Abstract
When conducting toxicology studies, the interpretation of drug-related neurological clinical signs such as convulsions, myoclonus/myoclonic jerks, tremors, ataxia, and salivation requires an understanding of the spontaneous incidence of those observations in commonly used laboratory animal species. The spontaneous incidence of central nervous system clinical signs in control animals from a single facility using cage-side observations or high definition video monitoring was retrospectively analyzed. Spontaneous convulsions were observed at low incidence in Beagle dogs and Sprague–Dawley rats but were not identified in cynomolgus monkeys and Göttingen minipigs. Spontaneous myoclonic jerks and muscle twitches were observed at low incidence in Beagle dogs, cynomolgus monkeys, and Sprague–Dawley rats but were not seen in Göttingen minipigs. Spontaneous ataxia/incoordination was identified in all species and generally with a higher incidence when using video monitoring. Salivation and tremors were the two most frequent spontaneous clinical signs and both were observed in all species. Data from the current study unveil potential limitations when using control data obtained from a single study for toxicology interpretation related to low incidence neurological clinical signs while providing historical control data from Beagle dogs, cynomolgus monkeys, Sprague–Dawley rats, and Göttingen minipigs.
Introduction
The identification of adverse effects associated with drugs in preclinical development 1 is crucial in toxicology and safety pharmacology studies.
In conventional toxicological studies, the neurotoxicity profile of a drug is assessed through the evaluation of cage-side observations for the emergence of adverse clinical signs but also with a pathology evaluation of neuronal tissues. 2 Amongst the adverse drug reactions most commonly related to drug effects on the central nervous system (CNS) are seizures. These are amongst the most frequently observed findings in preclinical safety assessments: 3 67% of participants from an industry survey listed seizure as a CNS issue encountered within the last 5 years. Indeed, several drug classes are known to induce epileptiform (or seizurogenic) activity and include cholinesterase inhibitors, 4 dopamine agonists, 5 quinolone antibacterial agents, 6 and gamma-aminobutyric acid (GABA) agonist, 7 but there are many other drug classes with similar profile of neurotoxicity. Withdrawal effects with opioids, amphetamines, and barbiturates have been extensively characterized. 8 Assessing neurological clinical signs is an essential part of the safety pharmacology core battery. 9
In a standard study design, the inclusion of a control (or vehicle) group allows for the differentiation between spontaneous clinical signs and drug-related neurological effects. Spontaneous clinical signs in healthy animals, especially at low incidence can confound the interpretation of test article-related effects. Characterizing species-specific spontaneous neurological clinical signs can help elucidate and interpret drug-related effects and differentiate these from species-specific variations in background findings. This can also support Refinement in the application of the 3Rs and could potentially Reduce the sample size in toxicology studies.
To determine the incidence of neurologically related clinical observations in control animals, we compiled observations of spontaneous neurological signs including tremors, myoclonic jerks, convulsion, ataxia, muscle twitches, and salivation10-17 for studies performed at Charles River Laboratories Laval. The studies were conducted either with cage-side observation (between 2009 and 2019) or with continuous video monitoring (between 2014 and 2022). The latter method allowed for a higher temporal resolution compared to cage-side observations and generally avoided any interference related to the presence of the technical staff in the room.
Neurological clinical signs were evaluated in standard safety pharmacology and toxicology preclinical species, including Beagle dogs, Sprague–Dawley rats, Göttingen minipigs, and cynomolgus monkeys. These control animals were administered formulation vehicle by various administration routes.
Materials and Methods
Animals
For cage-side observations, data sets of adult control animals from 85 studies comprised of 1015 Beagle dogs (Marshall Bioresources); from 131 studies comprised of 4569 Sprague–Dawley rats (Charles River); from 46 studies, comprised of 469 Cynomolgus monkeys (Charles River); and from 23 studies, comprised of 209 Göttingen minipigs (Marshall Bioresources) were compiled and analyzed. For video monitoring analyses, data sets from control adult Beagle dogs from 18 studies comprised of 174 Beagle dogs (Marshall Bioresources); from 10 studies comprised of 165 Sprague–Dawley rats (Charles River); and from 18 studies, comprised of 170 cynomolgus monkeys (Charles River) and 1 data set of 24 Göttingen minipigs (Marshall Bioresources) were compiled and analyzed. Data were retrospectively compiled for cage-side observations from studies conducted at a single facility (Charles River Laboratories Laval) between 2009 and 2019. Similarly, data for clinical signs observed by video monitoring were evaluated from 2014 to 2022 at the same facility. All the procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) prior to study initiation. The research facility where the studies were conducted is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) and the Canadian Council on Animal Care (CCAC). The current analysis did not include reproductive and developmental toxicology studies.
Selection of Studies and Control Animals
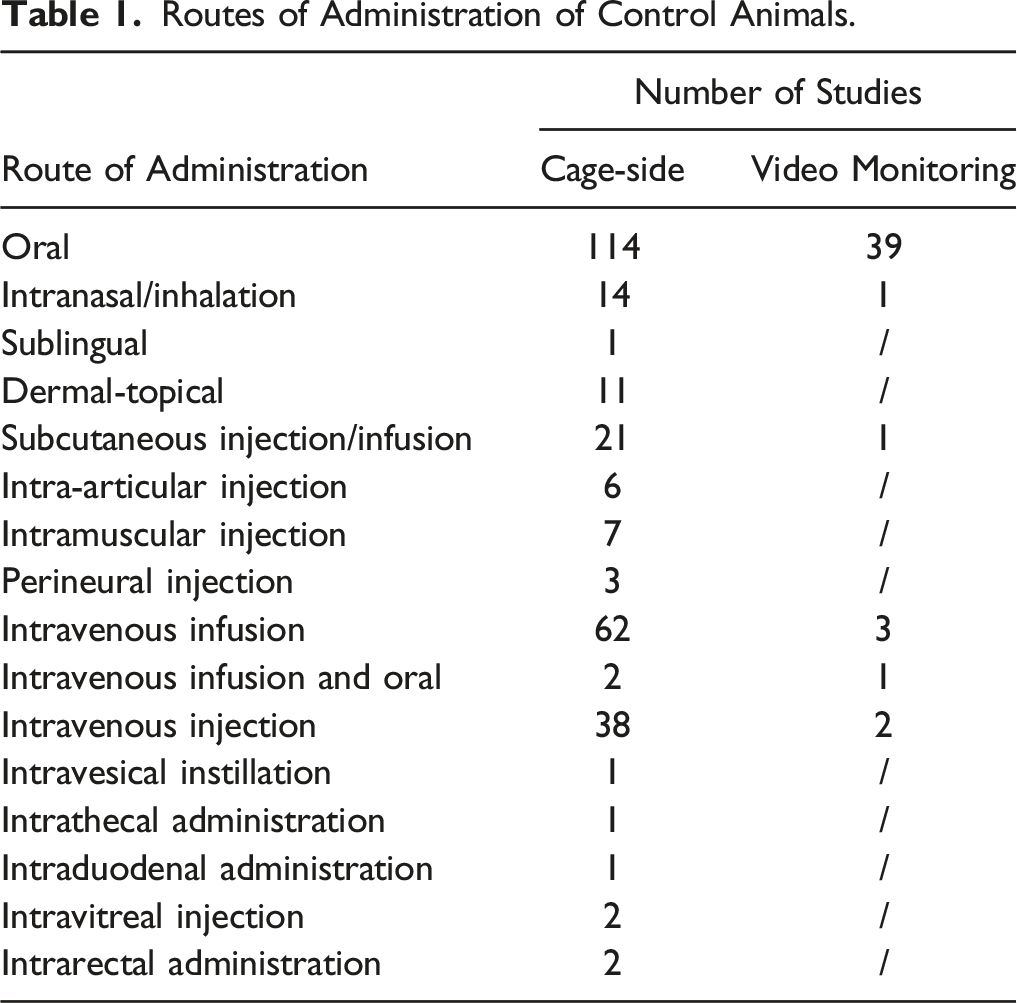
Routes of Administration of Control Animals.
Average Length of the Studies for Cage-side and Video Monitoring Observations by Species.
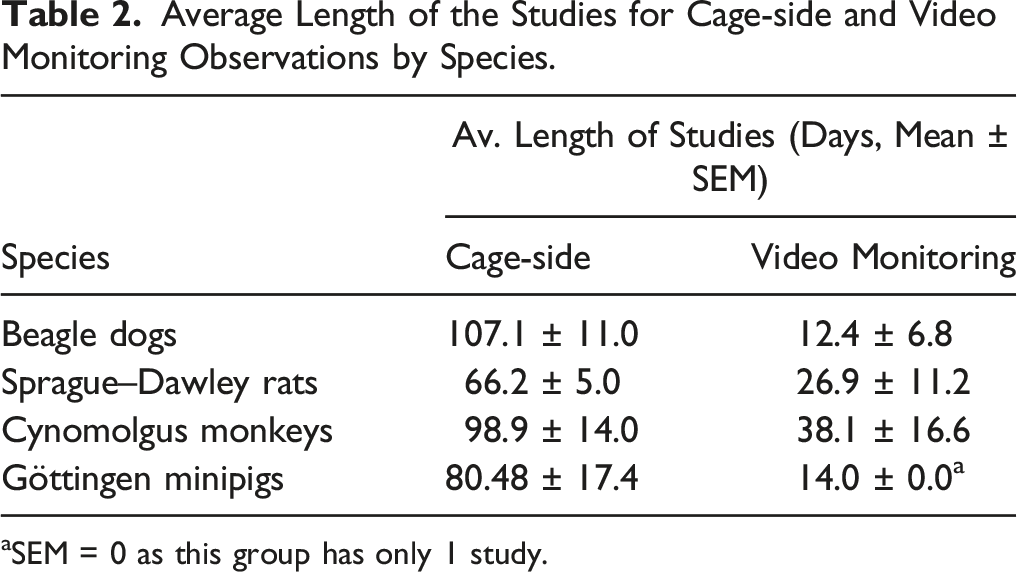
aSEM = 0 as this group has only 1 study.
Clinical Sign Evaluation
Clinical signs from control animals were recorded using Provantis® versions 9 and 10 (Instem, Stone, Staffordshire, UK) and compiled. The observations were identified and recorded by technicians, which were trained to detect clinical signs for each studies working in a contract research organization (CRO). In Provantis®, the clinical signs were defined as present, whether mild, moderate or high/severe in nature. Given the wide manifestations compatible with seizures in animals (motor, autonomic, and behavioral signs) and the difficulty to identify them without concomitant EEG, we chose to group these observations under the term “convulsions.” Tonic seizures are characterized by rigid, sustained muscular contractions, fixing the limbs or body part(s) in some strained position. Clonic seizures are characterized by repeated jerking movements of the pelvic and/or thoracic limbs on one or both sides of the body. Tonic-clonic seizures are a combination of those 2 types of movements.10,18 Myoclonus is defined as a sudden and involuntary brief movement of a body part due to an abrupt contraction of one or more muscle (positive myoclonus)15,16,19 or brief cessation of ongoing muscular activity (negative myoclonus). Tremors are defined as “involuntary, rhythmic oscillatory movements of a body part with symmetric velocity in both movement directions (ie, sinusoidal), around a joint axis.” 19 Ataxia is a term use for uncoordinated gait or movements while salivation was noted when an increased amount of saliva was present in the buccal cavity. Muscle twitches are defined as a sudden contraction of a muscle or group of muscles visible at the skin surface.
Video Monitoring
When applicable, animals were continuously monitored using video cameras. High-definition digital color cameras (Geovision, Irvine, CA, USA), with daylight and infrared night vision connected to a computerized system (IBM Intellistation Z pro, Xeon 3.8 Ghz, 3.5 TB hard drive, New Orchard Road Armonk, NY, USA) were used for behavioral video monitoring assessments.
Data Reporting and Statistical Analysis
The statistical analysis was conducted by collecting the occurrence of clinical signs for control groups in all the studies in Microsoft Excel. The incidence proportion expressed as a percentage (%) across the studies was calculated for each species as the number of animals showing a specific clinical sign/total number of control animals in all studies. The average length of the studies from which the data was derived was calculated for each species as the average number of days for all the studies (ie, testing and recovery period, if applicable) and expressed as mean ± standard error of the mean (SEM). No inferential statistics were performed between cage-side observations and video monitoring observations given methodology and collection time biases.
Results
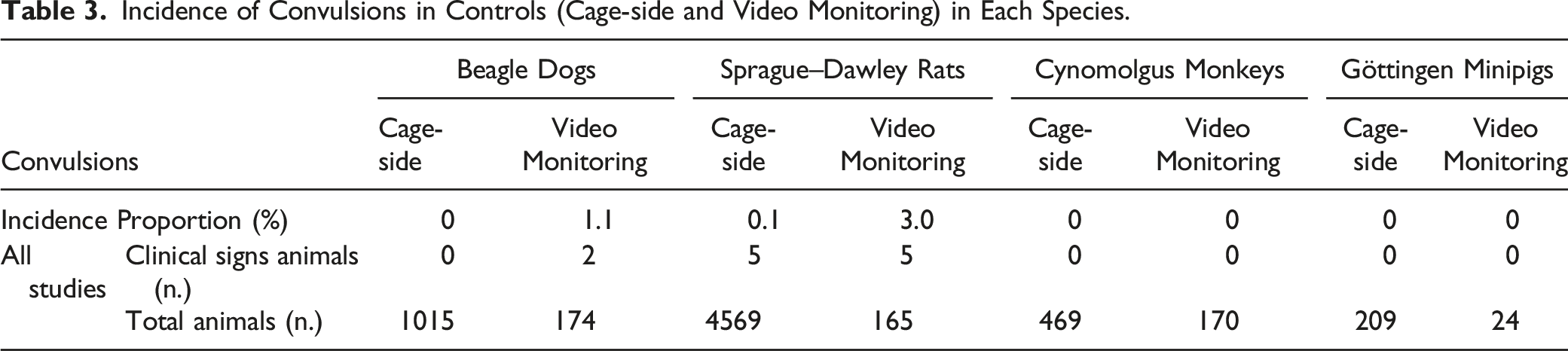
Incidence of Spontaneous Convulsions
Incidence of Convulsions in Controls (Cage-side and Video Monitoring) in Each Species.
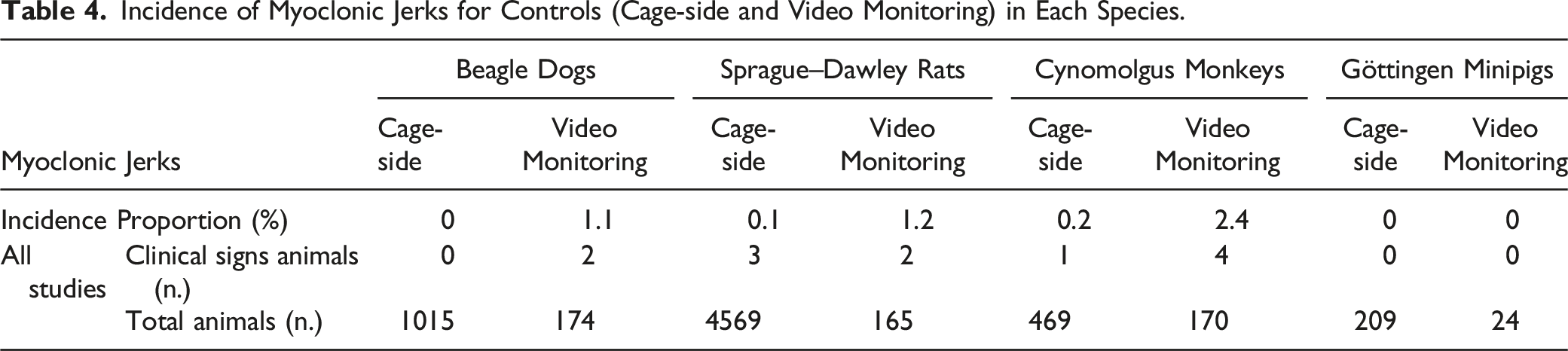
Incidence of Spontaneous Myoclonus
Incidence of Myoclonic Jerks for Controls (Cage-side and Video Monitoring) in Each Species.
Incidence of Spontaneous Tremors
Incidence of Tremors for Controls (Cage-side and Video Monitoring) in Each Species.
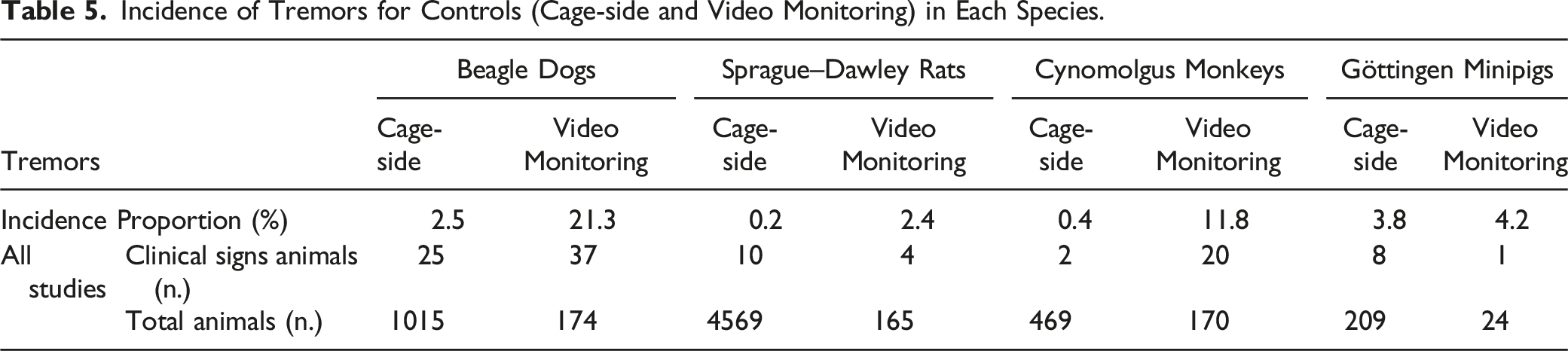
Incidence of Spontaneous Ataxia/Incoordination
Incidence of Incoordination Observations for Controls (Cage-side and Video Monitoring) for Each Species.
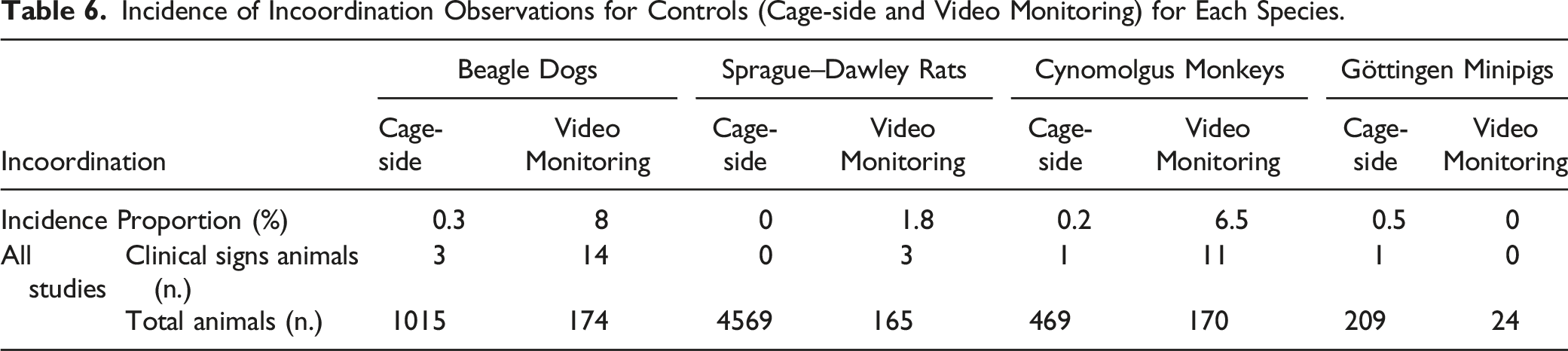
Incidence of Spontaneous Muscle Twitches
Incidence of Muscle Twitches Observations for Controls (Cage-side and Video Monitoring) in Each Species.
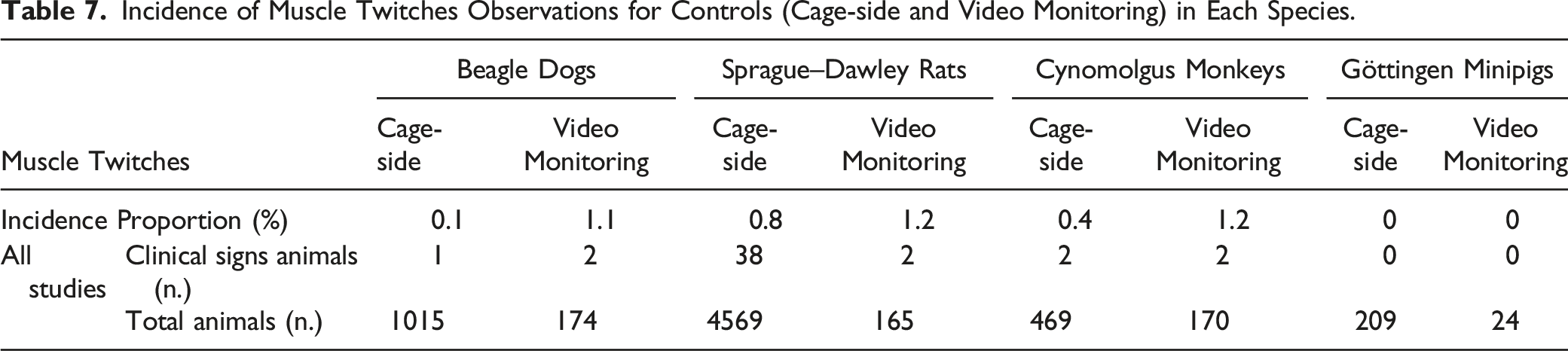
Incidence of Spontaneous Salivation
Incidence of Salivation for Controls (Cage-side and Video Monitoring) in Each Species.
Discussion
We aimed to evaluate the incidence of neurological clinical signs in control animals to create a larger species-specific reference dataset to facilitate interpretation, especially for low incidence neurological clinical signs. In toxicology and safety pharmacology, some spontaneous or drug-induced clinical signs can be exacerbated by a variety of factors, including stress, anxiety, or pain/discomfort 20 and experimental conditions can also contribute to the biological variability observed.
Video recording concomitant to EEG monitoring allows for observation of premonitory clinical signs of seizure, 21 and it is generally viewed as a pharmacologically relevant methodology to evaluate for the presence of seizure-related effects.22-24 As expected, an increased incidence of clinical signs following video monitoring was found for most clinical signs evaluated in this study when compared to cage-side observations and the difference is likely related to the higher density of observations with this modality. Such result highlights the interest in future applications that would associate video recording with artificial intelligence algorithm to conduct automatic detection of such clinical signs, in the absence of human interference (presence) eventually increasing data consistency and reducing the risk for human errors.
Susceptibility to Neurological Clinical Signs Across Species
Compared to most preclinical animal species, dogs have been extensively evaluated as it relates to their susceptibility to develop neurological clinical signs.25,26 It is suggested that the dog has a distinctly different cytoarchitecture of the cerebral cortex compared to other species. 27 Their higher susceptibility to seizure compared to rats, monkeys, and minipigs has been well documented and also demonstrated with ex-vivo models. 22
Although the comparison between Beagle dogs and human neurological clinical signs is facilitated by their similar epilepsy nomenclature), 25,28,29Beagle dogs present translational limitations when assessing human seizure risk, given their genetic predisposition to idiopathic epilepsy 22 which results in a higher risk for under-estimating test article safety. Irrespective of this increased susceptibility, dogs can be considered to characterize seizure premonitory clinical signs, including behavioral changes, present in up to 39% of human patients prior to seizure onset. 30
Although Sprague–Dawley rats are considered non-epileptic, 31 the increased use of long-term digital video-EEG recording systems applied to laboratory rodents demonstrated a widespread prevalence of spontaneous spike–wave discharges, recorded variably at 7-12 Hz. 32 Spike–wave discharges in Sprague–Dawley rats are relevant as this strain is commonly used in animal models of temporal lobe epilepsy 33 and for drug-induced seizure liability testing, and these spontaneous patterns can potentially confound seizure assessment in this species 33 in the absence of an appropriate baseline period.
Cynomolgus monkeys present a genetic polymorphism that is closer to the human population.34,35 They may represent a useful model to characterize the seizurogenic potential of pharmaceutical candidates when ethically justified and to increase the translational value of preclinical studies. 36
Lastly, as for non-human primates, considered a similar model to the human, minipigs have been argued to be a more suitable species for experimental models of various human brain disorders than rats since their brain anatomy and neuro-development are more closely related to that of humans. 37 However, seizure liability assays demonstrated the lowest susceptibility to seizures when compared to the other commonly used preclinical models. 22
Convulsions
Most dogs with epilepsy experience ictal EEG activity (ie, seizures), with localized or generalized motor activity (ie, convulsions).38-40 Many dog breeds, including Beagle, are reported to have a higher-than-average incidence of seizures41,42 and convulsions have been observed at low incidence in the absence of drug treatments. 43
Most canine epilepsies have been considered idiopathic, defined as primary generalized epilepsy of unknown cause but with a possible familial predisposition.44,45 Pedigree studies of several affected breeds indicate a high degree of heritability for canine idiopathic epilepsy, 46 although only few causative genes or genetic markers have been identified for Belgian Sheperd, Boerbel, and Lagotto Romagnolo.47-49
Canine idiopathic generalized epilepsies are similar to the same condition in humans, in which no known cause has been identified. Rare hereditary factors (such as a specific mutation of the sodium channel) 50 are reported to affect approximately 1% of the human population. 51 Searching for “common” genes is difficult as many susceptibility genes probably interact, leading to epilepsy. 50
One study reported the incidence of spontaneous convulsions in Sprague–Dawley rats assessed from 11 oral gavage carcinogenicity studies. 52 The incidence of spontaneous clonic convulsions was 2.4% during the 2-year study period (n = 2990 for each sex) and most of the convulsions were observed during the oral gavage procedure, during animal holding. 52
In the current retrospective study, spontaneous convulsions were not detected in either cynomolgus monkeys or in Göttingen minipigs which could be related to the low susceptibility reported in these species22,34,35 and the relatively low population of minipigs included.
In the context of drug toxicity in patients, between 6 and 9% of new-onset seizures are considered drug related 53 including drug withdrawal syndromes. 54 Drug-induced seizures can occur as a result of changes in specific excitatory or inhibitory transmitters inducing an alteration of neural pathways. 54 Antidepressants, diphenhydramine, stimulants (including cocaine and methamphetamine), tramadol, and isoniazid account for the majority of the drugs responsible for seizures in humans. 54
Myoclonic Jerks
Myoclonus can be classified in several ways, and their classification by etiology is subdivided into physiological myoclonus (eg, physiological myoclonic jerks during sleep onset), essential myoclonus (idiopathic or hereditary), epileptic myoclonus, or symptomatic myoclonus.15,19,26 While most myoclonus observed in toxicology and safety pharmacology are physiological, myoclonus can be associated with epileptiform EEG activity and generally represents a greater level of seizure liability concern 43 than tremors. In veterinary medicine, myoclonus and seizures may occur as part of a degenerative encephalopathy. Lafora disease in dogs, including Beagle, 55 is characterized by gene mutations, including a mutation to the laforin protein, which is postulated to prevent the accumulation of carbohydrates in neurons. 56 A mutation responsible for the disease has been identified in the Wire-Haired Dachshund and the Beagle. 19 Mutations have also been identified in the human disease where the laforin gene is dysfunctional.57-59 More frequent myoclonic movements and seizures occur in some subtypes of neuronal ceroid lipofuscinosis, a group of inherited progressive myoclonic epilepsies resulting from lysosomal storage disorders in dogs as well as in humans.19,26
Physiological myoclonus can be observed during sleep or physiological startle which may be induced by a stimulus such as a sudden noise. 15 Non-human primates are reported to present muscle atonia accompanied by intermittent saccades in the N2 and REM sleep stages, similar to humans, 60 and interestingly, in this retrospective study, cynomolgus monkeys presented a higher incidence of spontaneous myoclonic jerks in either cage-side observation or video monitoring. Some pharmacological agents have been shown to alter sleep stage duration and EEG power spectra in cynomolgus monkeys 61 potentially changing the incidence of physiological myoclonus. Although the neurological relationship between sleep and myoclonic jerks has not been completely characterized, an increase or a decrease in myoclonic jerks carries pharmacological importance, especially with neurological drugs.
A pharmacovigilance database study registering all compulsorily reported adverse drug reactions in France, 62 reported an incidence of drug-induced myoclonus of .2% over a 20-year period, 62 suggesting that drug-induced myoclonus is a movement disorder observed at an noticeable level. 16 The most important group of drugs linked to myoclonus are opiates, antidepressants, antipsychotics, and antibiotics; however, myoclonus may also be caused by a wide variety of other drugs. 16 Importantly, myoclonus is considered as a frequent precursor sign to seizures in all species. Similar to convulsions, the incidence of myoclonic jerks was relatively low in our study sample.
Tremors
Tremors can be divided into action and resting tremors. 14 Resting tremors happen when concerned muscles are at rest, that is, not even against gravity. They are not reported in companion animals. Action tremors can be divided into postural and kinetic tremors. 14 Kinetic tremors occur when a purposeful movement is performed.19,63 They have a broad etiology but are typically associated with cerebellar effects or changes (intention tremors). In humans, cerebellar tremors could be associated with multiple sclerosis, stroke, or brainstem tumors. 14
On the other hand, postural idiopathic tremors, which occur when the body part is voluntarily maintained against gravity, 63 fulfill many of the characteristics of essential tremor in humans, although it can also involve features of kinetic tremor.11,64 These tremors are believed to be an inherited condition in some canine breeds, including Beagle, 65 as a result of a physiological disturbance in the stretch reflex mechanism. 66 As a clinical parallel, in half of the essential tremor cases in humans, the disease is assumed to be transmitted in an autosomal dominant fashion, affecting .4 to 6% of the population. 14
In particular, canine idiopathic head tremor syndrome (IHTS) is a benign condition that manifests as episodic uncontrolled head tremors67-69 which may be of relevance for the animal in research facilities. Indeed, stress has been reported to be a precipitator of IHTS, with studies reporting an association in 7-50% of dogs.67-69 The incidence of IHTS has not been studied in Beagle dogs to date.
Interestingly, in this retrospective analysis, tremors were found at appreciable levels in all species following cage-side observations and video monitoring. While tremors are frequently identified as a premonitory sign to seizures, it should be emphasized that most tremors observed in preclinical drug safety testing studies are not associated with epileptiform EEG activity 43 and are simply considered as physiologically enhanced tremors most of the time related to stress or shivering.
Drug-induced tremors may manifest with all types of tremors and involve any body part. 70 Enhanced physiological tremors or postural/intentional tremors affecting the upper limbs and resembling essential tremors are also the most common clinical presentation in humans. 71 However, some drugs may induce resting tremors, which occur in body parts that are relaxed and completely supported against gravity, resembling the tremor of Parkinson’s disease. 71
Ataxia or Incoordination
Ataxia is a common clinical sign of nervous system dysfunction involving neurological structures responsible for the spatial perception of the body (proprioception) and balance, such as the vestibular system (vestibular nerve and nuclei, cerebellum), medulla, and/or spinal cord. 13 Causes of ataxia are numerous and are generally categorized into genetic17,72 and non-genetic; the latter category contains both degenerative and acquired causes,17,72 which include metabolic, neoplastic, infectious/inflammatory, traumatic, toxic, and vascular etiologies. 73 In drug safety testing, ataxia is commonly observed, often with an onset/offset kinetic that correlated with exposures.
Characteristic clinical signs can be identified in the presence of a vestibular dysfunction of either the central or peripheral nervous system. 74 Asymmetric ataxia is commonly reported75-77 in animals with vestibular dysfunction and approximately 92.5% of dogs affected have been reported to have gait abnormalities.13,75-77
For certain dog breeds, such as Beagle dogs, a deletion in a gene associated with cytoskeleton integrity (SPTBN2) has been described to cause neonatal onset of cerebellar cortical degeneration. 78
A study investigating cerebellar ataxia identified three main approved drug categories associated with this adverse reaction: antiepileptic drugs, benzodiazepines, and antineoplastics. 17 Some drugs may also cause ataxia when used for a different indication, such as antiepileptic drugs that can potentially be used to treat neuropathic pain or migraine. 17
In toxicology, the neurological mechanism(s) involved with ataxia/incoordination are typically not required to be identified. Non-specific or indirect mechanisms may contribute to the presence of drug-induced ataxia, especially at higher doses during dose range finding studies (eg, hypoglycemia, hypotension, and significant electrolyte imbalances). The incidence, severity, and duration of the ataxia are important to establish if the clinical signs should be considered adverse. The incidence of spontaneous ataxia reported in the current retrospective study highlights the necessity, particularly for Beagle dogs and cynomolgus monkeys, to integrate a control group of adequate size not to overestimate test article-related ataxia/incoordination. The drug label for a number of approved drugs contains a warning for potential ataxia and preclinical toxicology studies can be informative to identify and characterize this risk.
Muscle Twitches
Muscle twitches and contractions of various amplitude and frequencies above the spontaneous incidence for the species can be part of peripheral nerve hyperexcitability (PNH) syndromes. They correspond to spontaneous discharges generated by the motor neuron/axon.12,19 It results in a variety of clinical signs, including fasciculations (“brief spontaneous contraction resulting from the spontaneous activation of a small number of muscle fibers”), 19 myokymic discharges or myokymia (focal or generalized continuous contraction of facial or limb myofibers), 19 neuromyotonia (more severe form of myokymia with more pronounced rigidity and delayed relaxation), cramps (sudden contraction or shortening of a muscle, auto-resolutioning, more or less associated with pain), and tetany (focal or general muscle contraction without any relaxation).12,19
In many human disorders characterized by generalized PNH, including generalized myokymia, neuromyotonia, and fasciculation–cramp syndrome, 79 a potassium channel dysfunction of the nerve has been identified as the etiology. Indeed, peripheral nerve conduction relies on voltage-gated sodium currents responsible for depolarization and potassium channels are responsible for the termination of the action potential. 80 In humans with PNH, this potassium channel defect can be caused by genetic channelopathies,81,82 immune-mediated reactions targeted to the ion channel,83,84 or ion channel maldistribution due to axonal degeneration or demyelination. 85
In dogs, the exact cause of generalized myokymia and neuromyotonia remains unknown. However, it is possible that potassium channel dysfunction may also play a role in animals with generalized PNH. 86 Interestingly, antibodies against one of the proteins associated with the voltage-gated potassium channels have been identified in adult cats with partial seizures. 87
In the current retrospective study, we identified a low incidence of spontaneous muscle twitches with cage-side observations and video monitoring for all species, except minipigs where they were absent. When muscular toxicity is suspected or when an increased incidence of muscle twitches is observed, additional toxicology endpoints are typically considered. Serum creatine kinase elevation or myoglobinuria can be observed with relatively severe musculo-skeletal toxicity. Immuno-histochemistry can also be used to identify the presence of antibodies in muscle tissues of auto-immunity is suspected.
Salivation
Salivation can be present in humans with medications such as muscarinic agonists, antidopaminergic drugs, and benzodiazepines and with neurological diseases. 88 Salivation, before and after dosing in toxicology studies, is an expected reaction to the oral administration procedures. 43 Hypersalivation or sialorrhea is a common clinical sign observed in animals, particularly dogs 89 and it can represent a premonitory sign to seizures 43 in some cases. Dogs monitored with video presented a lower incidence of salivation than those evaluated at cage-side. This was likely due to a reaction also known as Pavlov conditioning, 90 mostly related to anticipation of feeding activities associated with the presence of the technical staff during cage-side observations. In this retrospective analysis, salivation was present in all species both following cage-side observations or video monitoring.
When investigating mechanisms for salivation, a nuance between hypersalivation and drooling can be considered. In the presence of normal saliva production, drooling represents an unintentional loss of saliva from the mouth, often due to motor dysfunction. Drooling was identified in a small proportion of individuals in human control groups. 88 In toxicology studies, hypersalivation and drooling are generally not differentiated at cage-side observation but most causes, where drooling could be present, would also be expected to present additional neurological clinical signs that would prompt a neurological examination.
Study Results: Perspectives, Implications, and Limitations
The current study suggests a higher sensitivity to detect neurological clinical signs in toxicology studies when using continuous video monitoring when compared to cage-side clinical signs across all species. This apparent increase in sensitivity with video monitoring compared to cage-side evaluation needs to be interpreted cautiously as the duration of observation per day with video was higher. Video monitoring also offers the possibility to involve subject matter experts (ie, neurologists) for a detailed interpretation of neurological clinical signs that can be difficult to classify for the technical staff present in the room. The development of neurological clinical sign automatic recognition using artificial intelligence may also be possible with video monitoring. Another potential advantage of video monitoring is the possibility to retrospectively quantify the precise kinetics and incidence of various drug-induced clinical signs over a period of several days or even weeks and to correlate with exposure data. In some cases, this can allow for the identification of early adverse clinical sign onset that can have an important impact on the clinical trial monitoring plan, as earlier clinical signs can serve as dose-stopping criteria. While advanced digital video monitoring can be relevant in specific scenarios when clinical signs with serious safety consequences can be observed (eg, seizures), it remains cost prohibitive and would not be justified for use in most toxicology programs for routine evaluation.
A cognitive bias where the technical staff expects control animals not to show clinical signs may be a limitation of the current study. Indeed, we cannot exclude that expectations that control group animals will be normal, possibly led to a lower sensitivity or minimizing clinical observations in some cases. 91 Age is also known to impact the incidence of spontaneous clinical signs in all non-clinical safety pharmacology and toxicology animal species. While this retrospective analysis used animals considered as young adults for all species, this factor was not precisely controlled between groups which was another limitation of the current analysis. Similarly, the study did not control for the number of days recorded per animal between monitoring methodologies, but as shown in Table 2, the number of days per animal for video monitoring studies was shorter on average for all species, yet the incidence of spontaneous neurological clinical signs was generally higher than with cage-side observations. Another limitation of our study, related to its retrospective nature, was that the precise timepoint of vehicle placebo administration (when present) was not included in the analysis. Therefore, it was not possible to compare the incidence of neurological clinical signs relative to placebo administration, where it would have been possible to evaluate for possible stress-related placebo effects. The incidence of clinical signs per species may also differ between animal suppliers. When serious spontaneous clinical signs are observed in control animals, the suppliers are to be notified to ensure genetic selection can be considered to remove breeders or lineages that were involved.
In summary, the data highlighted in this retrospective analysis show the importance of developing historical control databases under the experimental conditions that prevail on studies using standard non-clinical animal species to interpret neurological clinical signs, especially rare observations. Furthermore, this study shows the potential benefit of including an adequate pre-dose monitoring period so that the spontaneous incidence of clinical signs can be assessed and to later address individual variability.
Footnotes
Author Contributions
Chiara Orciani contributed to analysis and interpretation and drafted the manuscript; Cristina Ballesteros contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; Eric Troncy contributed to analysis and interpretation and critically revised the manuscript; Clemence Berthome contributed to acquisition and interpretation, drafted the manuscript, and critically revised the manuscript; Kim Bujold contributed to design, contributed to interpretation, and critically revised the manuscript; Nehla Bennamoune contributed to analysis and interpretation and critically revised the manuscript; Samantha Sparapani contributed to acquisition and analysis and critically revised the manuscript; Michael K. Pugsley contributed to analysis and interpretation and drafted the manuscript; Dominique Paquette contributed to analysis and interpretation and drafted the manuscript; Emmanuel Boulay contributed to analysis and interpretation and drafted the manuscript; Simon Authier contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The opinions presented here are those of the authors. No official support or endorsement by participating companies is intended or should be inferred.
