Abstract
The presence of health issues (diarrhea, poor body condition) in non-human primates can impact animal welfare, confound toxicity study data, and lead to animal exclusion from studies. A working group cosponsored by DruSafe and 3Rs Translational and Predictive Sciences Leadership Groups of the IQ Consortium conducted a survey to benchmark quarantine, pre-study screening, husbandry, and veterinary care practices and their impact on NHP health. Nineteen companies participated in the survey providing separate responses for studies conducted in-house and at Contract Research Organizations from 3 regions (North America (NA), Europe and Asia) for an aggregate of 33 responses. A majority of responding companies conducted studies at North America CROs (39%) or in-house (36%) using primarily Chinese (33%) or Cambodian (27%) and to a lesser extent Vietnam (18%) or Mauritian (15%) origin NHPs. Forty-Five percent of responses had pre-study health issues (fecal abnormalities, etc.) on ≥ 1 studies with the highest incidence observed in Vietnam origin NHPs (80%). The survey suggested variable pre-screening and quarantine practices across facilities. Husbandry practices including behavioral assessments, environmental enrichment and consistent diets were associated with a lower incidence of health issues. The survey also benchmarked approaches used to diagnose and manage abnormal feces in NHPs and has provided strategies to minimize impact on NHP health. The survey highlighted opportunities for harmonizing screening criteria across industry and for improving tracking and sharing of health screening results, leading to further refinement of NHP veterinary care practices, higher quality studies, and reduced NHP use.
Introduction
The concept of the Three Rs (3Rs) that includes Replacement, Reduction and Refinement is considered the guiding principle for ethical and humane care of animals in research.1,2 Scientists across the pharmaceutical industry integrate 3Rs approaches in preclinical studies aimed to refine animal husbandry practices, aid in improving animal welfare, and provide better quality scientific data. Nonclinical studies supporting drug development often require use of non-human primates (NHPs) as a non-rodent animal species based on their genetic and physiological similarity with humans. 3 NHPs are often the only suitable species available for use in preclinical safety studies with biologics, thus necessary for eventual regulatory approvals for new biotherapeutic drugs.4,5 Safeguarding the health and well-being of NHPs used in research is critical to ensure quality study outcomes and judicious use of NHPs.
Over the past few years, the already limited global supply of NHPs (including sexually mature animals) has been significantly impacted by the COVID-19 pandemic leading to a pronounced shortage of NHPs.6,7 This can result in the sourcing and placement of animals that do not meet ideal study criteria for study inclusion, such as age and/or physical condition, and in turn negatively impact the quality of study results and animal welfare. NHP shortages are likely to further contribute to an increase in the incidence of animals with abnormal fecal changes, poor body condition, or abnormal stress-related behaviors, which can have an impact on NHP study outcomes. NHP health issues, such as abnormal feces, during the pre-study/acclimation phase can lead to exclusion of animals from study, longer acclimation periods (to allow for appropriate treatments and recovery), and delays in study initiation. To avoid delays in study initiation and to ensure an adequate number of healthy animals for a study, additional animals may be ordered leading to increased animal usage. When animals with suboptimal health are inadvertently included in toxicity studies, health issues such as abnormal feces or body weight loss can resurface during the study phase, impacting the study quality and resultant data. This can include early removal of animal(s) after study initiation thus reducing the power of the study and/or confounding study interpretation with unintentionally attributing health-related issues as test article-related effects. In addition, medical intervention with therapeutic drugs may interact with the test article in an unknown way and alter study endpoints such as clinical or anatomical pathology.
In November 2019, the DruSafe and 3Rs Translational and Predictive Sciences (TPS) Leadership Groups of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ) formed a working group (WG) to determine the frequency of NHP health issues and benchmark current NHP husbandry practices across the pharmaceutical industry. To this end, an initial cross-industry survey was conducted that primarily focused on the pre-study period. In addition to determining the frequency of NHP health issues and benchmarking current NHP practices, the survey provides insights into NHP usage in terms of demographics and country of origin. Survey results were used to decipher the impact of current common NHP husbandry management practices on NHP health issues and identify opportunities to improve quality and consistency of NHP husbandry. Recommendations for best practices in NHP husbandry and pre-screening prior to transport are proposed that can minimize health issues, enhance animal welfare consistent with the principles of the 3Rs, and improve the quality of toxicity studies utilizing NHPs.
Material and Methods
The survey was submitted to member companies of the IQ DruSafe and 3Rs TPS Leadership Groups at the end of 2020. Companies were asked to share their experience and practices related to NHP husbandry. Responding companies were asked to fill out separate survey responses based on where their NHP studies were conducted (in-house, or externally at Contract Research Organizations [CROs] in North America, Europe, or Asia) Therefore, it was possible that a single company could have provided up to 4 separate responses if they conducted studies at CROs from all 3 regions as well as in-house studies. Respondents from each participating company were subject matter experts and largely included veterinarians, study directors, study monitors, toxicologists, or pre-clinical scientists. Survey feedback from individual companies submitted should not be considered as an “individual opinion” as collated responses were based on diverse working knowledge and husbandry practices that were internally agreed upon or in discussion with their respective CRO partners. The responses from individual companies were kept confidential and were anonymized and collated by an independent third-party secretariat (Faegre Drinker Biddle and Reath, LLP) before being provided to the authors.
The survey was composed of 35 main questions with 6 questions having sub-question(s) depending on response to the initial question (see Supplemental Material). The first 4 questions were focused on demographics involving where NHP studies were conducted, average number of studies conducted, and source of the NHPs used in toxicology studies. The respondents were then asked general questions about experience with NHP health issues and number of studies that were negatively impacted due to these health issues during the acclimation or pre-study period over the past 2-3 years. This was followed with questions about quarantine and screening processes. The final set of questions was focused on respondents experience with abnormal fecal observations that addressed veterinary engagement, treatment and diagnostic approaches, and any observed correlations with NHP age or source. Data analysis was based on the number of responses to each question and further stratified as responses with and without health issues. Diagnostic data was further classified as abnormal feces with compromised health or persistent abnormal feces but otherwise healthy.
Survey Limitations
As with all cross-industry surveys, there were several limitations that can impact the interpretation of the survey. A majority of the survey questions were multiple choice; therefore, responses were limited to the available choices. Additionally, many of the questions that had numerical responses provided a range. For instance, respondents were asked “On average, how many NHP studies do you conduct per year?” with the following answer choices a) 0-25, b) 25-50, c) 50-75, d) 75-100 and e) > 100. While this approach is acceptable for showing trends, it lacks precision, and this should be taken into consideration when interpreting responses and correlations. Data interpretation for a few questions was also limited due to free textbox and “other “categories. Another limitation is that the survey does not capture trends in health issues over time from respondents where husbandry practices were revised after health issues were noted. Finally, responses regarding CRO practices were provided by pharmaceutical companies whose level of familiarity with individual CRO practices were likely variable and potentially incomplete as demonstrated by respondents selecting “do not know/rely on CRO practices” for some questions. A follow up survey to complete the gaps identified in the current survey and directed to the CRO specifically could provide a more comprehensive picture of current practices.
Results and Discussion
Demographics
Thirty-three responses were received from 19 member companies. Eleven companies provided responses from a single CRO region or in-house only, 3 companies provided responses from 2 CRO regions or 1 CRO region and in-house, 4 companies provided responses from 3 CRO regions or 2 CRO regions and in-house, and 1 company provided responses from all 3 CRO regions and in-house.
From the 33 responses, the majority of studies were conducted at CROs in North America (CRO-NA; 39%; 13 of 33) and/or in-house (36%; 12 of 33) with small percentages in Asia (CRO-Asia; 15%; 5 of 33) and Europe (CRO-EU; 9%; 3 of 33) (Figure 1A). The majority of responses (73%; 24 of 33) conduct 0 to 25 studies per year on average (Figures 1B). The survey only captured ranges and percentages; thus, this survey did not capture the actual number of studies conducted in a specific region or by an individual respondent. Most NHPs were sourced from China (33%; 11 of 33) or Cambodia (27%; 9 of 33) with fewer NHPs coming from Vietnam (18%; 6 of 33) and Mauritius (15%; 5 of 33) (Figure 1C). One response indicated they defer to the CRO to source the NHPs on their studies and another response used domestically sourced Indian-origin rhesus. Panel A: Y and X-axis represents number of responses placed at CROs in different Regions or in- House. Panel B: Pie chart demonstrates percent of survey responses of average number of nonclinical NHP studies conducted per year at any site. Panel C: Histogram represents number of responses on Y-axis and countries of NHP origin on X-axis. Shaded region and non-shaded region of histogram depicts responses with and without NHP health issues.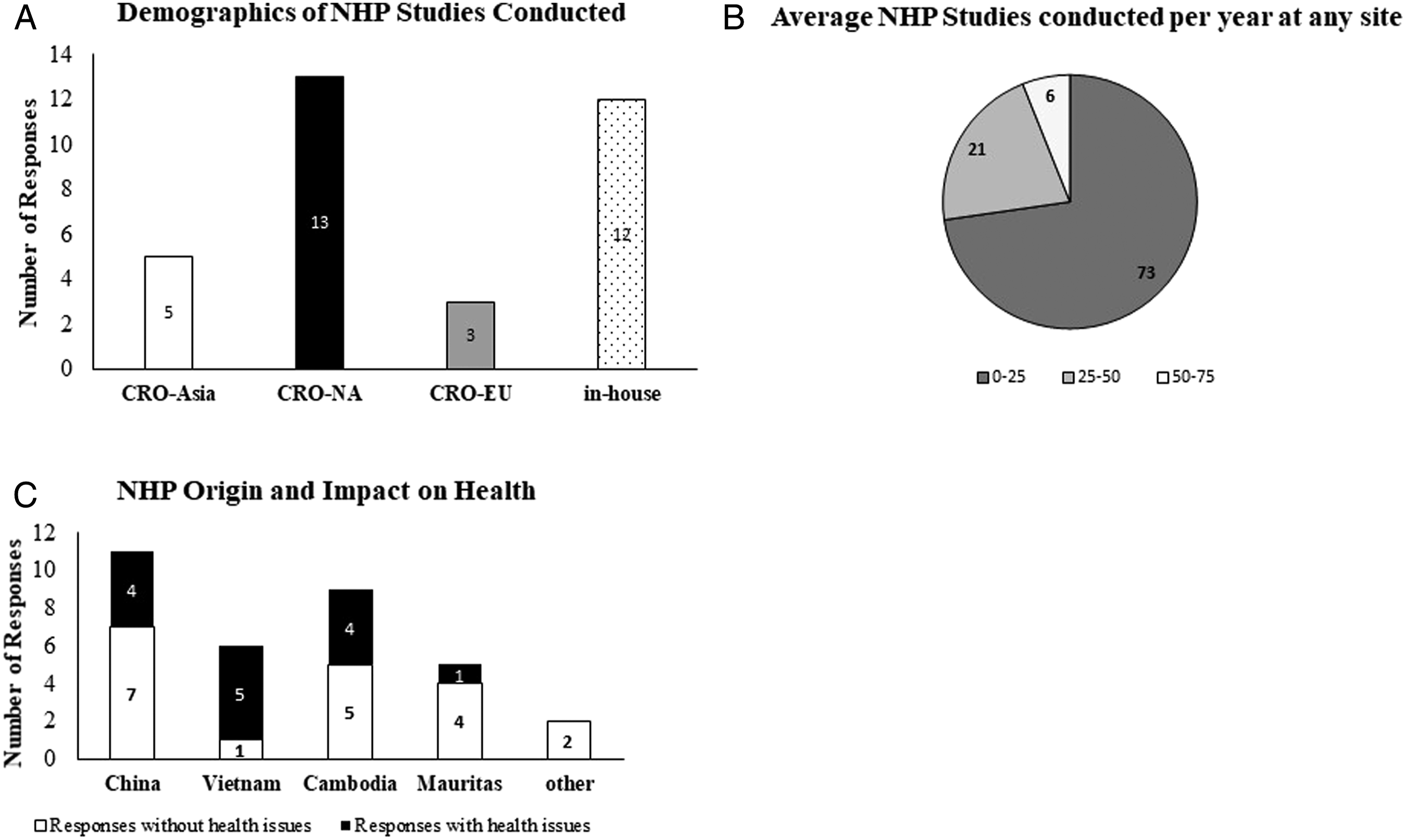
In addition to the questions on NHP study location and number, survey respondents were also asked about the number of studies impacted due to health issues (ie, abnormal feces, respiratory findings etc.) unrelated to test article that required veterinary intervention in the pre-study phase. The survey suggests that veterinary intervention during the acclimation/pre-study period is common (87%; 28 of 32); however, the percentage of studies impacted was relatively low impacting less than 25% of studies (78%; 25 of 32). Twenty-one percent (6 of 32) of responses had to euthanize an animal in the acclimation/pre-study period or control animals during the study for health-related issues (excluding trauma). For the respondents that had experience with euthanasia under these conditions, only one response (3%; 1 of 32) indicated this occurring in more than 4 studies. The other responses (15%, 5 of 32) indicated this occurred in 1 to 4 studies only (available predesignated responses: “0”, “1 to 4”, and “>4”). One respondent chose not to respond. When asked if studies were negatively impacted due to non-test article-related animal health issues (e.g., animal health issue made interpretation of study results more challenging), 44% (14 of 32) indicated having one or more studies (1-4 or >4 studies) impacted while the remainder of responses indicated no impact (56%; 18 of 32).
CROs in North America appear to have a greater incidence of NHP health issues compared to European and Asian CROs. However, the apparent increase could be attributed to the higher number of studies conducted at North American CROs and the limited sample size with uneven distribution of the NHP source. It’s unclear from the survey if animal origin/source contributes to incidence of health issues. Vietnam-sourced NHPs appear to have a higher incidence of health issues as compared to Cambodian or Chinese NHPs (Figure 1C). However, responses indicated that country of origin was not perceived to be a causative factor for increased frequency of persistent abnormal feces (34%; 11 of 32) or don’t know (59%; 19 of 32) with only 2 (6%) responses indicating Mauritius animals seem more affected. Published information on the impact of NHP origin and general health including susceptibility to gastrointestinal (GI) disorders such as persistent abnormal feces, are limited. Mauritius NHP are reported to be free of several simian viruses (Simian Immunodeficiency Virus (SIV), measles, Simian Retroviruses (SRV-1 and -2), Simian T-lymphotrophic Viruses (STLV-I and -III), and B-virus) that can be found in NHPs from other origins. 8 In one study, there were no relevant differences reported on clinical observations and pathogen screening (viral, fecal parasitology and fecal culture) between NHPs of Mauritius (N = 10/sex) and Asian origin (N = 20/sex), 9 although there were fewer spontaneous physiologic and anatomic abnormalities in the Mauritius origin animals compared to Asian origin. This is consistent with findings from another study where there was an increase in lymphoplasmacytic cell infiltrate in the GI tract from Asian origin NHPs. 10 This study was focused on clinical and anatomical pathology findings and did not provide any information on clinical observations such as abnormal feces associated with the pathology findings. Additionally, a study of a limited number of NHPs (N = 33) found Mauritius NHPs to be more resistant to Shigella dysenteriae than Indochina or Philippine origin NHPs, an effect that was attributed to differences in GI microbiota. 11 To appreciate the impact of CRO location and NHP origin more fully on NHP health issues including GI disorders, a more focused survey on these topics that includes responses from CRO representatives would be necessary.
NHP Quarantine Practices
NHPs are imported from suppliers and are transported and relocated to test facilities where they have an acclimation period before their use in pre-clinical studies. Data obtained during the acclimation period is highly variable and common physiological changes, including alterations in clinical pathology parameters, can return to baseline with an acclimation of 1 to 7 days following transportation; however, some changes (like circulating monocytes, B-cell numbers) may take greater than 30 days to normalize depending on the parameter measured, age of the NHP, and level of stress.12,13 While transportation practices were outside of the scope of this survey, attempts to minimize transportation-related stress including transporting established social pairs should be considered as part of the concerted effort to reduce stress during this period.
14
Most of the survey responses indicate a secondary quarantine for NHPs between 8-29 days (59%, 17 of 29) once animals arrive at the testing facility. Other responses (38%, 11 of 29) indicated 30 + days for quarantine and only 1 of 29 responses indicated quarantine for 0-7 days after animal arrival. Considering the risk posed to successful study outcomes and interpretation, as well as reduced animal use by emergent and/or pre-existing health issues, respondents were also surveyed on their quarantine practices in relationship to NHP health. It is widely believed that extended quarantine periods (15 + days) allow for increased monitoring and detection of latent or emerging health issues. Interestingly, the survey found the frequency of issues detected during the quarantine period was similar regardless of the quarantine duration; 44% (4 of 9), 50% (4 of 8), and 36% (4 of 11) of responses reported health issues for 8-14-day, 15-29 day and 30 + day quarantine period, respectively (Figure 2A). No health issues were reported to have been detected by the one response with a quarantine period of <7 days. It should be noted that quarantine periods of at least several weeks are standard recommended practice for NHPs and considered a critical part of facility biosecurity plans.
15
Notably, within these quarantine windows there was a clear decrease in the number of responses that had observed health issues when behavioral assessments and/or specialized training are in place, 29% (4 of 14) with vs. 58% (11 of 19) without health issues. This may be related to reductions in relative stress levels as well as increased monitoring that occurs when these practices are implemented (Figure 2B). Similarly, most responses (>75%) use environmental enrichment (entertainment, treats, changing of toys, etc.) during the quarantine period and correspondingly fewer observe health issues with environmental enrichment in place compared to without enrichment (40% [10 of 25] with vs. 63% [5 of 8] without). Interestingly, all other behavioral and husbandry management practices commonly employed (including social housing, use of positive reinforcement training and lack of an extended acclimation period) had no clear impact on NHP health issues (Figure 2B). This finding may be related to the wide implementation of these procedures and should not be interpreted as justification to stop their use. Behavioral indicators of well-being were not included within the scope of this survey. Importantly, higher incidences of health issues were noted in responses where no quarantine behavioral/handling training was provided (6 of 6) compared to responses (6 of 18) that indicated behavioral or handling training was provided during the quarantine period. Another outcome from this survey was the low answer rate (23 responses selected not applicable) for personal protective equipment (PPE), anteroom and separate exit/entry use, and other environmental controls to limit the spread and occurrence of common pathogens for respondents who directly import NHPs. This may reflect an issue in which the survey question was addressed towards “directly importing NHPs” and not necessarily reflects a lack of respondent’s familiarity with CRO or animal facility practices or a lack of these practices being utilized. Panel A: Y and X-axis represents number of responses and range of quarantine periods (0-7 days, 8-14 days, 15-29 days, and 30+ days). Shaded and non-shaded region of histogram depicts responses with and without NHP health issues. Panel B: Histogram represents percentage of responses on X-axis and refinement practices on Y-axis. Shaded and non-shaded region of histogram depicts percentage responses using refinement practices with and without NHP health issues. Panel C: Histogram represents percentages of responses on X-axis and criteria used for NHP exclusion from shipment on Y-axis. Shaded and nonshaded region of histogram depicts percentage responses with NHP health issues with and without using exclusion criteria.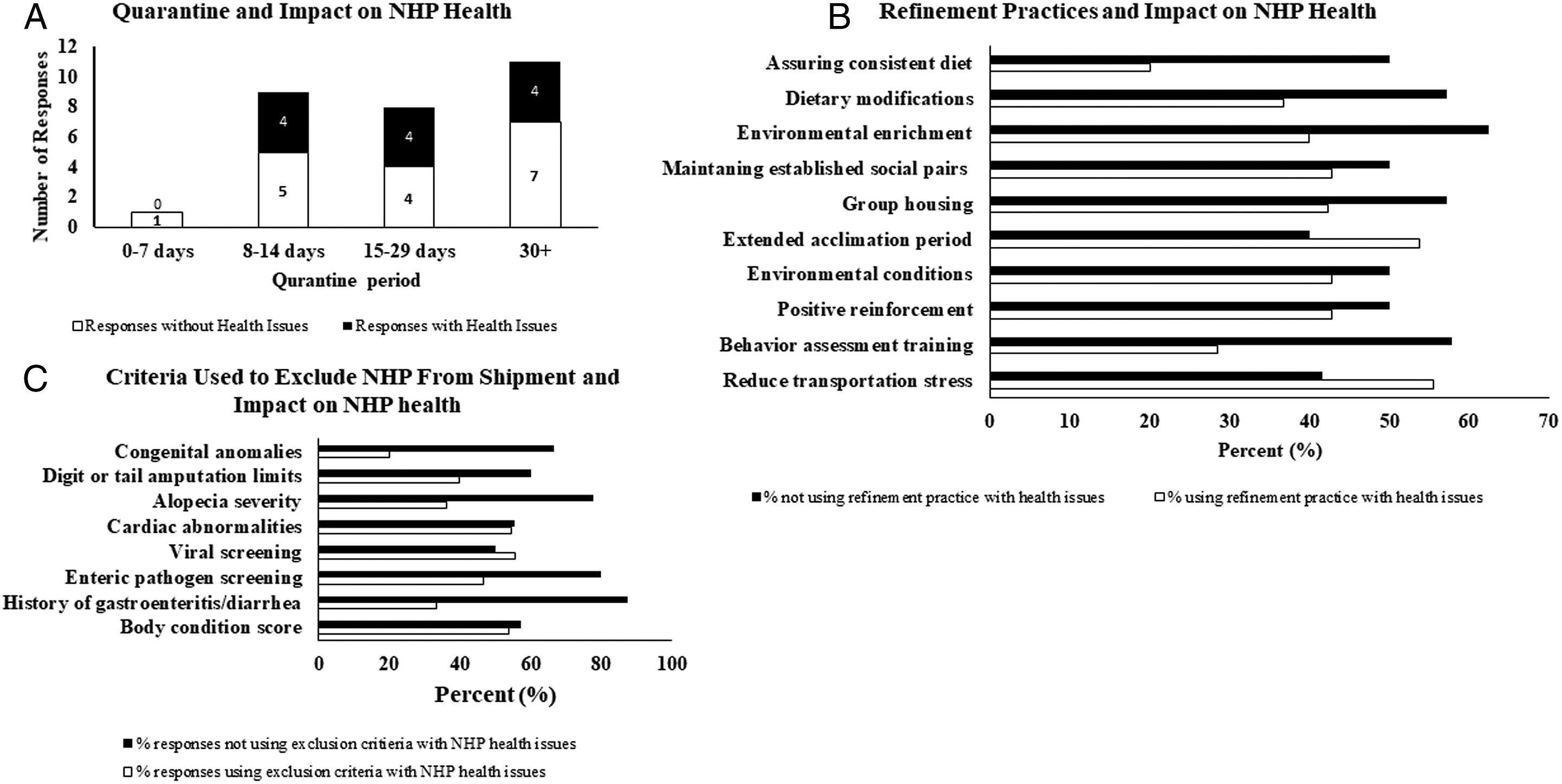
NHP Pre-study Exclusion Criteria
NHP exclusion via pre-screening is a critical step in eliminating confounding variables that can impact study data interpretation. 8 Sixty-two percent (62%; 20 of 32) of responses excluded NHPs from supplier shipments for one or more criteria as shown in Figure 2C, while 34% (11 of 32) indicated they did not know/deferred to CRO practices. Amongst the 20 responses that used pre-defined criteria for exclusion, a majority (>60%) used viral and enteric pathogen screening, followed by body condition score and history of gastroenteritis/diarrhea. Responses that indicated not using pre-defined exclusion criteria such as history of gastroenteritis/diarrhea, enteric pathogen screening, alopecia severity or congenital anomalies reportedly had a higher percentage of animals with health issues (Figure 2C).
The most common health problems leading to pre-study exclusion were abnormal feces and wasting/failure to maintain body weight (51%, 17 of 33 for each), stress-related behavior (36%, 12 of 33) followed by ECG and ocular abnormalities (21%, 7 of 33 for each). Respiratory infections were rarely a reason for exclusion (9%, 3 of 33). Overall, the survey indicates that the implementation of pre-study exclusion criteria especially those focused on general health (e.g., body condition and pathogen screening) and stress behaviors (e.g., severe alopecia) results in fewer health issues during the study.
NHP Health Screening Practices
A number of governmental regulations and general guidelines govern NHP health screening practices for microbial pathogens and viruses during quarantine due to their potential impact on public health and research. 16 However, these screening practices may vary widely between individual research facilities. The survey indicated that most responses (93%, 28 of 30) applied the same health screening practices for all study types, while 7% (2 of 30) indicated the criteria may vary and be more stringent for safety assessment studies than pharmacokinetic studies. The survey also collected details around serology and fecal screening testing. Of note, less than half of the responses (13 of 30) track health screening outcomes with the majority (17 of 30) relying on their CRO partners, CDC quarantine facilities, or suppliers from the country of origin to track this important metric. Closer tracking of this information by study sponsors would provide better insight into evolving trends in NHP health, early identification of emerging health issues and rapid implementation of preventative measures to limit the impact of emerging health issues on NHP studies.
NHP Serology Testing Practices
Table depicts serology testing and number of responses.
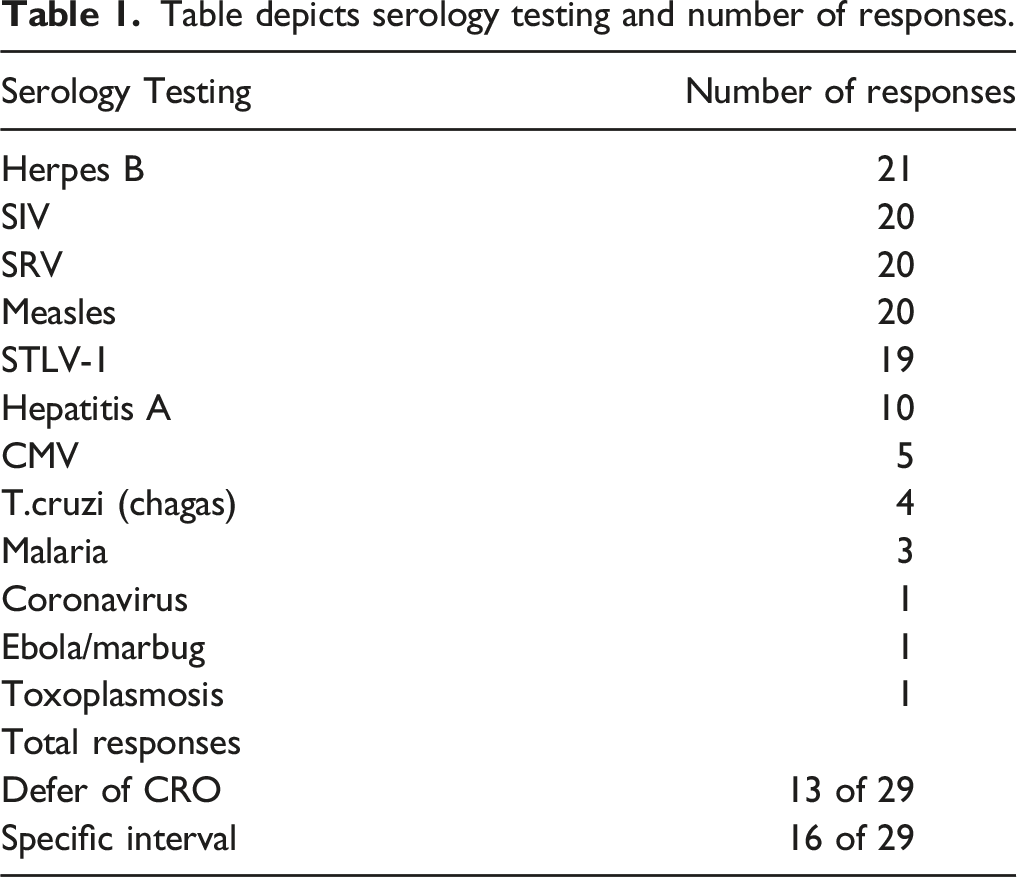
There was wide variation in the frequency of viral screening with more frequent screening being associated with fewer health issues. Thirty-six percent (11 of 31) of responses indicated that they screened only once pre-study or pre-shipment with 10% (3 of 31) screening more frequently and one response screening every 90 days before shipping. One response indicated that repeated testing occurs only in animals that test positive. Thirty-nine percent (12 of 31) of responses relied on their CRO partners for screening practices. For the responses that only perform serology screening once, 64% (9 of 14) have experienced health issues during the pre-study phase, whereas only 25% (1 of 4) with repeated screenings, have experienced health issues in the pre-study phase. This observation supports the recommendation to increase viral screening of animals prior to placement on studies. Additionally, this can act as a focus point retrospectively for future studies to compare screening panels and results during the last decade and in relationship to the country of origin.
As this survey was conducted at the start of the COVID-19 pandemic, a specific question regarding viral screening for SARS CoV2 was included. The majority (81%, 25 of 31) of the responses did not perform SARS CoV2 testing. Only 1 response (out of 32) utilized SARS CoV2 testing for general health surveillance, while five other responses indicated they would only use it for targeted programs or would defer to CROs.
NHP Fecal Screening and Testing Practices
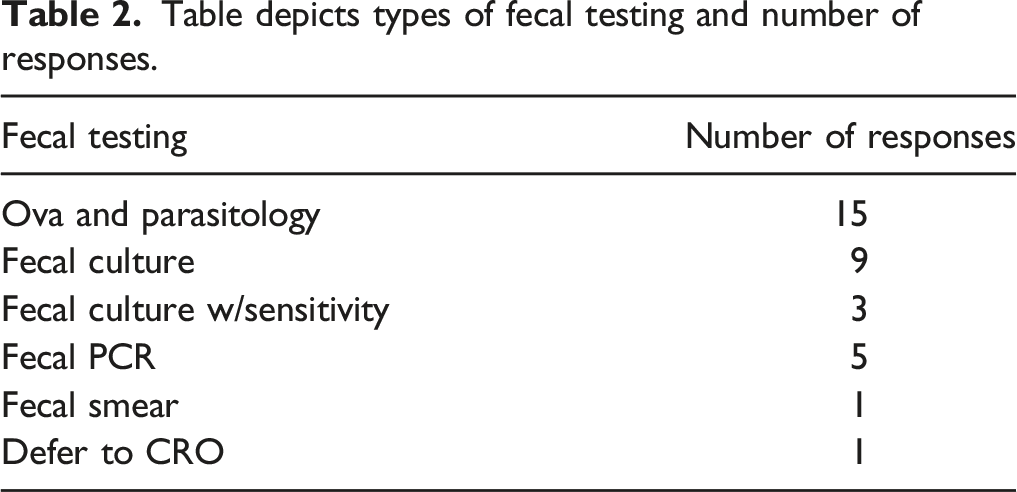
The survey identified that 47% (15 of 32) of the responses routinely perform fecal screening prior to placing NHPs on study, while 13% (4 of 32) only do so with abnormal fecal observations. Only 6% (2 of 32) of responses indicated that they do not perform fecal testing, while 34% (11 of 32) do not know or deferred to the CRO. Fecal testing was performed prior to arrival at the test facility (79%, 15 of 19), upon arrival to the test facility (47%, 9 of 19), during routine health exams (26%, 5 of 19), or before study start (11%, 2 of 19). Out of the 15 responses conducting fecal testing prior to placement, seven responses reported having health issues, while the 2 responses who do not perform fecal testing both reported having health issues. For the responses that routinely conduct fecal screening, 26% (8 of 31) reported excluding NHPs from study based on positive test results during the pre-study phase. Some responses perform fecal testing as needed when symptomatic (21%, 4 of 19), while others did not know or referred to CRO practices (11%, 2 of 19). These observations support the recommendation to increase fecal screening of animals prior to placement on studies. In addition to conducting fecal screening upon animal arrival to the facility, teams should consider pre-study fecal screening as well.
Table depicts types of fecal testing and number of responses.
Abnormal Fecal Observations in NHP
Abnormal feces are a common health observation in captive NHPs housed in breeder and research facility settings. It was unclear from the survey if age of the NHP correlated with persistent abnormal feces, as a majority of responses did not know/did not have sufficient information (56%; 18 of 32) while 34% (11 of 32) indicated no impact of age. Persistent or recurring abnormal feces can be observed in otherwise healthy animals but can also affect overall health and well-being. The survey found that 21% (6 of 29) of responses reported that they typically see no correlative clinical signs or findings with abnormal feces whereas for others body weight loss and dehydration are frequently observed (66% for each, 19 of 29) with inappetence and decreased body temperature seen less frequently in cases of abnormal feces (21%, 6 of 29). Other correlating findings included rectal prolapse, distended abdomen and hypoactivity (write in responses).
The majority of responses (71%, 22 of 31) indicated that persistent and/or unresolving abnormal fecal observations was justification for excluding animals from study. However, a small number of responses indicated that they do not exclude animals with abnormal feces (5 responses) or indicated that it depends on the clinical history of the animals, purpose of the study or if animal numbers are critical to execute the study (4 responses). For responses that do not always exclude animals with abnormal fecal observations, recurrence of fecal abnormalities was observed < 25% (5 responses), <50% (2 responses), or < 75% (3 responses) of the time.
The survey also included questions regarding the triggers for veterinary intervention and types of diagnostic testing and treatment modalities employed.
Veterinary Intervention for Abnormal Feces
Most responses indicated that veterinary intervention was required when an animal had abnormal fecal observations on 3 or more consecutive days (66%, 21 of 32) or after a single occurrence if associated with blood in feces or abnormal behaviors such as lethargy or poor appetite (63%, 20 of 32) (Figure 3A). Observation of abnormal feces 3 or more times per week was another reason to seek veterinary consultation by many (47%, 15 of 32). A number of responses were unsure or deferred to the CRO to make the decision on involvement of veterinary staff (28%, 9 of 32). Further analysis is needed of NHP medical record data to fully understand the initiation of veterinary intervention. Panel A: X and Y-axis represents number of responses and criteria used to determine when veterinary consultation is required during abnormal fecal observations in NHP. Panel B: Histogram represents number of responses on X-axis and patterns of fecal testing based on NHP health status on Yaxis. Shaded and non-shaded region of histogram depicts responses for NHP with abnormal feces with and without overall health being impacted. Panel C: Histogram represents number of responses on X-axis and veterinary treatment employed based on health status of NHP with abnormal feces on Y-axis. Shaded and non-shaded region of histogram depicts responses having NHPs with abnormal feces with and without compromised health.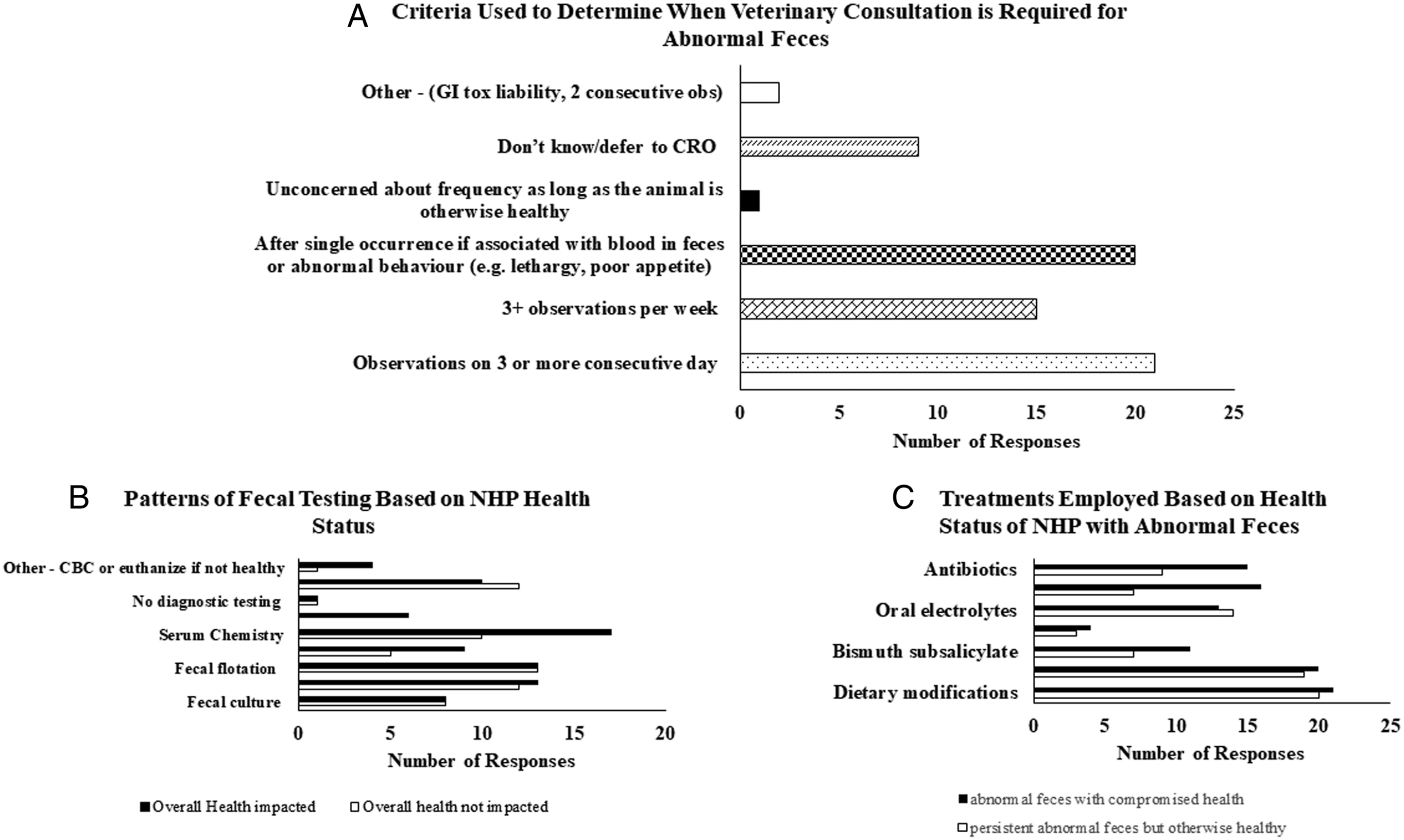
Diagnostic strategy for abnormal fecal changes
Diarrhea in macaques is a complex health issue that requires a multimodal diagnostic and treatment approach. Since bacterial, viral, or parasitic infectious agents have been implicated in causing disease directly or indirectly, close monitoring of study animals and feasibility of eliminating carrier animals should be investigated.
Diagnostic Strategy in Overall Healthy NHPs With Abnormal/Persistent Fecal Changes
The diagnostic tests most often used to evaluate otherwise healthy NHPs with persistent or recurring abnormal feces included fecal flotation (42%, 13 of 31), fecal culture and sensitivity (39%; 12 of 31), fecal culture alone (26%, 8 of 31), and serum chemistry (32%, 10 of 31) (Figure 3B). Many responses (39%, 12 of 31) were unsure of the diagnostic resources/capabilities of the facility or deferred to CRO practices. Fewer responses (16%, 5 of 31) indicated that PCR analysis is typically used for diagnostic workup.
Diagnostic Strategy When NHP Health Is Impacted due to Abnormal Fecal Changes
The survey also investigated which diagnostic tests are used for NHP whose health or overall well-being is impacted due to persistent and/or unresolving abnormal feces. Of the responses that did not defer to the CRO or reported known information about diagnostics, a majority (42%, 13 of 32 for both) employed fecal culture with sensitivity and fecal flotation. Interestingly, 25% (8 of 32) reported that sensitivity testing, which typically aids in the selection of antibiotic, was not conducted along with culture results. Sensitivity testing is a critical step in selection of appropriate antimicrobial therapeutic(s) for enteric bacterial infections. Over half of responses (53%; 17 of 32) used serum chemistry in their evaluations. This represents a higher percentage of responses using serum chemistry as a diagnostic tool when animal health or well-being is impacted than when abnormal feces are observed in otherwise healthy animals. Fecal PCR was used less frequently than culture and sensitivity, though a sizable number of responses (28%, 9 of 32) did use PCR. Postmortem necropsy and histopathology were used less frequently (19%, 6 of 32). A considerable percentage of responses (31%; 10 of 32) reported unfamiliarity with diagnostic testing or deferred to the CRO when animals showed compromised health and diarrhea, one of the gaps identified in the survey.
Usefulness of Fecal Diagnostic Testing and histopathology (microscopic) Evaluations
Fifty percent (14 of 28) and 36% (10 of 28) of responses reported that fecal diagnostic testing and histopathology evaluation, respectively, was not “helpful”. Twenty-one percent (6 of 28) indicated they do not routinely perform fecal diagnostic testing and 32% (9 of 28) don’t perform histopathology evaluation. Approximately a third of the responses found fecal diagnostic testing (29%, 8 of 28) or histopathology (microscopic) evaluation (32%, 9 of 28) “helpful”. When combining these two diagnostics (fecal testing and histopathology), nearly half of responses (46%, 13 of 28) found the fecal diagnostic testing and/or histopathology evaluation “helpful” suggesting the combination is better than either evaluation in isolation.
Of note, the way the question was posed may have impacted the response to how “helpful” fecal diagnostic testing and histopathology evaluations were as choices were limited to “helpful” in >75% of cases, “helpful” in >50% of cases, not “helpful” or do not routinely perform diagnostic testing. It is possible that respondents may have defaulted to “not helpful” when diagnostics were only useful in small percentage of cases (e.g., <50%).
NHP Husbandry Practices for Abnormal Feces
Most responses (71%, 20 of 28) did not take an altered approach if there was more than one animal in the same room presented with persistent, abnormal feces. Of the small percentage of responses that did alter diagnostic and treatment strategies (29%, 8 of 28), multiple factors were considered: separation of affected animals from unaffected animals, quarantine if an outbreak was suspected, additional diagnostics for more animals and treating the entire room including unaffected animals if an enteric agent with potential pathogenicity was identified.
Treatment and Enrichment for NHPs with Abnormal Feces
Respondents were asked a series of questions on typical regimens used to treat persistent or recurring abnormal feces both for when the NHP was otherwise healthy and when the overall health or well-being of the NHP was affected. The typical treatment regimens for persistent or recurring abnormal feces in an otherwise healthy animal often involve dietary modification (63%, 20 of 32), probiotics (59%, 19 of 32), and oral electrolyte supplements (44%, 14 of 32). Other commonly used treatments included antibiotics (28%, 9 of 32), bismuth subsalicylate (22%, 7 of 32), and subcutaneous fluids (22%, 7 of 32) (Figure 3C). Loperamide was a less commonly employed treatment (9%, 3 of 32). Some responses were unsure of typical treatments or deferred to CRO practices (25%, 8 of 32).
When the NHP’s overall health or well-being is compromised, typical treatment regimens involve dietary modification including bland diet or fiber supplements (68%, 21 of 31), probiotics (61%, 19 of 31), subcutaneous fluids (52%, 16 of 31), antibiotics (48%, 15 of 31), and oral electrolyte supplements (42%, 13 of 31). Some responses indicated that animals are typically separated from cage mates (16%, 5 of 31) or given loperamide (13%, 4 of 31). Other approaches included the use of dosing holidays, a tiered approach dependent on animal observations, and pain medications if needed. Some responses (23%, 7 of 31) were unsure of the typical treatment regimen used or deferred to CRO practices.
As discussed above, dietary modification is a common first-line treatment approach to cases of persistent abnormal feces. The most common responses related to changes in diet to address persistent and/or unresolving abnormal feces included increasing dietary fiber (66%, 21 of 32) and provision of more liquid diets (25%, 8 of 32). Twenty-two percent (7 of 32) of responses managed abnormal feces by using probiotics and high protein enrichment; restricting food enrichments offered; providing bland edible enrichment food items; supplementing with electrolyte-rich fluids and incorporating an exclusion diet (write-ins).
For studies that are conducted at CROs, sponsor teams weigh in on the treatment course of action for animals with persistent and/or unresolving abnormal feces in most or all cases 61% (19 of 31) of the time, with 16% (5 of 31) getting involved only in chronic or problematic cases. The remaining responses 23% (7 of 31) indicated that treatment plans are left to the discretion of clinical veterinarians or not applicable.
Pathogen Trends and Managing NHPs with fecal issues
When looking at the responses for the number of impacted studies due to non-test article-related disease, five out of six that were impacted also reported Yersinia sp. Positive colonization and four out of seven also reported Shigella sp. Positive colonization in Mauritian NHPs. Interestingly, these bacteria may be subclinical in macaques but are reported to cause disease in stressed or immune compromised animals or may vary with serotype. 8 It would be useful to obtain more detailed information from CRO partners to interpret the impact of subclinical colonization with bacterial agents used in studies with immunologic endpoints or gastrointestinal toxicity.
Maintaining a microbiologically defined colony status or excluding Salmonella, Shigella and Yersinia sp. May be realistic with proper screening, treatment, separation, and sanitization practices.20-22 On the other hand, elimination of some enteric organisms like Campylobacter sp. From an open colony with near 100% prevalence may not be feasible. 23 In addition, agreement between CROs and pharmaceutical industry processes and procedures must be in place to ensure negative status to avoid repeat treatment of animals for clinical disease and exposure of carriers to non-carriers that may experience severe weight loss and dehydration while on study. Adding to the complexity of managing recurrent diarrhea is the reported high prevalence of antimicrobial resistance in zoonotic bacteria including Shigella and Campylobacter24-26 as well as consideration given to the limitations of laboratory testing, sensitivity and specificity. 17
Goals of research and use of animals can also drive treatment decisions of individual animals or the decision and demands to maintain a microbiologically defined colony. For example, in some Good Laboratory Practices (GLP) toxicology studies the stress of experimental procedures, compounds targeting the gastrointestinal tract or immune system can precipitate clinical disease in animals with sub-clinical Shigella flexneri colonization.20,27 In another study, immunosuppression in rhesus macaques caused a change in the normal flora of Balantidium coli leading to pathogenic effects. 28 Another study indicated that the increased incidence of Campylobacter, Shigella flexneri and Yersinia enterocolita in NHPs (rhesus macaques and cynomolgus) with chronic diarrhea compared to NHPs without diarrhea suggested that 1 or more of these bacterial agents may be the source of chronic diarrheal disease.28,29 Some enteric agents can be isolated from clinically healthy animals. Other non-infectious causes have been identified such as indoor nursery reared infants are 7.5 times more likely to have diarrhea than in dam reared infants. 30 Diarrhea percentages among nursery raised animals are 3-4 times higher than any other housing type. 31 While macaques imported to research facilities may be subclinical carriers of microorganisms with pathogenic potential, elimination of the microorganisms could avoid possible precipitation of disease state due to other stressors while on study.
Conclusion
Forty-four percent (44%, 14 of 32) of the responses reported NHP health issues (1- 4 or >4 nonclinical studies impacted) in the pre-study phase highlighting the point that NHP health issues occur relatively frequently and can have an impact on toxicology studies. This benchmarking survey identified fecal issues and/or failure to maintain body weight as the primary causes of NHP abnormal health observations. Efforts to understand root causes of and define effective treatments and preventative strategies for these clinical issues would benefit NHP health and welfare on study. Vietnam-sourced NHPs appear to have a higher incidence of health issues as compared to Cambodian or Chinese NHPs; however, this cannot be concluded based on the limited data from this survey. Though, additional monitoring of Vietnam-sourced NHPs for health issues may be warranted. Overall, the survey did not identify a clear impact of animal origin/source on NHP health issues. Regardless of animal origin, maintenance of strict SPF source colonies would likely prevent occurrence of some if not most fecal issues seen at the recipient facility. Additionally, pre-study screening practices can positively impact the percentage of healthy NHPs on study. These pre-study practices include excluding NHPs with GI-related health issues (confirmed potential enteric pathogens or diarrhea) and signs of abnormal stress behavior (alopecia) while implementing preventive health practices such as diet modification, environmental enrichment, and behavior assessments.
The survey also benchmarked industry-wide husbandry practices and common approaches used to diagnose and manage abnormal feces in NHPs. The criteria most frequently used to determine the need for veterinary intervention was either abnormal feces on 3 or more consecutive days or after a single occurrence if associated with blood in feces and/or decline in NHP overall health (e.g., lethargy, poor appetite, etc.). When diagnostic testing was employed, the most common tests included fecal flotation, fecal culture with or without sensitivity and serum chemistry. The most frequently employed treatments included dietary modification (e.g., increasing dietary fiber, bland or liquid diet), probiotics and oral electrolytes. In cases of persistent abnormal feces in otherwise healthy NHPs, subcutaneous fluids and antibiotics are often used when there was a decline in overall health. The survey was not designed to determine the response to treatment and thus no conclusion can be drawn on the superiority of one treatment over another for managing abnormal feces.
One of the primary limitations to this survey was that all respondents were from pharmaceutical companies and, depending on the question, up to 38% of the respondents did not know how to respond as they deferred to their CRO partner’s standard practices. The survey also indicated less than half of survey respondents track health screening outcomes, limiting the ability to appreciate evolving trends in NHP health. Thus, increased monitoring and tracking of health screening outcomes is highly recommended. Presumably, for those respondents that do not conduct studies in-house, they are relying on their CRO partners to track this information. This highlights the need to conduct a similar survey with CROs to more fully appreciate NHP health issues, their impact on toxicology studies, and identify best practices for preventing, diagnosing, and treating NHP health issues.
Over the past 1-2 years, the focus on NHP health issues has intensified due to the COVID-19 pandemic and its impact on the availability of NHP. Due to the global shortage of NHP and the resultant pressures on the NHP supply chain continuing, issues related to NHP health can be expected. For instance, lack of available NHPs has resulted in use of younger animals and/or including animals of mixed origin on studies in order to expeditiously advance research on therapies for critical unmet medical needs. Therefore, there is a need to continue evaluating how these changes are impacting study quality and the resultant data.
Supplemental Material
Supplemental Material - Non-Human Primate Husbandry and Impact on Non-Human Primates Health: Outcomes From an IQ DruSafe/3RS Industrial Benchmark Survey
Supplemental Material for Non-Human Primate Husbandry and Impact on Non-Human Primates Health: Outcomes From an IQ DruSafe/3RS Industrial Benchmark Survey by Smita Salian-Mehta1, Jerry Poling, Joanne Birkebak, Susanne Rensing, Chris Carosino, Rosemary Santos, Wanda West6, Khary Adams, Kathy Orsted, Denise Fillman-Holliday, and Monika Burns in International Journal of Toxicology.
Footnotes
Acknowledgments
The author(s) would like to acknowledge the contributions of the following individuals during the survey drafting and analysis process: Todd Page and Jamie Blackbourne (Eli Lilly), Amber Lange (previously employed at Novartis), Hilton Klein (Incyte), Mila Kundu (previously employed at BMS), Annick Perfontaine (Biogen) and David Shaw (Roche). Authors would also like to extend they’re thanks to Fatou Sarr (IQ Secretariat) and Alexis Myers (IQ 3R TPS LG) for all their support for anonymizing and compiling survey results and their support during review of this manuscript.
Author Contributions
Smita Salian-Mehta contributed to conception and design and contributed to acquisition, analysis, and interpretation; Jerry Poling contributed to conception and design and contributed to acquisition, analysis, and interpretation; Joanne Birkebak contributed to conception and design and contributed to acquisition, analysis, and interpretation; Susanne Rensing contributed to conception and design and contributed to analysis and interpretation; Chris Carosino contributed to conception and design and contributed to analysis and interpretation; Rosemary Santos contributed to conception and design and contributed to analysis and interpretation; Wanda West contributed to conception and design and contributed to analysis and interpretation; Khary Adams contributed to conception and design and contributed to analysis and interpretation; Kathy Orsted contributed to conception and design and contributed to analysis and interpretation; Denise Fillman-Holliday contributed to conception and design and contributed to analysis and interpretation; Monika Burns contributed to conception and design and contributed to analysis and interpretation. All authors drafted manuscript, critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This publication was developed with the support of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ, ![]() ). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
