Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Polysilicone-11 as used in cosmetic formulations. This ingredient is reported to function as a film former. The Panel considered the available data and concluded that Polysilicone-11 is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
This is a safety assessment of Polysilicone-11 as used in cosmetic formulations. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), Polysilicone-11 functions as a film former in cosmetics.
1
Polysilicone-11 is the product of a reaction between bis-vinyldimethicone and hydrogen methicone (Figure 1). Polysilicone-11 reactants (wherein each instance of R may be hydrogen or methyl; x, y, and z not defined).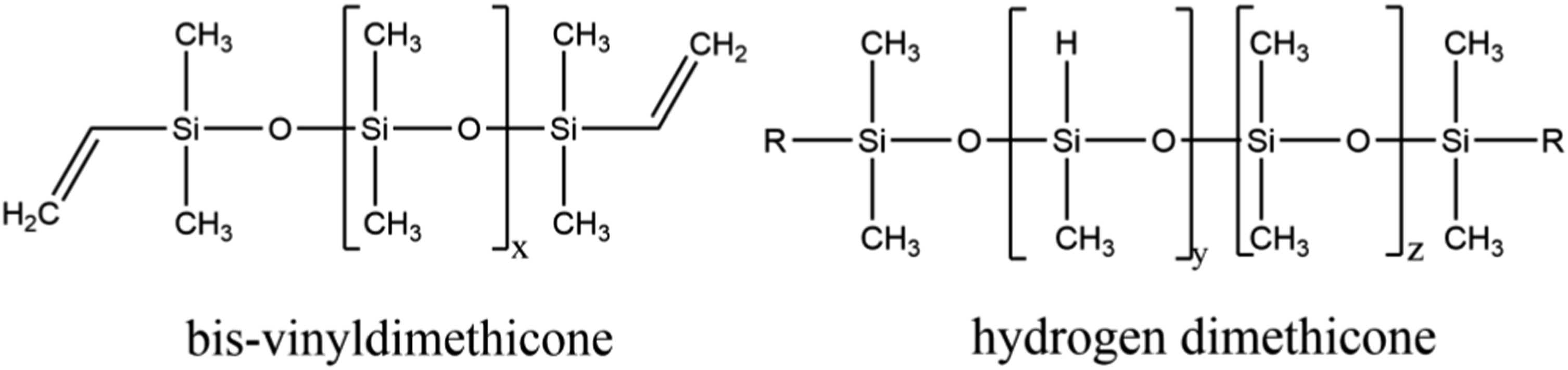
This safety assessment includes relevant unpublished data that are available for each endpoint that is evaluated. An exhaustive search of the world’s literature was performed, and very little published data were found regarding this ingredient. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints the Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and Structure
According to the Dictionary, Polysilicone-11 (CAS No: 63394-02-5, 156065-02-0) is a crosslinked dimethyl siloxane formed by the reaction of bis-vinyldimethicone and hydrogen dimethicone. 1
For use in cosmetics, copolymers such as Polysilicone-11, are typically supplied to finishing houses as swollen gels (i.e., trade name mixtures) that contain 1 or more solvents (e.g., cyclopentasiloxane). 2 The addition of the comonomer (i.e., the vinyl-substituted dimethicone) affects both the chemical and the rheological properties of the resultant ingredient. Furthermore, the degree of crosslinking could also significantly affect these properties. Accordingly, this one copolymer ingredient theoretically represents a wide variety of materials ranging from liquids to elastomeric solids.
Chemical Properties
The molecular weight of Polysilicone-11 has been reported to be greater than 1 million Da, in the form of an elastomer rubber, amorphous polymer. 3 For 3 different tradename mixtures, Polysilicone-11 was stated to comprise 10–20% of the mixture composition.4-6 The composition remainder of these mixtures (i.e., the other 80–90%) was reported to be isododecane, cyclopentasiloxane, or dimethicone. Each of these tradename mixtures is a clear liquid, with a viscosity ranging from 300 to 500 pascal second (Pa·s).
Method of Manufacture
According to a supplier, Polysilicone-11 is manufactured in cyclopentasiloxane (D5) solvent, preferably from low cyclotetrasiloxane (D4) feedstock using a hydrosilation catalyst. 3 This is reported to be a pure addition reaction in which no impurities are formed during the reaction and no residual monomers remain after completion.
Impurities
According to a manufacturer, Polysilicone-11 generally contains less than 20 ppm platinum catalyst from hydrosilation. 3 The same manufacturer also reported that heavy metal testing results for Polysilicone-11 typically include: below limits of detection for mercury, and less than 1 ppm for lead and arsenic.
Use
Cosmetic
The safety of the cosmetic ingredient addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aNot specified that this is used in spray or powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders.
NR – no reported use.
Additionally, Polysilicone-11 is used in cosmetic sprays and could possibly be inhaled; for example, it is reported to be used in suntan pump sprays at up to 0.04%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared with pump sprays.9,10 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.11,12 Polysilicone-11 was reportedly used in face powders at concentrations up to 3.5%, and could possibly be inhaled. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the air.13-15
Polysilicone-11 is not restricted from use in any way under the rules governing cosmetic products in the European Union. 16
Toxicokinetic Studies
Toxicokinetics studies on Polysilicone-11 were not found in the published literature, and unpublished data were not submitted.
Toxicological Studies
Acute Toxicity Studies
Oral
An acute oral toxicity study was performed on Sprague Dawley rats (5/sex) using a test substance consisting of 6% Polysilicone-11 and 94% cyclotetrasiloxane. 17 The test substance was administered undiluted. The LD50 was reported to be >5 g/kg. No other details regarding this study were provided.
Short-Term, Subchronic, and Chronic Toxicity Studies
Short-term, subchronic, and chronic toxicity studies on Polysilicone-11 were not found in the published literature, and unpublished data were not submitted.
Developmental and Reproductive Toxicity (DART) Studies
DART studies on Polysilicone-11 were not found in the published literature, and unpublished data were not submitted.
Genotoxicity Studies
The genotoxic potential of a mixture consisting of 14% Polysilicone-11, 47% dimethicone, and 39% cyclopentasiloxane, was evaluated in an Ames assay. 17 Bacterial cell lines (Salmonella typhimurium strains TA98 and TA100) were tested with and without metabolic activation. The test substance was tested at concentrations of 50, 100, 500, 1000, and 5000 μg/plate, and was considered to be non-mutagenic.
Carcinogenicity Studies
Carcinogenicity studies on Polysilicone-11 were not found in the published literature, and unpublished data were not submitted.
Other Relevant Studies
Cytotoxicity
An agar diffusion test was performed in vitro to determine the biological reactivity of a mammalian cell culture (not specified) following indirect contact with the test substance (a trade name mixture containing 12–16% Polysilicone-11, 43–50% dimethicone, and 36–42% cyclopentasiloxane). The test substance exhibited no reactivity after the 24-h observation period, and did not induce cytotoxicity.
Dermal Irritation and Sensitization Studies
Dermal Irritation and Sensitization Studies.
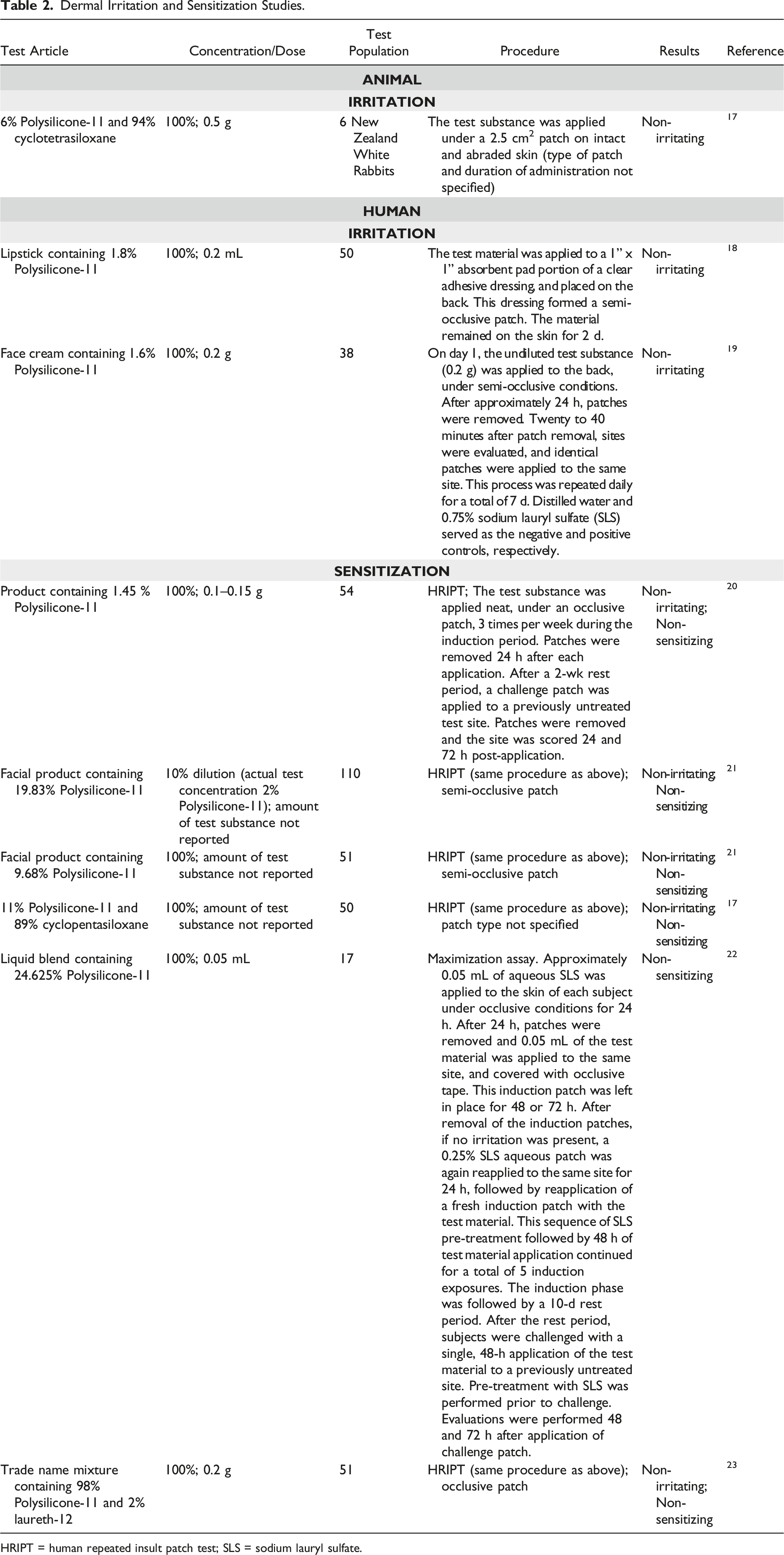
HRIPT = human repeated insult patch test; SLS = sodium lauryl sulfate.
A skin irritation study was performed on 6 New Zealand white albino rabbits. 17 The test substance (6% Polysilicone-11 and 94% cyclotetrasiloxane) was applied, undiluted, under a patch (type of patch not specified), on intact and abraded skin. The test substance was not considered to be a primary irritant. A 48-h patch test was performed on 50 human subjects using a lipstick containing 1.8% Polysilicone-11 under semi-occlusive conditions. 18 No dermal irritation was observed. Similarly, a 7-d dermal irritation study was performed on 38 subjects using a face cream containing 1.6% Polysilicone-11 under semi-occlusive conditions. 19 On day 1, patches were applied for 24 h and removed. After evaluation of the site, identical patches were applied to the same site, and the process was repeated for 7 d. The subjects showed no evidence of irritation to the test substance.
A human repeated insult patch test (HRIPT) was performed to evaluate the sensitization potential of a product containing 1.45% Polysilicone-11. 20 The test article was placed on the skin of 54 subjects, under an occlusive patch. No evidence of irritation or sensitization was observed. Another HRIPT was performed on 110 subjects using a facial product containing 19.83% Polysilicone-11. 21 Applications were made using a 10% dilution of the test substance (2% Polysilicone-11) under a semi-occlusive patch. No sensitization or irritation was observed. The amount of test substance used was not stated in either study. No sensitization or irritation was observed in an HRIPT performed on 51 subjects using a facial product containing 9.68% Polysilicone-11. 21 The product was applied neat, under semi-occlusive conditions. An HRIPT was performed on 50 subjects using a test substance consisting of 11% Polysilicone-11 and 89% cyclopentasiloxane. 17 All applications were performed neat (type of patch used not specified). The test substance was considered to be non-irritating and non-sensitizing. A maximization assay was performed on 17 subjects to evaluate the sensitization potential of a test substance containing 24.625% Polysilicone-11 (applied undiluted). 22 No instances of contact allergy were recorded at either 48 or 72 h after the application of the challenge patch. The test substance was not considered to possess a detectable contact-sensitizing potential. No signs of sensitization or irritation were observed when an HRIPT was performed on 51 subjects using a trade name mixture consisting of 98% Polysilicone-11 and 2% laureth-12. 23
Ocular Irritation Studies
In Vitro
A tissue equivalent assay was performed with EpiOcularTM cultures to evaluate the ocular irritation potential of a face cream containing 1.6% Polysilicone-11. 24 The face cream was tested neat (100 μl), the test samples were treated in duplicate, and the exposure periods were 8, 16, 20, and 24 h. Appropriate negative and positive controls were used. The ET50s (i.e., the time at which the tissue viability was reduced 50% compared to negative control tissues) for Polysilicone-11 and the positive control were 18.2 h and 30.3 min, respectively.
A MatTek EpiOcular™ methyl thiazole tetrazolium (MTT) viability assay was also performed to evaluate the ocular irritation potential of a test substance containing 98.5% Polysilicone-11. 25 The chemical was tested neat (100 μl), the test samples were treated in duplicate, and the exposure periods were 64, 256, and 1200 min. Appropriate negative and positive controls were used. The ET50 was 12 h, and the ocular irritancy classification for this test substance was “non-irritating, minimal.”
Animal
An acute eye irritation study was performed on 6 New Zealand albino rabbits using a test substance consisting of 6% Polysilicone-11 and 94% cyclotetrasiloxane. 17 Approximately 0.1 mL of the test substance was applied to the eye, undiluted. No other details regarding this study were provided. The test substance was reported to be minimally irritating.
Summary
This is a safety assessment of Polysilicone-11 as used in cosmetics. According to the Dictionary, Polysilicone-11 is a crosslinked dimethyl siloxane formed by the reaction of bis-vinyldimethicone and hydrogen dimethicone, and is reported to function as a film former in cosmetics.
According to 2020 VCRP data, Polysilicone-11 is reported to be used in 440 formulations, 432 of which are leave-on formulations. The majority of these uses are in face and neck (excluding shave) products, moisturizing products, eye lotions, and foundations. Results of the concentration of use survey conducted by Council in 2018, and updated in 2019, indicate Polysilicone-11 is used at a maximum concentration of up to 19.9% in other skin care preparations.
An LD50 of >5 g/kg was established in an acute oral toxicity study performed on Sprague-Dawley rats given a test substance consisting of 6% Polysilicone-11 and 94% cyclotetrasiloxane.
No mutagenicity was reported in an Ames assay performed using a mixture consisting of 14% Polysilicone-11, 47% dimethicone, and 39% cyclopentasiloxane. The test substance was tested on S. typhimurium (TA98 and TA100) at concentrations of up to 5000 μg/plate.
No cytotoxicity was observed in an agar diffusion test using a test substance consisting of 12–16% Poylsilicone-11, 43–50% dimethicone, and 36–42% cyclopentasiloxane.
No irritation was observed in a skin irritation study performed on New Zealand white albino rabbits using a test substance consisting of 6% Polysilicone-11 and 94% cyclotetrasiloxane. A 48-h patch test was performed on 50 subjects using a lipstick containing 1.8% Polysilicone-11. No irritation was observed. In addition, no dermal irritation was observed in a 7-d dermal irritation study (24-h patches) performed on 38 subjects using a face cream containing 1.6% Polysilicone-11.
No sensitization was observed in multiple HRIPTs using the following test materials: product containing 1.45% Polysilicone-11 (54 subjects), a 10% dilution of a facial product containing 19.83% Polysilicone-11 (2% Polysilicone-11 as actual test concentration; 110 subjects), a facial product containing 9.68% Polysilicone-11 (51 subjects), a mixture of 11% Polysilicone-11 and 89% cyclopentasiloxane (50 subjects), or a trade name mixture containing 98% Polysilicone-11 and 2% e laureth-12 (51 subjects). In a maximization assay performed on 17 subjects using a pre-treatment with SLS, the test substance (containing 24.625% Polysilicone-11) was considered to be non-sensitizing.
An in vitro tissue equivalent assay was performed in order to evaluate the ocular irritation potential of a face cream containing 1.6% Polysilicone-11. The ET50s for Polysilicone-11 and the positive control were 18.2 h and 30.3 min, respectively. A MatTek EpiOcular™ MTT viability assay was also performed to evaluate the ocular irritation potential of a test substance containing 98.5% Polysilicone-11. The ET50 was 12 h, and the ocular irritancy classification for this test substance was “non-irritating, minimal.” In an ocular irritation study in New Zealand white rabbits, a test substance consisting of 6% Polysilicone-11 and 94% cyclotetrasiloxane applied to the eyes was considered to be minimally irritating.
Discussion
The Panel determined that the available acute toxicity, genotoxicity, irritation, and sensitization data were adequate for assessing the safety of Polysilicone-11 as used in cosmetics. There was a lack of chronic toxicity and mammalian genotoxicity data in this safety assessment; however, the Panel was not concerned about these gaps because Polysilicone-11 is reported to have a large molecular weight, and therefore skin penetration would not occur. According to a supplier, this ingredient is reported to be the product of a pure addition reaction, forming no impurities and resulting in no residual monomers. In addition, safety of this ingredient was supported by the lack of adverse clinical reports.
The Panel discussed the issue of incidental inhalation exposure from powders and suntan pump sprays. The Council survey results indicate that Polysilicone-11 is being used in face powders at concentrations up to 3.5%. In addition, Polysilicone-11 is used in spray suntan products at up to 0.04%. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Polysilicone-11 is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.

