Abstract
Gastric cancer is one of the most common cancers worldwide, particularly in China, with over half a million new cases and over 400 thousand deaths in 2022. Zolbetuximab, a first-in-class investigational monoclonal antibody (mAb) targeting tumor-associated antigen CLDN18.2 which is highly expressed on gastric cancer cells, was recently reported to meet the primary endpoint in Phase III trial as first-line treatment in CLDN18.2 positive and HER2-negative gastric cancers. In the present study, we developed a humanized bispecific antibody (bsAb) CLDN18.2/4-1BB named PM1032. PM1032 activates immune cells via CLDN18.2 mediated crosslinking of 4-1BB, a potent stimulator of T/NK cells. It induced strong immunological memory in multiple tumor-bearing animal models, indicating significant potential as an effective treatment for CLDN18.2 positive cancers such as gastric cancer. Since liver and gastrointestinal (GI) related toxicities were reported in 4-1BB and CLDN18.2 targeting programs during the clinical development, respectively, extensive pharmacokinetics (PK) and safety profile characterization of PM1032 was performed in rhesus monkeys. PM1032 had a half-life comparable to a conventional IgG1 mAb, and serum drug concentration increased in a dose-dependent pattern. Furthermore, PM1032 was generally well tolerated, with no significant abnormalities observed in toxicity studies, including the liver and stomach. In summary, PM1032 demonstrated good PK and an exceptional safety profile in rhesus monkeys supporting further investigation in clinical studies.
Introduction
Gastric cancer is one of the most common cancers worldwide especially in China. Over half a million new cases and over 400 thousand deaths were reported in 2022, making it the third most commonly diagnosed cancer and the third leading cause of cancer death in China. 1 Moreover, despite significant achievements in targeted therapy for cancer treatment over the last few decades, chemotherapy remains the dominant treatment in advanced gastric cancer, resulting in poor outcomes and a high recurrence rate.
Claudin-18 isoform 2 (CLDN18.2), a membrane-bound protein involved in tight junction formation, has been identified to be expressed in various types of cancer, including gastric and pancreatic cancer, while its physiological expression is limited to the stomach. 2 Zolbetuximab (also known as IMAB362), a first-in-class chimeric monoclonal antibody (mAb) with relatively low affinity for CLDN8.2, was recently reported to have met the primary endpoint (OS) in a gastrointestinal (GI) cancer Phase III trial when combined with mFOLFOX6 (a combination regimen that includes oxaliplatin, leucovorin, and fluorouracil) compared to mFOLFOX6 chemotherapy alone.
GI-related toxic responses such as nausea, vomiting, and decreased appetite were the most common treatment-emergent adverse events (TEAEs) in patients treated with zolbetuximab plus mFOLFOX6. 3 Moreover, significant GI toxicity was also observed in various clinical studies targeting CLDN18.2 including CD3 bispecific, ADC, and CAR-T therapy, indicating potential safety concerns when directly eliminating CLND18.2+ cells on stomach epithelium.4,5
Lymphocytes express co-stimulatory receptors, which improve effector and memory responses when activated. Therefore, agonist antibodies have been used to target tumor necrosis factor receptors (TNFRs), such as CD27, 4-1BB (CD137), OX40 (CD134), and GITR (CD357). 6 As a potent T and NK cell stimulator, 4-1BB agonist showed improved anti-cancer responses in combo with immune checkpoint inhibitors, particularly in immunogenic-poor tumors. 7 Unfortunately, inherent on-target-related toxic effects in the liver were observed during the clinical development of urelumab (an agonistic mAb targeting 4-1BB developed by Bristol Myers Squibb). It is most likely due to the super-agonistic activity of urelumab and/or ultra-activation upon crosslinking of urelumab by Fc-receptors (FcRs) expressed on liver resident FcRs expressing cells, such as kupffer cells.8,9 To reduce liver-related on-target off-tumor toxicity of 4-1BB targeting therapeutics, we developed PM1032, a bispecific antibody (bsAb) conditionally crosslinking 4-1BB only in the presence of tumor-associated antigen (TAA) CLDN18.2 thus specifically activates T cells in the TAA-expressing tumor microenvironment. Furthermore, Leu234Ala/Leu235Ala (LALA) mutations, 10 a well-demonstrated mutations that eliminates the interaction between FcRs and antibody, were also introduced to the Fc region of PM1032 to further reduce potential FcRs-mediated off-tumoral T-cell activation.
We investigated the PK and toxicity of PM1032 in rhesus monkeys with special attention to liver and GI-related toxicity. In the rhesus monkey, PM1032 demonstrated an IgG1 mAb-like PK profile with a half-life of approximately 100 hours, similar to a conventional IgG antibody. Its serum concentration also displayed a dose-dependent pattern. Moreover, PM1032 illustrated an exceptional safety profile with no significant abnormalities observed during the toxicity studies including clinical observation, clinical pathology, immunotoxicity parameters, and pathological examination. In conclusion, PM1032 demonstrated an excellent PK and safety profile in rhesus monkeys, and the results support its further clinical development.
Materials and Methods
PM1032 Sample Preparation
CHO-S cell line producing PM1032 was generated by transfection of cell with pCHO2.0-GS-Puro-PM1032-H-L plasmid and selected under MSX and puromycin pressure according to the manufacturer's protocol (Life Technologies). A Biotheus in-house platform cell culture process and purification process were employed to manufacture PM1032 at the 200L scale. The final product was stored in the histidine-containing buffer in sealed glass vials.
Biolayer Interferometry (BLI)
The kinetics and affinity of PM1032 interaction with antigens were determined using biolayer interferometry (BLI), an optical technique for measuring macromolecular association. Briefly, anti-hIgG Fc capture (AHC) biosensors were first loaded with PM1032 and incubated for 150 s with 100 nM 4-1BB from the indicated species (human, rhesus/cyno monkey, mouse, and rat) to measure antigen association. The biosensors were then incubated in a wash buffer for 200 s to measure antigen dissociation. The Fortebio Data Analysis 12.0 software computed the KD (koff/kon) value.
Cell Binding Assay
CHO cells steadily expressing human or monkey 4-1BB or HEK293 cells steadily expressing human/monkey, mouse, or rat CLDN18.2 were adjusted to an appropriate concentration before being seeded at approximately 1 × 105 cells per well a 96-well plate. Cells were then incubated for 30 min at room temperature with blocking buffer (1% BSA in PBS), followed by 1 -h incubation with testing antibodies. Cells were then incubated in a blocking buffer for 30 min with appropriate secondary antibodies before being detected on a CytoFlex S system (Beckman).
Validated Enzyme-Linked Immunosorbent Assay (ELISA) for PM1032 Concentration Measurement
The ELISA plate was coated with 4-1BB ECD (Fc), and PM1032 was captured on the ELISA plate. The test reagent Goat F(ab')2 Anti-Human IgG-(Fab')2 (HRP), pre-adsorbed, was added to detect the captured PM1032 specifically. Tetramethylbenzidine (TMB) was added as the substrate. TMB and peroxide in the substrate solution produced colorimetric signals directly proportional to the PM1032 concentration in samples when HRP was applied. The color intensity (OD value) was measured at 450 nm (reference wavelength is 620 nm).
Validated Electrochemiluminescence Method (ECL) for anti-PM1032 Antibody Detection
Biotinylated PM1032, ruthenylated PM1032, tris buffer and acid-dissociated samples were incubated together to form a “Bio-PM1032-ADA--Ru-PM1032” bridging complex and then added to the Meso Scale Discovery (MSD) plate coated with Streptavidin. Biotin in the bridge complex binds specifically to streptavidin. Ruthenium is excited by voltage to generate electrochemical signals, whose signal value is proportional to the amount of anti-drug antibody in the sample.
Pharmacokinetic Analysis
A total of 18 rhesus monkeys were divided into three groups, each with three animals of each sex. PM1032 was administered intravenously to monkeys in a single dose of 3, 10, and 30 mg/kg. Blood samples were collected for PK analysis at pre-dose, 2 min, and 1, 3, 8, 24, 48, 72, 96, 120, 144, 168, 240, 336, 504, and 672 h post-dosing. Blood samples were collected for anti-drug antibody (ADA) analysis before and on days 8, 15, 22, and 29 after dosing. Supernatant from centrifuged samples (without anticoagulant) was collected and stored until analyzed. The ELISA assay was used to determine the PM1032 concentration in the serum, while the MSD assay was used to determine the ADA of PM1032. The PK parameters, such as the area under the curve (AUC), maximum concentration (Cmax), and the volume of distribution at steady-state (Vss) and clearance (Cl), were calculated using the WinNonlin 8.1 non-compartmental model.
Good Laboratory Practice (GLP) Toxicity Study in Rhesus Monkeys
A repeat-dose toxicity study in rhesus monkeys was performed in accordance with GLP at TriApex Laboratories Co., Ltd to determine the PM1032 toxicity. PM1032 at the dosage of 0 (vehicle), 50, 100, and 150 mg/kg were administered intravenously once a week for a 4-week dosing period, with scheduled necropsies performed on three monkeys/sex in each group at the end of the dosing phase and on the remaining animals at the end of the 4-week recovery phase. General clinical signs, body weight, food consumption, body temperature, electrocardiograms, ophthalmology examination, clinical pathology parameters (hematology, coagulation, serum chemistry, serum electrolyte, and urinalysis) and immunotoxicity parameters (lymphocyte populations, cytokines, and circulating immune complex), organ weight, and macroscopic and microscopic pathology examination were monitored regularly throughout the study as represented in Supplementary Table 1.
Moreover, toxicokinetics (TK) and ADA were assessed to further validate the toxicity study. Blood samples were collected at the pre-determined time points (Supplementary Table 1) during the study. The samples were centrifuged at 1500 g for 15 min at room temperature, and the supernatant was collected and stored below −65°C until analyzed. A validated ELISA method was performed to measure the drug concentration. AUC and Cmax were calculated using WinNonlin 8.1 software using a non-compartmental analysis. A validated ECL method was performed to measure the ADA.
Regulatory Compliance
The laboratories of TriApex are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International. In the present study, the animal experiment-related contents and procedures adhere to the relevant requirements of the Institutional Animal Care and Use Committee (IACUC) at TriApex Laboratories. The study protocols were reviewed and agreed by IACUC (IACUC No.: PK-AP-2138-209N and Toxicity-AP-2138-210).
Toxicity studies comply with GLP for Non-Clinical Studies of Drugs (September 2017) issued by the National Medical Products Administration (NMPA), GLP for Non-clinical Laboratory Studies in 21 CFR Part 58 of the United States Food and Drug Administration (FDA) and Organization for Economic Co-operation and Development Principles of Good Laboratory Practice, ENV/MC/CHEM (98)17.
The monkeys were euthanized according to procedures recommended by the AVMA Guidelines for the Euthanasia of Animals (2013 Edition). The monkeys were euthanized with Zoletil 50 (5 mg/kg, intramuscular injection) combined with xylazine hydrochloride (2 mg/kg, intramuscular injection) and exsanguinated by severance of the descending aorta.
Statistical Analysis
All data for the in vitro assay came from at least three independent repeats, and a difference of P < .05 was considered significant. GraphPadPrism was used for statistical analysis. The appropriate test, such as Mann–Whitney (unpaired) t test and one-way or two-way ANOVA, was used based on the data. For the PK study, statistical analysis was performed in Microsoft Excel 2010 using the F-Test and t test. For repeated toxicity studies, data were processed, statistical analysis was performed using SAS statistical software built-in Provantis 10.2.3, and one-way ANOVA or Kruskal–Wallis was applied.
Results
PM1032 Binds to Human/Monkey CLDN18.2 and 4-1BB with Similar Affinity
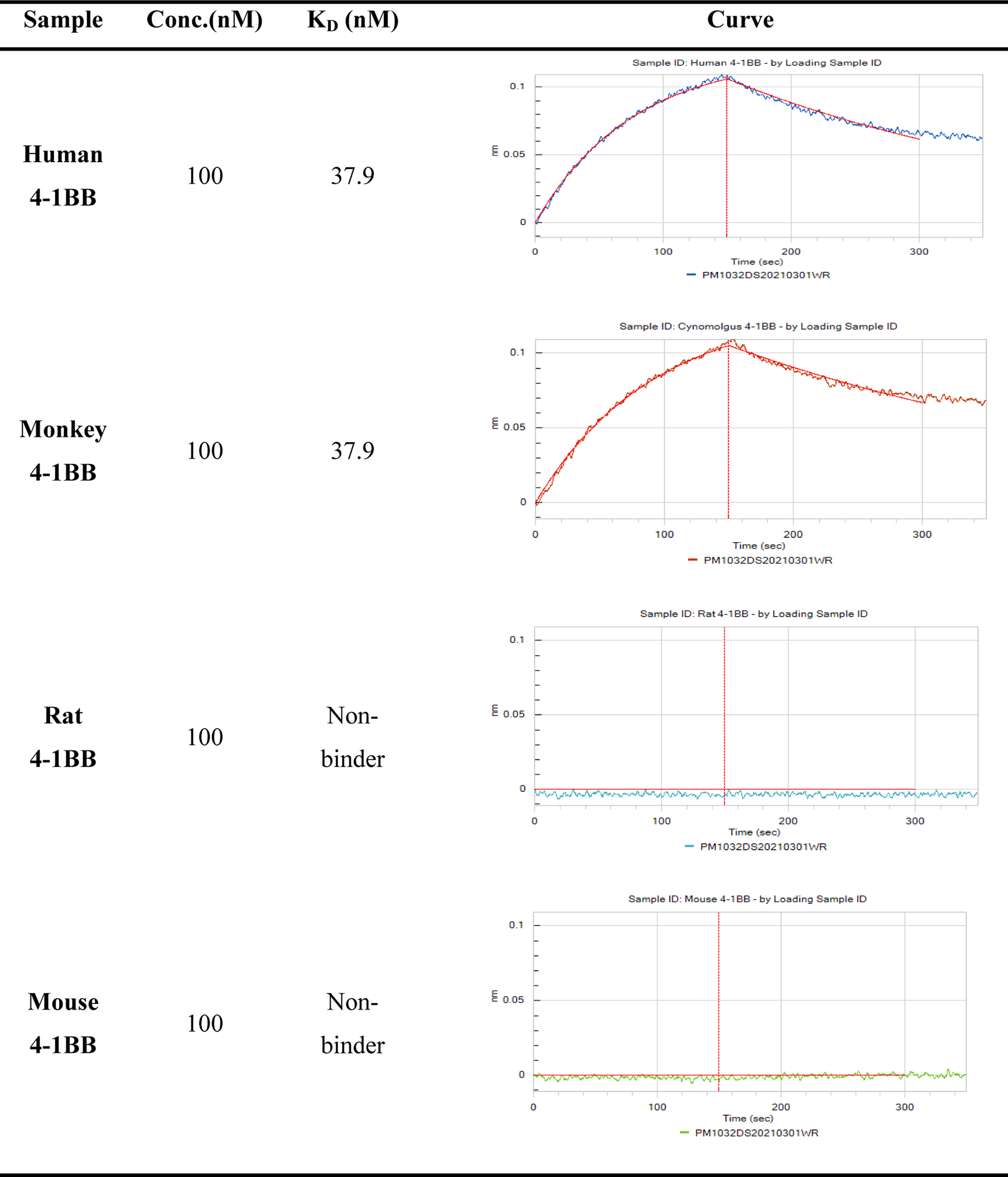
Affinity Analysis of PM1032 and 4-1BB in Different Species.
Furthermore, we generated the CHO cell line that steadily expresses human or monkey 4-1BB, with the EC50 of PM1032 binding to cells was 6.7 nM and 8.3 nM, respectively (Figure 1A). As shown in Supplementary Table 3, the sequence of CLDN18.2 ECD is completely conserved between humans and monkeys but differs from mouse or rat CLDN18.2 on ECD2 by a few amino acids. We then developed HEK293 cells expressing human/monkey, mouse or rat CLDN18.2. PM1032 showed a similar binding profile on these three cell lines with the EC50 of 2.2 nM, 1.1 nM, and 3.1 nM, respectively (Figure 1B). These findings demonstrated that PM1032 interacts with CLND18.2 and 4-1BB with a similar affinity and cell binding profile between humans and rhesus monkeys, supporting the use of rhesus monkeys for PK and toxicity studies. Moreover, since PM1032 does not interact with rodent 4-1BB, rhesus monkey has been selected as the only relevant species for further toxicity study. Species cross-reactivity of PM1032 to 4-1BB and CLDN18.2. (A) The binding profiles of PM1032 on CHO cells expressing human or monkey 4-1BB. (B) PM1032 binding profiles on HEK293 cells expressing human/monkey, mouse, or rat CLDN18.2.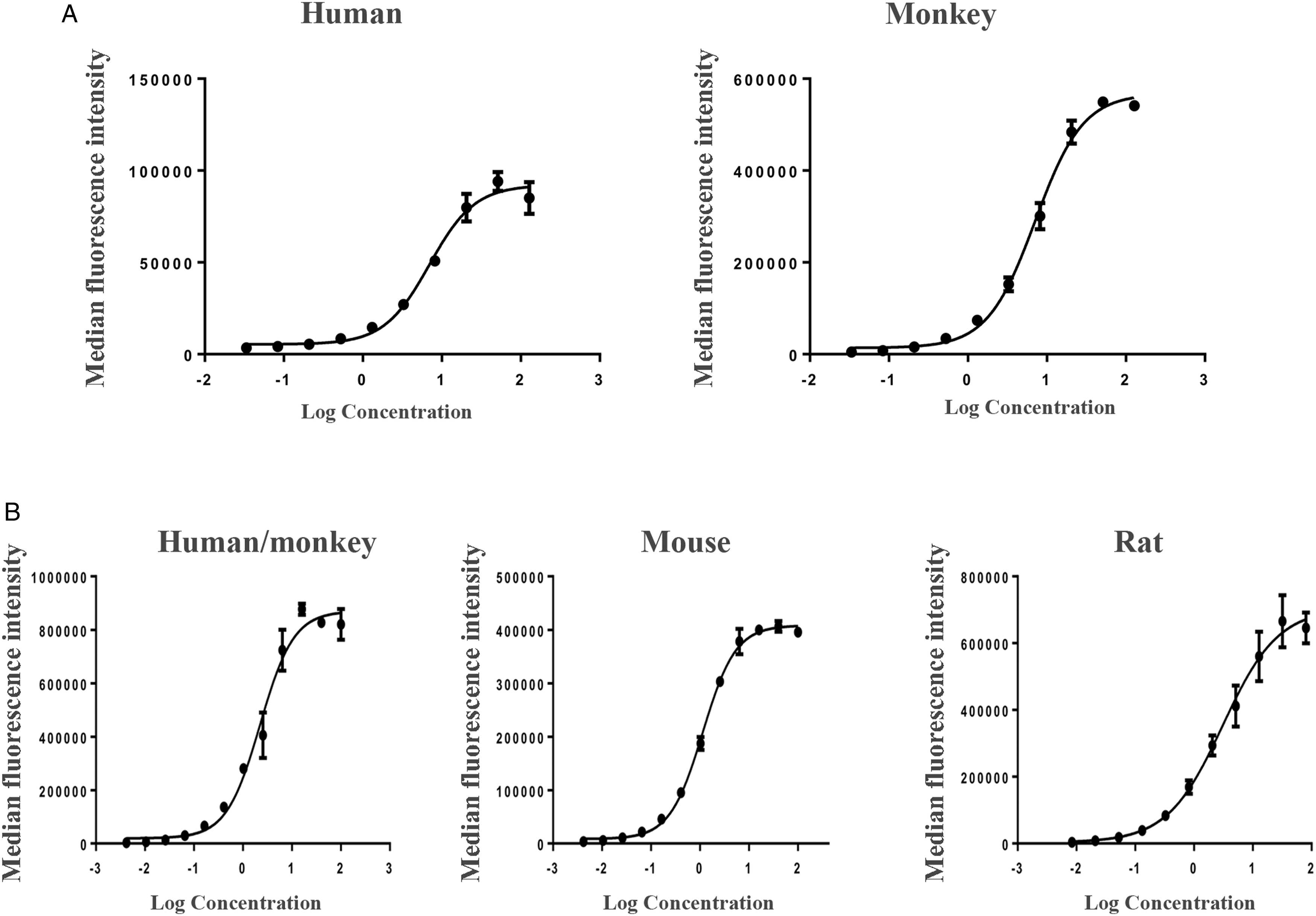
PM1032 Displayed an IgG1 mAb-like PK Profile
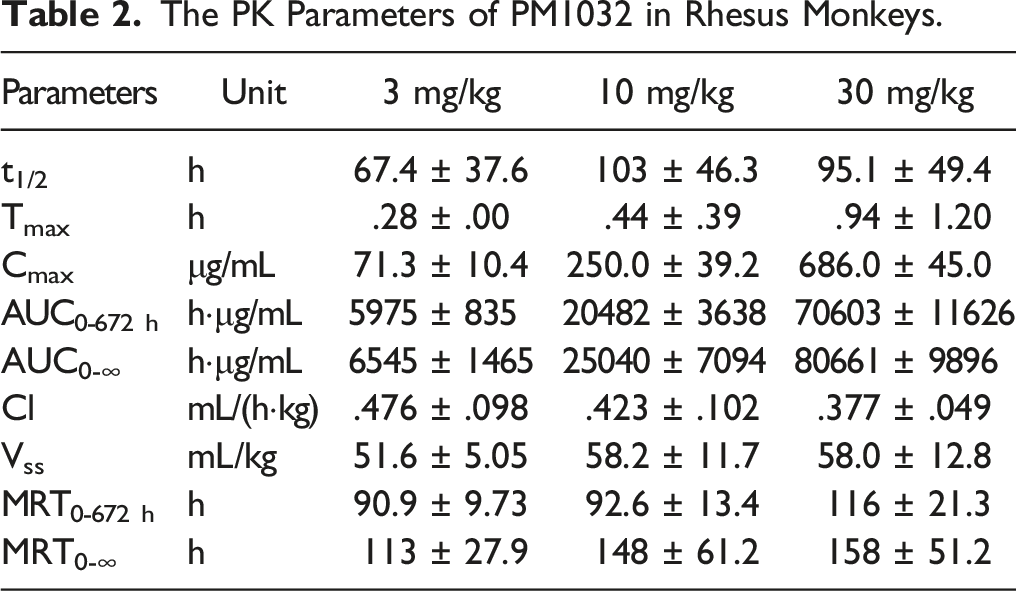
PM1032 was intravenously (iv) injected to rhesus monkeys with a single dose of 3 mg/kg, 10 mg/kg, and 30 mg/kg, and the blood samples were collected at indicated time points up to 29 days as described in method. The serum concentration of PM1032 from these three dosing groups displayed a dose-dependent pattern. Moreover, the drug concentration remained relatively high, with calculated half-lives (T1/2) of 67.4 ± 37.6 h, 103 ± 46.3 h, and 95.1 ± 49.4 h at 3, 10, and 30 mg/kg, respectively, comparable to conventional monoclonal antibody (Figure 2). Furthermore, as shown in Table 2, the time to peak drug concentration (Tmax) was .28 ± .00 h, .44 ± .39 h, and .94 ± 1.20 h, respectively; while the maximum drug concentration (Cmax) was 71.3 ± 10.4 μg/mL, 250.0 ± 39.2 μg/mL, and 686.0 ± 45.0 μg/mL, respectively. The clearance (Cl) of PM1032 was .476 ± .098 mL/(h·kg), .423 ± .102 mL/(h·kg), and .377 ± .049 mL/(h·kg) in the corresponding group while drug exposure (AUC0-672 h) was 5975 ± 835 h·μg/mL, 20482 ± 3638 h·μg/mL, and 70603 ± 11626 h·μg/mL, respectively. There was no gender difference observed. All the data (Cmax and AUC0-672h) were approximately present at the 1:3.3:10 ratio, which was consistent with the injection dose of PM1032. In conclusion, PM1032 demonstrated a typical IgG1 mAb-like half-life after a single intravenous administration, with serum concentration and drug exposure exhibiting an excellent correlation with administration dose. Pharmacokinetic concentration-time profiles of PM1032 in rhesus monkeys. A total of 18 monkeys were divided into three groups, each with three animals of each sex. Monkeys in each group received a single intravenous dose of PM1032 at 3, 10, or 30 mg/kg. Serum drug concentration was detected and plotted at the indicated time points (pre-dose and 2 min and 1 h, 3 h, 8 h, 24 h, 48 h, 72 h, 96 h, 120 h, 144 h, 168 h, 240 h, 336 h, 504 h, and 672 h). The PK Parameters of PM1032 in Rhesus Monkeys.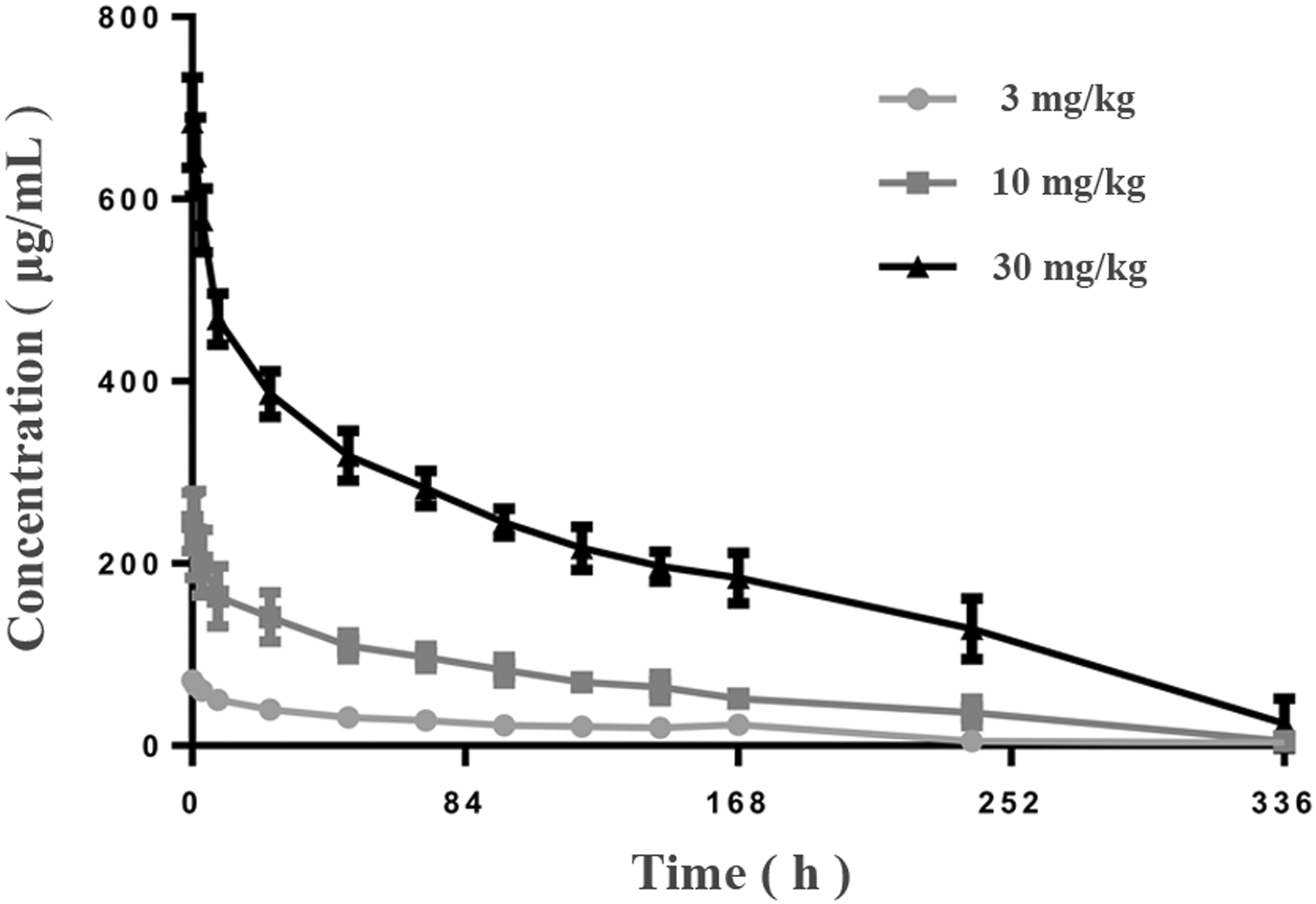
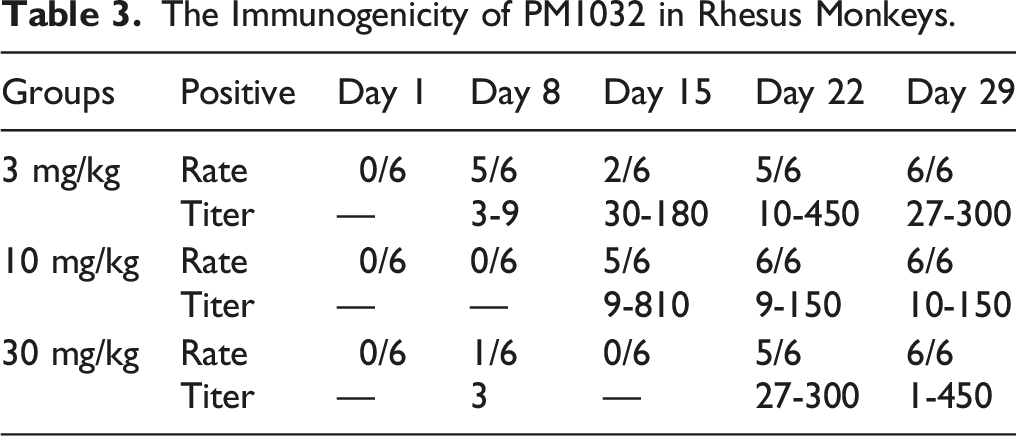
The Immunogenicity of PM1032 in Rhesus Monkeys.
PM1032 did Not Induce Significant Abnormalities in the GLP Toxicity Studies
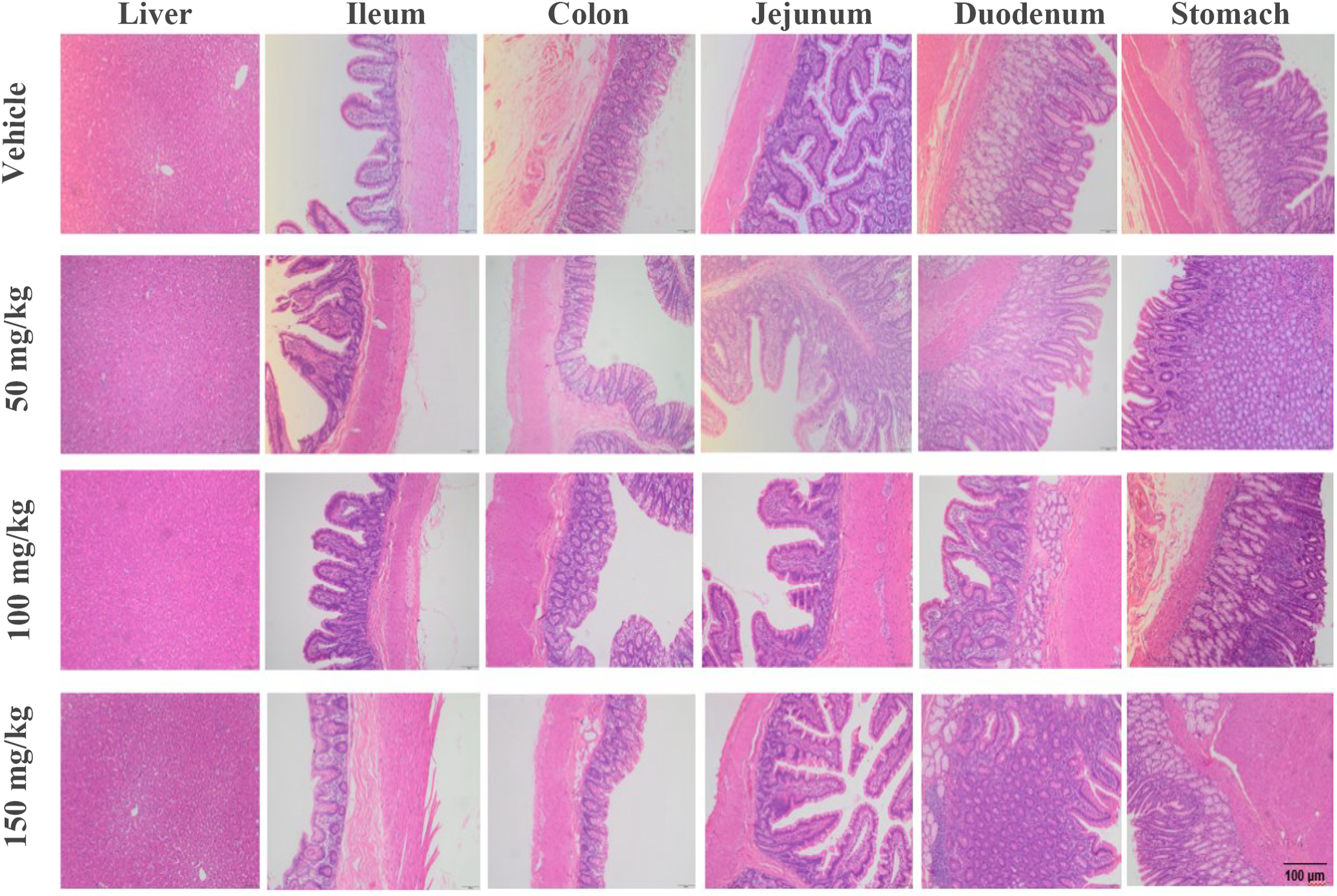
PM1032 was intravenously injected to rhesus monkeys once a week for four times followed by a 4-week recovery phase as described in the methods. Clinical observations such as behavior, food consumption, body weight, body temperature, and electrocardiograph (ECG) were monitored, and blood samples were collected at the specified time points (Supplementary Table 1, full list of testing items are following the Technical Guidelines for drug on Repeated Dose Toxicity Study (May 2014) issued by NMPA and ICH S6 Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals (supplementary, Jun. 2011)). Animals were then sacrificed, and organs each as heart, lung, liver, stomach, kidney, brain, and other organs were collected and further examined by pathologists (Supplementary Table 1). Treatment of 50, 100, or 150 mg/kg PM1032 did not induce any test article-related findings compared to either the control group (saline) or the pre-dose baseline. For example, PM1032 did not cause a decrease in food consumption or weight loss (Figure 3A), which was reported as one of the most common side effects of IMAB362 in clinical trials. However, after a single dose of 20 mg/kg, the CLDN18.2 mAb using the same antigen binding domain as PM1032, reduced food consumption and weight loss (∼10% in one animal) (Supplementary Figure 1(a)). Furthermore, PM1032 had no significant impact on the biochemical profile, including AST, ALT, TBIL, BUN, TP, ALB, GLOB, and CREA (Figure 3B–I, Supplementary Table 4) or urinalysis (Supplementary Table 4). Finally, we observed no treatment-related histopathological changes in any of the examined organs during the microscopic examination compared to the control group (Figure 4). Furthermore, no significant weight difference was observed of all the examined organs across treatment groups (Supplementary Table 5). In conclusion, repeated injections of PM1032 at doses of up to 150 mg/kg did not raise any significant safety concerns in the GLP toxicity study. Body weight and biochemical results of repeat-dose toxicity study of PM1032 in rhesus monkeys. Monkeys were randomly divided into four groups, each with five animals of each sex. They were administered intravenously with doses of 0 (vehicle) or 50, 100, and 150 mg/kg of PM1032 once a week for four weeks (four doses in total) followed by a 4-week recovery phase. (A) Body weight for all animals was recorded once a week as indicated. (B)-(I) Biochemical profiles including AST, ALT, TBIL, BUN, TP, CREA, ALB, and GLB were measured at indicated time points. Examples of the H&E stain stomach, rectum, and liver from PM1032 repeat-dose toxicity study in rhesus monkeys. The above tissues and organs were trimmed, embedded in paraffin, sectioned, stained in hematoxylin and eosin (H&E), and examined microscopically. The samples from each dosing group were presented as labelled, and no relevant histopathological changes were observed in any PM1032 treated groups.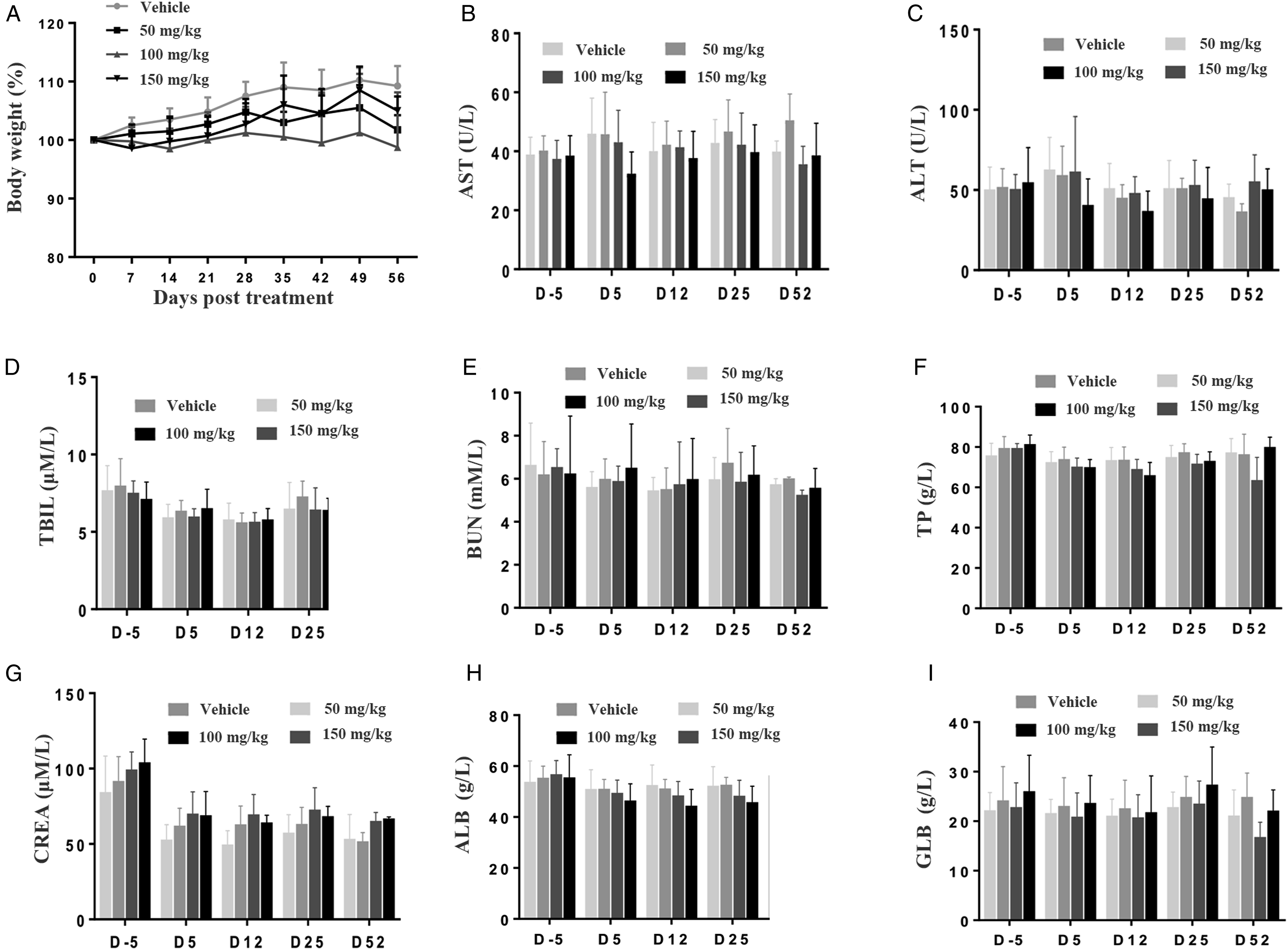
The TK Profile of PM1032 Was Not Influenced by ADA
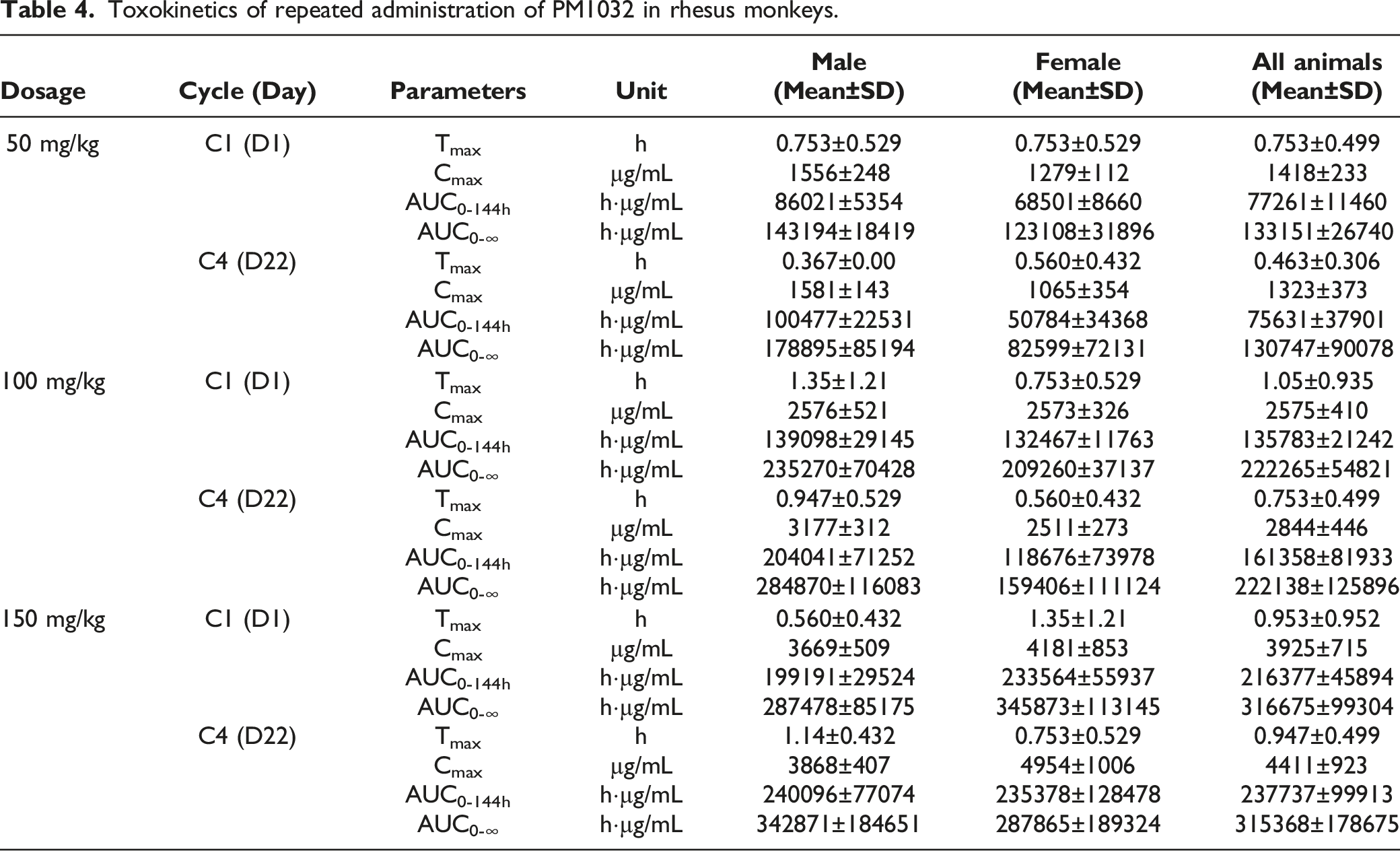
TK analysis was performed to further confirm that PM1032 has an exceptional safety profile in rhesus monkeys. No obvious changes in drug serum concentration were observed between each cycle including Cmax, Tmax, and AUC, which validated sufficient PM1032 exposure during the toxicity study (Figure 5). Moreover, as shown in Table 4, the Cmax of cycle 1 was 1418 ± 233 μg/mL, 2575 ± 410 μg/mL, and 3925 ± 715 μg/mL in 50 mg/kg, 100 mg/kg, and 150 mg/kg groups, respectively, and the ratio of Cmax was 1.0:1.82:2.77, which were consistent with the dosing ratio. The Cmax of cycle 4 was 1323 ± 373 μg/mL, 2844 ± 446 μg/mL, and 4411 ± 923 μg/mL, respectively, and was proportional to the injection dose. In agreement with the Cmax data, the drug exposure parameter AUC0-144 h illustrated a similar pattern, with the ratio at cycle 1 being 1.0:1.76:2.80 and 1.0:2.13:3.14 at cycle 4. Finally, all animals developed ADA after repeated dosing of PM1032, but there was no effect of ADA on the drug TK profile. This is consistent to the knowledge that ADA developed during toxicity study in monkey does not usually affect the PK/TK profile of the drug.
11
In conclusion, PM1032 exposure increased dose-dependently in each dosing cycle, and the TK profile from the repeated dosing toxicity study indicated sufficient PM1032 exposure. The TK profile of PM1032 from the repeated-dosing toxicity study in rhesus monkeys. Blood samples from each group were collected at the indicated time (pre-dose and 2 min, 1 h, 3 h, 6 h, 24 h, 48 h, 96 h, and 144 h post-dosing from the 1st and 4th dosing period, and pre-dose and 2 min post-dosing from the 2nd and 3rd dosing period). The drug concentration was measured and plotted as indicated. Toxokinetics of repeated administration of PM1032 in rhesus monkeys.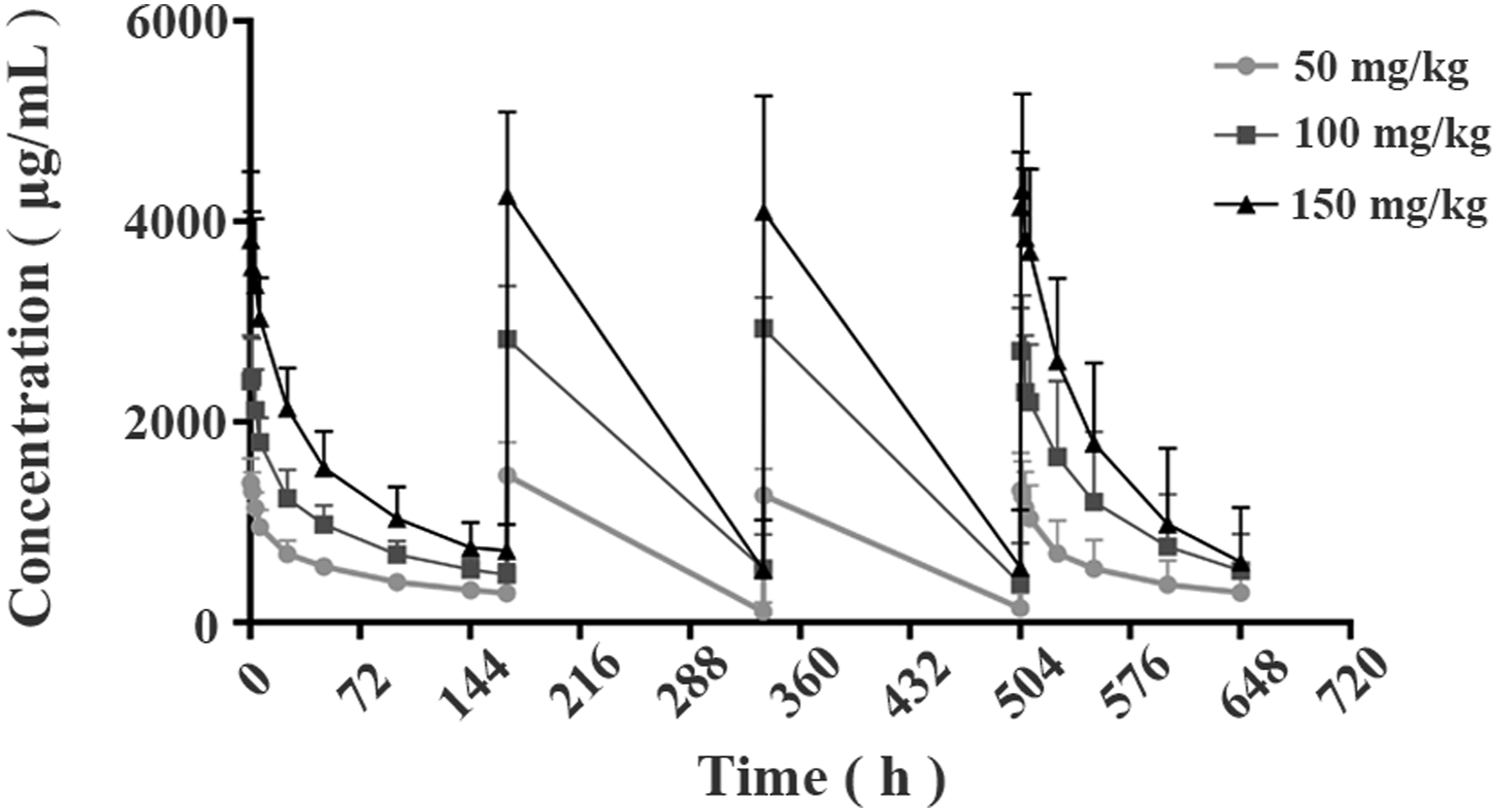
Discussion
Despite advancements in cancer research over the last two decades, gastric cancer still has a poor 5-year survival rate which is approximately 30% globally. 12 In addition to chemotherapy, only human epidermal growth factor receptor 2 (HER2) targeted agent trastuzumab and some immune checkpoint inhibitors such as nivolumab and pembrolizumab were approved in the advanced stage. 13 Therefore, there is still a significant unmet medical need in gastric cancer therapy that demanding the development of effective treatment such as novel targeted therapy.
CLDN18.2, a highly conserved protein across species, expresses only in the stomach and is only involved in tight junction formation under physiological conditions. During cancer development, the expression of CLDN18.2 was identified in over 70% of primary gastric cancer samples and various metastatic cancers, including pancreatic cancer, lung cancer, ovarian cancer, and esophagus cancer2,14 When combined with chemotherapy, targeting CLND18.2 with a monoclonal antibody, zolbetuximab significantly improved overall survival more than chemotherapy alone. 15 However, the efficacy is only moderate. Instead of inducing antibody-dependent cell-mediated cytotoxicity (ADCC) against CLDN18.2+ cancer cells such as zolbetuximab, other CLDN18.2 targeting agents including ADC, CD3-bispecific antibody, or CAR-T were actively under investigation at both preclinical and clinical stages.4,5 Although most preclinical studies demonstrated improved anti-tumor efficacy compared to zolbetuximab, higher GI-related toxicities were also observed raising safety concerns about directly eliminating CLDN18.2+ cells (both cancer cell and normal epithelia cell on the GI tract).4,5
In the present study, we developed PM1032, a CLDN18.2/4-1BB bispecific antibody that induces 4-1BB signal transduction, which activates T cells via tumor-associated antigen CLDN18.2 mediated crosslinking. PM1032 does not significantly cause direct CLDN18.2 positive cell elimination due to loss of Fc-effector function after LALA mutations (data not shown). Instead, PM1032 stimulates tumor-experienced T cells which frequently have upregulated 4-1BB expression on their surface. The resulting activation and proliferation through CLDN18.2+ cell-mediated crosslinking in turn induces potent anti-tumor efficacy with immunological memory. However, in clinical trials, the therapeutic monoclonal 4-1BB antibody urelumab was found to cause severe liver toxicity by activating T cells via 4-1BB signal transduction. 9 Therefore, PM1032 was well designed to avoid potential systemic (e.g., liver) toxicity at indicated doses. First, a single-domain antibody fragment (vHH) targeting the membrane-proximal region of 4-1BB was prepared and used in PM1032, where binding at the unique 4-1BB epitope induces T-cell activation at an optimized level after crosslinking, allowing for the balance of efficacy and toxicity. 14 The first bispecific antibody based on this unique 4-1BB vHH binder PM1003, an anti-PD-L1/4-1BB bispecific, demonstrated a good safety profile in a phase I dose-escalation study. Furthermore, LALA mutations were also introduced into PM1032 to eliminate any unwanted 4-1BB signal transduction via FcR-mediated crosslinking. 10 Therefore, PM1032 was designed to specifically activate tumor-experienced T cells in tumor microenvironment via cancer cell surface CLDN18.2 instead of mediating direct killing against CLDN18.2 expressing cells, for example, epithelial cells on normal tissue of the stomach. Importantly, as an upregulated co-stimulatory receptor, 4-1BB expresses on antigen-experienced T cells of which their TCR likely recognizing tumor-associated and/or tumor-specific antigens, and thus unlikely to induce immune response against normal tissues. This unique mechanism of PM1032 is thought to be the reason for its low GI-related and liver toxicity. Indeed, in the GLP toxicity study, we observed that PM1032 has an excellent safety profile. For instance, we found no evidence of any liver or GI-related toxicity, such as weight lost, reduced food consumption, and changes in the clinical pathology profile, including AST, ALT, TBIL, BUN, TP, CREA, ALB, and GLB (Figure 3). In contrast, CLDN18.2 mAb, which shares the same antigen binding domain as PM1032, induced GI toxicity in monkeys after a single 20 mg/kg injection (Supplementary Figure 1(a)). Moreover, PM1032 did not induce any detectable signs of toxicity in tissues other than the liver and gastrointestinal tract such as the heart, kidney, and lung further supporting the unique molecular and biological properties as the reason for minimum systemic activation of T cells. This is consistent with a recently published study of another CLDN18.2x4-1BB bispecific antibody, givastomig, of which it also displayed a good safety profile in NHP. 16 Importantly, the TK profile from the repeated dose toxicity study indicated good drug exposure after each injection confirming the safety profile of PM1032. Finally, PM1032 demonstrated an IgG1 mAb-like PK profile with an approximate half-life of 70–100 h, indicating that a relatively long dosing interval is feasible. Although ADA was detected in all animals, no significant changes in the PK profile were observed. These findings strongly supported the advancement of PM1032 to the clinical stage.
Collectively, our data demonstrated that PM1032 has an excellent safety profile in rhesus monkeys, supporting its development as a promising therapeutic agent in clinical applications such as gastric cancer. Based on our preclinical studies, a starting dose of .3 mg/kg PM1032 was used in a first-in-human (FIH) trial with a biweekly or triweekly administration schedule. To date, the phase I escalating study in solid tumors is ongoing, and the PK and safety data collected so far were in consistent with the findings in preclinical studies, indicating a very promising clinical development potential. Dose-escalation studies in CLDN18.2-positive gastric cancer patients are planned. PM1032, a novel targeted immunotherapeutic agent with high anti-tumor activity and the ability to induce immune memory induction while maintaining a good safety profile, is hoped to bring a new therapeutic option to gastric cancer patients where unmet medical needs remain high.
Supplemental Material
Supplemental Material - Safety and Pharmacokinetic Assessment of the FIC CLDN18.2/4-1BB Bispecific Antibody in Rhesus Monkeys
Supplemental Material for Safety and Pharmacokinetic Assessment of the FIC CLDN18.2/4-1BB Bispecific Antibody in Rhesus Monkeys by Jing Wang, Tiantian Dong, Xinjiang Gong, Deli Li, Joanne Sun, Yi Luo, and Huazhang Wu in International Journal of Toxicology.
Footnotes
Author Contributions
W.J. and D.T.T. designed and preformed the experiments, derived the models and analyzed the data. G.X.J. contributed to the project design and data analysis. L.D.L. assisted experiment conduction and data management. S.J. helped supervise the project. L.Y. took the lead in writing the manuscript and helped shape the research. W.H.Z. supervised the findings of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was partially supported by the Natural Science Foundation of Bengbu Medical College (2022byfy002) and the Natural Science in Higher Education of Anhui Province (KJ2020A00561).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.