Abstract
The NCG triple immunodeficient mice on a NOD/Nju background lack functional/mature T, B, and NK cells, and have reduced macrophage and dendritic cell function. This study characterized the NCG mouse model for toxicity, engraftment and tumorigenicity assessments of cell therapies, using CD34+ hHSPC adult mobilized cells with two myeloablation regimens.
Mice received sub-lethal irradiation or busulfan and were then injected intravenously with CD34+ hHSPCs (1.0 x 106 cells/mouse) or PBS (control), while positive control animals received 2 x 106 HL-60 cells/mouse. hCD34+ cell donors were treated with the mobilizing agent G-CSF prior to leukapheresis. Following injections, mouse blood samples were collected to assess engraftment rates by flow cytometry with body weights recorded periodically up to 20 weeks post-cell injection. No significant clinical signs or body weight changes were observed. At week 10 post-cell injection, the peripheral blood chimerism of hCD45+ cells was above 20%. While mCD45+ concentration was constant between week 10 and 17 in whole blood samples, hCD45+ concentration and chimerism slightly decreased at week 17. However, chimerism remained above 10%, with busulfan-treated mice presenting higher values. Chimerism was further assessed by quantifying human Alu sequences in blood and multiple organs using qPCR. Alu sequences were most abundant in the spleen and bone marrow, while lowest in the testes. In the positive control group, expected mortalities due to tumorigenesis were observed between days 27 and 40 post-cell injection. Overall, study results may be used to inform study design and potential toxicological endpoints relevant to non-clinical cell therapy development.
Introduction
Cell therapy is a rapidly growing therapeutic modality ranging across multiple indications, with applications in treating cancer, infections, allogenic transplantation, and treating autoimmune disorders among other ailments. 1 For example, chimeric antigen receptor (CAR) cell therapy clinical trials increased 4-fold between 2014 and 2018. 2 Then between 2017 and 2019, 3 cell therapies had the highest number of new drugs added to the pharmaceutical industry pipeline, when compared to other common cancer drug classes. Furthermore, the recent FDA approval of CAR-T cell therapy for hematological malignancies in 2017 has only increased the pre-clinical need for assessing these and other cell therapies. In 2019, a press release from the FDA correctly estimated at least 200 investigational new drug (IND) applications in addition to the 800 cell-based or gene therapy INDs in 2019. 4 Furthermore, the FDA predicted that by 2025, they would approve 10 to 20 cell and gene therapy products per year according to the pharmaceutical industry pipeline and clinical success rates observed in this class. 5 In response to this rapid development, the FDA increased allocation of resources to support cell and gene therapy studies. This increase in IND applications for cell therapies also reflects the growing need for pre-clinical models for assessing safety and efficacy of cell therapies.
Across the vast therapeutic indications that cell therapies apply to, a major challenge is addressing the complexity of the human immune system and the consequential variability in patient responses to treatments. 6 Safety concerns pertaining to allogenic CAR-T therapies will differ compared to autologous CAR-T safety assessments. In the former, graft versus host disease (GvHD) is a significant concern, while in the latter, GvHD is less of a concern due to developmental thymic negative selection leading to tolerance. 7 Similarly, for regenerative medicine indications, an important safety assessment relates to tumorigenicity. It is common practice to conduct non-clinical studies that combine a range of objectives including efficacy and biodistribution but also general toxicity and tumorigenicity.6,8 A relatively limited number of animal models were characterized to assess cell therapies pre-clinically, such as the BALB/c SCID 9 or the NOD immunodeficient mouse strains that lack the IL-2r γ chain. Some of the mouse strains used in cell therapy studies include the BALB/c-Rag2 null IL2rγ null or the NSG (NOD/SCID/IL-2Rγ-null). Other mouse models that are used include the NSGW41 (NOD/SCID Il2rg−/− KitW41/W41) that harbor an additional mutant kit receptor decreasing the competition with human HPSCs, 10 and the NBSGW (NOD.Cg-KitW−41J Tyr + Prkdcscid Il2rgtm1Wji/Thom) which can support human cell engraftment without conditioning or myeloablation. 11 The current needs in this field have provided impetus for designing new, alternative animal models in order to improve safety assessment of cell therapies.
The objective of the study was to characterize the NCG mouse model as a pre-clinical model to assess toxicity, engraftment, biodistribution, and tumorigenicity for the development of cell therapies. NCG mice are triple immunodeficient with a NOD/Nju background and CRISPR/Cas9 edited genes Prkdc and Il2rg. Consequently, these mice lack functional T, B, and NK cells and have reduced macrophage and dendritic cell function. Importantly, previous work demonstrated that NCG mice were a stable host for human immune cells and tumors in various disease contexts including bladder, colon, and breast cancer.12-15
The present study consisted of two phases, the first phase optimized total body irradiation dose for myeloablation in NCG mice, while the second phase characterized the model for toxicity, engraftment, biodistribution, and tumorigenicity endpoints with different conditioning regimens. In Phase 1, the optimized irradiation dose was established to be 200 cGy. This dose was well tolerated with no mortalities, clinical observations or changes in body weight observed. In Phase 2, in order to condition the NCG mice for receiving CD34+ human hematopoietic stem and progenitor cells (hHSPCs), two types of myeloablation were employed, namely, total body irradiation at 200 cGy or intra-peritoneal busulfan administration. Notably, both of these myeloablation strategies have been investigated for hematopoietic cell transplantation in patients. 16 Mice were then administered a slow bolus intravenous injection of adult mobilized CD34+ hHSPCs as a cell product. As a conditioning, the donors providing the hHSPCs were treated with granulocyte colony-stimulating factor (G-CSF) in order to increase (mobilize) peripheral blood CD34+ hHSPCs. 17 As a positive control, selected animals received an injection of the tumor cell line HL-60 following irradiation.
Methods
Animals
NCG triple-immunodeficient mice (Mus musculus, NOD-Prkdc em26Cd52 Il2rg em26Cd22 /NjuCrl) were co-developed by Nanjing Galaxy Biopharma in 2014 and transferred to Charles River in 2016. This model was created by simultaneous CRISPR/Cas9 editing of the Prkdc and Il2rg loci in the NOD/Nju mouse, generating a mouse coisogenic to the NOD/Nju. A 52-base-pair deletion in exon 37 (starting in codon 1619) was created in the Prkdc gene and a 22-base-pair deletion in exon 3 (codon 156) was created in the Il2rg gene. Both male and female NCG mice are homozygous for the Prkdc deletion, while female mice are homozygous and males are hemizygous for Il2rg deletion, since Il2rg is located in the X chromosome.
Conditioning Regimens
Total body irradiation: Prior to sub-lethal irradiation conditioning, the radiation dose was calibrated using an acrylic phantom placed in the same experimental set up to be used for animal irradiation. The measurements were taken using an ion chamber with a solid water phantom build-up. Animals in Phase 1 and Phase 2 were exposed to total body irradiation (Day 1) as follows: Dosimetry measurements were taken with scanned inLight nanodot from Landauer and using a Farmer electrode chamber. The theoretical dose rate, duration of irradiation and the actual time of irradiation for each subset of animals were documented. Body measurements parallel to the radiation beam were taken from 10 selected animals in each phase to calculate the midplane dose rate and radiation exposure times. Chest depth was measured from the sternum to equivalent point in the intrascapular area in millimeter with 2 digits with a caliper. Animals in Phase 1 received a total body irradiation dose of either 140, 160, 180 or 200 cGy (±5% of target dose). The animals in Phase 2 received a total irradiation dose of 200 cGy (±5% of target dose), based on the data obtained in Phase 1. The dose rate of the 60Co gamma source (Theratron 1000) was approximately 160 cGy per minute. The optimal irradiation dose for myeloablation was considered to be the highest sublethal dose.
Myeloablation: Busulfan (6 mg/mL) was prepared in 0.9% Sodium Chloride for Injection USP solution and administered via intra-peritoneal injection at a concentration of 15 mg/kg for males and 20 mg/kg for females on Day −2 and Day −1 relative to CD34+ hHSPCs administration. The day that hHSPCs were administered was considered as Day 1 for the purposes of this study.
CD34+ human hHSPCs (Adult Mobilized)
Adult mobilized CD34+ hHSPCs from three different donors at the concentration of 5.0 x 106/mL were obtained cryopreserved in liquid nitrogen vapor phase from HemaCare BioResearch Products (catalog Nos. M34C-GCSF-2 (donor 1 and 2) and M34C-2 (donor 3)). Cell formulations were prepared on the day of dosing, prior to dose administration. The vials of cells were thawed in a water bath at 37°C and resuspended in CellGenix® GMP SCGM Medium/0.1% HSA (thawing media) up to 10 mL. Cells were centrifuged at 300 x g for 5 minutes at room temperature. The cell pellet was resuspended with the PBS 1X pH 7.4/0.1% HSA (injection buffer), counted, and further diluted to achieve ∼4x10 6 viable cells/mL. Animals were irradiated at 200 cGy or conditioned with busulfan and then received ∼1x10 6 cells (in a total volume of 0.25 mL) of adult mobilized CD34+ hHSPCs. Control groups (non-irradiated, irradiated or busulfan-treated) were administered 0.1% Human Serum Albumin in PBS 1X (Phosphate buffered saline 1X, pH 7.4 without calcium and magnesium and 25% HSA).
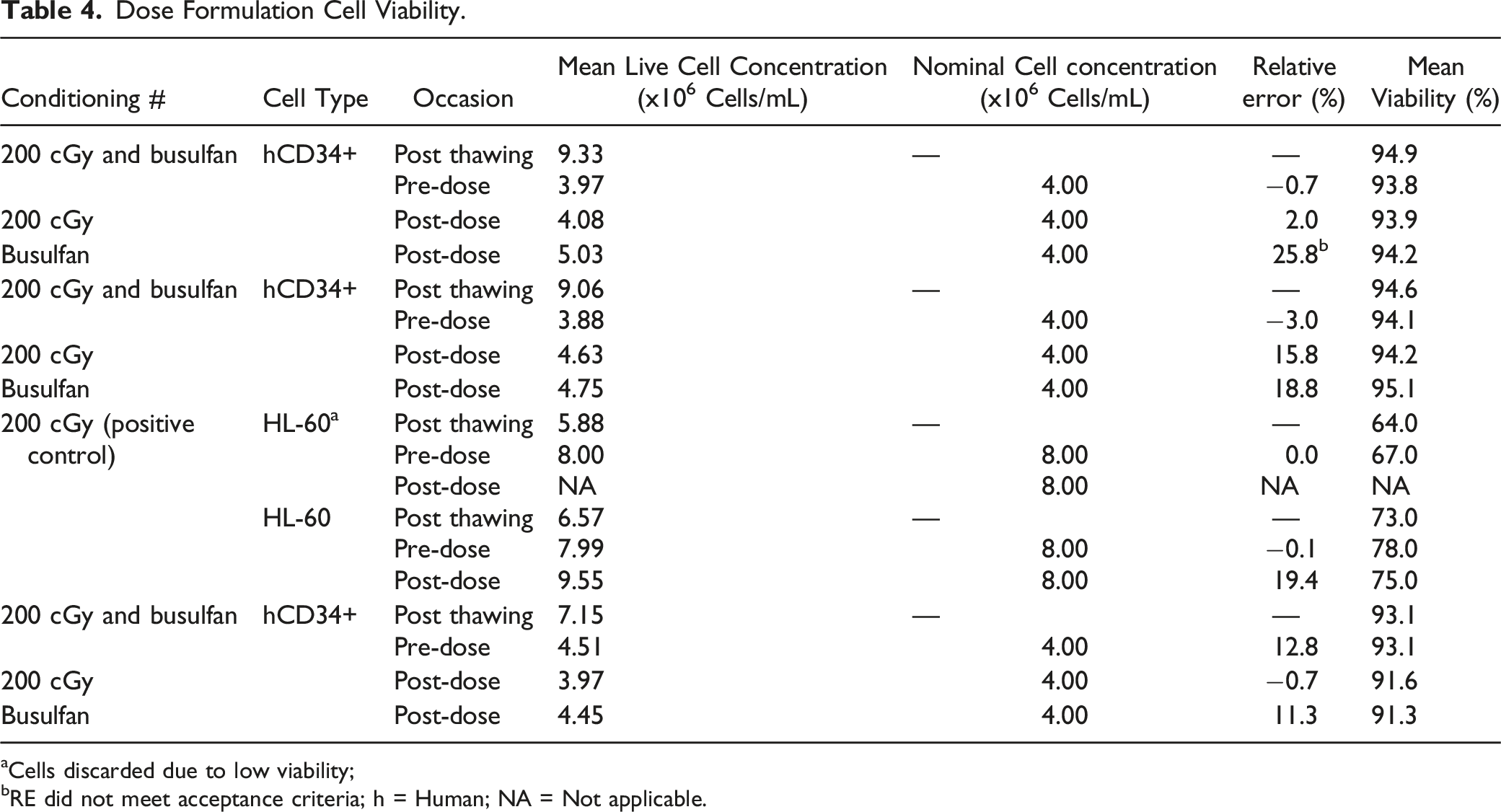
Cell counts and viability were performed prior and after dosing. After dosing completion, an aliquot from the remaining volume of the preparation that was drawn up in the syringe was used for cell count and viability. The viability CD34+ cells was performed by mixing the cell suspension with acridine orange/propidium iodide solution (AO/PI). The AO/PI allowed the differential staining of live and dead cells. CD34+ cell counts were determined with an automated cell counter. Cell count and viability of HL-60 cells was performed by mixing the cell suspension with trypan blue and manual counting with an hemacytometer. Acceptance criteria for viability was considered to be ≥ 70%. The cell formulations were administered once by intravenous slow bolus injection (i.e. 0.25 mL administered over 30-60 seconds) in the tail vein using a 27G needle attached to a syringe on Day 1. Based on unpublished data obtained at the facility, all injections of CD34+ hHSPCs were administered within 8 hours of irradiation and 24 ± 2 hours of busulfan, when applicable. The number of cells administered intravenously to each animal was ∼1x10 6 cells ±20% for test groups irradiated or busulfan-treated. Negative control animals (non-irradiated, irradiated or busulfan-treated) received a single intravenous slow bolus tail vein injection of injection buffer (0.25 mL) with the same conditions as the treated groups. Cell formulations were dosed within 3 hours post-concentration adjustment in the syringe.
Experimental Design
Phase 1 – Radiation dose optimization.

Phase 2 –Characterization of the NCG for cell therapy assessments.
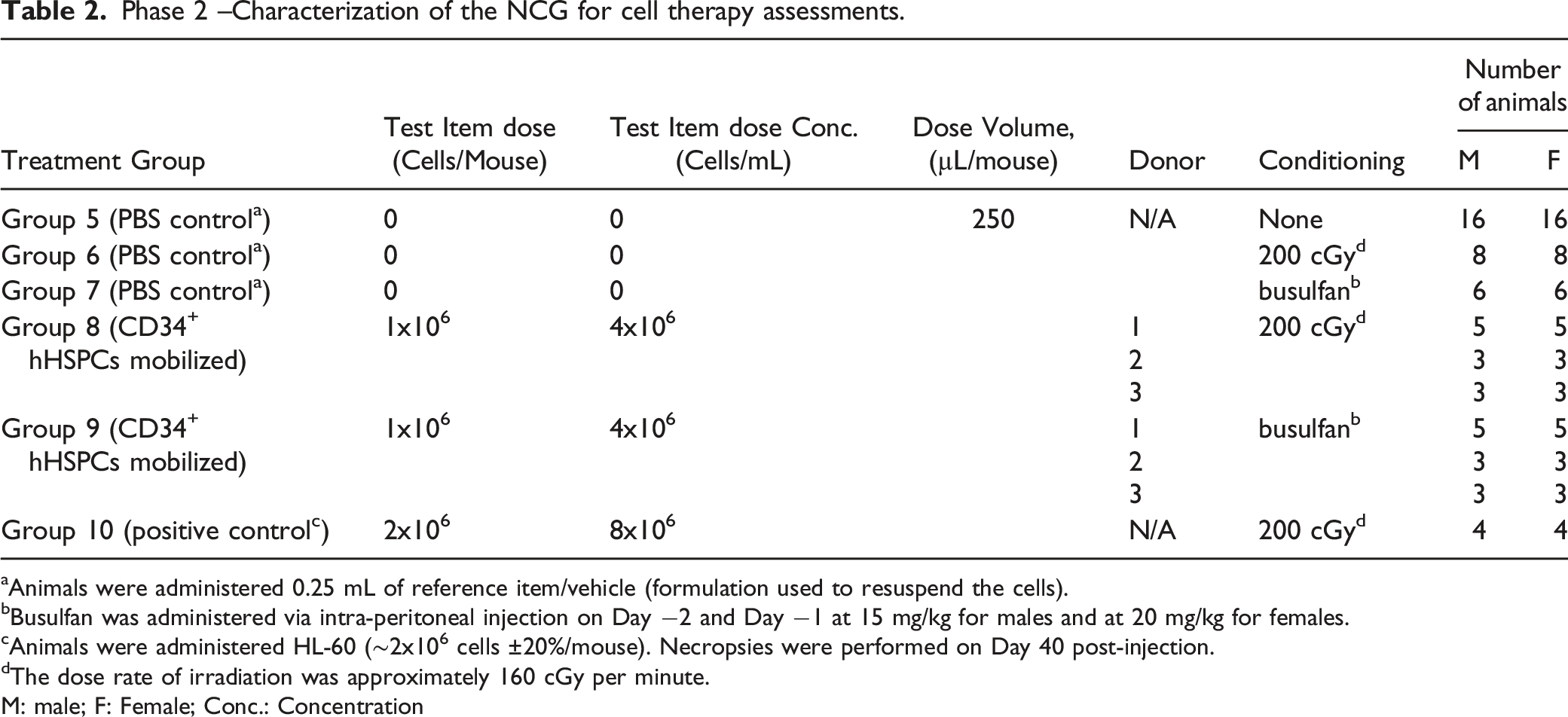
Animals were administered 0.25 mL of reference item/vehicle (formulation used to resuspend the cells).
Busulfan was administered via intra-peritoneal injection on Day −2 and Day −1 at 15 mg/kg for males and at 20 mg/kg for females.
Animals were administered HL-60 (∼2x106 cells ±20%/mouse). Necropsies were performed on Day 40 post-injection.
The dose rate of irradiation was approximately 160 cGy per minute.
M: male; F: Female; Conc.: Concentration
Supportive Care
Acidified water was given throughout the study to prevent bacterial growth. Sterile water gel packs were provided at least two days before irradiation or busulfan administration, and for the duration of the study.
In-Life Assessments
Mean Weekly Body Weights in Animals From Phase 2.
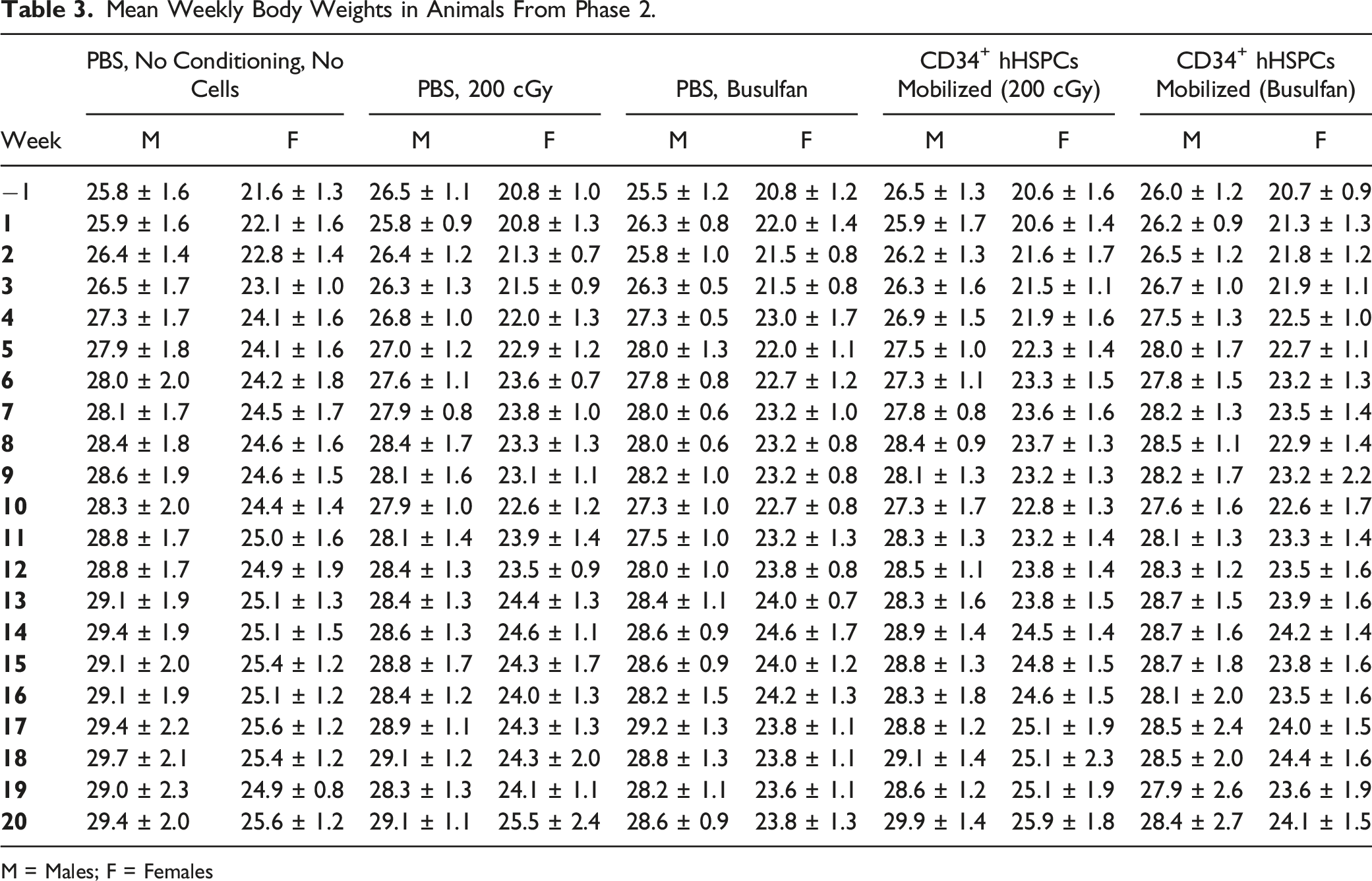
M = Males; F = Females
Hematology
Whole blood (125 µL) was collected in K2EDTA 0.5 mL tubes on Weeks 12 and 19 post-injection and analyzed using the Sysmex hematology analyzer in Low Aspiration Mode on the same day of collection. The following parameters were assessed: hematocrit, hemoglobin, mean corpuscular hemoglobin, mean corpuscular volume (MCV), mean corpuscular hemoglobin concentration (MCH), red blood cell count (RBC), red blood cell distribution width (RDW), total white blood cell count (WBC), WBC differential (neutrophils, lymphocytes, monocytes, eosinophils, basophils and large unstained cells; absolute and relative), platelet count and reticulocyte (absolute and relative).
Flow Cytometry
Non-terminal whole blood samples were collected in K3-EDTA tubes, on Weeks 10 and 17 post-injection from negative control groups (non-irradiated, irradiated or busulfan-treated) and from groups that received CD34+ hHSPCs following irradiation or busulfan. The blood samples were kept at room temperature with orbital shaking pending flow cytometry analysis. On Week 20 post-injection, terminal spleen and bone marrow samples were collected from the same groups. Following euthanasia, the spleen was removed, and one-third of the spleen was placed on wet ice or at 2 to 8°C then used for flow cytometry analysis. Femurs were excised, trimmed (muscle removed) and placed on wet ice or at 2 to 8°C pending flow cytometry. The identification of the mouse cell (mCD45+hCD45-) and human cell (mCD45-hCD45+) populations by flow cytometry was performed by direct staining of whole blood samples, as well as spleen and bone marrow cell suspensions with APC mouse anti-human CD45 and PE rat anti-mouse CD45. Fixable Viability Dye eFluor 506® (FVD506, Invitrogen) was also included in the staining panel to exclude dead cells from the analysis.
Splenocytes were isolated from spleen using an initial 100 μm cell strainer at 2 – 8°C conditions, followed by dissociation using a gentleMACSTM Dissociator. The cell suspension was placed in a GentleMACS C tube with ice cold MACS buffer followed by a 70 μm cell strainer. For bone marrow cell preparation, femurs, with the epiphysis cut, were placed in a perforated 0.5 mL tube in a second 1.5 mL tube with PBS 1X and centrifuged at 10,000 x g until the bone marrow was flushed to the collecting tube. Any clumps were dissociated with a pipette and the cell suspension was then passed through a 70 μm cell strainer, rinsed with ice-cold PBS 1X and filtered again. Cell concentrations in whole blood and absolute cell counts in spleen and bone marrow samples were determined using AccuCheck counting beads (Life Technologies/Invitrogen) with a known stock concentration. For whole blood samples, erythrocytes were lysed (PharmLyse Lysing Buffer, BD Biosciences), then cells were blocked and washed. Cells were then stained with the appropriate antibody mix and viability stain for 30 ± 5 minutes on wet ice protected from light. For spleen and bone marrow samples, erythrocytes from the cell suspension were lysed and cells were counted prior to flow cytometry staining. Spleen and bone marrow cell suspensions were lysed with PharmLyse 1X, blocked with TruStain FcXTM (anti-mouse CD16/32) and Human TruStain FcXTM (Fc receptor blocking solution) solutions for 10 minutes on wet ice and then incubated with the appropriate antibody mix and viability stain for 30 minutes on wet ice protected from light. Antibodies used were APC mouse anti-human CD45 and PE rat anti-mouse CD45 (BD Biosciences) diluted in PBS 1X with 2% iFBS. Single events were gated based on their height and area in the forward scatter (FSC) parameter. Then, leukocytes were gated based on granularity (side scatter, SSC) and size (FSC) while dead cells were excluded based on FVD506 positivity. Samples were acquired on FACS Lyric flow cytometer (BD Biosciences).
qPCR for Human Alu Sequences
Quantitative real-time PCR (qPCR) was used to determine the expression of human Alu sequences in whole blood (Weeks 12 and 19 post-injection) and tissues (Week 20 post-injection) from NCG mice. Following genomic DNA extraction (gDNA), qPCR reaction was performed using 500 ng of gDNA per well and ALU expression reported as pg/500 ng of mouse gDNA. The following primers and probes were used: forward primer nsP1: 5′- TGG TGG CTC TCT CCT GTA AT -3′, reverse primer nsP1: 5′- GAT CTC GGC TCA CTG CAA GC -3′ and TaqMan Probe nsP1: 5’-/56-FAM/TGA GGC AGG AGA/ZEN/ATC GCT TGA ACC/3IABKFQ/-3' (Integrated DNA technologies) in a QuanStudio 5 Real-Time PCR system with Quantstudio 5 Design and analysis software. The total fluorescence emitted by the released fluorophore was measured at each cycle. A passive reference dye (ROX dye) was included in the qPCR reaction and provided a stable baseline reference to which samples were normalized to compensate for variations in fluorescence between wells or between experiments. Human peripheral blood mononuclear cells (hPBMCs) were used as a positive control and the number of copies per well in samples was extrapolated for ALU qPCR assays from a standard curve of hPBMC in water containing NCG mouse DNA. The following list of tissues were analyzed: liver, brain, kidney, heart, ovaries, testes, spleen, lung, and bone marrow.
Histopathology
The masses, spleen, femur and bone marrow were fixed and preserved in neutral buffered 10% formalin for at least 16 hours and up to 32 hours then processed into paraffin blocks. Hematoxylin and eosin staining, and microscopic analysis were performed on these tissues.
Statistics
Pairwise comparisons of interest were performed comparing each group with PBS control animals that were irradiated with 200 cGy. For each data set with more than two groups, a one-way analysis of variance (ANOVA) was performed and the residuals were tested for normality using a Shapiro-Wilk test. When the distribution of the residuals was significantly different from a normal one (P ≤ 0.05), the results from the ANOVA were rejected and a second ANOVA was performed using square root transformed data. For data sets with negative values, the absolute lowest observed value was added to the data before the transformation. The normality distribution of the residuals from the second ANOVA was tested using a Shapiro-Wilk test.
When the Shapiro-Wilk test was not significant (P > 0.05) a Levene test was performed on the residuals to assess the homogeneity of the group variances. If differences between group variances were not found to be significant (P > 0.05) then the results from the related ANOVA were retained. When significant differences among the group means are indicated by the ANOVA overall F-test (P ≤ 0.05), a Dunnett test was used to perform the group mean comparisons of interest.
When the Shapiro-Wilk test on the residuals of the second ANOVA was significant or when heterogeneous group variances (P ≤ 0.05) were revealed by the Levene test, ANOVA results were rejected and the groups were compared using a non-parametric Kruskal-Wallis test. When the Kruskal-Wallis test was significant (P ≤ 0.05), a Dunn test was used to perform the pairwise group comparisons of interest.
Each group comparison of interest was conducted via a two-sided test at the 5% significance level and the significant results were reported as either P ≤ 0.001, P ≤ 0.01 or P ≤ 0.05, where p represents the observed probability.
Separately for the 2 phases and on combined sexes, the survival function of each group was estimated using the Kaplan-Meier product-limit method applied on daily intervals.
Results
Phase 1
NCG mice well tolerated all irradiation dose levels, and all animals survived until scheduled euthanasia. For hematology parameters, minimal and non-dose related decreases in mean corpuscular volume were noted in males and females irradiated at ≥ 160 cGy at termination on Day 30. Decreases in mean corpuscular volume likely resulted in minimal and non-dose related increases in red blood cell distribution width in males irradiated at ≥ 180 cGy and females irradiated at 200 cGy when compared to animals irradiated at the lowest irradiation dose at 140 cGy. These changes (decreases in mean corpuscular volume and increases in red blood cell distribution width) were not associated with any changes in red blood cell mass parameters (red blood cell count, hemoglobin and hematocrit), and thus were considered not clinically significant (data not shown).
The pathology evaluation did not reveal any irradiation-related findings or organ weight changes. Although, the thymus was not identified in 12/16 male and in 10/16 female animals, which was considered a consequence of the NCG mice phenotype and unrelated to the irradiation dose. Two males that received 200 cGy had minimally decreased cellularity of the femoral bone marrow hematopoietic compartment, which was considered incomplete regeneration following irradiation induced depletion. There were no residual irradiation effects on the femoral bone marrow at 140, 160 or 180 cGy, indicating that these animals had completely recovered from the irradiation conditioning.
There were no significant body weight changes or clinical observations in Phase 1 and Phase 2 related to irradiation, busulfan conditioning or administration of CD34+ hHSPCs.
Phase 2
In light of Phase 1 results, the irradiation dose in Phase 2 for myeloablation conditioning was set as 200 cGy. Following conditioning with irradiation or busulfan-treatment, Figure 1 NCG mice were administered a slow intravenous bolus injection of CD34+ hHSPCs, with a pre-injection and post-injection viability above 70%. Details on dose formulation cell viability are presented on Table 4. Irradiation or administration of busulfan only or injection of CD34+ hHSPCs to mice myeloablated with either conditioning regimens did not result in significant body weight changes or clinical observations. Animals in the positive control group administrated with HL-60 cells exhibited expected tumor-related clinical signs, such as palpable masses (Table 5 below), decreased activity, hunched back posture, thinness, weakness, closed eyes, erected fur and increased respiratory rate. Clinical observations in this group increased in incidence and severity over the duration of the observation period and had to be humanely euthanized. Body weights of Phase 1 and Phase 2 male and female animals. Average body weights from Week −2 to Week 5 for Phase 1 animals that underwent irradiation and Week −2 to Week 20 for Phase 2 animals that were either irradiated with 200 cGy or treated with busulfan and then injected with CD34+ hHSPCs. Data points represent means and error bars represent standard deviation. Dose Formulation Cell Viability. Cells discarded due to low viability; RE did not meet acceptance criteria; h = Human; NA = Not applicable. Palpable Masses Observed in Animals Administered HL-60.

Hematology
Conditioning and mobilized CD34+ hHSPCs-related hematology changes.
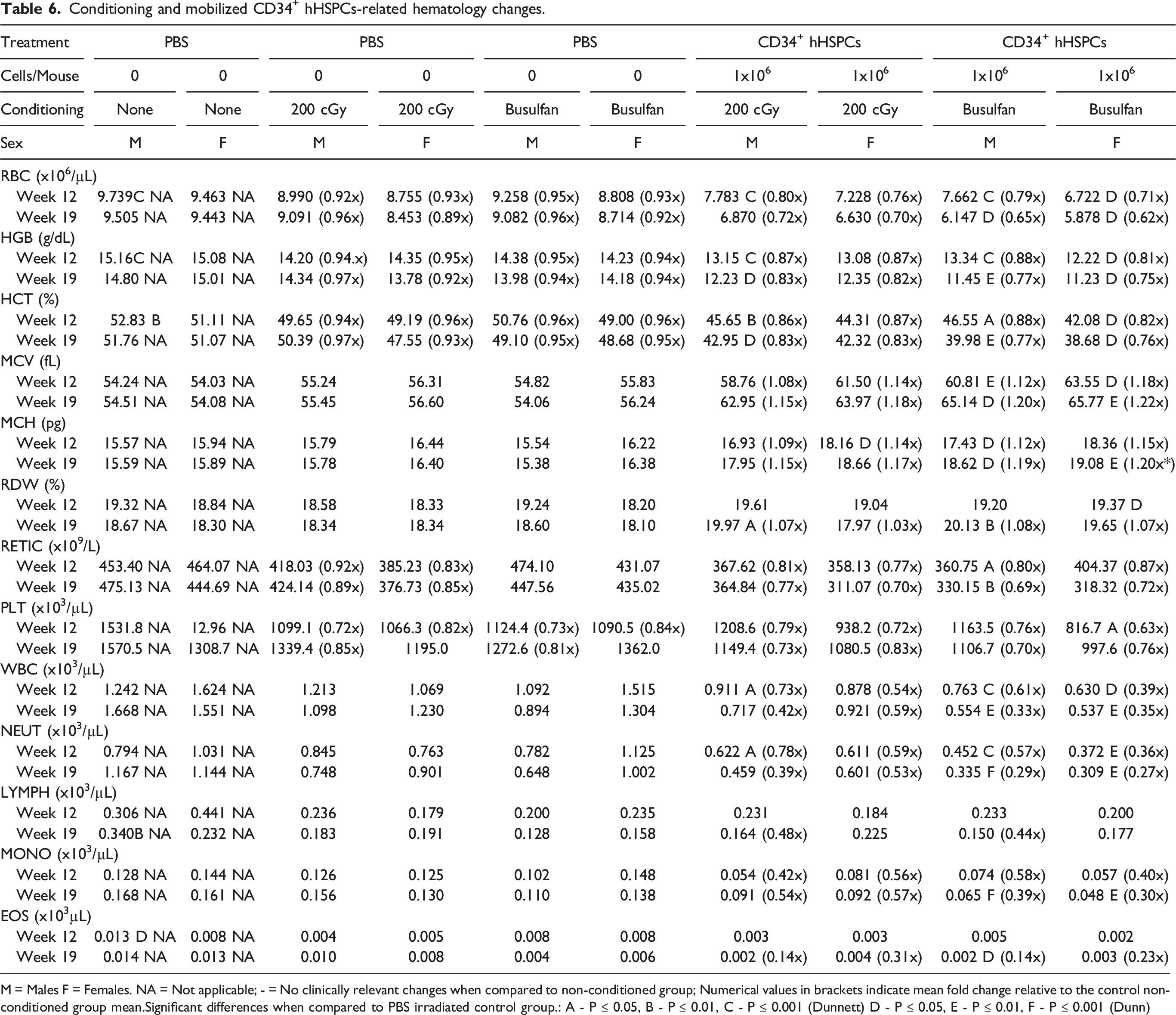
M = Males F = Females. NA = Not applicable; - = No clinically relevant changes when compared to non-conditioned group; Numerical values in brackets indicate mean fold change relative to the control non-conditioned group mean.Significant differences when compared to PBS irradiated control group.: A - P ≤ 0.05, B - P ≤ 0.01, C - P ≤ 0.001 (Dunnett) D - P ≤ 0.05, E - P ≤ 0.01, F - P ≤ 0.001 (Dunn)
Conditioning
Administration of busulfan or irradiation at 200 cGy resulted in comparable minimal decreases in red blood cell mass parameters (RBC, HGB, and HCT) and platelets in mice of both sexes at Weeks 12 and 19 post-injection relative to the PBS irradiated control group (Figure 2A, D, and E). Minimal decreases in reticulocyte counts were observed in irradiated mice only at Weeks 12 and 19 post-injection (Figure 2B). Hematology parameters in NCG mice conditioned either with irradiation or busulfan-treatment and injected with adult mobilized CD34+ hHSPCs. Changes in hematology parameters at Weeks 12 and 19 post-injection of adult mobilized CD34+ hHSPCs, as reported in Table 6. P values were calculated with Dunnett test (* = P ≤ 0.05, ** = P ≤ 0.01, and *** = P ≤ 0.001) and Dunn test (# = P ≤ 0.05, ## = P ≤ 0.01, and ### = P ≤ 0.001. Values represent the means of each group and error bars represent standard deviation.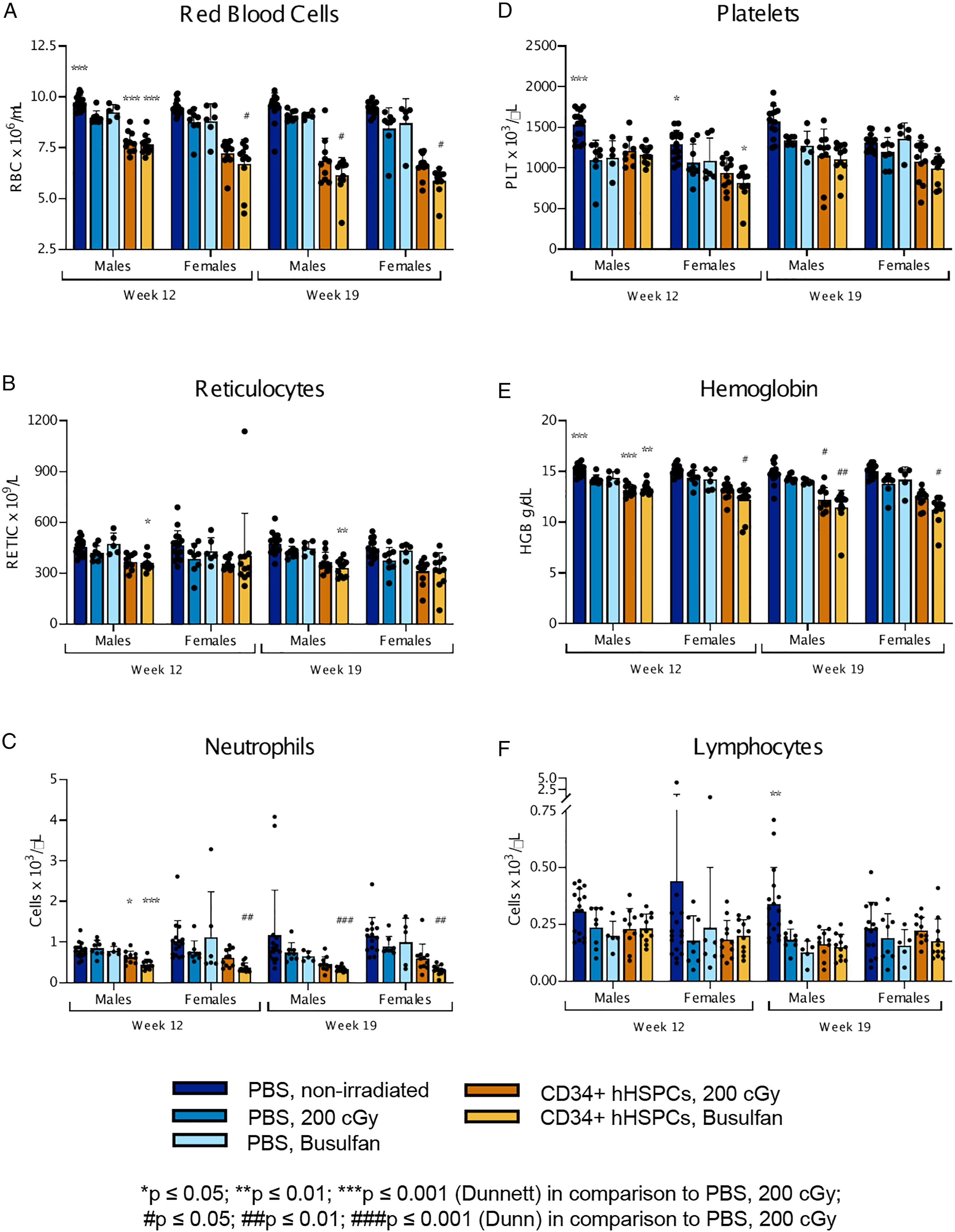
Mobilized CD34+ hHSPCs
Administration of CD34+ hHSPCs in mice irradiated at 200 cGy or busulfan-treated was associated with minimal to moderate and progressive decreases in red blood cell mass parameters (RBC, HGB, and HCT) and reticulocyte counts from Week 12 to 19 (Figure 2A, B, and E). Decreases in red cell mass and reticulocytes were comparable in magnitude between irradiated or busulfan-treated, while mice injected with CD34+ hHSPCs had more marked decreases compared to mice irradiated or busulfan-treated only. These exacerbated decreases in red cell mass and reticulocyte in mice that received mobilized CD34+ hHSPCs were noted with concurrent minimal and progressive increases in MCV and MCH in all groups from Week 12 to 19, which resulted in minimally increased RDW in all groups at Week 19 only. In addition, there were minimal to mild and generally progressive decreases in platelets in mice irradiated or busulfan-treated and injected with CD34+ hHSPCs at Weeks 12 and 19 (Figure 2D). This change, of comparable magnitude between groups that received CD34+ hHSPCs irrespective of their conditioning, exhibited a generally greater magnitude than in mice irradiated or busulfan-treated only. Other CD34+ hHSPCs-related changes in hematology parameters included mild to moderate and generally progressive decreases in WBC, neutrophils (Figure 2C), and monocytes in all groups at Weeks 12 and 19, mild decreases in eosinophils in all groups at Week 19 and mild decreases in lymphocytes in male mice irradiated or busulfan-treated at Week 19 (Figure 2F). These changes were attributed to the administration of CD34+ hHSPCs as they were not observed in mice irradiated or busulfan-treated only. Positive control animals that received HL-60 cells that survived until scheduled termination only included two males, where in one male, hematology changes consisted of moderately decreased red blood cell mass parameters (RBC, HGB, and HCT) and leucocytes (absolute and differential) on Day 40. These changes were comparable to those observed in other mice from this group that were terminated early.
Flow Cytometry
Whole Blood
Chimerism and cell concentration or absolute cell counts of hCD45+ and mCD45+ cell populations in peripheral whole blood, spleen and bone marrow of NCG mice engrafted with adult mobilized CD34+ hHSPCs
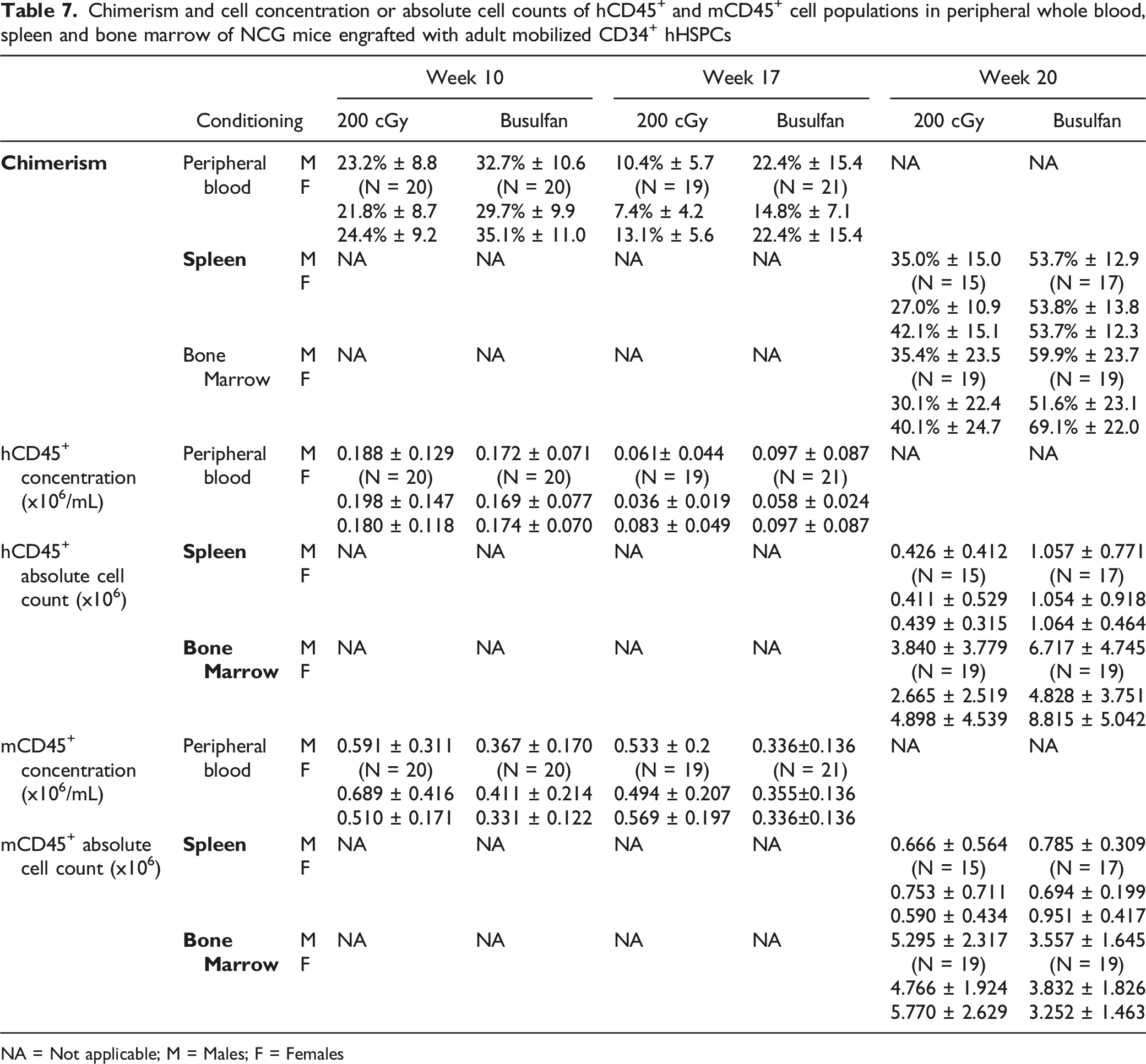
NA = Not applicable; M = Males; F = Females
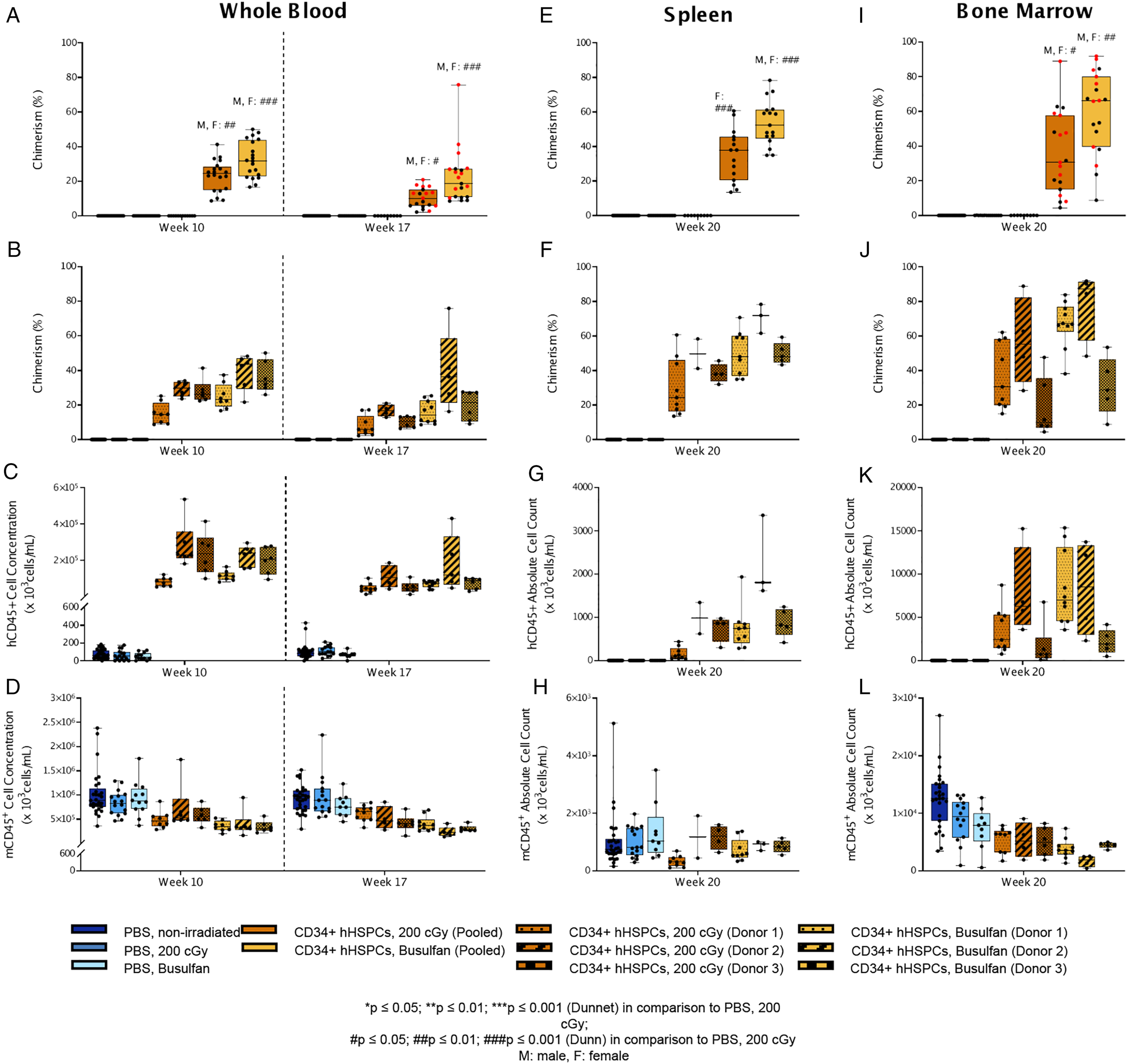
Percent chimerism of hCD45+ cells and absolute counts of hCD45+ and mCD45+ cells across different CD34+ hHSPC donors in various tissues. Orange datasets represent irradiation while yellow datasets represent busulfan treatment. Male (black) and female (red) data points were only indicated separately when there were significant differences between sexes (
Spleen
Human CD45+ cells were observed in spleen samples at Week 20 in animals that received CD34+ hHSPCs following irradiation or treatment with busulfan. The chimerism (Figure 3E, F), absolute cell count (Figure 3G) and the frequency of this cell population seemed higher in busulfan-treated animals compared to irradiated animals. The chimerism ranged from 13.6% to 60.6% in irradiated animals injected with CD34+ hHSPCs, and from 34.9% to 78.3% in busulfan-treated animals injected with CD34+ hHSPCs, with an absolute human CD45+ cell count ranging from 36.13x10 3 to 1,343x10 3 cells in irradiated animals, and from 284.2x10 3 to 3,360x10 3 cells in busulfan-treated animals (mean values presented in Table 7), suggesting a higher engraftment in the busulfan group. However, this difference seemed to be CD34+ hHSPC donor dependent. Only two out of the three CD34+ hHSPC donors demonstrated a higher hCD45+ absolute cell count with busulfan-treatment compared to irradiation, while one CD34+ hHSPC donor (Donor 3) demonstrated similar counts in both groups (Figure 3G). The absolute mCD45+ cell count was comparable between irradiated and busulfan-treated groups, unlike what was observed in whole blood samples (Figure 3H). No differences were observed between males and females in spleen samples.
Bone Marrow
Human CD45+ cells were observed in bone marrow samples at Week 20 in both groups of animals that received CD34+ hHSPCs following irradiation or treatment with busulfan. Similar to spleen samples, the chimerism (Figure 3I, J), absolute cell count (Figure 3K) and the frequency in bone marrow for this cell population tended to be slightly higher in the busulfan-treated group compared to the irradiated group, although these were more similar in range.
The chimerism ranged from 4.5% to 88.8% in irradiated animals, and from 8.8% to 91.8% in busulfan-treated animals, with an absolute human CD45+ cell count ranging from 91.66x10 3 to 15,244x10 3 cells in the irradiated group, and from 482.7x10 3 to 15,349x10 3 cells in the busulfan-treated group. Even when the range of chimerism was similar between the two groups, the mean was higher for busulfan-treated animals at 59.9% compared to the irradiated group at 35.4% (Table 7). The extent of this difference between irradiated and busulfan-treated groups seemed to be driven by CD34+ hHSPC Donor 1 and 2 since with Donor 3 (Figure 3J and K), the levels of hCD45+ cells and chimerism for busulfan and irradiation conditioning were similar.
Differences between males and females were also observed, particularly in the busulfan-treated group, where the chimerism for females was higher than males (irradiated animals receiving CD34+ hHSPCs males: 30.1%, females: 40.1%; busulfan-treated animals receiving CD34+ hHSPCs males: 51.6%, females: 69.1%) (Figure 3I).
Similar to whole blood samples, the absolute cell count and frequency of mCD45+ seemed lower in busulfan-treated animals compared to the irradiated group (Table 7, Figure 3L), except for one CD34+ hHSPC donor (Donor 3). However, slightly higher absolute hCD45+ cell counts were present in the busulfan-treated group when compared to the irradiated group. Therefore, it was concluded that the slightly higher chimerism in busulfan-treated animals was not an artefact as in whole blood samples.
Biodistribution (qPCR)
Concentrations of human Alu sequences detected by qPCR in the blood, spleen, bone marrow, ovaries, and testes.
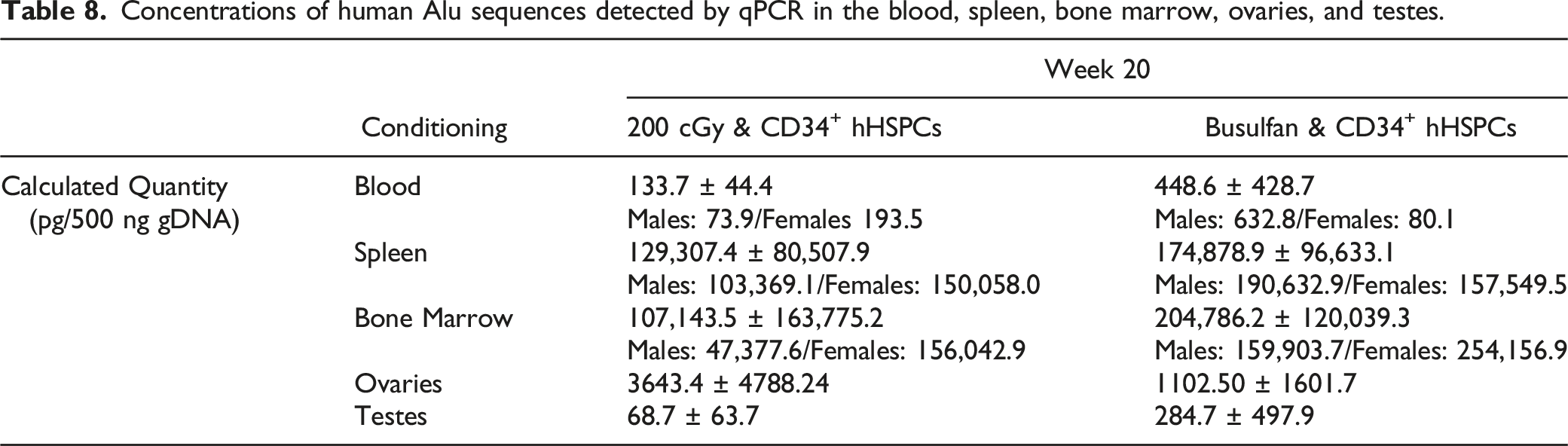
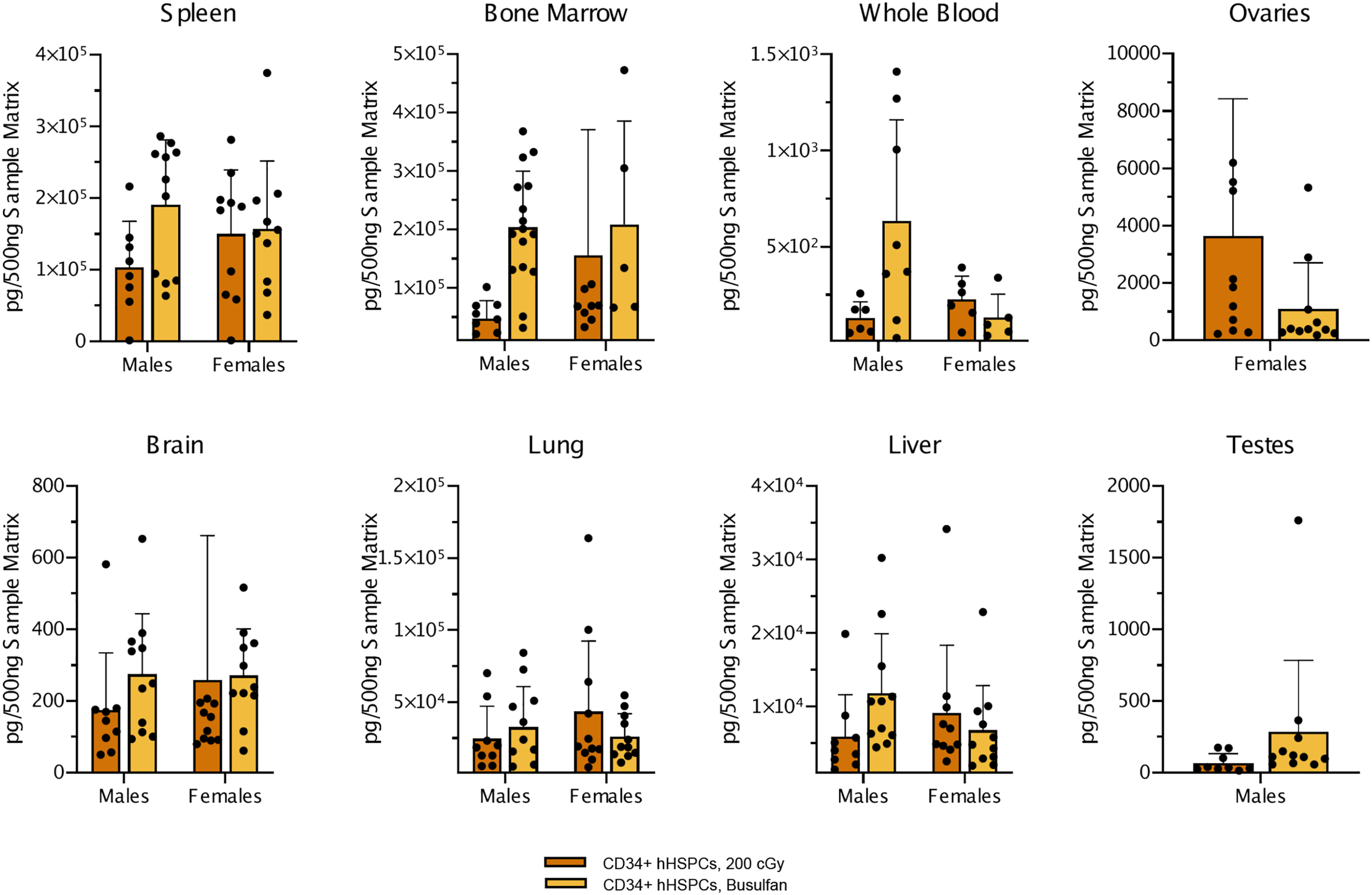
Biodistribution of human Alu sequences in males and females throughout various tissues. Alu sequence quantification by qPCR in various tissues from males and females either conditioned by irradiation with 200 cGy or treatment with busulfan before injection with CD34+ hHSPCs. Values represent means of each group and error bars represent standard deviation.
Pathology Evaluation
Organ Weights
Mobilized CD34+ hHSPCs-related irradiation and busulfan-treatment mean absolute and relative organ weight differences compared to irradiated controls.
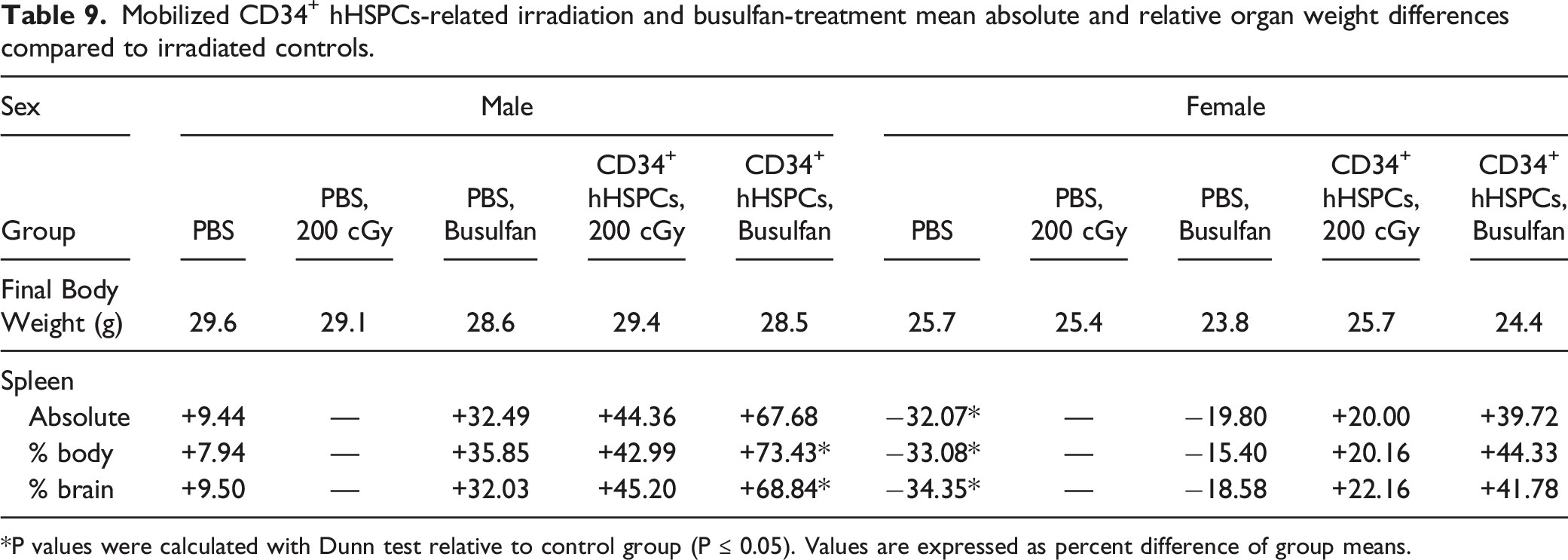
P values were calculated with Dunn test relative to control group (P ≤ 0.05). Values are expressed as percent difference of group means.
Macroscopic Pathology
Macroscopic changes related to the injection of CD34+ hHSPCs with 200 cGy or busulfan-treatment were present in the spleen. Three female animals from each group had an enlarged spleen that correlated microscopically with minimal to moderate engrafted round cells.
Microscopic Evaluations
Group incidence and severity of mobilized CD34+ hHSPCs related irradiation and busulfan-treatment microscopic changes in the spleen and femoral bone marrow.
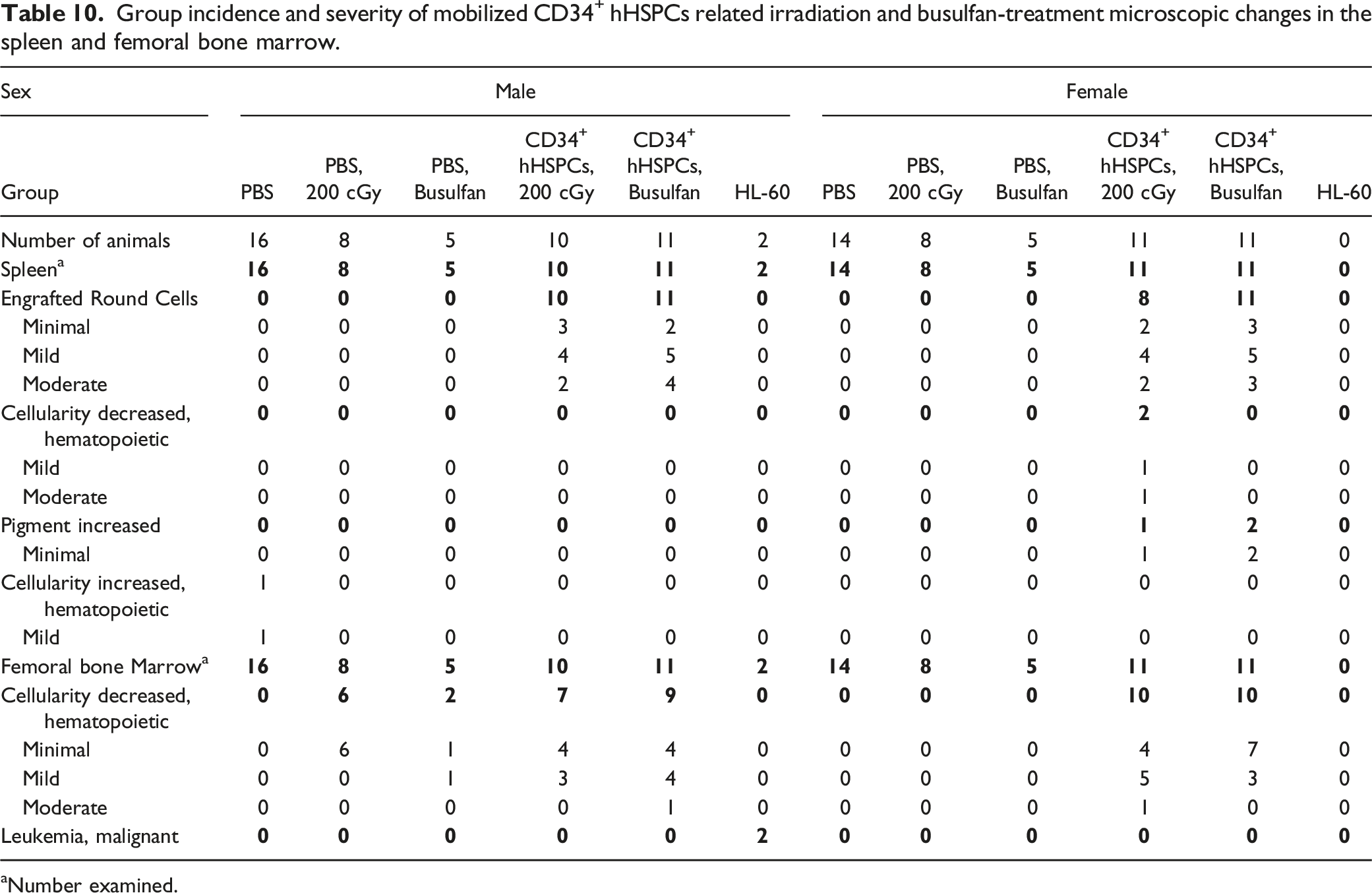
Number examined.
Mortality
Group incidence of neoplastic findings during Phase 2.
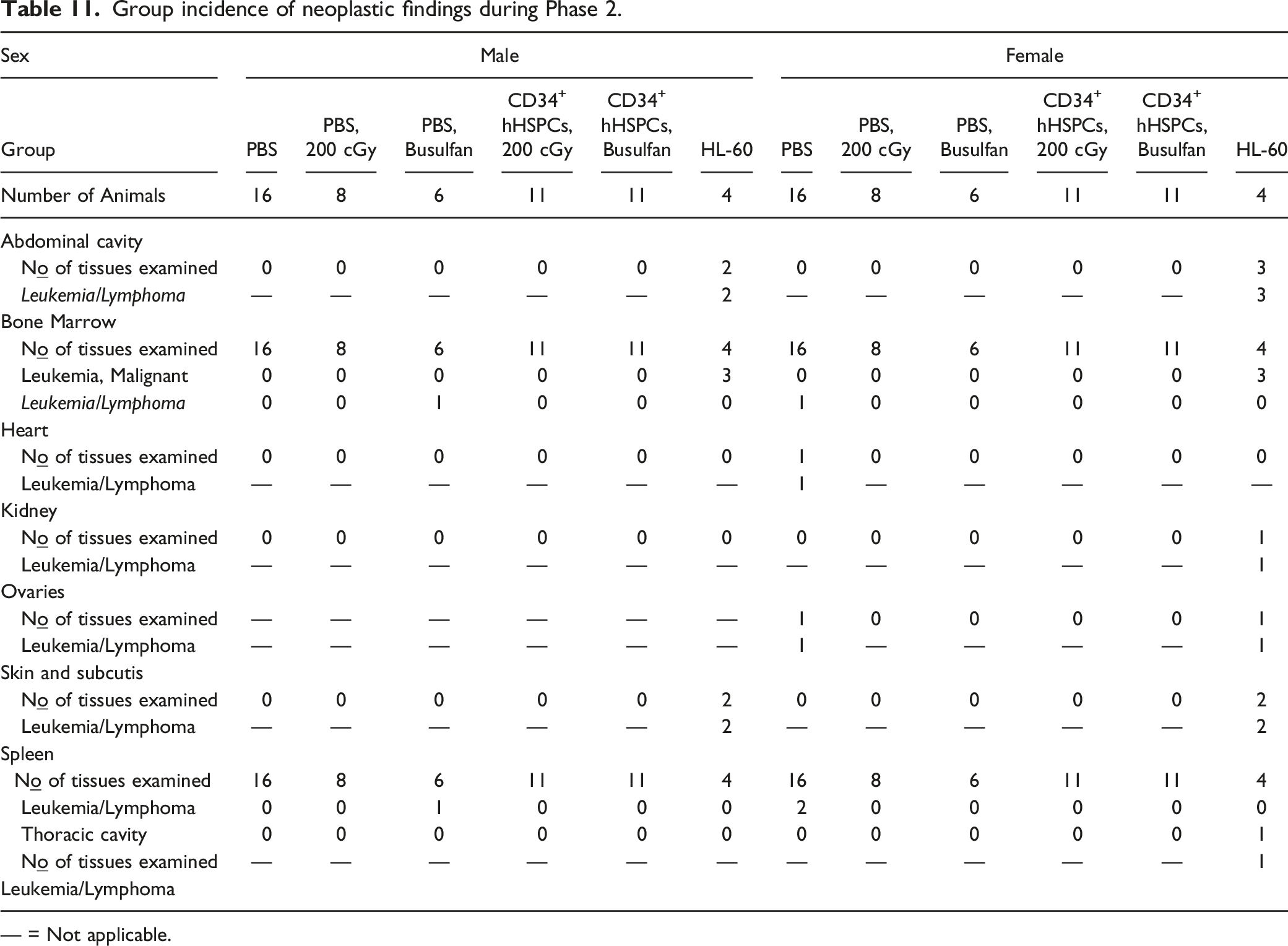
— = Not applicable.
Discussion
In the first phase of this study, the optimized irradiation dose for myeloablation in NCG mice was identified as 200 cGy. This dose corresponds to the historical irradiation dose largely used in preclinical cell therapy research18,19 and was well tolerated with no mortalities, clinical observations or changes in body weights observed. In the second phase, the NCG mouse model was developed as a test system to assess toxicity, engraftment, biodistribution, and tumorigenicity with different myeloablation regimens: irradiation at 200 cGy or intra-peritoneal busulfan administration.
Pre-Clinical Models for Cell Therapy
Immunodeficient animal models are essential to cell therapy development since mice with intact immune systems will systematically reject human cell engraftments. For a review of humanized mouse models see Shultz et al. 2007 and 2019.20,21 Currently, there are mouse models that exhibit varying degrees of immunodeficiency. However, the NCG mouse model bearing a NOD background with edits in the genes Prkdc and Il2rg overcomes some of the limitations of previously engineered immunodeficient mouse models such as SCID (severe combined immune deficiency) and NOD (nonobese diabetic) SCID models. For example, while NOD mice have decreased NK cell activity, 22 the NK cell activity in NCG mice are absent, 23 thus further decreasing innate immune function and increasing the probability of successful human cell engraftment. This is especially beneficial since NK cell activity is a known deterrent of cell engraftment.24,25
By having a NOD/Nju background, NCG mice contain a mutation in the gene SIRPα which allows for enhanced engraftment of human hematopoietic stem cells 26 while the mutation in the Prkdc gene (protein kinase, DNA-activated, catalytic polypeptide) confers an SCID-like (severe combined immunodeficiency) phenotype which was first characterized in 1983. 27 Prkdc is essential for repairing double-stranded DNA breaks, and for generating multiple unique antigen receptors during T and B cell maturation via recombination of V (variable), D (diversity), and J (joining)28,29 DNA segments. The former function in DNA repair makes these mice sensitive to irradiation while the latter recombination function for mature B and T cell development is reduced in these mice. 20 Lastly, the mutation in the Il2rg (interleukin-2-receptor γ-chain) gene results in defective cytokine signaling since the gene product, IL2Rγ, is a necessary component for multiple cytokine receptors including IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21 leading to additional deficits in innate immunity.20,21 Notably, mice deficient in Il2rg also have defective NK cell activity. 20 In sum, the combination of these genetic modifications makes the NCG mouse model a useful tool for pre-clinical assessment of cell therapy safety and efficacy.
The NSG mouse model is often used for cell therapy toxicology, biodistribution and tumorigenicity assessments.18,30 Similar to the NSG, the NCG presents defective function for Prkdc and IL2rg genes which renders the strain permissive to human cell engraftment and consequently safety assessment.
Factors Impacting Engraftment
In addition to the mouse model used, other experimental parameters related to animal procedures influence engraftment success to varying degrees including, method of cell injection31-33 (which also appears to influence risk of graft versus host disease (GvHD) when cord blood is used)34,35 the interval between myeloablation and cell injection,36-38 conditioning method,39,40 conditioning dose, 41 and sex 25 and importantly the age of the mice at cell treatment. 42 Our observation that female mice exhibited increased engraftment rates compared to male mice has been reported in previous studies. 25 Indeed, this observation could in part be due to the hemizygous nature of the Il2rg mutation in male mice. When evaluating myeloablation methods, under certain contexts, busulfan-treatment has been shown to be equivalent 39 or superior 40 to irradiation. Furthermore, irradiation may influence subsequent differentiation and lineage fate of the engrafted hHPSCs (see Dorshkind et al. 2020 for review). 43
Aside from impacting engraftment success, there are clinically relevant, potential negative side effects associated with irradiation and busulfan-treatment. For example, because irradiation-based conditioning is not cell-specific, especially at high doses, it damages faster replicating cells such as those in gastrointestinal crypts,44-46 thus increasing the risk of bacterial translocation. To reduce this risk, antibiotics have been used to regulate the gut microflora following irradiation. In addition to preventing infection, antibiotic treatment can reduce GvHD in some experimental contexts. However, antibiotics may also reduce mobilization by impacting endotoxin content in the gut which seems to play a role in mobilization. 47 Similarly, busulfan which is often used for its myeloablative properties, is known to produce potential adverse effects on vascular endothelial cells. 48
Engraftment success can also be influenced by experimental parameters associated with the human donors including the type of human cells administered, 49 source of the cells (for a review comparing peripheral blood or bone marrow stem cell sources see Amouzegar et al. 2019)), 50 method of collection (venous or arterial access), 51 characteristics of the human donors (including age, sex, body mass index, and genetic background),52,53 concentration of the cells administered, 25 and mobilization strategies. 54 For example, stem cells from cord blood present a few advantages including permissibility of HLA mismatch and a lower risk for graft versus host disease. However, a major limitation of cord blood is the finite number of HSPCs relative to the amounts found in bone marrow or mobilized peripheral blood. As reviewed by Lund et al. (2015), cord blood has a median time to engraftment longer than 3 weeks, while mobilized peripheral blood stem cells can engraft in 13-15 days. 49 To overcome these challenges, research has focused on improving ex vivo expansion of cord blood cells to increase their numbers before injection, and on increasing homing of cells to the bone marrow by using intra-bone marrow injections or pathway-mediated approaches through complement, chemokine, and prostaglandin pathways.49,55
Conversely, when cells are obtained from adults, as mentioned, characteristics including age and sex of the donor impact the success of CD34 cell mobilization response, whereby mobilization has been shown to be less efficient in females51,52,56 and in older individuals.51,52 Furthermore, for donor cell mobilization, there is evidence that a combination of G-CSF and plerixafor (Mozobil®) is more effective. 54 Importantly, these variables do not exist in isolation and can be compounding in nature, for example, busulfan may be metabolized more rapidly in young individuals, 57 and thus, if using young mice, a higher dose may be considered. 58 Here, both age and the myeloablation method must be considered. For a review of considerations related to autologous and allogenic hematopoietic cell transplantation, see Duong et al. (2014). 59
Characterization of the NCG Mice as a Test System for Cell Therapy Assessment
In the present study with NCG mice, we compared irradiation or busulfan-treatment followed by the injection of CD34+ hHSPCs from three adult donors then assessed toxicology, engraftment, biodistribution and tumorigenicity.
Conditioning-related clinical pathology changes included minimal decrease in erythrocyte mass and platelets (irradiation or busulfan) and minimal decrease in reticulocytes in irradiated mice. Administration of CD34+ hHSPCs in conditioned mice was associated with greater decreases in erythrocyte mass (with secondary increased mean corpuscular volume, mean corpuscular hemoglobin and red blood cell distribution width), reticulocytes and platelets when compared to conditioned mice. Injection of CD34+ hHSPC in conditioned mice (irradiation or busulfan-treated) resulted in mildly to moderately decreased total white blood cells, neutrophils, monocytes and eosinophils in all groups while moderately decreased lymphocytes were observed in conditioned male mice.
Engraftment was evaluated by flow cytometry data of hCD45+ cell concentration in whole blood at Weeks 10 and 17 post-injection, and absolute hCD45+ cell counts in spleen and bone marrow at Week 20. Chimerism obtained with cells collected in adult donors ranged from 2.2% to 75.8% for whole blood, from 13.6% to 78.3% for spleen, and from 4.5% to 91.8% for bone marrow. Interestingly, in whole blood, the level of human cells in females was approximately double that in the males, especially at Week 17. Engraftment was also higher in bone marrow samples of females and differences in chimerism level appeared to be dependent on the CD34+ hHSPC donor. Moreover, the chimerism was higher in the busulfan-treated mice compared to irradiated mice, which in whole blood, may be explained in part by the lower level of mCD45+ cells in the busulfan-treated group compared to their irradiated counterparts in whole blood samples.
Biodistribution data was assessed by qPCR of human Alu sequences which are ubiquitously spread throughout the human genome 60 and thus offer a robust method for determining the level of chimerism achieved following CD34+ hHSPC injection. We observed Alu sequences across various tissues, in both busulfan-treated and irradiated mice, with highest values in spleen and bone marrow, and lowest in testes.
Spleen organ weights were higher in busulfan-treated mice and irradiated male mice, indicative of round cell engraftment which corroborated with microscopic findings. In bone marrow of conditioned mice injected with CD34+ hHSPCs, there was decreased cellularity of the hematopoietic compartment possibly due to delayed recovery and/or the presence of engrafted cells. As expected, neither conditioning group injected with CD34+ hHSPCs presented with neoplasms by termination at Week 20 nor did body weights change over the course of the study.
Future Outlook
We observed donor-dependent variation in engraftment as reflected by the varying hCD45+ chimerism between CD34+ hHSPC donors across tissues. Indeed, donor variability will remain an important consideration in future pre-clinical and clinical trials, and essential to report. Regarding the dose of cells used in the present study (1 x 106 cells/mouse), unlike typical toxicological studies where multiple doses are tested and the pre-clinical doses relate in a predictable manner to the clinical doses, pre-clinical cell therapy dose regimens typically investigate a single dose given limitations with dose translation from murine models to the clinic. It is unclear whether mouse cell doses are even relevant to human doses as the physiological environments and cell signaling cascades are significantly different when studying human cells in a murine model. As an example, mice are lymphocyte predominant amongst the total leukocyte population while humans have a higher population of polymorphonuclear cells. 61 In humans, studies have investigated varying doses of CD34+ cells in multiple contexts and patient populations62-64 with variable results, ultimately demonstrating that the optimal cell dose is heavily dependent on the donor and the stem cell source. 62
Rather than informing future dose levels in humans, the pertinence of murine toxicology studies is mostly related to identifying the interaction of gene edited human cells in various organs and confirming that malignancies do not develop over the observation period. Pre-clinical and clinical cell therapy safety assessments will also likely incorporate the typical toxicological paradigm where more significant toxicological effects are accepted for more severe diseases. With autologous CAR-T safety assessments, GvHD is not as much of a concern as with allogenic T cell therapies. However, in both cases, a tissue cross reactivity study is required to assess whether the chimeric TCR reacts with endogenous human tissues. In this assessment, a surrogate that carries the TCR (T cell receptor) binding area, such as a mouse monoclonal antibody, needs to be generated, then applied to healthy human tissues for assessing immunoreactivity. For indications in regenerative medicine, tumorigenicity is an important safety assessment to conduct. 65 However, the time frame to assess tumorgenicity with confidence remains limited since lymphomas are the most common tumors in mice, and risk increases with age.66,67 Therefore, when assessing tumorgenicity for extended periods of time post-engraftment, this factor must be considered. Although NSG and NCG mice have some resistance to developing lymphomas when compared to other immunodeficient mouse strains (NOD SCID mice), lymphomas still can occur, 68 thus, appropriate controls are necessary to help differentiate gene editing related malignancies from spontaneous occurrences.
In future studies, it would be of great interest to precisely characterize histological endpoints in NCG mice. Notably, histopathological signs of GvHD appear to precede clinical signs, pointing towards an increased sensitivity for histopathology to detect GvHD in NSG engrafted mice. 69 Additionally, it will be relevant to explore cell and gene therapy in unison with the concurrent advancement of gene therapy and the improvement of viral vectors such as lentiviral (LV) and adeno-associated viral (AAV) vectors. LVs insert into DNA potentially causing insertional mutagenesis and increasing the risk of tumors, while AAVs have much lower incidence of integration into DNA, which was further diminished by the generation of recombinant AAVs. 70 As a result, AAVs are regarded as a safer option for gene therapy approaches with reduced risk of tumorgenicity, in addition to low pathogenicity, and low immunogenicity. 70 However, depending on whether the target cell is mitotically active, integrating vectors would be necessary for sustained gene expression as non-integrating vectors are preferrable for non-dividing cell targets, therefore LVs are useful for ex vivo gene transfer into hematopoietic and other stem cells, while AAVs are useful for in vivo gene therapy in post-mitotic cells. 26 Future studies will likely focus on long-term effects of LV and AAV administration to confidently rule out any potential genotoxicity caused by vector genome integration.71,72 Whether LV and AAV integration can solely cause increased tumorgenicity remains to be seen, especially since conditioning methods such as busulfan and irradiation, as well as individual comorbidities found in patients can also lead to increased risk of certain cancer types. 71 However, both pre-clinical assessments of tumorgenicity and long-term follow-up clinical studies will be helpful for determining the risk/benefit ratio of these cell and gene therapy strategies.73-75
Conclusion
The irradiation dose for cell myeloablation in NCG mice targeting the highest sublethal dose which was determined as 200 cGy in Phase 1. The engraftment, biodistribution, and tumorigenicity of mobilized adult CD34+ hHSPCs following a single intravenous slow bolus injection into pre-conditioned irradiated or busulfan-treated NCG mice was determined in Phase 2. Human CD34+ cells were observed in whole blood, spleen and bone marrow samples in groups injected with mobilized adult CD34+ hHSPCs, displaying a chimerism ranging between 2.2% and 91.8%. Biodistribution showed the presence of human Alu sequences at Week 20 across various tissues with the highest levels in the spleen and bone marrow and the lowest in the testes while no Alu sequences were identified in non-injected animals. As expected, there was no neoplasm associated with the administration of mobilized adult CD34+ hHSPCs with either 200 cGy irradiation or with busulfan-treatment. Overall, this work illustrates key considerations when designing and interpreting toxicology studies for cell therapies with murine test systems.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
