cosmetics, and the data needs include: • Impurities • Phototoxicity
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the available data are insufficient to make a determination of safety for Capryloyl Salicylic Acid under the intended conditions of use and concentration in cosmetic formulations.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
