Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Hydrogen Peroxide for use in cosmetics. This ingredient is reported to function in cosmetics as an antimicrobial agent, cosmetic biocide, oral health care agent, and oxidizing agent. The Panel reviewed the data relevant to the safety of this ingredient and concluded that Hydrogen Peroxide is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
This is a review of the safety of Hydrogen Peroxide as used in cosmetics. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), this ingredient is reported to function in cosmetics as an antimicrobial agent, cosmetic biocide, oral health care agent, and oxidizing agent. 1 The Dictionary also lists oral health care drug as a function of Hydrogen Peroxide. However, in the United States (US), this is not considered a cosmetic function, and therefore the Panel will not evaluate safety in relation to this function.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
There are several studies in this report that evaluate the safety of Hydrogen Peroxide mixed with an oxidative hair dye or hair dye ingredient in a 1:1 mixture. The resulting product is not a mixture containing the ingredients in proportional amounts, but instead is a reaction product of the two substances with little to no residual Hydrogen Peroxide. These studies are included in this safety assessment to acknowledge that Hydrogen Peroxide is an ingredient in hair dyes, particularly as an oxidizer.
Some of the data included in this safety assessment were found on the European Chemicals Agency (ECHA) website. 2 In this safety assessment, ECHA is cited as the references for summaries of information obtained from this website. Also referenced in this safety assessment are summary data found in reports made publically available by the European Commission’s (EC) Scientific Committee on Consumer Products (SCCP), 3 EC Scientific Committee on Cosmetic Products Non-Food Products Intended for Consumers (SCCNFP), 4 Australia’s National Industrial Chemicals Notification and Assessment Scheme, 5 and numerous other organizations. Reports by these organizations are cited in this assessment to identify the source of the summary data.
Chemistry
Definition and Structure
Hydrogen Peroxide is the inorganic oxide that conforms to the structure in Figure 1.
1
Hydrogen peroxide.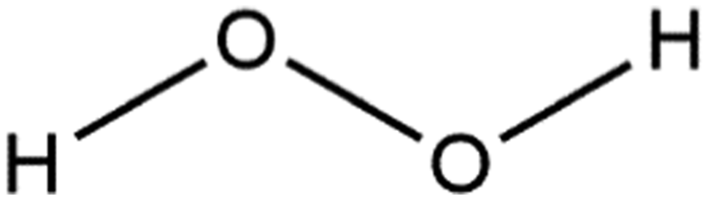
Chemical Properties
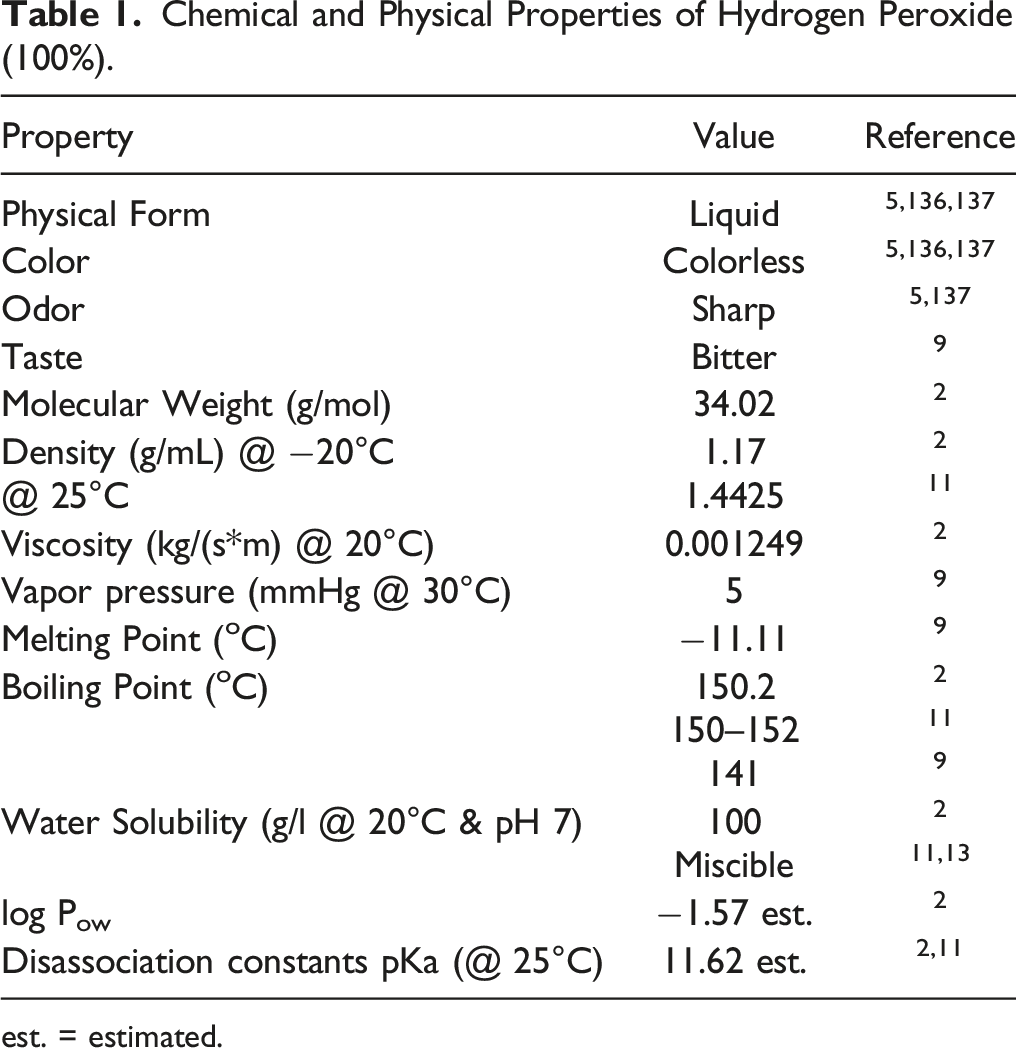
Chemical and Physical Properties of Hydrogen Peroxide (100%).
est. = estimated.
Aqueous solutions containing 35, 5, 70, or 90% Hydrogen Peroxide are the most commonly used solutions for industrial applications and in laboratory settings, and require a stabilizer (commonly acetanilide) to prevent rapid decomposition to water and molecular oxygen. Aqueous solutions of 3 to 6% are used for cosmetic and medical applications.
Hydrogen Peroxide and water do not form an azeotropic mixture (two or more liquids whose proportions cannot be altered or changed by simple distillation), and are completely separable. 6 The dissociation of Hydrogen Peroxide can be a violent and exothermic reaction.9,10 Hydrogen Peroxide is nonflammable, but it is a powerful oxidizing agent that can accelerate combustion when it comes in contact with organic material. Aqueous solutions of Hydrogen Peroxide, at low concentrations in clean inert containers, are relatively stable. 8 Stability is at a maximum in mildly acidic solutions between pH 3.5 to 4.5.
Natural Occurrence
The concentration of Hydrogen Peroxide in the environment results from a dynamic equilibrium between its production and degradation. 11 Hydrogen Peroxide may be formed in photochemical, chemical, or biochemical processes.
Hydrogen Peroxide is produced metabolically in intact cells and tissues. 8 It is formed by reduction of oxygen either directly in a two-electron transfer reaction, often catalyzed by flavoproteins, or via an initial one-electron step to a superoxide anion, followed by dismutation to Hydrogen Peroxide.
Method of Manufacture
Hydrogen Peroxide can be manufactured by anthraquinone autoxidation. 11 The anthraquinone derivate is hydrogenated to corresponding anthrahydroquinone using a palladium or nickel catalyst. Hydrogen Peroxide is formed when anthrahydroquinone solution is oxidized back to anthraquinone by bubbling air or oxygen through the solution. Crude Hydrogen Peroxide is extracted with water from the organic solution and the redox cycle is repeated with the generation of additional Hydrogen Peroxide. The extracted crude aqueous solution contains approximately 20 to 40% Hydrogen Peroxide and is normally purified in two or three stages by extraction with organic solvent. Finally, the aqueous solution is concentrated to give 50 to 70% Hydrogen Peroxide solutions.
Several other methods of manufacture have been reported. Hydrogen Peroxide can be manufactured by the electrolytic oxidation of sulfuric acid or a sulfate to persulfuric acid or a persulfuric acid salt with subsequent hydrolysis and distillation of the Hydrogen Peroxide that is formed; by decomposition of barium peroxide with sulfuric or phosphoric acid; by hydrogen reduction of 2-ethylanthraquinone, followed by oxidation with air, to regenerate the quinone and produce Hydrogen Peroxide; or by electrical discharge through a mixture of hydrogen, oxygen, and water vapor. [21CFR184.1366]
High concentration commercial Hydrogen Peroxide grades are stabilized to prevent or slow down decomposition and prevent possibly violent decomposition due to catalytic impurities or elevated temperatures and pressure.6,11 The stabilizers are of several types: mineral acids to keep the solution acidic (stability is at a maximum at pH 3.5 to 4.5); complexing/chelating agents to inhibit metal-catalyzed decomposition; or colloidal agents to neutralize small amounts of catalysts or adsorb/absorb impurities. The types of stabilizers used in Hydrogen Peroxide vary between producers and product grades and may have additional purposes. 12 For example, nitrate (sodium and ammonium) is used for pH adjustment and corrosion inhibition, and phosphoric acid is also used for pH adjustment. Colloidal silicate is used to sequester metals and thereby minimize Hydrogen Peroxide decomposition in certain applications that depend on the bleaching ability of Hydrogen Peroxide in alkali. In some applications, a high degree of stabilization is needed; whereas, in others (e.g., drinking water treatment or semiconductor manufacture) product purity is more important.
Chemicals Used to Stabilize Aqueous Hydrogen Peroxide. 11

Impurities
In the US, to meet the requirements of the Food Chemicals Codex, 30 to 50% aqueous solutions of Hydrogen Peroxide must pass an identification test and meet the following specifications: acidity (as sulfuric acid), 0.03% max; phosphate, 0.005% max; lead, 0.0004% max; tin, 0.001% max; and iron, 0.00005% max. 14
In commercial Hydrogen Peroxide manufactured for the purposes of medical and food biocides in Finland, none of the reported impurities were at concentrations greater than 0.1%. 6 The sum of organic and inorganic impurities in aqueous solution is reported to be below 0.2 w/w %. Calculated from a 35% aqueous solution of Hydrogen Peroxide, the theoretical total impurity contents is below 0.5 w/w %. In biocidal products, heavy metals in aqueous Hydrogen Peroxide are limited to a maximum of 1 mg/kg each of lead, mercury, cadmium, and arsenic.
Use
Cosmetic
The safety of the cosmetic ingredient included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentration by product category.
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for Bath Product Uses.
Note: Because this ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
aIt is possible these products
bIt is possible these products
cThe 15% hair-coloring product is a professional developer that is diluted as needed. The next highest maximum concentration of use in this category is 12.4% in hair dyes and colors.
The results of the concentration of use survey conducted by the Council in 2017 indicate that Hydrogen Peroxide is used at a maximum concentration of 15%; this use is in the category of “other” hair coloring preparations. 16 The product that contains 15% Hydrogen Peroxide is a professional 50 volume developer, and standard dilutions include 10, 20, 30, and 40 volume (i.e., 3, 6, 9, and 12% Hydrogen Peroxide, respectively).
The highest maximum concentration of use reported in hair dyes and colors is 12.4%. 16 Permanent hair dyes, also called oxidative dyes, are the most common type of hair dye. 17 The hair is dyed by oxidation of precursors which penetrate the hair fiber, where they react with Hydrogen Peroxide to produce dyes. Since Hydrogen Peroxide is an excellent decolorizing agent for melanin, the hair’s natural coloring matter, manufacturers can balance the amounts of Hydrogen Peroxide and of dye precursors in such a way as to produce lightening, darkening, or matching of the natural color of the hair.
According to the Council survey, Hydrogen Peroxide is being used at up to 12% in hair bleaches. 16 Hair bleaching methods are oxidative processes, 17 and Hydrogen Peroxide is the most common oxidant used in hair bleaching. Hydrogen Peroxide can be used alone to bleach hair, but in hairdressing salons, it is mixed with an alkaline solution, typically comprising aqueous ammonia (in part), before use in order to accelerate the process.
When using hair dyes or relaxers, the FDA recommends that consumers follow all directions in the package, perform a 48-h patch test on the skin before using the dye on hair, wear gloves, and rinse the scalp well with water after use. 18 Consumers should not dye eyebrows or eyelashes, or leave the product on longer than the directions say. For more information, the FDA’s informational website is https://www.fda.gov/forconsumers/byaudience/forwomen/ucm118527.htm.
Hydrogen Peroxide is also used in products that can result in incidental oral ingestion; the highest reported maximum concentration of use in oral hygiene formulations is in dentifrices at up to 4.6%. Formulations containing Hydrogen Peroxide can come in contact with the skin, and the maximum concentration of use for leave-on dermal exposure is 2.5% in “other skin care preparations.” Hydrogen Peroxide is also reported to be used in the category of baby lotions, oils and creams at up to 0.0019% and in formulations that are used near the eyes at up to 0.000002% (eye lotions).
Additionally, Hydrogen Peroxide is used in cosmetic sprays and could possibly be inhaled; for example, it is reported to be used at up to 4% in aerosol hair sprays. In practice, 95 to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared with pump sprays.19,20 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.21,22
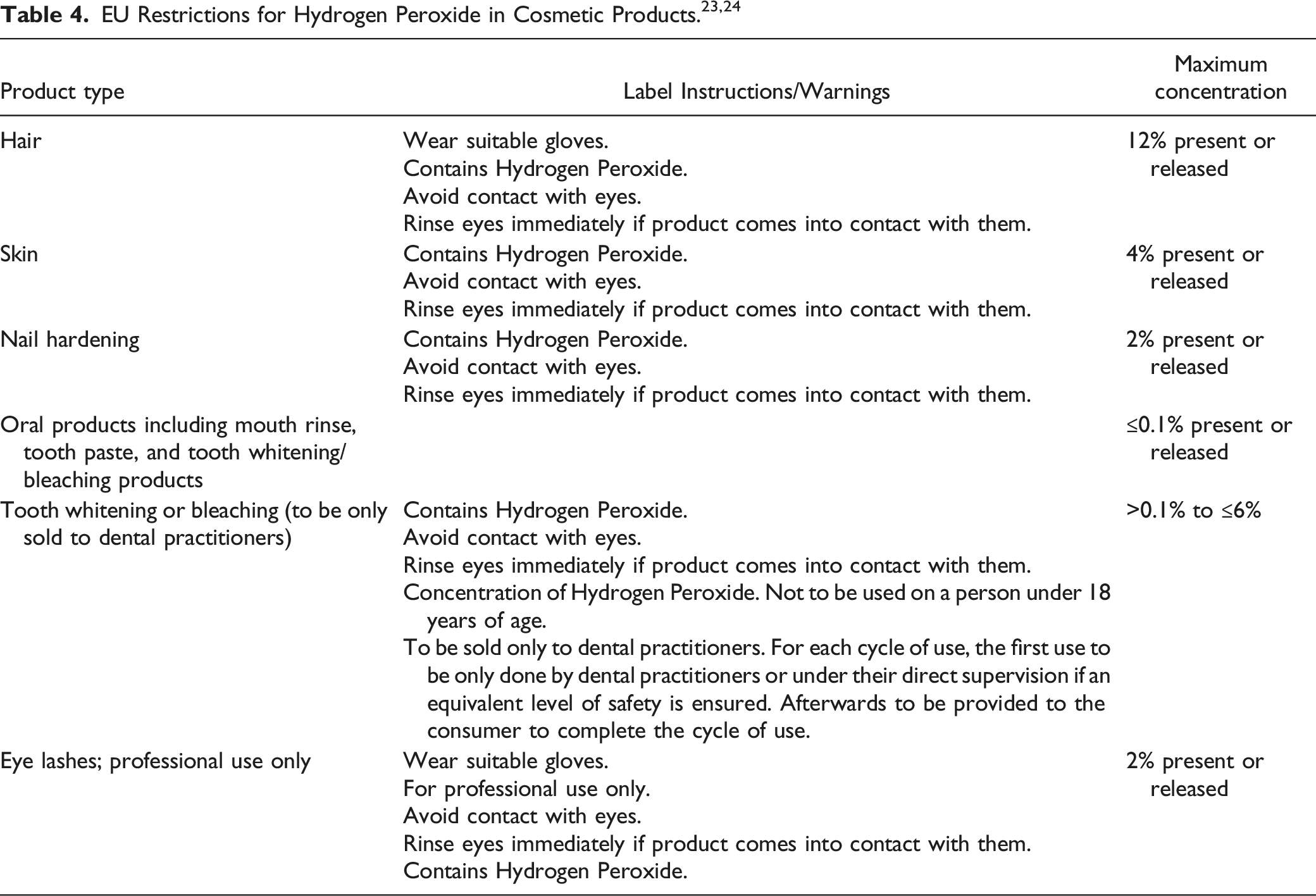
Limits for Hydrogen Peroxide in oral care products are included in European Union (EU) cosmetic regulations 25 ; an SCCP opinion formed the basis of these limits. 3 According to Part 1 of Annex III to Directive 76/768/EEC, the maximum authorized concentration of Hydrogen Peroxide in finished oral products in the EU, including mouth rinse, tooth paste and tooth whitening or bleaching products, is ≤ 0.1% (present or released). 25 In addition, tooth whitening or bleaching products containing more than 0.1%, but less than 6%, Hydrogen Peroxide should only be sold to dental practitioners and used by those over the age of 18. Based on a no-observable-adverse-effects level (NOAEL) of 20 mg/kg/day (Hydrogen Peroxide (concentration not specified) in a 100-day rat gavage study and an estimated daily exposure to toothpaste of 480 mg/day Hydrogen Peroxide (0.1% aq.)), the estimated margin of safety (MOS) was calculated to be 2500. 3 Based on an estimated daily exposure to mouth-rinse of 3000 mg/day Hydrogen Peroxide, the MOS was calculated to be 400.
NICNAS conducted a Tier II assessment (evaluation of risk on a substance-by-substance or chemical category-by-category basis) on Hydrogen Peroxide under the Multi-tiered Assessment and Prioritisation Framework (IMAP). 5 In that assessment, it was noted that Hydrogen Peroxide, in hair dyes containing 3, 6, or 12% Hydrogen Peroxide, is both a Schedule 5 (caution – substances with a low potential for causing harm, the extent of which can be reduced through the use of appropriate packaging with simple warnings and safety directions on the label) and a Schedule 6 (poison – substances with a moderate potential for causing harm, the extent of which can be reduced through the use of distinctive packaging with strong warnings and safety directions on the label) substance, according to the Australian Government Poisons Standards. 26 It is advised that consumers using products containing Hydrogen Peroxide follow the directions on the label to avoid harm.
Non-Cosmetic
Food
U.S. FDA and EPA Regulations on Hydrogen Peroxide in Food Preparation.

EPA = Environmental Protection Agency; FDA = Food and Drug Administration.
In the US, the FDA recognizes Hydrogen Peroxide as generally recognized as safe (GRAS) to treat food under specific conditions outlined in the Code of Federal Regulations (CFR); maximum treatment levels range from 0.04% to 1.25%, or as an amount sufficient for the purpose. [21CFR184.1366] Hydrogen Peroxide may be used in several capacities in food preparation (bleaching agent, emulsifier, epoxidizing agent). [21CFR172.182, 21CFR172.814, 21CFR172.892, 21CFR172.723] It may be used in adhesives that come in contact with food. [21CFR175.105] Hydrogen Peroxide is also permitted to be used as an antimicrobial agent in bottled water (in a silver nitrate solution), to sterilize food-contact surfaces, and in solutions to clean food-processing equipment and utensils. [21CFR172.723, 21CFR178.1005]
The US Environmental Protection Agency (EPA) stipulates that Hydrogen Peroxide, when used as an ingredient in an antimicrobial pesticide formulation, may be applied to food-contact surfaces in public eating places, and food-processing equipment and utensils; when it is ready for use, the end-use concentration is not to exceed 91 ppm (0.0091%) Hydrogen Peroxide. [40CFR180.940]
Hydrogen Peroxide (not to exceed 200 ppm; 0.02%) is used to reduce the bisulfite aldehyde complex in distilling materials for processing spirits. [27CFR24.247]
Over the Counter (OTC)
Aqueous Hydrogen Peroxide has been historically present in oral mucosal injury drug products for use as an oral wound healing agent. Oral wound healing agents have been marketed as aids in the healing of minor oral wounds by means other than cleansing and irrigating, or by serving as a protectant. Hydrogen Peroxide in aqueous solution is safe up to 3% for use as oral wound healing agents, 27 but there are inadequate data to establish general recognition of its effectiveness for this purpose [21CFR310.534]
Hydrogen Peroxide has a history of use as a first aid antiseptic. 28 Based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of Hydrogen Peroxide for the specified uses of external analgesic drug products to treat dermal poison ivy, poison oak, and poison sumac reactions. [21CFR310.545]
Medical
Hydrogen Peroxide (20% aq.) has been used to treat corneal ulcerations, particularly in herpetic dendritic keratitis. 29 A product containing Hydrogen Peroxide (40%) has been approved for the treatment of seborrheic keratoses that are raised. 30
In an assessment to establish a permissible daily exposure (PDE) of Hydrogen Peroxide, the FDA Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER), in its guidance for the use of reactive chemicals in drugs, stated that even though Hydrogen Peroxide is genotoxic, Hydrogen Peroxide is endogenously produced in the body at such high levels as to exceed the levels encountered in oral care and other personal care products. 31 Therefore it was not considered appropriate to derive a PDE based on carcinogenicity data. Even an intake 1% of the estimated endogenous production of 6.8 g/day, that is, 68 mg/day (or 68,000 μg/day) would not significantly add to the background exposure of Hydrogen Peroxide in the body.
In veterinary medicine, Hydrogen Peroxide is commonly used as an emetic at a concentration of 3%. 32
Agricultural
Hydrogen Peroxide is GRAS as a general purpose food additive for animal feed, when used as a bleaching agent in accordance with good manufacturing practices (GMP) or feeding practices. [21CFR582.1366; 40CFR180.940] An exemption from the requirement of a tolerance is established for residues of Hydrogen Peroxide in or on all food commodities at the rate of ≤ 1% Hydrogen Peroxide per application on growing and postharvest crops. [40CFR180.1197]
Other
In a safety assessment of Hydrogen Peroxide in household products, the Human and Environmental Risk Assessment (HERA) program concluded that the use of Hydrogen Peroxide in household cleaning products raises no safety concern for consumers. 7 In the US, Hydrogen Peroxide is used as a 90% solution in rocket propulsion. 11
Toxicokinetic Studies
Dermal Penetration
Hydrogen Peroxide is reactive, and degrades rapidly, due to reactions with all classes of organic biomolecules. 6 The rapid degradation upon contact with skin explains the absence of systemic effects from dermal exposure to Hydrogen Peroxide. However, it is possible that application of Hydrogen Peroxide solutions to damaged skin, or exceptional cases with excessive amounts of exogenous Hydrogen Peroxide on skin, may result in some systemic exposure. If Hydrogen Peroxide does penetrate the skin, it is presumed to degrade rapidly into molecular oxygen and water when in contact with blood or other body fluids; therefore, measurement of dermal penetration would not be possible. Despite the fact that Hydrogen Peroxide is a normal metabolite in cell metabolism and that Hydrogen Peroxide metabolism is understood (e.g., through catalase and glutathione peroxidase enzymes), data on the effects of exogenous Hydrogen Peroxide exposure in humans or animals are limited and mainly consist of case reports of oxygen embolization following the degradation of Hydrogen Peroxide after exposure to large amounts. No standard dermal penetration studies with Hydrogen Peroxide have been successfully conducted. Based on the physico-chemical properties of Hydrogen Peroxide, 100% dermal penetration should be used in the absence of more accurate information. 6
After application of 5 to 30% solutions of Hydrogen Peroxide on rat skin in vivo, some Hydrogen Peroxide could be localized in the excised epidermis within a few minutes. 11 By contrast, with human cadaver skin in vitro, only after the application of high Hydrogen Peroxide concentrations for several hours, or after pretreatment with hydroxylamine (inhibitor of catalase), was Hydrogen Peroxide detectable in the dermis. Based on histochemical analysis, Hydrogen Peroxide was not metabolized in the epidermis, and the passage was transepidermal, avoiding the “preformed pathways” of skin appendages. The localization of dermal emphysema, caused by liberation of oxygen, correlated for the most part with the distribution of catalase activity within the tissue.
Absorption, Distribution, Metabolism, and Excretion (ADME)
Hydrogen Peroxide is a normal metabolite in aerobic cells. 11 Hydrogen Peroxide passes readily across biological membranes. Under normal, physiological conditions, the concentration of Hydrogen Peroxide in tissues is 1 to 100 nM (0.034 to 3.4 μg/L) depending upon the organ, cell type, oxygen pressure, and cell metabolic activity. 33
In biological systems, Hydrogen Peroxide is metabolized by catalase and glutathione peroxidases. 33 The highest activities are found in highly vascularized tissues such as the duodenum, liver, kidney, and mucous membrane. 34 In the metabolism of Hydrogen Peroxide to water and oxygen, the decomposition rate in human plasma is approximately 0.01 to 0.05 M/min. Catalase is more efficient at the decomposition of higher concentrations of Hydrogen Peroxide; glutathione peroxidase is more efficient at decomposing lower Hydrogen Peroxide concentrations. 35 Glutathione peroxidase is present in cytosol and mitochondria but not in peroxisomes. A high glutathione peroxidase reduction activity of Hydrogen Peroxide is found in liver and erythrocytes; moderate levels are found in the heart and lungs, and a low activity is present in muscle.
In the presence of transition metals in cells, Hydrogen Peroxide can be reduced via the Haber-Weiss reaction. 36 This reaction produces hydroxyl radicals (free radicals) which are highly reactive and can result in lipid peroxidation.
At high uptake rates, Hydrogen Peroxide can pass the absorption surface and enter the adjacent tissues and blood vessels, where it is rapidly degraded by catalases and molecular oxygen is liberated.11,33 Consequently, mechanical pressure injury and oxygen embolism may be produced. In the view of the high degradation capacity for Hydrogen Peroxide in blood, it is unlikely that it is systemically distributed; therefore, the endogenous steady state levels of the substance in tissues are unlikely to be affected.
In rat blood diluted 1000 times, the half-life of Hydrogen Peroxide was less than 5 min at both 5 and 10 mg/L. 6 For 20 mg/mL, the half-life was more than 4 h. In the study, concentrations of Hydrogen Peroxide were much greater than the range of aqueous solutions in products or in-use concentrations. The study demonstrates the high efficacy of the antioxidative system in blood. Furthermore, it supports the view that if Hydrogen Peroxide is entering blood circulation, it is rapidly decomposed in blood and will not be systemically available. For this reason, the distribution of Hydrogen Peroxide in the body is expected to be very limited after exposure to Hydrogen Peroxide solutions. Due to the rapid endogenous transformation into water and oxygen, there is no specific excretion of Hydrogen Peroxide or a determinable degradation product. 33
Inhalation
Anesthetized rabbits (number and strain not specified) were administered aerosolized 1 to 6% aq. Hydrogen Peroxide by inhalation. 11 The left atrial blood was found to be supersaturated with oxygen up to levels that corresponded to oxygen administration at 3 atm. When the amount of Hydrogen Peroxide was increased, small bubbles began to appear in the blood samples. The amount of arterial oxygen was the same with both 1 and 6% Hydrogen Peroxide. No further details were provided.
Mucosal
Administration of Hydrogen Peroxide solutions to body cavities lined by mucous membranes, such as via sublingual, intraperitoneal, and rectal administration, resulted in increased oxygen content of the draining venous blood and, if the amounts of Hydrogen Peroxide were sufficiently high, formation of oxygen bubbles. 11 Mongrel dogs were treated with dilute saline solutions of Hydrogen Peroxide by colonic lavage or by lavage of the small and large bowel via an enterotomy. Small amounts of a more concentrated solution (1.5% aq. or higher) produced immediate whitening of the mucosa, with prompt appearance of bubbles in the circulation. More dilute (0.75% to 1.25% aq.) solutions had the same effect when left in contact with the bowel for a longer time (not specified) or when introduced under greater pressure or in greater volume for a given length of bowel. Venous bubbling was never observed at concentrations less than 0.75% aq. Hydrogen Peroxide. In none of the dogs did mesenteric thrombosis or intestinal gangrene develop. Application of 1% aq. Hydrogen Peroxide to the serosal membrane caused whitening due to gas-filled small vessels; higher concentrations (up to 30% aq.) on the skin and mucous membranes (of various species) caused lasting damage when subcutaneous emphysema and disturbances of local blood circulation impaired tissue nutrition.
In two cats, sublingual application of 1.5 mL of 9% aq. 18O-labeled Hydrogen Peroxide or .1 mL 19% aq. 18O-labeled Hydrogen Peroxide was followed up with mass spectrometric analyses in arterial (femoral artery) blood and exhaled air. Within approximately 1 h in the former case, and within half an hour in the latter case, one-third of the labeled oxygen was exhaled. There was a rapid initial rise of the arterial blood 18O concentration, but the arterial blood oxygen saturation gradually declined, probably because of impaired gas exchange in the lung due to oxygen embolism. 11
Toxicological Studies
Acute Dose Toxicity
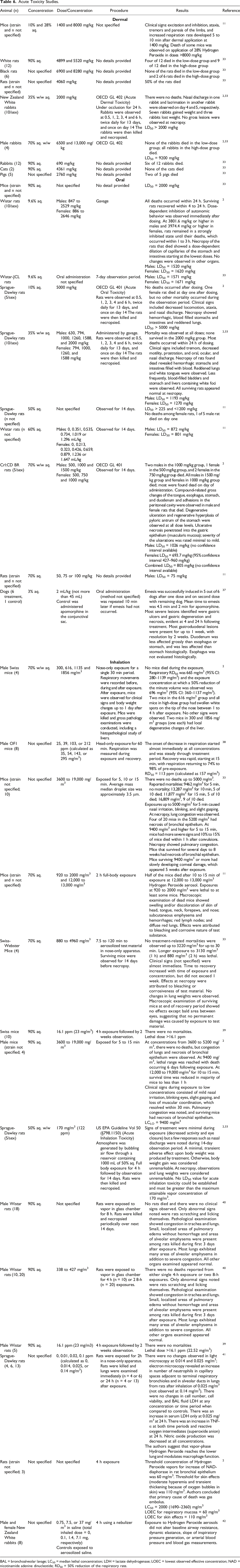
Acute Toxicity Studies.
BAL = bronchoalveolar lavage; LC50 = median lethal concentration; LDH = lactate dehydrogenase; LOEC = lowest observed effective concentration; NAD = nicotinamide adenine dinucleotide; RD50 = 50% reduction of the respiratory rate.
Animal
Dermal
In general, the acute dermal toxic effects of Hydrogen Peroxide were dependent on concentration as well as dose. The dermal LD50 was >8000 mg/kg Hydrogen Peroxide in mice; more mice died when the dose was applied at a higher concentration (28 vs 10% aq.). 11
Dermally administered Hydrogen Peroxide (90% aq.) caused 4 of 12 rats to die at 4899 mg/kg and 9 of 12 to die at 5520 mg/kg. 33 In one study, dermally administered Hydrogen Peroxide (concentration not specified) did not cause any (n = 6) rats to die at 6900 mg/kg and 2 of 6 to die at 8280 mg/kg. In another study, 50% of the rats (n not specified) died at 4060 mg/kg (concentration not specified).
Dermal LD50s in rabbits were >2000 mg/kg, 9200 mg/kg, and 690 mg/kg with 35% aq., 70% aq., and 90% Hydrogen Peroxide, respectively, that was administered under occlusion for 24 h.2,33 Clinical signs included lacrimation and nasal discharge.
No cats died when 90% aq. Hydrogen Peroxide was dermally administered at 4361 mg/kg 33 Two of 5 pigs died when dermally administered 2760 mg/kg Hydrogen Peroxide (concentration not specified). 33
Oral
In general, the acute oral toxic effects of Hydrogen Peroxide were dependent on concentration as well as dose. The oral LD50 of Hydrogen Peroxide (90% aq.) for mice was reported to be 2000 mg/kg. 33
Oral LD50s in rats ranged from 1520 mg/kg to >5000 mg/kg with approximately 10% aq. Hydrogen Peroxide. 2,33 The LD50s at 35% aq. Hydrogen Peroxide were 1193 mg/kg in male rats and 1270 mg/kg in females.2,33 At 60% aq. Hydrogen Peroxide, the LD50s in rats were 872 mg/kg in males and 801 mg/kg in females. 11 At 70% aq. Hydrogen Peroxide, the LD50 in rats ranged from 75 mg/kg to 1026 mg/kg.2,5,11 Clinical signs in rats administered 35% aq. Hydrogen Peroxide and greater included tremors, decreased motility, prostration, and oral, ocular, and nasal discharge. Most rats that died had reddened lungs, hemorrhagic and white stomachs, and blood-filled intestines; some had white tongues.2,5,33
Dogs administered one or two oral doses (manner of administration not specified) of Hydrogen Peroxide (2 mL/kg; 3% aq.) vomited within 4.5 min; the most severe lesions identified were gastric ulcers and gastric degeneration and necrosis, evident at 4 and 24 h following treatment. 37
Inhalation
In inhalation studies, increasing concentrations of Hydrogen Peroxide and times of exposure had increasingly corrosive effects on the pulmonary tract. Mortality rates increased accordingly.
In mice, the concentration at which a 50% reduction of the respiratory rate was observed (RD50) was 665 mg/m3 Hydrogen Peroxide (70% aq.) and the exposure concentration at which a 50% reduction of the minute volume was observed was 696 mg/m3. 2 In another experiment, the RD50 in mice (n = 8) was 113 ppm (calculated as 157 mg/m3; concentration of Hydrogen Peroxide not specified) when exposure was 60 min. 38 At up to 5000 mg/m3 Hydrogen Peroxide (concentration not specified) there were no deaths but there was necrosis of the bronchial epithelium and pulmonary congestion; half of the 10 mice died after 10 to 15 min of exposure at 11,877 and 13,287 mg/m3. 33 Exposure to 920 to 2000 mg/m3 Hydrogen Peroxide (70% aq.) via inhalation was lethal to at least some mice (n not specified). 11 At necropsy, subcutaneous emphysema and hemorrhages, red lymph nodes, and diffuse red lungs were observed. No treatment-related mortalities were observed in mice exposed to up to 3220 mg/m3 Hydrogen Peroxide (70% aq.) for up to 30 min; longer exposure to 3130 mg/m3 (1 h) and 880 mg/m3 (2 h) was lethal to all 4 mice. 33 One study reported the lethal dose for mice to be >16.1 ppm (calculated as 23 mg/m3; only test dose) Hydrogen Peroxide (90% aq.) when exposed for 4 h. 39 In mice, concentrations of 3600 to 5200 mg/m3 Hydrogen Peroxide (90% aq.), there was no mortality, but congestion of lungs and necrosis of bronchial epithelium were observed; at 9400 mg/m3, lethal range (LCLO) was reached with death occurring 6 days following exposure. 2
In rats, at 170 mg/m3 Hydrogen Peroxide (50% aq.), clinical signs were minimal during inhalation exposure (decreased activity and eye closure), but a few responses, such as nasal discharge, were observed.2,33 There were no deaths in rats exposed to vaporized 90% aq. Hydrogen Peroxide for 8 h; at necropsy, most lungs exhibited many areas of alveolar emphysema and severe congestion. 40 There were no deaths when rats were exposed to 338 to 427 mg/m3 of 90% aq. Hydrogen Peroxide vapor in a glass chamber. 40 In another study, the lethal dose in rats exposed to 90% aq. Hydrogen Peroxide by inhalation was >16.1 ppm (only test dose; calculated as 23 mg/m3). 39 In rats exposed to vapor-phase Hydrogen Peroxide in a nose-only apparatus for 2 h, there were no changes observed in light microscopy at 0.014 and 0.025 mg/m3; however, electron microscopy revealed an increase in the number of neutrophils in capillary spaces adjacent to terminal respiratory bronchioles and in alveolar ducts in lungs from rats after inhalation of 0.025 mg/m3. 41 In one study in which rats were exposed to Hydrogen Peroxide vapors for 4 h, the median lethal concentration (LC50) was 2000 mg/m3, the lowest-observed-effective-concentration (LOEC) for respiratory mucosa effects was 60 mg/m3, and the LOEC for skin effects (moderate hyperemia and transient thickening because of oxygen bubbles in skin) was 110 mg/m3. 2 Exposure to Hydrogen Peroxide aerosols (up to 37 mg/m3) for 4 h did not alter baseline airway resistance, dynamic elastance, slope of inspiratory pressure generation, or arterial blood pressure and blood gas measurements in rabbits. 42
Human
Dermal
In humans, Hydrogen Peroxide administered to the skin has been reported to cause transient (lasting 10 to 15 min after 1 min exposure) dermal blanching starting at 3% aq. 43
Inhalation
Human exposure by inhalation may result in extreme irritation and inflammation of the nose, throat and respiratory tract, pulmonary edema, headache, dizziness, nausea, vomiting, diarrhea, irritability, insomnia, hyper-reflexia, tremors and numbness of extremities, convulsions, unconsciousness, and shock. 29 The latter symptoms are a result of severe systemic poisoning.
Subjects (n = 11) were exposed to Hydrogen Peroxide (30% aq.; 0, 0.5, and 2.2 ppm; calculated as 0, 0.7, and 3.08 mg/m3) vapors for 2 h at rest in an exposure chamber (20 m3). 44 Symptoms related to irritation and central nervous system (CNS) effects were rated with Visual Analog Scales. The ratings varied considerably but were generally low and with no significant differences between exposure conditions, although the ratings of smell, nasal irritation, and throat irritation showed borderline tendencies to increase at 3.08 mg/m3, but not at 0.7 mg/m3. Nasal airway resistance increased after exposure to 3.08 mg/m3, but not at 0.7 mg/m3. No exposure-related effects on pulmonary function, nasal swelling, breathing frequency, and blinking frequency were detected. No clear effects were seen on markers of inflammation and coagulation (e.g., interleukin-6, C-reactive protein, serum amyloid A, fibrinogen, factor VIII, von Willebrand factor, and Clara cell protein in plasma). The authors concluded that Hydrogen Peroxide was slightly irritating at 3.08 mg/m3, but not at 0.7 mg/m3.
In 32 subjects, the threshold of detection for irritation through inhalation exposure was 10 mg/m3 (independent of the exposure time, which was from 5 min to 4 h) when Hydrogen Peroxide (concentration not provided) vapor was inhaled through the nose using a face mask. 5
Short-Term Toxicity Studies
Dermal
No published short-term dermal toxicity studies were discovered and no unpublished data were submitted.
Oral
Oral Repeated Dose Studies of Hydrogen Peroxide.

DFG = Deutsche Forschungsgemeinschaft; LOAEL = lowest observed adverse effects level; LOEL = lowest observed effects level; NOEL = no observed effects level; NOAEL = no observed adverse effects level; SGOT = serum glutamic oxaloacetic transaminase; SGPT = serum glutamic pyruvic transaminase.
In general, orally administered Hydrogen Peroxide caused inflammation of and erosion to the upper digestive tract of mice and rats. Mice administered Hydrogen Peroxide (0.3 and 0.6%) in drinking water had decreased body weights over 2 weeks; mice died when administered 1% Hydrogen Peroxide or greater. 33 In another 2-week study in mice exposed to Hydrogen Peroxide in drinking water, the mice had reduced water consumption and weight gains at 1000 mg/L and greater; at necropsy, degenerative (minimal to mild erosions) and regenerative (minimal to mild hyperplasia) changes in the mucosa of the stomach and/or duodenum in the 3000 and 6000 mg/L groups in both sexes were observed. 33 The overall NOAEL for pathology was 1000 mg/L for both sexes.
In a 3-week drinking water study in rats of 0.45% Hydrogen Peroxide, there was a decrease in fluid consumption and body weights; there were no differences in relative weights in testes, kidneys, spleen, or heart. 45 In a 40-day oral study in rats, no deaths were reported and no toxic effects were observed at doses <30 mg/kg/day (via gavage), but blood effects (reduction of hematocrit values, blood plasma proteins concentrations, and plasma catalase activity) were observed at 60 mg/kg; the lowest-observed-adverse-effects-level (LOAEL) was 30 mg/kg/day. 46 In another gavage study in rats, administration of 1/5 and 1/10 of the LD50 (actual dose not specified) for 45 days caused blood effects (increased blood peroxidase activity) and inflammatory responses in the stomach wall. 33 In an 8-week drinking water study, 7 of 24 rats died in the 1.5% Hydrogen Peroxide group; dose-dependent extensive carious lesions and pathological changes in the periodontium were observed. 47 In a 12-week oral gavage toxicity study in rats, there were no mortalities at up to 506 mg/kg of 5% aq. Hydrogen Peroxide, but there were blood effects (reduced hemoglobin concentration, erythrocyte count, blood corpuscle volume, serum glutamic-oxaloacetic transaminase (SGOT), and serum glutamic pyruvic transaminase (SGPT)) at this dose; there were also changes to the weights of kidneys, livers, and hearts (decreased) and to adrenal glands and testes (increased).3,33
Inhalation
Inhalation Repeated Dose Studies of Hydrogen Peroxide.

LOAEL = lowest observed adverse effect level; MAO = monoamine oxidase; NOAEL = no observed adverse effects level; NOEL = no observed effects level; OECD = Organisation for Economic Co-operation and Development; SDH = succinate dehydrogenase activity.
Mice exposed to Hydrogen Peroxide (90% aq.; 79 or 107 mg/m3) for 6 h per day for 2 to 3 day per week, for up to 4 weeks, had nasal discharge, edematous feet, and irritation of the skin at week 2 and hair loss around the nose (probably due to scratching due to irritation) at week 5; seven of nine mice died after eight exposures in the low-dose group, and in the high dose group, five of 10 mice died after eight exposures and eight of 10 died after 18 exposures. 40 Rats exposed to aerosolized Hydrogen Peroxide (50% aq.) 5 days per week, 6 h per day, for 28 days showed clinical signs at 14.6 mg/m3 (including reddened nose, salivation, irregular breathing), but not at 2.88 mg/m3; the no-observed-effects-level (NOEL) was 2.9 mg/m3 and the LOAEL was 14.6 mg/m³. 2 Rats exposed to 93 mg/m3 Hydrogen Peroxide (90% aq.) for 6 h per day for 2 to 5 days per week for 7 weeks (30 exposures) showed signs of nasal irritation and profuse discharge at 2 weeks, lung congestion and hair loss (probably due to scratching due to irritation) at 5 weeks. 40 In black rabbits exposed to 90% Hydrogen Peroxide (30 mg/m3) vapor for 6 h per day, 5 days per week for 12 weeks, there were no effects observed except for the bleaching of the fur and some irritation around the nose. 40
Subchronic Toxicity Studies
Dermal
Shaved rats (strain and n not specified) were exposed to Hydrogen Peroxide vapor (0.1 to 10.1 mg/m3) 5 h per day, 5 day per week, for up to 4 months in whole body chambers.2,33 After 2 months at 1 mg/m3, examination of the epidermis of the backs of the rats revealed an increase in the activity of monoamine oxidase (MAO) and nicotinamide adenine dinucleotide (NAD)-diaphorase, and after 4 months, an increase in MAO, NAD-diaphorase, succinate dehydrogenase activity (SDH), and lactate dehydrogenase. At 4 months, there was significant dysfunction of the horny layer of the skin. The lowest-observed-effect-level (LOEL) was 1.0 mg/m3 and the NOEL was 0.1 mg/m3 for enzyme activities in the skin.
Nine hair dye formulations (1 mL/kg) each in 1:1 mixtures with Hydrogen Peroxide (6% aq.) were applied to the clipped dorsal lateral skin of the thoracic-lumbar area of New Zealand White rabbits (n = 6/sex) twice daily for 13 weeks. 48 The applications were alternated between the sides of the rabbits to minimize dermal irritation. The skin of three rabbits/sex in each group was abraded before the first treatment. The rabbits were restrained for 1 h after application, and then the application sites were shampooed, rinsed, and dried. Three separate control groups (n = 12/sex) were treated the same as the treatment groups without the hair dye.
There was no evidence of test substance-induced toxicity observed. Body weight gains of all test groups were similar to controls. Five control and five test rabbits died during the study due to complications during cardiac puncture to collect blood. There were some differences in the clinical chemistry and hematologic values between test and control groups at the various sampling intervals that were not considered to be of toxicological significance because of either the direction or continuity of the differences or the fact that they fell within the range of historical control values. There were a few instances when there were differences in relative organ weights between a test group and the combined controls, however there were no differences when the group was compared with each control group separately. In no instance were any of the relative organ weight differences accompanied by histological evidence of toxicity. The results of the urinalyses were unremarkable. The treated skin showed slight thickening in some groups, which was expected due to the frequency of dye application. No gross abnormalities were seen at necropsy, and no microscopic lesions were seen that were deemed to be due to the administration of the hair dye formulations containing Hydrogen Peroxide. The incidence and severity of disease processes common to laboratory rabbits was not affected by the experimental treatments. 48
Oral
Subchronic oral toxicity studies summarized below are presented in Table 7.
In an approximately 90-day drinking water study in mice, the overall LOEL was 300 ppm and the overall NOEL was 100 ppm (26 and 37 mg/kg/day for males and females, respectively) based on dose-related reductions in feed and water consumption and duodenal mucosal hyperplasia. 2 All effects noted during the treatment period were reversible. In a 100-day dietary study in rats, there were no deaths reported when Hydrogen Peroxide was administered in feed at up to 60 mg/kg. 46
Inhalation
Subchronic inhalation studies summarized below are presented in Table 8.
In rats exposed to Hydrogen Peroxide (concentration not specified) in whole body chambers for 5 h per day, 5 days per week for up to 4 months, the threshold for lung effects was 10 mg/m3; the NOEL was 1 mg/m3 and the LOEL was 10 mg/m3.2,33 There were no mortalities when rats were exposed to Hydrogen Peroxide (50% aq.) up to 10.3 mg/m3 for 6 h per day, 5 days per week, for 13 weeks; the NOAEL was 3.6 mg/m3 for male and female rats for decreased liver and thymus weights. 2 Irritation was noted around the nose of rabbits exposed to 90% aq. Hydrogen Peroxide at 22 ppm (calculated as 30.77 mg/m3) for 3 months. 29
Chronic Toxicity Studies
Oral
Chronic oral toxicity studies summarized below are presented in Table 7.
In a 100-week drinking water study of Hydrogen Peroxide (0.1% and 0.4% aq.) in mice, erosion in the stomach occurred after 40 weeks, duodenal hyperplasia after 55 weeks.49,50 In a 6-month gavage study in rabbits, the NOAEL was 0.005 mg/kg/day due to changes in hematology and enzyme activities. 2
Inhalation
The chronic inhalation study summarized below is presented in Table 8.
In two dogs exposed to aerosolized 90% Hydrogen Peroxide (10 mg/m3) for 6 h per day, 4 to 5 days per week for 26 weeks, the only observed effects were fur bleaching and loss at 14 weeks, and sporadic sneezing and lacrimation at 23 weeks. 40 At necropsy at 26 weeks, the lungs had areas of atelectasis and emphysema, and there was some hyperplasia in bronchial musculature.
Developmental and Reproductive Toxicity (dart) Studies
Dermal
The teratogenicity of nine oxidative hair dye formulations was tested using Charles River CD rats (n = 20). 48 The oxidative formulations were each mixed 1:1 with Hydrogen Peroxide (6% aq.) immediately prior to application (2 mL/kg/day) to shaved backs. The test materials were applied to the shaved dorsoscapular area of pregnant rats on every third gestation day (GD days 1, 4, 7, 10, 13, 16, and 19). Positive controls were administered oral acetylsalicylic acid (250 mg/kg) on GD 6 to 16. Three separate negative control groups were shaved, but not treated. No maternal toxicity was observed, there were no treatment effects on implantation or intrauterine growth, or survival, and there was no evidence of external, visceral, or skeletal malformation.
Six composite test materials, representative of commercial oxidative hair dye formulations, were evaluated in Sprague-Dawley rats (n = 20/sex) in a two-generation study of reproduction. 51 The dyes were each mixed 1:1 with Hydrogen Peroxide (6% aq.) and then applied (0.5 mL) twice weekly to the clipped backs of the rats. The treatment of the F0 rats began at 6 to 8 weeks of age, and rats of the second litter (F1b) began treatment at weaning. Breeding for both generations began at 100 days of age, and dermal applications continued throughout mating, gestation, and lactation periods. Occasional mild dermatitis was the only adverse effect noted. Body weight gain, feed consumption, survival, and reproductive indices (fertility, gestation, live birth and survival, and weaning weight) in F1a, F1b, F2a, and F2b litters were similar to controls.
Oral
Hydrogen Peroxide (30% aq.; 0.33% and 1%) was administered to male mice (strain and n not specified) in drinking water for 7, 21, or 28 days premating.2,4 After mating, the female mice were also administered Hydrogen Peroxide (0, 0.33% or 1%) in drinking water. All mated female mice became pregnant, the pups were healthy, and the litters were of normal size. Pregnant mice in the high-dose group had some delay in parturition compared to controls; however, the effect was small and inconsistent. The concentration, morphology, and motility of the spermatozoa (tested in three mice) after 3 weeks of treatment appeared normal.
Female and male Osborne-Mendel rats (n not specified) were administered Hydrogen Peroxide (0.45%) in drinking water for 5 months prior to mating. 45 The females continued to be treated through parturition. Six of the male offspring were also administered Hydrogen Peroxide (0.45%) in drinking water for 9 months. The litters of the treated females were normal. The only observed effect in the male offspring was a statistically significant reduction in weight (411 g vs 521 g in controls).
Aqueous solutions of Hydrogen Peroxide (0.02%, 0.1%, 2%, or 10%) were mixed with powdered feed and administered to pregnant Wistar rats (n = 7 to 11) for 1 week “during the critical period of pregnancy” (no further explanation was provided; no control group was specified).
11
Most of the dams (n = 5 to 8) were killed and the pups removed and examined on gestation day 20; some (n = 2 to 3) were allowed to deliver their pup and were followed for 4 weeks. The body weights of the dams in the high-dose group did not increase markedly. Fetal resorptions were increased and the fetal body weights were decreased. Most of the fetuses were close to death
It was reported that the concentration of Hydrogen Peroxide in the feed decreased to 1/10 after 24 h and to almost zero at 72 h. The authors of the study stated that “the amount of residue was determined and consumption was estimated;” however, it is not stated how frequently fresh feed was prepared. Nevertheless, it seems likely that the dams did ingested Hydrogen Peroxide, evidenced by that there was not much of an increase in dam body weight at the high-dose level. The authors proposed that the observed effects on fetal development were due to the breakdown of essential nutrients in food by Hydrogen Peroxide.
Male and female rats (strain and n not specified) were administered Hydrogen Peroxide (0.005 to 50 mg/kg; 1/10 to 1/5 LD50; vehicle not specified) by gavage for 6 months. 52 Females had modified estrus cycles and the males had decreased sperm mobility. The rats were then mated. At the highest dose, 3 out of 9 females produced litters, compared to 7 out of 9 in the control group (not described). Body weights of the offspring in the high-dose group were reduced compared to controls.
Genotoxicity Studies
Genotoxicity Studies of Hydrogen Peroxide.

2 -AAF = acetamidfluorene; CHL = Chinese hamster lung; CHO = Chinese hamster ovary; DMEM = Dulbecco’s modified Eagle’s medium; DMN = dimethylnitrosamine; ER = erythrocyte ratio; HPRT = hypoxanthine-guanine phosphoribosyltransferase; i.p. = intraperitoneal; LC-MS/MS = liquid chromatography-tandem mass spectrometry; MEM = minimal essential medium; MPE = micronucleated polychromatic erythrocytes; NE = normochromatic erythrocyte; NNG = net nuclear grain count; OECD GL = Organisation of Economic Co-operation and Development Guideline; PBS = phosphate buffered saline; PE = polychromatic erythrocyte; SCE = sister chromatid exchange; SHE = Syrian hamster embryo; SOD = superoxide dismutase; UDS = Unscheduled DNA Synthesis.
In Vitro
There are numerous genotoxicity studies of Hydrogen Peroxide. A representative sample is presented here. When available, the starting concentration of the Hydrogen Peroxide tested is stated in Table 9.
The results in Ames assays conducted on Hydrogen Peroxide were not consistent. In most of the Ames assays presented, Hydrogen Peroxide (concentrations not specified in most assays) increased the number of revertant colonies in Salmonella typhimurium strains without metabolic activation (3% or 30% in those assays with concentrations of Hydrogen Peroxide provided)53-58; however, there were a few assays where the results were negative for genotoxicity (3% in two of these assays).33,56,58-60 In one assay with metabolic activation, 3% Hydrogen Peroxide was mutagenic in strain TA100, but not in TA98, TA1535, TA1537, and TA1538, 58 and in another, Hydrogen Peroxide (concentrations not specified) without metabolic activation was weakly mutagenic in strain TA102, but less genotoxic with metabolic activation.33,59 In Ames assays in Escherichia coli, Hydrogen Peroxide was positive for genotoxicity in one assay (concentrations not specified), 61 and negative for genotoxicity in another assay (3% Hydrogen Peroxide). 58 Results were ambiguous for S. typhimurium TA100 up to 7.5 μmol/plate 62 and positive for TA102 at 75 μg/plate without metabolic activation. 61 In one other assay, Hydrogen Peroxide was genotoxic to E. coli without metabolic activation and not mutagenic with metabolic activation. 63 In other Ames-type assays, Hydrogen Peroxide (concentrations not specified) was mutagenic in E. coli, Bacillus subtilis, and Saccharomyces cerevisiae.64-66
In bacterial forward mutation assays, Hydrogen Peroxide (30% aq.) was genotoxic to E. coli K12 kat(−) and kat(+) strains at 75 and 600 nmol/ml, respectively,
67
and to E. coli (DB2) starting at 24 μg/ml (concentrations not specified).
68
Hydrogen Peroxide was genotoxic to B. subtilis (168DB) at 0.0005% aq.
65
Hydrogen Peroxide (concentration not specified) was not genotoxic to Chinese hamster lung fibroblast (V79) cells up to 300 μmol.
69
In an
In a chromosomal aberration test, Hydrogen Peroxide (concentration not specified) was genotoxic to Chinese hamster ovary (CHO) cells starting at 10 nL/mL with metabolic activation and 25.31 nL/mL without metabolic activation, 2 and in a second test, Hydrogen Peroxide (30% in saline) was genotoxic to Chinese hamster fibroblasts at 0.25 mg/plate. 71 Hydrogen Peroxide (concentration not specified) was also mutagenic to murine splenocytes, 72 V79 cells,69,73 and Syrian hamster embryo (SHE) cells69,74 in chromosomal aberration tests. Hydrogen Peroxide (concentration not specified) was mutagenic to human leukocytes and embryonic fibroblasts.33,75 Hydrogen Peroxide increased the number of abnormal metaphases in CHO-K1 cells without, but not with, metabolic activation. 69
In mouse lymphoma assays, Hydrogen Peroxide increased the mutation frequency in mouse lymphoma cells without metabolic activation.2,76 Hydrogen Peroxide did not increase the mutation frequency in mouse lymphoma cells with metabolic activation.
In various assays, Hydrogen Peroxide had mixed results in V79 cells.69,77-80 In sister chromatid exchange (SCE) assays, Hydrogen Peroxide (concentrations not specified) was mutagenic in V79 cells without metabolic activation, but was not mutagenic, or was mutagenic to a lesser extent, with metabolic activation.73,78,81,82 Hydrogen Peroxide (concentrations not specified) was mutagenic in CHO cells starting at 10 to 20 μM without metabolic activation, but was not mutagenic, or was mutagenic to a lesser extent, with metabolic activation.78,83-87 Hydrogen Peroxide (concentrations not specified) increased the number of SCEs at 300 μM in SHE cells. 88 Hydrogen Peroxide (concentrations not specified) was mutagenic at 20 μM in human lymphocytes but not in whole blood; metabolic activation reduced Hydrogen Peroxide-induced SCEs. 84 Hydrogen Peroxide (concentrations not specified), without metabolic activation, was mutagenic to D98/AH2 human cells. 89
In an endo-reduplicated cells assay, Hydrogen Peroxide (concentrations not specified) was mutagenic to CHO AUXB1 cells starting at 160 μM in a dose-dependent manner. 85 In an unscheduled DNA synthesis (UDS) in mammalian cells assay using rat hepatocytes, Hydrogen Peroxide (35.7% aq.) caused a dose-dependent increase in net nuclear grain (NNG) values at 6.25 to 25 μg/ml. 2
In comet assays, Hydrogen Peroxide was genotoxic in mouse lymphoma cells, 90 rat hepatocytes, 91 S. cerevisiae, 92 and V79 cells (40 μM; 37% aq.). 93 Hydrogen Peroxide was also mutagenic to human breast adenocarcinoma cell lines MCF-7 and MCF-10A, 94 human lymphocytes, 93,95 human fibroblasts (30 μM; 37% aq.),93,96 immortalized cervical cancer (HeLa) cells (30 μM),93,97 and liver hepatocellular carcinoma (HEP G2) cells (40 μM; .3 M).93,98 In a combined comet assay/micronucleus assay using human lymphoblastoid TK6 cells, Hydrogen Peroxide (concentration not specified) was genotoxic in the comet assay at 50 μM and at 100 μM in the micronucleus assay. 99 In another combined comet assay/micronucleus assay in V79 cells, Hydrogen Peroxide was genotoxic at 80 μM and at 40 μM, respectively. 100
In a multi-test assay, Hydrogen Peroxide (concentration not specified) was not genotoxic in two DNA adduct assays up to 500 μM (but was genotoxic at 500 μM in a comet assay), at 20 μM in a micronucleus test, and at 100 μM in a tk+/− gene mutation assay, all using L5178Y tk+/− mouse lymphoma cells. 90 Hydrogen Peroxide (37% aq.) was not mutagenic at 110 μM in V79 cells in a hypoxanthine-guanine phosphoribosyltransferase (HPRT) assay. 96
In Vivo
Animal
In a mammalian erythrocyte micronucleus test using mice, Hydrogen Peroxide, administered by intraperitoneal injection (i.p.), was not genotoxic at up to 2000 mg/kg. 2 Hydrogen Peroxide, administered in drinking water, was not genotoxic to mice at up to 536 and 774 mg/kg/day for males and females, respectively, in a second mammalian erythrocyte micronucleus test. 2 In an UDS test, intravenously (i.v.) administered Hydrogen Peroxide (50 mg/kg) did not induce UDS in rats. 2 Hydrogen Peroxide (200 mmol) was not genotoxic to mice in a dermal genotoxicity assay. 2
Human
The gingival tissue of the teeth (central incisors) of subjects (n = 30) was isolated with a light-polymerized resin dam, and a whitening gel containing Hydrogen Peroxide (35%) was administered three times for 15 min over 45 min. 101 This procedure was repeated 1 week later. Exfoliated oral mucosa gingival epithelial cells and upper lip lining were collected at baseline and 1 month after the second treatment. The scraped cells were placed on clean glass slides, smears prepared, and two blinded examiners performed cell and micronuclei counts. The frequency of micronuclei was not increased after administration of Hydrogen Peroxide in both the gingival tissue and upper lip. The authors concluded that the test material did not induce DNA damage to the gingival and lip tissue.
In conjunction with an epidemiological study where the treatment group (n = 6 women, 4 men) had their hair dyed 13 times at intervals of 3 to 6 weeks with commercial preparations, SCEs were examined in peripheral lymphocytes. 102 Blood samples were taken 3 weeks before the hair-dyeing treatment, and 24 h after each of the first three and last three dying treatments. There were no increases in SCEs at any point in the experiment. [See Epidemiological Studies for more information.]
Carcinogenicity Studies
Dermal, Oral, and Mucosal Carcinogenicity Studies.
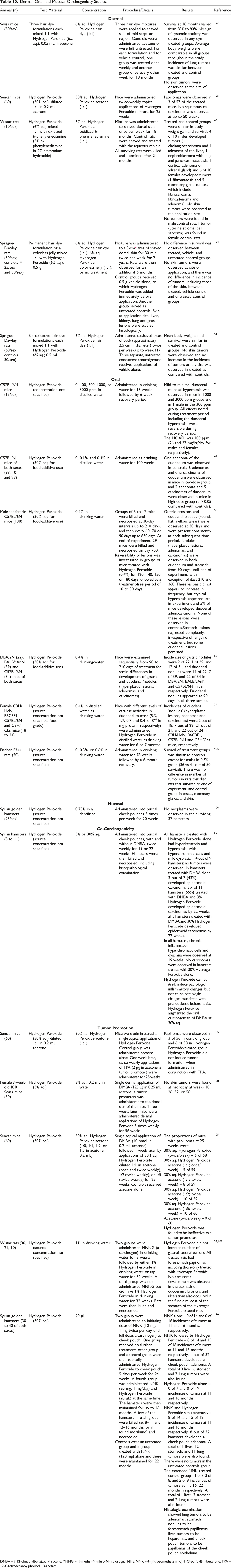
DMBA = 7,12-dimethylbenz(a)anthracene; MNNG = N-methyl-N’-nitro-N-nitrosoguanidine; NNK = 4-(nitrosomethylamino)-1-(3-pyridyl)-1-butanone; TPA = 12-O-tetradecanoylphorbol 13-acetate.
Animal
Dermal
Hydrogen Peroxide (at up to 15% aq.) was not carcinogenic when dermally administered to mice and rats.17,51,103-105 Three different hair dye formulations, each mixed 1:1 with Hydrogen Peroxide (6% aq.), did not cause skin tumors when applied once weekly or every other week to mice for 18 months. 103 Mice dermally administered Hydrogen Peroxide (30% aq. in a 1:1 mixture with acetone) twice-weekly for 25 weeks did not develop squamous-cell carcinoma when observed up to 50 weeks. 105
Hydrogen Peroxide (6% aq.) in a 1:1 mixture with oxidized p-phenylenediamine (5% p-phenylenediamine in 2% ammonium hydroxide) did not cause any skin tumors, but caused an increase in other types of tumors in rats when administered subcutaneously and topically once per week for 18 months. 60 Multiple hair dye formulations, each mixed 1:1 with Hydrogen Peroxide (6% aq.), applied to the skin of rats twice weekly for at least 2 years did not cause any skin tumors.51,104
Oral
In general, Hydrogen Peroxide in drinking water caused duodenal nodules in mice. Hydrogen Peroxide caused mild to minimal duodenal mucosal hyperplasia (reversible during the recovery period) in mice at 1000 and 3000 ppm in drinking water; the NOAEL was 100 ppm (26 and 37 mg/kg/day for males and females, respectively). 4 Hydrogen Peroxide (0.1%) administered in drinking water for 100 weeks did not increase the numbers of adenomas and carcinomas of the duodenum in mice. 49 However, at a higher concentration, Hydrogen Peroxide (0.4%) in drinking water caused gastric erosions and duodenal plaques at 30 days that were present consistently at each subsequent time period up to 700 days. 50 Nodules were observed in both the duodenum and stomach from 90 days until the end of the experiment; lesions were reversible after stopping treatment with Hydrogen Peroxide. In another experiment, Hydrogen Peroxide (0.4%) in drinking water caused duodenal nodules at 90 days in three strains of mice. 50 Hydrogen Peroxide (0.4%) in drinking water caused duodenal nodules at 6 or 7 months in four strains of mice. 34 Hydrogen Peroxide administered in drinking water at up to 0.6% for 78 weeks did not increase the number of tumors in testes, mammary glands, or skin in rats. 52
Mucosal
Hydrogen Peroxide (0.75% in a dentifrice) was administered into the buccal cheek pouches of hamsters for 20 weeks. 106 No neoplasms were observed.
Human
IARC determined that there is inadequate evidence in humans to come to a conclusion on the carcinogenicity of Hydrogen Peroxide and that there is limited evidence in experimental animals on the carcinogenicity of Hydrogen Peroxide. 107 IARC concluded that Hydrogen Peroxide “is not classifiable as to its carcinogenicity to humans (Group 3).”
Co-Carcinogenicity
Dermal
Hydrogen Peroxide (3% aq.) was administered to the skin of mice for up to 56 weeks after a single application of 7,12-dimethylbenz[a]anthracene (DMBA; a tumor initiator). 108 There were no skin tumors observed at necropsy at weeks 10, 26, 52, or 58. In another study, Hydrogen Peroxide (up to 30% aq.) was dermally administered for 25 weeks after a single application of DMBA. Hydrogen Peroxide was found to be an ineffective skin tumor promotor. 105
Hydrogen Peroxide (30% aq.) was administered once, followed by weekly applications of 12-O-tetradecanoylphorbol 13-acetate (TPA; a tumor promoter) for 25 weeks. 105 The initial administration of Hydrogen Peroxide did not cause or increase dermal tumors in the skin of mice.
Oral
Two groups of rats were administered N-methyl-N-nitro-N-nitrosoguanidine (MNNG; a carcinogen) in their drinking water for 8 weeks followed by either Hydrogen Peroxide (1%) in their water or plain water for 32 weeks. Hydrogen Peroxide did not increase the number of gastrointestinal tumors.33,109
Mucosal
Hydrogen Peroxide (3% and 30% aq.) was administered to the buccal cheek pouches of hamsters, concurrently with, or without, DMBA for 19 and 22 weeks. 52 Hydrogen Peroxide at 3% did not cause or increase the instances of preneoplastic lesions alone or with DMBA. However, at 30% Hydrogen Peroxide with DMBA, epidermoid carcinomas were observed by 22 weeks.
In hamsters administered Hydrogen Peroxide (30% aq.) for 24 weeks to the cheek pouches after a single dose of 4-(nitrosomethylamino)-1-(3-pyridyl)-1-butanone (NNK; a carcinogen), 1 out of 32 hamsters developed a cheek pouch adenoma and there was an increase in the instances of other tumors. 110 In hamsters that were administered NNK and Hydrogen Peroxide simultaneously, 23 out of 32 developed tumors. There were no instances of tumors in control groups that were administered NNK alone or Hydrogen Peroxide alone.
Other Relevant Studies
Dermatitis and Alopecia
Hair dye ingredients, including Hydrogen Peroxide (concentration not specified), were applied to the dorsal region of female C57BL/6 mice (n = 5 or 6) daily for 3 consecutive days and evaluated for dermatitis and hair loss. 111 The test materials were applied to the backs of the mice (2 × 5 cm) for 30 min; the test sites were then washed with tap water. The applied concentrations were selected based on their respective contents in commercial hair dye products. Three days after final hair-dyeing, photographs of mice were taken and the extent of hair loss was determined by measuring the area without hair using image processing and analysis software. Signs of hair loss and dermatitis (epidermal hyperplasia and inflammatory cell infiltration in the deep dermis and subcutaneous fat layer) developed in all groups treated with the component combinations containing Hydrogen Peroxide and the neutralized dye mixture (containing monoethanolamine (MEA) and toluene-2,5-diamine sulfate/p-phenylenediamine/resorcinol). The groups without either Hydrogen Peroxide or the neutralized dye mixture did not have hair loss or dermatitis, suggesting that both Hydrogen Peroxide and the neutralized dye mixture were necessary for inducing hair loss and dermatitis.
The experiment was repeated with the following test materials: (a) control (treated with a basic formulation of hair dye); (b) 6% MEA +10% neutralized dye mixture; (c) 4.5% aq. Hydrogen Peroxide; and (d) 6% aq. Hydrogen Peroxide +4.5% MEA +10% neutralized dye mixture. The group that contained Hydrogen Peroxide and MEA had increased hair loss compared to controls. Hydrogen Peroxide or MEA alone or in other combinations did not increase hair loss.
In a third experiment, Hydrogen Peroxide (6% aq.) was administered to the mice with MEA (0%, 3%, or 6%) and MEA (6%) was administered to the mice with Hydrogen Peroxide (0%, 3%, or 6% aq.). There was no hair loss or dermal damage when Hydrogen Peroxide or MEA were administered alone. There was a concentration-dependent increase in hair loss and dermal damage for Hydrogen Peroxide with the addition of MEA and for MEA with the addition of Hydrogen Peroxide. The authors concluded that hair dye-induced hair loss and dermatitis were caused by the combination of Hydrogen Peroxide and MEA. 111
Dermal Irritation and Sensitization Studies
Irritation
Animal
Dermal Irritation Studies of Hydrogen Peroxide in Animals.

OECD GL = Organisation of Economic Co-operation and Development Guide Lines; PII = primary irritation index.
In rabbits, generally, Hydrogen Peroxide was not irritating at up to 10% aq. and mildly irritating to irritating at 35% aq.; at approximately 50% aq. and above, Hydrogen Peroxide was severely irritating and corrosive.2,33 Hydrogen Peroxide was not irritating to intact and abraded skin at 3% and 6% aq. in rabbits.2,33 At 8% and 10% aq., erythema and edema were observed, but Hydrogen Peroxide was still rated as a non-irritant.2,33 At 35% aq. Hydrogen Peroxide, erythema, edema, and blanching of the test sites were observed in rabbits, and the test substance was found to be a dermal irritant.2,33 At 49.2% aq., Hydrogen Peroxide was corrosive to rabbit skin. 2 Dermal exposure to 50% aq. Hydrogen Peroxide for 1 or 4 h exposure was corrosive. 2 Dermal exposure to Hydrogen Peroxide at 70% aq. for 3 min caused moderate erythema and mild edema, and exposure for 30 min was corrosive. 33
A single application of 15% or 30% aq. Hydrogen Peroxide caused extensive epidermolysis, inflammation and vascular injury to mouse skin. 105 In rats, skin exposed to 3% to 10% aq. Hydrogen Peroxide had mild focal epidermal thickening, which had keratinocytes with signs of pyknosis. 112 Intracytoplasmic edema was sporadically observed at and around thickened skin, especially in the basal layer. The severity of the effects increased in a dose-dependent manner. Different solutions of 3% or 6% Hydrogen Peroxide were non-irritating or mildly irritating to intact guinea pig skin; in abraded skin, one of these solutions at 3% and one at 6% were strongly irritating, while the other preparations were at most only mildly irritating. 2
Human
The hands of subjects (n = 32) were exposed to Hydrogen Peroxide vapor (how the vapor was produced was not specified). 113 The lowest-observed-adverse-effects concentration (LOAEC) for skin irritation was 20 mg/m3 (14.2 mL/m3) after 4 h and 180 mg/m3 (128 mL/m3) after 5 min. This study was not used for evaluation by the MAK-Value Documentaries because of the inadequate documentation, but is included here for informational purposes. (A MAK value is the maximum permissible concentration of a substance as a gas vapor or aerosol in the air at the workplace.) 114
Sensitization
Animal
In a Magnusson-Kligman assay of Hydrogen Peroxide (0.1 mL), six induction applications at 0.1% aq. (intradermally) or at 3% aq. (epicutaneously to abraded skin) were administered to guinea pigs (n = 5). 2 Two weeks after the last induction, the test substances were applied once using the same concentrations and application routes. Dermal reactions were observed during the induction phase and at 1, 24, and 48 h after challenge patch. Two or three of the guinea pigs in each group (in both induction and challenge phases) had mild (faint pink) reactions to the application of Hydrogen Peroxide in the induction phase. One had a hemorrhagic reaction in the intradermal group. There were no signs of sensitization.
In a sensitization assay in guinea pigs, Hydrogen Peroxide (3% or 6% aq.) was administered to intact or abraded skin and by intradermal injection 10 times over a 2-week period. 33 After an unspecified time, the challenge under same conditions was administered. There were no signs of sensitization.
Ocular Irritation Studies
In Vitro
A 21-day porcine corneal opacity reversibility assay (PorCORA) was conducted on a hair colorant mixed 1:1 with Hydrogen Peroxide (12% aq.). 115 Ocular irritation effects were observed (microscopic changes only in the superficial squamous cell layer), but were fully reversible.
Animal
Ocular Irritation Studies for Hydrogen Peroxide.
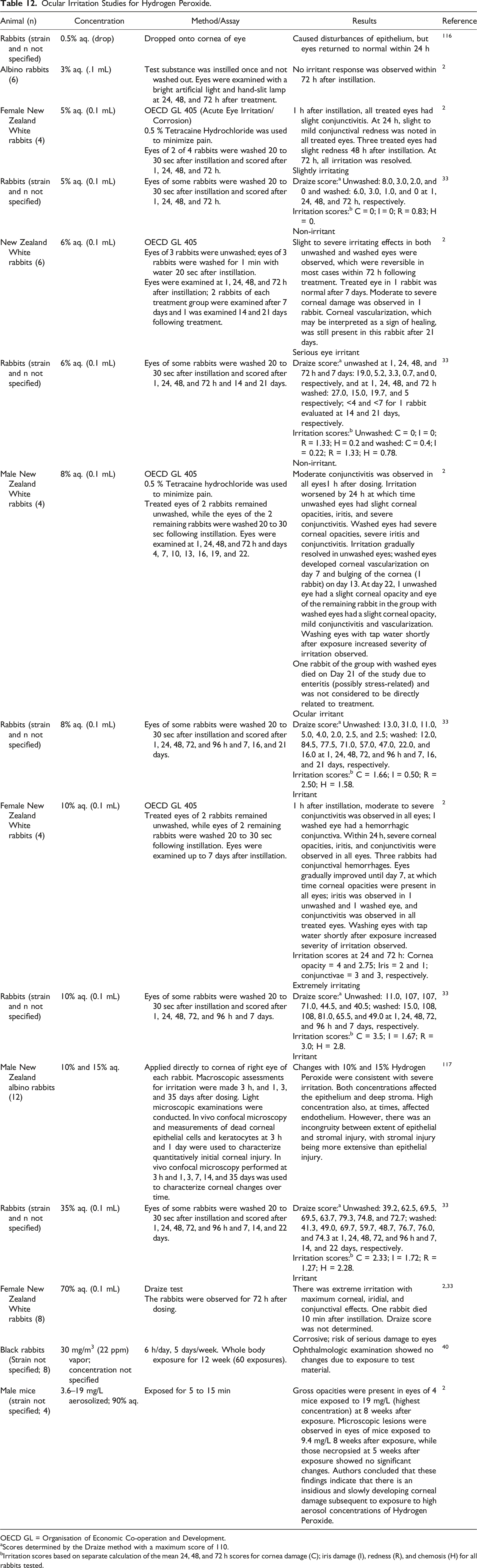
OECD GL = Organisation of Economic Co-operation and Development.
aScores determined by the Draize method with a maximum score of 110.
bIrritation scores based on separate calculation of the mean 24, 48, and 72 h scores for cornea damage (C); iris damage (I), redness (R), and chemosis (H) for all rabbits tested.
In general, when rabbit eyes were treated with Hydrogen Peroxide, corneal injury depended not only on the concentration of Hydrogen Peroxide, but also on the integrity of the corneal epithelium, which had a protective influence. 116 Hydrogen Peroxide at 0.5% to 5% aq. instilled into the eyes caused superficial corneal haze and much conjunctival reaction in rabbits, but these effects were resolved in 24 hr.2,116 Hydrogen Peroxide at 6% aq. had mixed results when instilled into the eyes of rabbits and was found to be both an ocular non-irritant and an irritant,2,33 while at 8% aq., Hydrogen Peroxide was an ocular irritant.2,33 Instillation of 10% to 30% aq. Hydrogen Peroxide caused superficial corneal haze, and, if there were defects in the epithelium, could cause localized swelling and opacities in the corneal stroma.2,33 At 70% aq., Hydrogen Peroxide was corrosive to the rabbit eye.2,33 In one study, Hydrogen Peroxide (10% and 15% aq.) was severely irritating; both concentrations affected the epithelium and deep stroma. 117
Rabbits exposed to 90% Hydrogen Peroxide vapor (30 mg/m3 for 6 hours day, 5 days/week) showed no ocular changes due to exposure to test material. 40 Mice exposed to Hydrogen Peroxide (90% aq.) vapors (19 mg/L) for 5–15 minutes showed gross opacities and microscopic lesions in the eyes. 2
Human
Instilling 1% to 3% aq. Hydrogen Peroxide solution on the human eye has been reported to cause severe pain, which soon subsides. 116 In contrast, Hydrogen Peroxide has been historically used at these concentrations as an ocular antibacterial agent, as much as three to five times per day, without significant injury. While the threshold for irritation in many subjects is considered to be 100 ppm (0.01% aq.), even at 800 ppm (0.08% aq.), Hydrogen Peroxide has been shown to not cause corneal or conjunctival epithelial staining; higher levels may result in greater discomfort. It is possible that pH may play a role in the variation in irritation levels and why similar concentrations of Hydrogen Peroxide may cause severe pain or be tolerated.
Solutions (isotonic and pH-balanced for the human eye; 300 milliosmoles (mOsm), pH = 7) containing Hydrogen Peroxide (30 ppm to 490 ppm; 0.003% to 0.049% aq.) were dropped into one eye of each subject (n = 10; a single group of subjects) and a control dose containing Hydrogen Peroxide (30 ppm; 0.003%) was dropped into the other eye in a single blinded study. 118 The tests started with the lowest concentration (30 ppm). The subjects reported their comfort response at 1 and 10 min after the drops were administered. Subjects were asked to compare the discomfort in their eyes and to report if they felt stinging. If there was no stinging reported, the next concentration was administered at the next session. No more than two test sessions were run per day. The mean detection threshold for drops of dilute Hydrogen Peroxide was 812 ppm (0.0812% aq.). The intersubject variability was quite large. If the lowest reported threshold for any stimulus is considered as an individual’s threshold level for sensation, the mean value is 247 ppm (0.0247% aq.).
Mucous Membrane Irritation Studies
Animal
Hydrogen Peroxide (30% aq.; 5 μL) was dropped on the tip of the tongues of anesthetized male albino Osborne-Mendel rats (n = 18) four times at 15 min intervals. 119 The tongues were rinsed with water 15 min after the last application. Three to six rats were killed either right after rinsing or 1 or 7 days after the last application, and their tongues examined. Hydrogen Peroxide caused an almost immediate marked edema on the anterior part of the tongue. After 1 day, this edema turned into a large ulceration that healed almost completely after 7 days.
Hydrogen Peroxide (1% or 1.2% aq.) was administered to the gingiva or tongues of anesthetized dogs by continuous drip (time and volume not specified). 33 Edema developed, followed by destruction and sloughing of the cornified epithelial layer of the gingiva.
Human
Repeated use of Hydrogen Peroxide topical solution as a mouthwash or gargle may produce a condition known as “hairy tongue” or may cause irritation of the buccal mucous membrane. 29 Concentrated solutions (20% to 30% aq. or more) of Hydrogen Peroxide are strongly irritating to mucous membranes.
A mouth rinse containing Hydrogen Peroxide (concentration not specified) was tested in a double-blind, stratified, two-treatment, parallel trial (n = 48) over a 4-week period in cancer patients undergoing chemotherapy from two cancer treatment centers. 120 Controls were administered baking soda and water rinses or a lemon glycerin solution. There were no adverse effects reported during this study. The original source document, published in 1999, does not provide the concentration of Hydrogen Peroxide in the mouth rinse.
Mucosal Irradiation
Syrian hamsters (n = 15) were used in an oral mucosal irritation assay to study the effects of Hydrogen Peroxide with irradiation. 121 The cheek pouches of the hamsters were treated as follows: group 1 was administered pure water (control); group 2 was administered laser irradiation at 80 mW; Group 3 was administered 3% aq. Hydrogen Peroxide; and groups 4 to 6 were administered 3% aq. Hydrogen Peroxide and laser irradiation at 80, 40, and 20 mW, respectively. The total treatment time was set at 7 min and treatment was repeated three times at approximately 1-h intervals. Macroscopic and microscopic histologic observations of the treated sites were performed immediately after each treatment and/or 24 h after the last treatment. The mean scores in macroscopic and histologic examinations in all six groups were 0. The authors concluded that treatment with 3% Hydrogen Peroxide and/or irradiation had no mucosal irritation potential in hamster cheek pouches under these test conditions.
Clinical Studies
Retrospective and Multicenter Studies
Dermal
Results for dermatitis patients (n = 210) who underwent patch testing with a standard allergen series (including 15 hairdressing chemicals) and a supplementary ‘‘hairdresser series’’ (18 additional hairdressing chemicals) were reviewed. 122 The most common sites of dermatitis were the scalp, face, and hands. Patients had widely varying occupations. The most common occupations were cosmetologist (10.5%), housewife (9.5%), and beautician (5.2%); 14.3% were retired. Positive allergic reactions to Hydrogen Peroxide (3% aq.) were observed in 1% of the subjects tested; 1.4% of the subjects were positive for irritation.
Subjects (n = 121) who worked as hairdressers, and were suspected to have allergies to chemicals that were used in their profession, were administered a patch tests or prick tests (European Standard Series and Hairdressing Series), depending on the center where they were tested. 123 One subject (0.9%) had a positive reaction to Hydrogen Peroxide.
During 1991 to 1997, patients (n = 130), mainly hairdressers, were patch tested, including for Hydrogen Peroxide (concentration not specified), at the Finnish Institute of Occupational Health because of suspected occupational skin disease. 124 None of the patch tests were positive for an allergic reaction to Hydrogen Peroxide; one patient had an irritant patch test reaction. At the Department of Dermatology, University of Turku, during 1995 to 1996, 59 patients who were suspected of having eczema caused by hairdressing compounds were patch tested. None of the patients had an allergic or irritant patch test reaction to Hydrogen Peroxide. Data from the Finnish Register of Occupational Diseases showed that the total number of occupational allergic dermatoses reported during 1975 to 1997 was 10,806 cases; none of these were shown to be caused by Hydrogen Peroxide with patch testing. In the same period, a total of 29,803 occupational dermatoses were reported to the Finnish Register of Occupational Diseases; four were shown to be caused by Hydrogen Peroxide. The concentrations tested were not specified. In a retrospective study of hairdresser’s with contact dermatitis during 1974 to 1993 in Finland, none of the patients (n = 355) tested positive for sensitization for Hydrogen Peroxide. 125
Clinical Trials
Dermal
A mixture of Hydrogen Peroxide (40% w/w) in an aqueous solution of isopropyl alcohol and water was tested in two double-blind, vehicle-controlled clinical trials, in subjects with four clinically typical seborrheic keratoses that were raised on the face, trunk, or extremities. 126 The subjects were treated with either the Hydrogen Peroxide mixture (n = 467) or the vehicle (n = 470). Subjects ranged from 42 to 91 years of age (mean 68.7 years), 58% percent were female, and 98% were Caucasian. A total of 925 of 937 subjects completed the trials; no reason was given for withdrawals from the study. Each lesion was treated with four applications on day 1 and then again on day 22, if needed, and were followed through day 106. The local skin reactions observed 10 minutes after treatment with the test substance included erythema (98% of the reactions), stinging (93%), edema (85%), pruritus (32%), and vesiculation (18%). The local skin reactions observed 1 week after treatment were scaling (72%), erythema (66%), crusting (67%), pruritus (18%), erosion (9%), and ulceration (4%). The local skin reactions observed 15 weeks after the initial treatment were erythema (21%), hyperpigmentation (18%), scaling (16%), crusting (12%), and hypopigmentation (7%). Less common adverse effects occurring in ≤ 0.5% of subjects treated with the Hydrogen Peroxide mixture included eyelid edema (0.6%) and herpes zoster (0.6%).
Oral/Mucosal
In a randomized, placebo-controlled, 1-year clinical trial conducted to evaluate safety of 6% Hydrogen Peroxide whitening strips under continuous use conditions, subjects (n = 40) were administered either Hydrogen Peroxide (6%; approximately 9 mg/strip) strips or placebo strips. 127 Strips were worn 5 min daily for 12 months. The first application was done under supervision and the rest were done at home. Safety and tolerability were assessed via oral status interviews and oral examinations at baseline and after 1, 2, 3, 6, 9, and 12 months of use. Oral irritation and tooth sensitivity were the two most common adverse events reported during the study. Perceived oral irritation was reported by 2.5% of subjects in the placebo group and 0% of subjects in the Hydrogen Peroxide strip group; however, upon examination, irritation was observed in 22.5% and 25.0% of the subjects, respectively. Tooth sensitivity was reported by 5% of subjects in the placebo group and 10% of subjects in the Hydrogen Peroxide strip group. The majority of subjects (86%) reported adverse events during the first 3 months of the clinical trial. There were no serious adverse events in the study. Two subjects (5%) in the treatment group left the study because of treatment-related tooth sensitivity. No subjects in the placebo group dropped from the study due to treatment-related adverse events.
The safety of Hydrogen Peroxide (6%) tooth-whitening strips was examined using a clinical trials database accumulated over a 4-year period conducted at a single site. 128 Each maxillary strip carried approximately 12 mg total Hydrogen Peroxide. The subjects (total n = 148) used the strips twice daily for 30 min over a 2-week period. Safety was assessed by examination and interview methods in all studies. Pooled subject-level data were analyzed. Overall, oral irritation occurred, on average, in 22% (range, 4% to 31% in the clinical trials) of the subjects and tooth sensitivity occurred, on average, in 20% (range, 10% to 28%) of the subjects. Other side effects were unremarkable, and only 1 subject (0.7%) discontinued treatment early due to an adverse event, which in this case, was moderate soft tissue pain that resolved fully 1 day after discontinuing the study. In virtually all circumstances, adverse events were transient in duration. Onset was typically early and resolved during treatment, without affecting strip use. Clinical examination was unremarkable, and other side effects were infrequent.
Hydrogen Peroxide (0.75% or 1.5%) or saline was used as an oral rinse in healthy subjects (n = 11 to 12) four times daily for 2 weeks.4,129 Mucosal status, buccal microbial adherence, salivary flow rate (SFR), and subjective reactions were assessed weekly. In the normal saline group, no significant changes were noted in any of the observed parameters and subjective reports were unremarkable. In both Hydrogen Peroxide groups, significant mucosal abnormalities were observed and subjective complaints included discoloration of the mucosal surfaces and the tongue. Bacterial adherence was significantly reduced in the 0.75% group, but not in the 1.5% group.
Other Clinical Studies
In patch tests using a standard hairdressers’ series in subjects (n = 54) who were hairdressers in Australia with occupational contact dermatitis of the skin and nails, none of the subjects had an allergic reaction to Hydrogen Peroxide (concentration tested not specified). 130 In patch tests in subjects who were hairdressers (n = 164) in Australia with occupational contact dermatitis, none of the subjects had an allergic reaction to Hydrogen Peroxide (concentration tested not specified). 131 In patch tests in subjects who were hairdressers (n = 44) in Bangkok with contact dermatitis of the hands, three of the subjects had an allergic reaction to Hydrogen Peroxide (3% aq.). 132
Case Reports
Case Reports on Exposure to Hydrogen Peroxide.

ECG = electrocardiogram; MRI = magnetic resonance imaging.
Occupational Exposure
The Occupational Safety and Health Administration (OSHA) permissible exposure limit (PEL) for inhalation of Hydrogen Peroxide is 0.0001% (1.4 mg/m3) averaged over an 8-h work shift. [29CFR1910.1000] The National Institute for Occupational Safety and Health (NIOSH) immediately dangerous to life or health (IDLH) level for Hydrogen Peroxide is 0.0075% and the recommended exposure limit (REL) is 0.0001% (1.4 mg/m3).9,133 According to the American Industrial Hygiene Association (AIHA) emergency response planning guideline (ERPG-2), the maximum airborne concentration below which nearly all individuals could be exposed for up to 1 h without experiencing or developing irreversible or other serious health effects or symptoms which could impair an individual’s ability to take protective action is 50 ppm (0.0050%).
The Scientific Committee on Occupational Exposure Limits (SCOEL) concluded that an occupational exposure limit (OEL) of 0.0001% (1.4 mg/m3) for Hydrogen Peroxide, as and 8-h time-weight average (TWA), is recommended. 134
NICNAS conducted a Tier II assessment on Hydrogen Peroxide under IMAP (see Non-Cosmetic Use section for more related information). 5 It is advised that industries should use measures to minimize the risk of oral, dermal, ocular, and inhalation exposure to Hydrogen Peroxide by workers.
Epidemiological Studies
Chromosomal aberrations in lymphocytes from subjects (n = 6 women, 4 men) who had their hair dyed were studied. 135 The treatment group had their hair dyed 13 times at intervals of 3 to 6 weeks with commercial preparations containing mixtures of aminotoluenes, aminophenols, and hydroxybenzenes and, in some cases, naphthol, as active ingredients; the coloring products used was chosen according to each subject’s hair color, and the same colorant was used on each subject throughout the study. The coloring preparations were each mixed (1:1) with Hydrogen Peroxide (3% to 6% aq.). The subjects in the control group (n = 10) were matched for sex and age and were subjected to sham hair dyes at the same times as the treatment group. Records were taken of smoking habits, alcohol consumption, and medical drug use and, during the experiment, exposure to X-rays, illness, and vaccinations. There were more smokers in the test group. None of the subjects had used hair dyes or shades for at least 1 year before entering the study, and the control group did not use hair colorants during the study. Nine blood samples were taken: 3 weeks before the first treatment, 24 h after a sham dyeing (no dye or Hydrogen Peroxide), and 24 h after each of the first 3 and last 4 dyeing procedures. There were no differences observed between the control and treated groups in the percentage of cells with one or more structural aberration (excluding gaps) before treatment, after sham dyeing, or after treatment. Subdivision of the groups according to sex revealed no differences. A significant increase in aberration rate with age was observed among the male but not the female subjects. Neither smoking nor X-ray exposure had an effect. No clastogenic effect of repeated hair dying was established in this study.
Summary
This is a review of the safety of Hydrogen Peroxide as used in cosmetics. According to the Dictionary, this ingredient is reported to function in cosmetics as an antimicrobial agent, cosmetic biocide, oral care agent, and oxidizing agent.
Hydrogen Peroxide is always used in cosmetics as an aqueous solution. It can behave both as an oxidizing and as a reducing agent. Hydrogen Peroxide is produced metabolically in intact cells and tissues.
According to VCRP survey data received in 2018, Hydrogen Peroxide is reported to be used in 390 cosmetic formulations (18 leave-on products and 372 rinse-off products). The results of the concentration of use survey conducted by the Council indicate that Hydrogen Peroxide is used at up to 15% in the category of other hair coloring preparations; this formulation is a professional 50 volume developer, and standard dilutions include 10, 20, 30, and 40 volume (i.e., 3%, 6%, 9%, or 12% Hydrogen Peroxide, respectively). The highest maximum concentration of use in hair dyes and colors is 12.4%. The highest maximum concentration of use reported for products resulting in leave-on dermal exposure is 2.5% in “other” skin care preparations. Hydrogen Peroxide is used in oral hygiene formulations, which may be incidentally ingested and come in contact with mucus membranes, at up to 4.6% (dentifrices).
In the US, the FDA recognizes Hydrogen Peroxide as GRAS to treat food. Hydrogen Peroxide may be used as a microbial agent, a bleaching agent, and a component of emulsifiers, and it may have other uses in the preparation and packaging of food for human use.
Hydrogen Peroxide is reactive, and it degrades rapidly when in contact with organic material. The rapid degradation upon contact with skin explains the absence of systemic effects from dermal exposure to Hydrogen Peroxide. However, application of Hydrogen Peroxide solutions to damaged skin, or excessive amounts of Hydrogen Peroxide on skin, may result in some systemic exposure. Administration of Hydrogen Peroxide solutions to body cavities lined by mucous membranes resulted in increased oxygen content of the draining venous blood. If the amounts were sufficiently high, Hydrogen Peroxide caused the formation of oxygen bubbles.
In general, acute dermal and oral toxic effects are dependent on concentration as well as dose. The concentration of Hydrogen Peroxide used is dependent upon its intended use. For example, industrial uses utilize concentrations as high as 90%, while in cosmetic use, Hydrogen Peroxide concentration is not reported to exceed 15%. The dermal LD50 was >8000 mg/kg Hydrogen Peroxide in mice; in this study, more mice died when 28% aq. Hydrogen Peroxide was administered than when 10% aq. Hydrogen Peroxide was used. Dermally administered Hydrogen Peroxide (90% aq.) caused 4 of 12 rats to die at 4899 mg/kg and 9 of 12 to die at 5520 mg/kg. The dermal LD50 in rabbits was >2000 mg/kg at 35% aq. Hydrogen Peroxide, 9200 mg/kg using 70% aq., and 690 mg/kg using 90% aq. No cats died when administered 4361 mg/kg of a 90% aq. Hydrogen Peroxide solution, dermally. Two of 5 pigs died when 2760 mg/kg (concentration not specified) Hydrogen Peroxide was applied to the skin.
The oral LD50 for Hydrogen Peroxide (90% aq.) in mice was reported to be 2000 mg/kg. Oral LD50s in rats ranged from 1520 to >5000 mg/kg at approximately 10% aq. Hydrogen Peroxide while at 70% aq. Hydrogen Peroxide, the LD50 in rats ranged from 75 to 1026 mg/kg. Rats that died had reddened lungs, hemorrhagic and white stomachs, and blood-filled intestines; some had white tongues.
In acute inhalation studies in mice, more mice died the longer they were exposed to Hydrogen Peroxide, while rats had only systemic effects. No treatment-related mortalities were observed in mice exposed to up to 3220 mg/m3 Hydrogen Peroxide (70% aq.) for up to 30 min; exposure to 3130 mg/m3 (1 h) and 880 mg/m3 (2 h) was lethal to all 4 mice tested. Rats exposed to saturated vapors of 90% aq. Hydrogen Peroxide survived exposure for 8 h; necropsy showed severe pulmonary congestion and emphysema, but no necrosis of pulmonary mucosa. In rats exposed to vaporized Hydrogen Peroxide for 4 h, the LC50 was 2000 mg/m3, the LOEC for respiratory effects was 60 mg/m3, and the LOEC for skin effects was 110 mg/m3.
Hydrogen Peroxide causes transient dermal blanching in human subjects starting at 3% aq. Hydrogen Peroxide was slightly irritating to the nose and throat at 3.08 mg/m3, but not at 0.7 mg/m3, in humans exposed to vapors for 2 h. In humans, the threshold of detection for irritation through inhalation exposure was 10 mg/m3 when Hydrogen Peroxide vapor was inhaled through the nose.
In general, orally administered Hydrogen Peroxide in repeated dose studies caused inflammation and erosion to the upper digestive tracts of mice and rats, which increased with time and concentration. In repeated-dose drinking water studies (0.3% to 1.5% aq. Hydrogen Peroxide), rats and mice had decreased body weights at 2 to 3 weeks, decreased organ weights at 3 weeks or longer, and duodenal mucosal hyperplasia, duodenal carcinomas, and/or extensive pathological changes in the periodontium in subchronic and chronic studies. Effects observed during treatment were reversible after stopping treatment. In a feeding study using rats, there were no observed effects at up to 60 mg/kg/day in feed.
In mice and rats, systemic effects become more pronounce with time when exposed by inhalation to Hydrogen Peroxide. For example, mice exposed to Hydrogen Peroxide (79 or 107 mg/m3; 90% aq.) had nasal discharge and irritated skin at week 2; seven of nine mice died after eight exposures at 79 mg/m3; and, in the high-dose group, 5 of 10 mice died after eight exposures, and eight of 10 after 18 exposures. Rats exposed to 93 mg/m3 Hydrogen Peroxide (90% aq.) for 7 weeks showed signs of nasal irritation and profuse discharge at 2 weeks, and lung congestion at 5 weeks. There were no mortalities when rats were exposed to Hydrogen Peroxide (50% aq.) up to 10.3 mg/m3 for 13 weeks; the NOAEL was 10.3 mg/m3 for male and female rats for decreased liver and thymus weights.
Dermal effects of aerosolized Hydrogen Peroxide included bleaching of the fur of rabbits and dogs that increased over time. Shaved rats exposed to Hydrogen Peroxide vapor (0.1 to 10.1 mg/m3) in whole body chambers for 4 months had significant dysfunction of the horny layer of the skin. The LOEL in rats was 1.0 mg/m3 and the NOEL was 0.1 mg/m3 for changes in enzyme activities in the skin.
Studies were performed to evaluate the safety of Hydrogen Peroxide mixed with an oxidative hair dye or hair dye ingredient in a 1:1 mixture (up to 6% aq.) The resulting mixture was a reaction product of the two substances with little to no expected residual Hydrogen Peroxide. In these studies, no signs of toxicity, teratogenicity, maternal toxicity, or carcinogenicity were present. There was no evidence of test substance-induced toxicity when Hydrogen Peroxide (6% aq.), in nine different hair dyes, were administered to the skin of rabbits for 13 weeks. Nine different hair dyes containing Hydrogen Peroxide (6% aq.) administered to female rats caused no maternal or developmental toxicity; the same results were obtained when six different hair dyes, also containing Hydrogen Peroxide (6% aq.), were administered to both the male and female rats.
In human subjects, hair dyes in 1:1 mixtures with Hydrogen Peroxide (3% to 6% aq.) did not cause chromosomal aberrations in lymphocytes collected from subjects after dying their hair 13 times at 3- to 6-week intervals. There were no increases in SCEs at any point in this experiment. Multiple hair dye formulations in 1:1 mixtures with Hydrogen Peroxide (6% aq.) did not cause skin tumors when applied to mice for 18 months and rats for at least 2 years. Hydrogen Peroxide (6% aq.) in a 1:1 mixture with oxidized p-phenylenediamine did not cause any skin tumors, but caused an increase in other types of tumors in rats when dermally administered once per week for 18 months.
Orally administered Hydrogen Peroxide caused maternal and fetal effects at higher doses (2% and 10% aq.). Male and female mice administered Hydrogen Peroxide (up to 1% aq.) in drinking water, prior to mating through parturition, produced normal litters. Hydrogen Peroxide (2% and 10% aq.) mixed with feed caused reduced body weights of the dams, fetal resorptions, decreased fetal body weights, and skeletal hypoplasia in rats. All of the neonates in the 10% aq. Hydrogen Peroxide group died. In rats, Hydrogen Peroxide (0.005 to 50 mg/kg) administered for 6 months caused modified estrus cycles in females and decreased sperm mobility in males.
The results in Ames assays conducted on Hydrogen Peroxide were not consistent. In most of the Ames assays presented, Hydrogen Peroxide (concentrations not specified in most assays) increased the number of revertant colonies in S. typhimurium strains without metabolic activation (3% or 30% in those assays with concentrations of Hydrogen Peroxide); however, there were a few assays where the results were negative for genotoxicity. In Ames assays, the results for Hydrogen Peroxide (3% in one of these assays) were also mixed in E. coli. In other Ames-type assays, Hydrogen Peroxide (concentrations not specified) was mutagenic in E. coli, B. subtilis, and S. cerevisiae. In bacterial forward mutation assays, Hydrogen Peroxide (30% in the one assay) was genotoxic to various strains of S. typhimurium and E. coli. In chromosomal aberration tests, Hydrogen Peroxide (30% in the one assay; as low as 0.25 to 10 μM) was genotoxic to multiple cell types, including CHO cells murine splenocytes, V79 cells, SHE cells, and human leukocytes and embryonic fibroblasts. Hydrogen Peroxide (concentration not specified; 500 μM) increased the number of abnormal metaphases in CHO-K1 cells without, but not with metabolic activation. In mouse lymphoma assays, Hydrogen Peroxide (30% in the assays with a concentrations; 0.075 μg/ml) increased the mutation frequency in mouse lymphoma cells without metabolic activation, but not with metabolic activation; Hydrogen Peroxide (concentrations not specified) had mixed results in V79 cells in mammalian cell gene mutation assays. In SCE assays, Hydrogen Peroxide (concentrations not specified) was mutagenic in V79, CHO cells, and human lymphocytes without metabolic activation, but was not mutagenic, or was mutagenic to a lesser extent, with metabolic activation. In an UDS assay in rat hepatocytes, Hydrogen Peroxide (35.7% aq.) caused a dose-dependent increase in NNG values at 6.25 to 25 μg/ml. In comet assays, Hydrogen Peroxide was genotoxic in mouse lymphoma cells (concentration not specified; 500 μM), rat hepatocytes (concentration not specified; 1 μM), S. cerevisiae (concentration not specified; 20 μM), V79 cells (37% aq.; 40 μM), and HepG2 cells (0.3 M aq.). Hydrogen Peroxide was also mutagenic to breast cancer cells (concentration not specified; 200 μM), human lymphocytes (concentration not specified; 10 μM), human fibroblasts (concentration not specified; 30 μM), HeLa cells (37% aq.; 40 μM), and HEP G2 cells (0.3 M aq; 40 μM). In a combined comet assay/micronucleus assay using human lymphoblastoid cells, Hydrogen Peroxide (concentration not specified) was genotoxic in the comet assay (50 μM) and in the micronucleus assay (100 μM). In another combined comet assay/micronucleus assay in V79 cells, Hydrogen Peroxide (concentration not specified) was genotoxic at 80 μM and 40 μM, respectively. In a multi-test assay using mouse lymphoma cells, Hydrogen Peroxide (concentration not specified) was not genotoxic in two DNA adduct assays up to 500 μM (but was genotoxic at 500 μM in a comet assay), at 20 μM in a micronucleus test, and at 100 μM in a tk+/− gene mutation assay. Hydrogen Peroxide (37% aq.) was not mutagenic at 110 μM in V79 cells in a HPRT assay.
Hydrogen Peroxide was not genotoxic in multiple in vivo assays. In a mammalian erythrocyte micronucleus test using mice, 35% aq. Hydrogen Peroxide, administered i.p., was not genotoxic at up to 2000 mg/kg. In a mammalian erythrocyte micronucleus test, Hydrogen Peroxide (35% aq.) administered in drinking water was not genotoxic to mice at up to 536 and 774 mg/kg/day for males and females, respectively. Hydrogen Peroxide (70% aq; ≤ 200 mmol) was not genotoxic to mice in a dermal genotoxicity assay. In an UDS assay, i.v. administered Hydrogen Peroxide (50 mg/kg) did not induce unscheduled DNA synthesis in rats. In human subjects, a whitening gel containing Hydrogen Peroxide (35% aq.) administered to gingival tissue during a teeth-whitening procedure did not induce DNA damage to the gingival and lip tissue.
Mice dermally administered Hydrogen Peroxide (15% aq.) twice-weekly for 25 weeks did not develop squamous-cell carcinoma. In oral carcinogenicity studies, Hydrogen Peroxide in drinking water was not carcinogenic at 0.1%, but at 0.4%, caused duodenal nodules in mice. Lesions observed in the duodenum and stomachs after 90 days of treatment were fully reversible when treatment was terminated. Hydrogen Peroxide administered in drinking water at up to 0.6% did not increase the number of tumors in testes, mammary glands, or skin in rats. In mucosal studies, no neoplasms developed when Hydrogen Peroxide (0.75% in a dentifrice) was administered into the buccal cheek pouches of hamsters for 20 weeks.
When administered to the buccal cheek pouches of hamsters. Hydrogen Peroxide (30% aq.) dermally administered once, followed by applications of TPA for 25 weeks, did not cause or increase dermal tumors in the skin of mice. In two studies, Hydrogen Peroxide (up to 30% aq.) did not promote tumors or cause squamous-cell carcinoma in mice after the administration of DMBA. In an oral study, after the administration of MNNG, Hydrogen Peroxide (1% aq.) in drinking water did not increase the number of gastrointestinal tumors. Hydrogen Peroxide (30% aq.) administered for 24 weeks to the cheek pouches of hamsters after a single dose of NNK did not increase the instance of cheek pouch adenoma, but did increase the instances of other tumors.
Hydrogen Peroxide (4.5% aq.) alone applied to the dorsal region of mice did not cause hair loss or dermatitis. However, when Hydrogen Peroxide (6% aq.) was administered in combination with MEA, there was a concentration-dependent increase in hair loss and dermatitis.
In rabbits, Hydrogen Peroxide was not irritating at up to 10% aq. and mildly irritating to irritating at 35% aq.; at approximately 50% aq. and above, Hydrogen Peroxide was severely irritating and corrosive. In rabbits, Hydrogen Peroxide was not irritating to intact and abraded skin at 3% and 6% aq.; rated a non-irritant, but with erythema and edema at 8% and 10% aq.; was a dermal irritant at 35% aq. Hydrogen Peroxide with erythema, edema, and blanching of the test sites; and was corrosive at 49.2% aq. Dermal exposure to Hydrogen Peroxide at 70% aq. for 3 min caused moderate erythema and mild edema in rabbits; exposure to 50% aq. Hydrogen Peroxide for 1 or 4 h exposure was corrosive. Dermal exposure to Hydrogen Peroxide at 70% aq. for 30 min exposure was corrosive in rabbits. A single application of 15% or 30% aq. Hydrogen Peroxide caused extensive epidermolysis, inflammation, and vascular injury to mouse skin. In rats, 3% to 10% aq. Hydrogen Peroxide caused mild focal epidermal thickening. A single application of 3% or 6% aq. Hydrogen Peroxide was non-irritating or mildly irritating to guinea pig skin. Hydrogen Peroxide was not sensitizing in guinea pigs at up to 6% aq.
When the hands of human subjects were exposed to Hydrogen Peroxide vapor for 4 h, the LOAEC was 20 mg/m3 (14.2 mL/m3) for skin irritation. The LOAEC was 180 mg/m3 (128 mL/m3) after 5 min.
In a 21-day PorCORA, Hydrogen Peroxide (12% aq.) in a 1:1 mixture with a hair dye caused microscopic changes only in the superficial squamous cell layer; the effects were fully reversible. In rabbit eyes treated with Hydrogen Peroxide, corneal injury generally depended not only on the concentration of Hydrogen Peroxide, but also on the integrity of the corneal epithelium. Hydrogen Peroxide at 0.5% to 5% aq. instilled into the eyes of rabbits caused superficial corneal haze and conjunctival reaction; these effects were resolved in 24 hr. Hydrogen Peroxide at 6% aq. had mixed results in rabbits, while at 8% aq., Hydrogen Peroxide was an ocular irritant. Instillation of 10% to 30% aq. Hydrogen Peroxide caused superficial corneal haze, and, if there were defects in the epithelium, could cause localized swelling and opacities in the corneal stroma. At 70% aq., Hydrogen Peroxide was corrosive to the rabbit eye. Rabbits exposed to Hydrogen Peroxide vapors (30 mg/m3) showed no changes due to exposure to test material. The eyes of mice exposed to Hydrogen Peroxide vapors (90% aq.) showed gross opacities and microscopic lesions.
The threshold for ocular irritation in many human subjects is 0.01% aq., but at 0.08% aq., Hydrogen Peroxide may not cause corneal or conjunctival epithelial staining. It is possible that pH may play a role in irritation levels. The mean detection threshold for drops of dilute Hydrogen Peroxide was 812 ppm in human subjects.
In mucosal studies, Hydrogen Peroxide dropped on the tongues of rats (30% aq.) and dogs (1 or 1.2% aq.) caused marked edema. In rats, edema evolved into large ulcerations that almost resolved in 7 days. In dogs, there was destruction and sloughing of the cornified epithelial layer of the gingiva. Hydrogen Peroxide (3% aq.) was not a mucosal irritant when administered to hamster cheek pouches and then irradiated.
In humans, repeated use of Hydrogen Peroxide as a mouthwash or gargle may cause irritation of the buccal mucous membrane. Concentrated solutions (20% to 30% aq. or more) of Hydrogen Peroxide are strongly irritating to mucous membranes. In a double-blind, stratified, two-treatment, parallel trial over a 4-week period, a mouth rinse containing Hydrogen Peroxide (concentration not specified) caused no adverse effects.
In multiple retrospective studies, there were few or no positive reactions to Hydrogen Peroxide in subjects with occupational dermatitis or occupational allergic dermatoses. In a retrospective study of Hydrogen Peroxide (6%) in tooth-whitening strips, oral irritation occurred in and average of 22% of the subjects. In virtually all circumstances, adverse events were transient.
In a clinical trial, the local skin reaction of a mixture of Hydrogen Peroxide (40% w/w) in an aqueous solution of isopropyl alcohol and water administered to subjects seborrheic keratoses included erythema, stinging, edema, scaling, and crusting. In a clinical trial of tooth-whitening strips containing Hydrogen Peroxide (9 mg/strip), irritation was observed in 25.0% of the subjects; there were no serious adverse events. In a clinical trial of Hydrogen Peroxide (0.75% or 1.5%) as an oral rinse, mucosal abnormalities were observed and subjective complaints included discoloration of the mucosal surfaces and the tongue.
Discussion
The Panel determined that the available genotoxicity, dermal, inhalation, carcinogenicity, and reproductive/developmental toxicity data were sufficient to issue the conclusion that Hydrogen Peroxide is safe in the present practices of use and concentration. The potential for dermal absorption of Hydrogen Peroxide was noted, but was considered negligible due to the low concentrations used for cosmetic products. In addition, Hydrogen Peroxide is reactive, and degrades rapidly, due to reactions with all classes of organic biomolecules. The rapid degradation upon contact with skin explains the absence of systemic effects from dermal exposure to Hydrogen Peroxide. The Panel also noted the positive genotoxicity studies, but determined the results are not relevant to cosmetic use due to rapid consumption of Hydrogen Peroxide by reaction with the proteins on the skin surface.
Generally, the toxic potency of Hydrogen Peroxide is positively correlated with the concentration used. Hydrogen Peroxide comes in a variety of grades for different uses. For example, the 3% grade is commonly used topically and for pharmaceutical purposes, while a 90% grade is used as an oxygen source for rocket fuel. For cosmetics, the maximum concentration of Hydrogen Peroxide in leave-on hair products is reported to be relatively low (4%), and the maximum reported concentration of use for Hydrogen Peroxide is 15% in a professional hair coloring formulation.
The Panel noted the positive irritation studies in rabbits dermally exposed to concentrations of 35–70% Hydrogen Peroxide. However, the concentrations used in these studies are higher than the concentrations used in cosmetics. Hydrogen Peroxide was non-irritating in rabbits and non-sensitizing in guinea pigs at concentrations of 10 and 6%, respectively. Due to these factors, the concern of dermal irritation and sensitization resulting from dermal cosmetic application is considered negligible.
In addition, Hydrogen Peroxide is inevitably synthesized in the body (e.g., oral cavity, urinary tract, respiratory system). When 16 urine samples were examined in healthy human volunteers from the ages of 18–49, an average of 16.9 μM Hydrogen Peroxide was detected. The robust natural production and clearance of Hydrogen Peroxide in the body further mitigates any concern of toxic potential resulting from cosmetic use.
Hydrogen Peroxide is an oxidizer in permanent hair coloring formulations and hair bleaches. The highest maximum concentration of use in hair dyes and colors is 12.4%; however, Hydrogen Peroxide is commonly reacted (e.g., 1:1 with an oxidative hair dye or hair dye ingredient) in preparation of the hair dye formulation to be applied. Accordingly, it should be noted that FDA has issued certain safety precautions to be followed when using permanent hair dye formulations, https://www.fda.gov/Cosmetics/ResourcesForYou/Consumers/ucm167436.htm.
The Panel noted the potential for incidental inhalation exposure with aerosol hair sprays (4%). The available inhalation data suggest little potential for respiratory effects at relevant doses. It should also be noted that inhalation toxicity studies on test animals are often conducted using high concentrations of droplets/particles with size distributions well within the respirable range and long exposure durations to ensure that the potential for pulmonary or systemic toxicity will be detected. In contrast, however, the concentrations of respirable droplets/particles and the inhalation exposure durations from the use of cosmetic products will be much less than those of the animal studies. Thus, the adverse effects reported in such studies may have little or no relevance for evaluating the inhalation safety of cosmetic ingredients. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient concluded that Hydrogen Peroxide is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.