Abstract
Aromatic L-amino acid decarboxylase deficiency is a genetic disorder of enzyme loss with decreased neurotransmitter synthesis, and it is characterized by symptoms of impaired motor development and cognitive function, hypotonia, dystonia, and oculogyric crises. Though symptomatic severity varies, the majority of patients experience severe motor impairments, including an inability to sit, stand, or walk. One approved therapy for Aromatic L-amino acid decarboxylase deficiency involves intraputaminal delivery of an adeno-associated virus packaging the human Aromatic L-amino acid decarboxylase enzyme (hAADC) cDNA. The objective of this study in monkeys was to determine the acceptability of ICV/IT as minimally invasive dosing options by evaluating hAADC biodistribution and expression following intraputaminal, intracerebroventricular (ICV), or intrathecal (IT, lumbar) administration. Results show that all routes produced comparable CSF transgene levels and were well-tolerated. The intraputaminal route yielded the highest levels of transgene-derived mRNA expression in the putamen, caudate, and globus pallidus, while expression levels in the spinal cord and dorsal root ganglia (DRG, a target of special toxicological concern) were undetectable. In contrast, the highest transgene levels in ICV/IT groups were observed in the spinal cord and DRG, but levels were too low to result in expression in the putamen, caudate, and globus pallidus. Unlike ICV/IT, the intraputaminal route produced no transgene in blood, suggesting a lower likelihood of off-target toxicities. Additionally, intraputaminal dosing resulted in the lowest anti-AAV2 antibody (anti-drug antibody) levels. Together, these data demonstrate the superiority of intraputaminal administration over ICV/IT routes in achieving AAV2-hAADC transgene DNA distribution and mRNA expression in target therapeutic areas while minimizing risk of toxicity.
Introduction
Aromatic
Pharmacological treatment options for AADC deficiency include attempts to increase the availability of the affected monoamine neurotransmitters via increased production, decreased catabolism, or the use of agonists to mimic the physiologic effects of the affected neurotransmitters. Other pharmacological treatments only treat specific symptoms of the disorder, such as autonomic dysfunction, insomnia, and nasal congestion. 2 However, patient responses to these treatments are variable and these agents can potentially give little or no benefits because they do not directly target the underlying genetic cause of the disease.1,2,5
Eladocagene exuparvovec, a targeted gene therapy consisting of adeno-associated viral vector serotype 2 containing a healthy copy of the human DDC gene (AAV2-hAADC) to drive production of the AADC enzyme, is administered directly into the putamen and has been assessed in AADC deficiency. 6 It was recently (July 2022) approved by the European Commission for use in the European Union member states, as well as Iceland, Liechtenstein, and Norway.7-12 Recently published work demonstrated that intraputaminal infusions of eladocagene exuparvovec increase AADC activity in the putamen, as visualized with 18F-DOPA positron emission tomography imaging. This gene therapy was well tolerated and resulted in significant improvements in motor development, cognitive function, body weight, and severity of symptoms including mood, temperature instability, sweating, and oculogyric crises that were sustained for >5 years following administration. 12 An independent investigation of an intraputaminal gene therapy similar to eladocagene exuparvovec similarly demonstrated improvements in motor function, cognition, dystonia, autonomic symptoms, and oculogyric crises.9,10
The putamen was chosen as the therapeutic target for this gene therapy in part owing to its role in the corticostriatal circuit involved in motor control.
8
Additionally, rates of AADC enzymatic activity are highest in the putamen under normal circumstances, as measured in homogenates of 31 distinct human brain regions taken from deceased patients with no history of neurological disorders. The caudate is the second highest site of activity, with AADC enzymatic activity 85% of that in the putamen. Notably, activity levels in the globus pallidus and substantia nigra were low, both being only 5% that of the putamen.
13
Similarly, 6-[18F]fluoro-
Clinical trials conducted in children aged ≥18 months with AADC deficiency have demonstrated the efficacy and safety of intraputaminal administration of AAV2-hAADC.7-10,12 The objective of this current study was to investigate additional administration options by comparing the transgene biodistribution and mRNA expression of single dose AAV2-hAADC delivered via intraputaminal, intracerebroventricular (ICV), and intrathecal (IT, lumbar) methods in cynomolgus monkeys, with the specific goal of determining whether ICV and/or IT administration might serve as a viable alternative to intraputaminal administration.
Materials and Methods
Ethics Statement
This study was conducted in accordance with the recommendations of the Public Health Service Policy on Humane Care and Use of Laboratory Animals from the Office of Laboratory Animal Welfare and the Guide for the Care and Use of Laboratory Animals from the National Research Council (US) Committee.
Subjects
Twelve cynomolgus monkeys, (Macaca fascicularis, 6 males and 6 females) were included in the study. Only those with body weights within ±20% of the mean body weight for each sex were selected. They were provided Lab Diet (Certified Primate Diet #5048, PMI Nutrition International Inc.) twice a day except during designated fasting periods. Enrichment foods were provided regularly, while supplemental foods were provided as warranted by clinical signs or other changes. At dosing initiation, monkeys were aged between 1 year, 4 months and 1 year, 8 months.
Each monkey was identified by a subcutaneously implanted electronic chip and permanent tattoo All were observed daily during the 18- to 20 day acclimation period for signs of disease and were given a physical examination prior to selection.
Intraputaminal, ICV, and IT (Lumbar) Dosing and Surgical Procedures
The intraputaminal dose of AAV2-hAADC was selected based on data from clinical development of AAV2-hAADC for the treatment of AADC deficiency. A human intraputaminal dose of 1.8 × 1011 vg/patient
12
was allometrically scaled to a monkey dose of 1.2 × 1010 vg/animal based on total brain weight, assuming a brain weight of 1000 g for a 1- to 2 year- old human15,16 and a brain weight of 74 g for the monkey
17
(Figure 1). Doses were increased 10-fold for intracerebroventricular (ICV) and intrathecal (IT, lumbar) routes to achieve comparable parenchymal biodistribution. This arbitrary increase in dose for ICV/IT routes was to compensate for the limited distribution of vector by these routes to deep parenchymal regions, specifically the putamen as the primary therapeutic target. Comparable exposures to the putamen across dosing routes, even if requiring a reasonably increased dose level, would give reason to further explore an alternative route. Study Design and Treatment Groups Summary. The study design and treatment groups, including dosing information, is summarized here. AAV2, adeno-associated virus type 2; ICV, intracerebroventricular; IT, lumbar intrathecal; rAAV2-hAADC, recombinant adeno-associated virus vector type 2 containing the human AADC gene.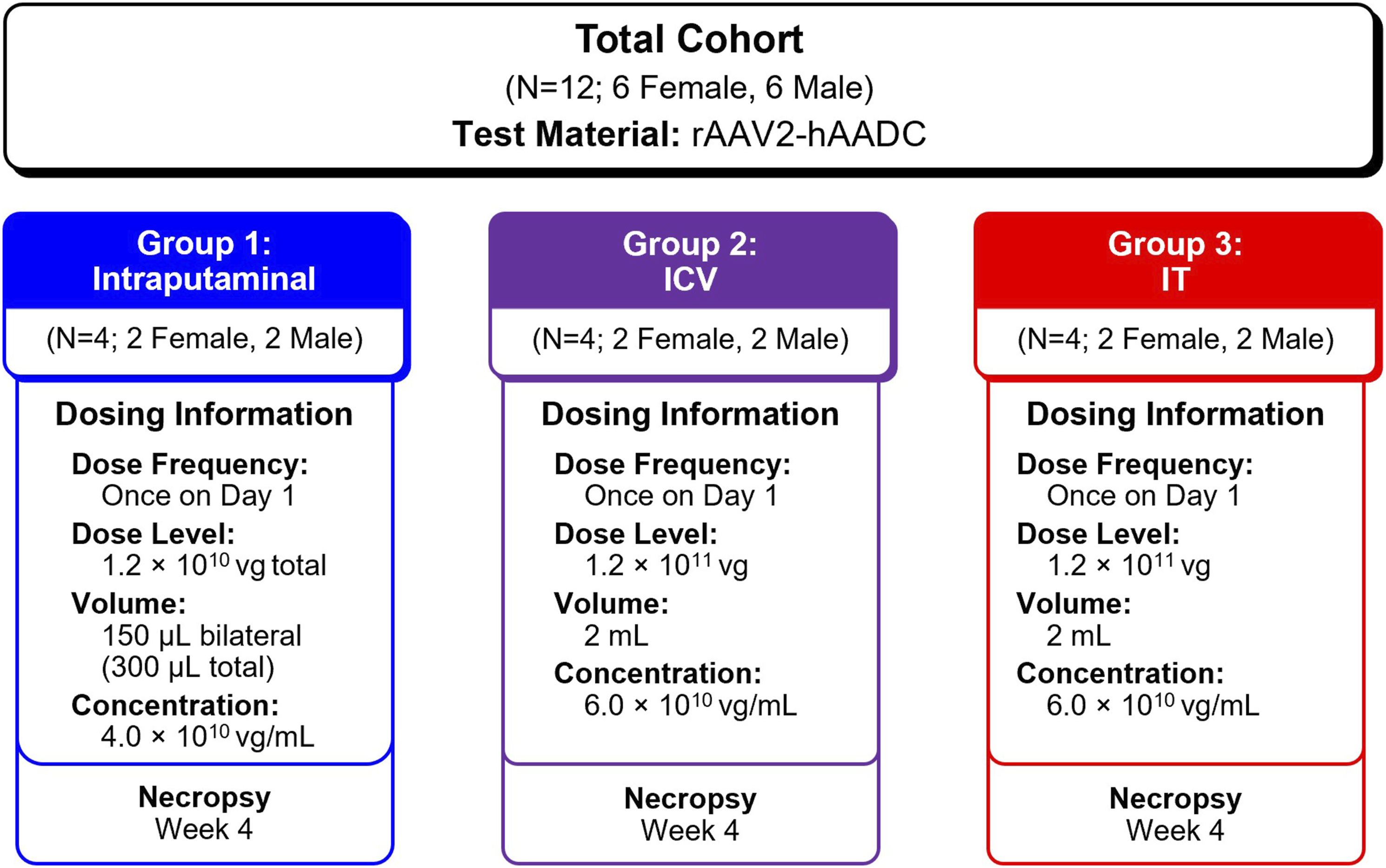
Details of the surgical procedures used to administer AAV2-hAADC via intraputaminal, ICV, and IT routes can be found in the Supplemental Materials (Supplemental Methods).
In-Life Assessments
Mortality or cage-side observations were performed at least twice daily (morning and afternoon) upon arrival and throughout the study (Supplemental Table 1). Detailed clinical observations were also conducted at predose on Day 1 and weekly throughout the study. Observations included, but were not limited to, evaluations of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs, and feet; respiratory and circulatory effects; and autonomic effects such as salivation. Nervous system effects including tremors, convulsions, reactivity to handling, and unusual behavior were noted. For observations that could not be attributed to an individual because of social housing, the observation was noted to each animal in the socialized group. All monkeys were observed for change in body weight at transfer, Day 1, and once weekly throughout the study.
Clinical Pathology
Blood samples (1.0-1.8 mL) for clinical pathology including hematology, coagulation, and chemistry were collected for all monkeys before dosing, on Day 1, and prior to necropsy. All animals were fasted for at least 8 hours prior to scheduled blood collections.
Anti-AAV2 Antibody (Anti-drug Antibody [ADA]) Analysis
Blood samples for anti-AAV2 antibody analysis were collected in a fasted state prior to dosing and prior to necropsy in vacutainer tubes with gel separator and centrifuged at controlled room temperature. The resulting serum was divided into 2 aliquots of 200 μL each. All aliquots were stored frozen at −60°C to −90°C.
ADA to AAV2 was detected using MSD plates coated with the capture antibody, AAV2 capsid-empty (Virovek). Each plate was blocked with blocking buffer, incubated, and washed prior to sample plating. Positive control anti-AAV2 A20 mouse monoclonal antibody and blood samples from monkeys were added to the appropriate wells of each coated plate. After the plate was incubated and washed, Sulfo-Tag labeled Protein A/G detection antibody was added. After an additional wash, 2X Read Buffer T was added to the wells, and the plate was read within 10 minutes of the addition of the buffer. The plate cut points for screening and titer were calculated by multiplying the applicable cut point factor by the median of the negative control wells on each plate. A cut point factor of 5.00 was used for both screening and titer analysis. The minimum dilution required was 1/100 since at this point the interfering components of the serum were appropriately diluted. Samples with titers <10.0 were considered to be negative.
Biodistribution in CSF and Blood
CSF samples (∼0.5 to 1.0 mL) were collected for determination of recombinant AAV2-hAADC transgene DNA concentrations and biodistribution at baseline, on Day 2, and at necropsy via the cisterna magna or lumbar cistern. All CSF samples were collected in cryovials and placed on wet ice. Each CSF sample was divided into 2 approximately equal aliquots and stored frozen at −60°C to −90°C.
Blood samples (∼1 mL) were collected for AAV2-hAADC biodistribution analyses of transgene DNA at baseline, on Day 2, and prior to necropsy. Blood samples were collected in tubes containing dipotassium ethylenediaminetetraacetic acid, placed on wet ice in 1 aliquot, and stored frozen at −60°C to −90°C. Monkeys were not fasted prior to CSF and serum collection unless sample collection coincided with fasting periods for surgery or clinical pathology collections.
Samples of CSF and blood were analyzed for AAV2-hAADC biodistribution via a TaqMan®-based qPCR assay. All qPCR plates for sample analysis met the acceptance criteria for assay precision, accuracy, correlation coefficient, and amplification efficiency. Each standard was run in 3 replicate 50-μL reactions in a 96-well plate. Total DNA was isolated from samples using QIAsymphony DSP DNA mini kits. DNA isolated from each sample was assayed for AAV2-hAADC in triplicate. When possible, blood DNA samples were tested at 100 ng per reaction and CSF DNA samples were tested at 5 μL per reaction. A smaller amount of sample DNA was analyzed if it was not possible to load the amounts specified. In addition, 1 random sample was spiked with 200 copies of the vector standard to evaluate for potential qPCR inhibition. If qPCR inhibition was observed, as indicated by low reaction (<110 copies of the targeted DNA) in the spiked well, the sample DNA were reanalyzed at lower amount. For CSF samples, generally speaking, results may be reported either on a per volume basis (ie, 20 μL) or per mass (eg, 1 μg) of genomic DNA (gDNA). However the extraction of CSF samples often results in very little or no genomic DNA, which is expected since normal CSF is nearly cell free. For those samples without any quantifiable amount of DNA, the reportable result is copies/well which can be scaled to a set volume but cannot be extrapolated to the total amount of DNA in the sample. Thus, for this study, the assay results were reported based on copies per 20 μL volume of CSF.
Tissue biodistribution and expression
All monkeys were euthanized on Day 28 or 29 while under sedation. Complete gross necropsy and tissue examinations were performed by qualified personnel with appropriate training and experience. For tissue collection, animals were transcardially perfused with chilled saline at 300 mL/min until they were clear of blood. Brains were removed and cut into 4-5 mm coronal sections in a brain matrix. Brain regions (prefrontal cortex, frontal cortex, caudate, putamen, nucleus accumbens, globus pallidus, amygdala, hypothalamus, hippocampus, thalamus, substantia nigra, midbrain, pons, medulla, and dentate nucleus), dorsal root ganglion (DRG; cervical, thoracic, lumbar, and sacral), and spinal cord sections (cervical, thoracic, and lumbar) were collected bilaterally with aseptic techniques and frozen for qPCR (left hemisphere) and reverse transcriptase qPCR (RT-qPCR) (right hemisphere) analyses. The remaining brain slabs were preserved in 10% neutral buffered formalin, unless otherwise indicated. RT-qPCR was used to assess transgene mRNA expression since other techniques (eg, immunohistochemistry, in situ hybridization) to directly assess the expressed AADC protein was not possible because of a lack of an AADC antibody at the time of study conduct.
AAV2-hAADC biodistribution was measured in left brain, spinal cord, and DRGs via a TaqMan®-based qPCR assay as described above. When possible, tissue DNA samples were tested at 1000 ng per reaction. The primers and probe used for the qPCR assay bind within the transgene portion of the vector and are specific for human DDC. Sequences are detailed below:
PCR Primer/Probe-hDDC-F1 Forward Primer: 5′-TTGGCTGCATCGGCTTCT-3′
PCR Primer/Probe-hDDC-R1 Reverse Primer: 5′-CGAGCCAGTCCATCATCACA-3′
PCR Primer/Probe-hDDC-P1 Probe: 5′-CATGCACAGAGCTGG-3′
Vector-derived messenger RNA (mRNA) gene expression in tissue was measured via RT-qPCR. The primers and probe (for DDC) of the RT-qPCR assay bind 5′ to the start codon of the transgene. Sequences are detailed below:
RT-PCR Primer/Probe-hDDC-F5 Forward Primer: 5′-TCCCCGTGCCAAGAGTGA-3′
RT-PCR Primer/Probe-hDDC-R5 Reverse Primer: 5′-GAAAGCATTTTTTGTGGGCCTAT-3′
RT-PCR Primer/Probe-hDDC-P5 Probe: 5′-TAAGTAACCGCCTATAGAGTC-3′
RNA was purified from tissue samples, and complementary DNA (cDNA) was synthesized from the RNA samples. RT-qPCR was performed using TaqMan-based RT-qPCR assay on 96-well plate. Each plate was run with a standard curve, 2 sets of QC samples, and study samples cDNA or RNA. RNA samples were analyzed in singlet wells, while cDNA samples were analyzed in duplicate. Tissue cDNA or RNA samples were tested at 10 ng per reaction when possible or at smaller amount if it was not possible to load the specified amount. To monitor for vector DNA contamination in the isolated RNA samples, each RNA sample was analyzed in a single well by qPCR at 10 ng of total RNA per well. The hAADC vector-derived transgene RNA expression in cynomolgus monkey samples was completed across a total of 15 plates, while an additional 7 plates were analyzed using non-reverse transcribed RNA samples to monitor for the contamination of undigested vector. All monkey samples for RT-qPCR analysis were also evaluated for possible contamination with vector DNA. In addition, RNA integrity was also monitored by analyzing for monkey housekeeping gene Hprt1 cDNA in duplicate qPCR wells.
Results
Dosing Procedures and Outcome Measurements
Summary of ADA, Biodistribution of Transgene DNA, and Transgene mRNA Expression Analyses.

ADA, anti-drug antibody; BLOD, below level of detection; DRG, dorsal root ganglion; GP, globus pallidus; ICV, intracerebroventricular; LLOQ, lower limit of quantitation.; mRNA, messenger RNA.
Anti-Drug Antibody Analysis
Summary of Anti-AAV2 Antibody Analysis Pre-dose and Prior to Necropsy in Intraputaminal, ICV, and IT Dose Groups.
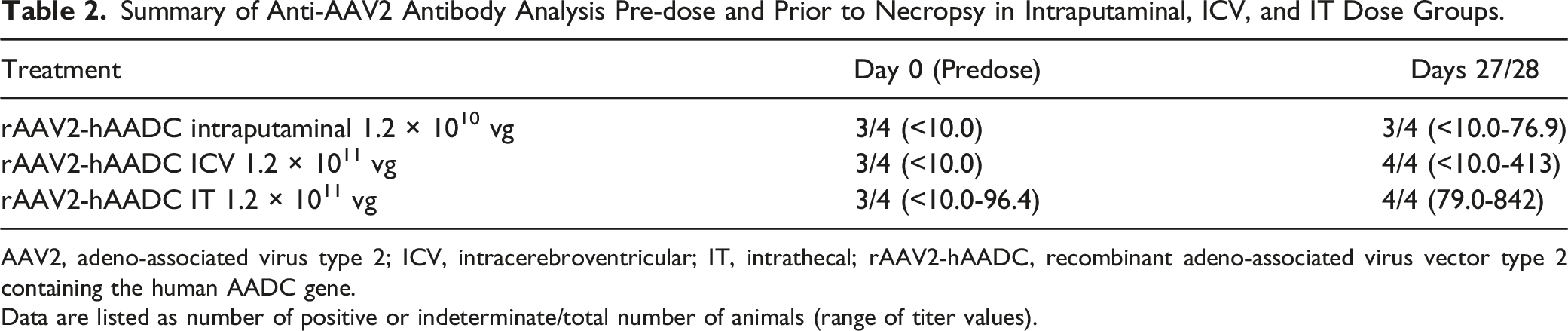
AAV2, adeno-associated virus type 2; ICV, intracerebroventricular; IT, intrathecal; rAAV2-hAADC, recombinant adeno-associated virus vector type 2 containing the human AADC gene.
Data are listed as number of positive or indeterminate/total number of animals (range of titer values).
Biodistribution Analysis
Animals in the intraputaminal dose group exhibited no transgene DNA in blood on either Day 2 or at necropsy. In contrast, animals from both ICV and IT dose groups showed detectable concentrations of transgene DNA of up to 757 and 1513 copies/μg gDNA, respectively, in blood on Day 2, which decreased to below the limit of detection (BLOD) by necropsy. All 3 routes of administration resulted in slight (<1000 copies/20 μL) to moderate (<100 000 copies/20 μL) levels of transgene DNA in CSF samples. On Day 2, concentrations of transgene DNA ranged from 314 to 33 838 copies/20 μL for the intraputaminal dose group, 29 483 to 58 560 copies/20 μL for the ICV group, and 224 to 57 531 copies/20 μL for the IT group. Based on mean values for each group, the ICV route of administration was associated with the highest concentration of transgene DNA in the CSF, followed by IT, and intraputaminal (42 293 copies/20 μL, 18 186 copies/20 μL, and 11 235 copies/20 μL, respectively).
Among the intraputaminal dose group the highest concentrations of transgene DNA were detected in most of the target therapeutic areas, defined as the putamen and caudate, plus distribution to the globus pallidus as shown in Figure 2. Mean concentration ranged from 40 to 1.1 million copies/μg gDNA in the putamen, 20 to 1 million copies/μg gDNA in the caudate, and 1000 to 220 000 copies/μg gDNA in the globus pallidus. Moderate distribution was also observed in the nucleus accumbens, along with mild distribution (<10 000 copies/μg gDNA) in the amygdala, prefrontal cortex, and substantia nigra. Interestingly, biodistribution to the globus pallidus and to a lesser extent the substantia nigra in humans has been suggested to be due to AAV2 anterograde transport.
10
The remaining tissues across all 4 monkeys, including spinal cord and DRG, yielded slight (<1000 copies/μg gDNA) or minimal (<200 copies/μg gDNA) to BLOD concentrations of transgene DNA. Though the presence of transgene DNA in the globus pallidus and substantia nigra may be related to anterograde AAV2 transport, the mechanism of biodistribution to the prefrontal cortex (∼1500 copies) is unclear, though transduction is critical to the therapeutic effect which is dependent on the corticoputaminal network.
10
Biodistribution of Transgene DNA (Copies/μg genomic DNA) in Brain, Spinal Cord, and DRG Samples Obtained from Monkeys in Intraputaminal Dose Group. Transgene DNA concentration in tissue samples from animals in the intraputaminal dose group are shown. Intraputaminal administration produced the highest mean concentrations of transgene DNA in the putamen, caudate, and globus pallidus. Values for individual monkeys are represented by blue squares; group means for each sample are indicated by black horizontal bars. DRG, dorsal root ganglion.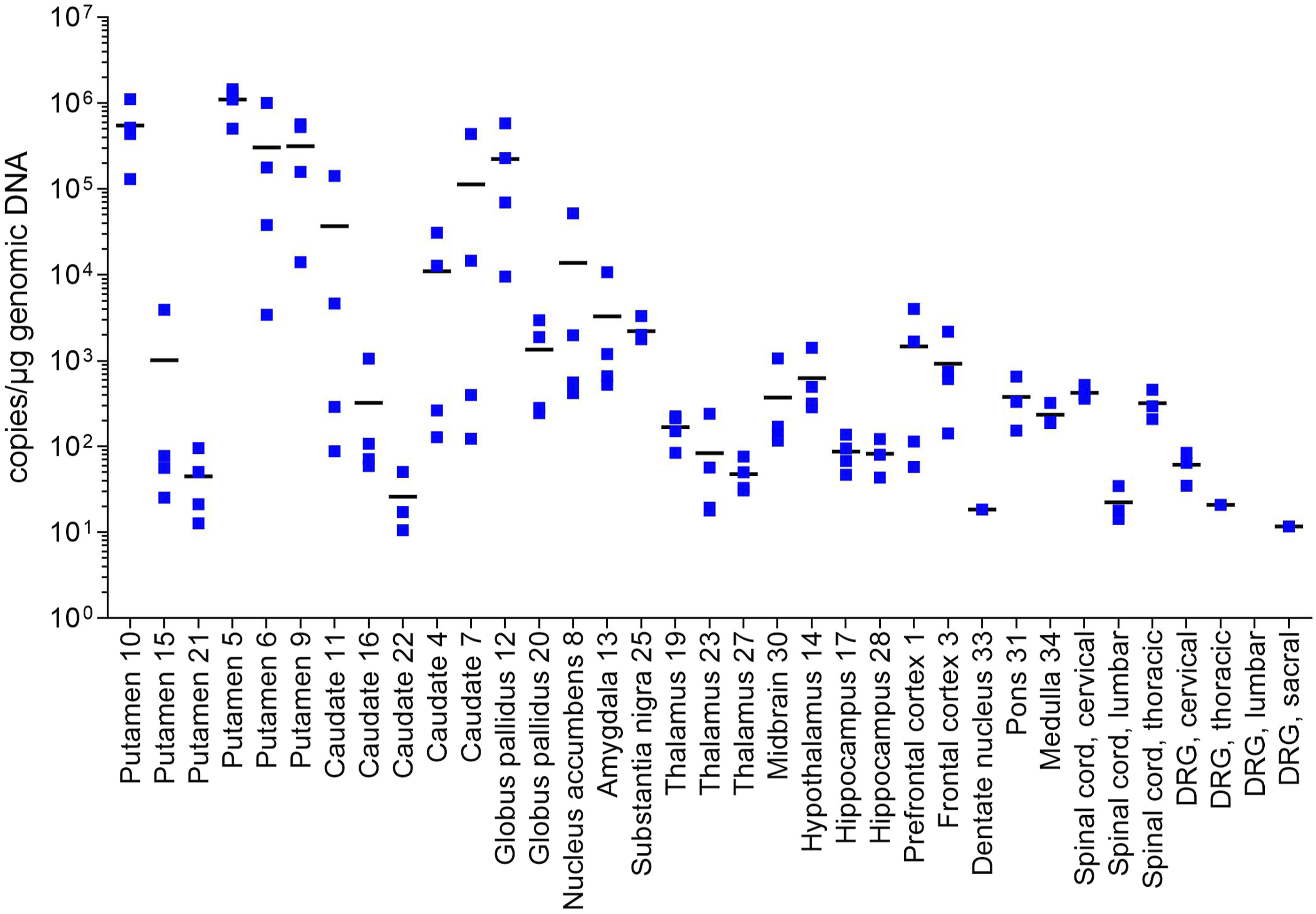
Tissues taken from the ICV dose group showed the highest brain concentration of transgene DNA (copies/μg gDNA) in the medulla (∼33 000 copies), pons (∼29 000 copies), and thalamus (∼22 000 copies) Higher concentrations of transgene DNA were detected in the spinal cord (∼36 000 to ∼143 000) and DRG (up to ∼87 000). The putamen and globus pallidus showed the lowest concentrations of transgene DNA, with mean concentrations <200 copies/μg gDNA, while the caudate was ∼3500 copies/μg gDNA, as shown in Figure 3. Biodistribution of Transgene DNA (Copies/μg genomic DNA) in Brain, Spinal Cord, and DRG Samples Obtained from Monkeys in ICV Dose Group. Transgene DNA concentration in tissue samples from animals in the ICV dose group are shown. The ICV route of administration produced the highest mean concentrations of transgene DNA in the medulla, pons, and thalamus. Values for individual monkeys are represented by purple circles; group means for each sample are indicated by black horizontal bars. DRG, dorsal root ganglion; ICV, intracerebroventricular.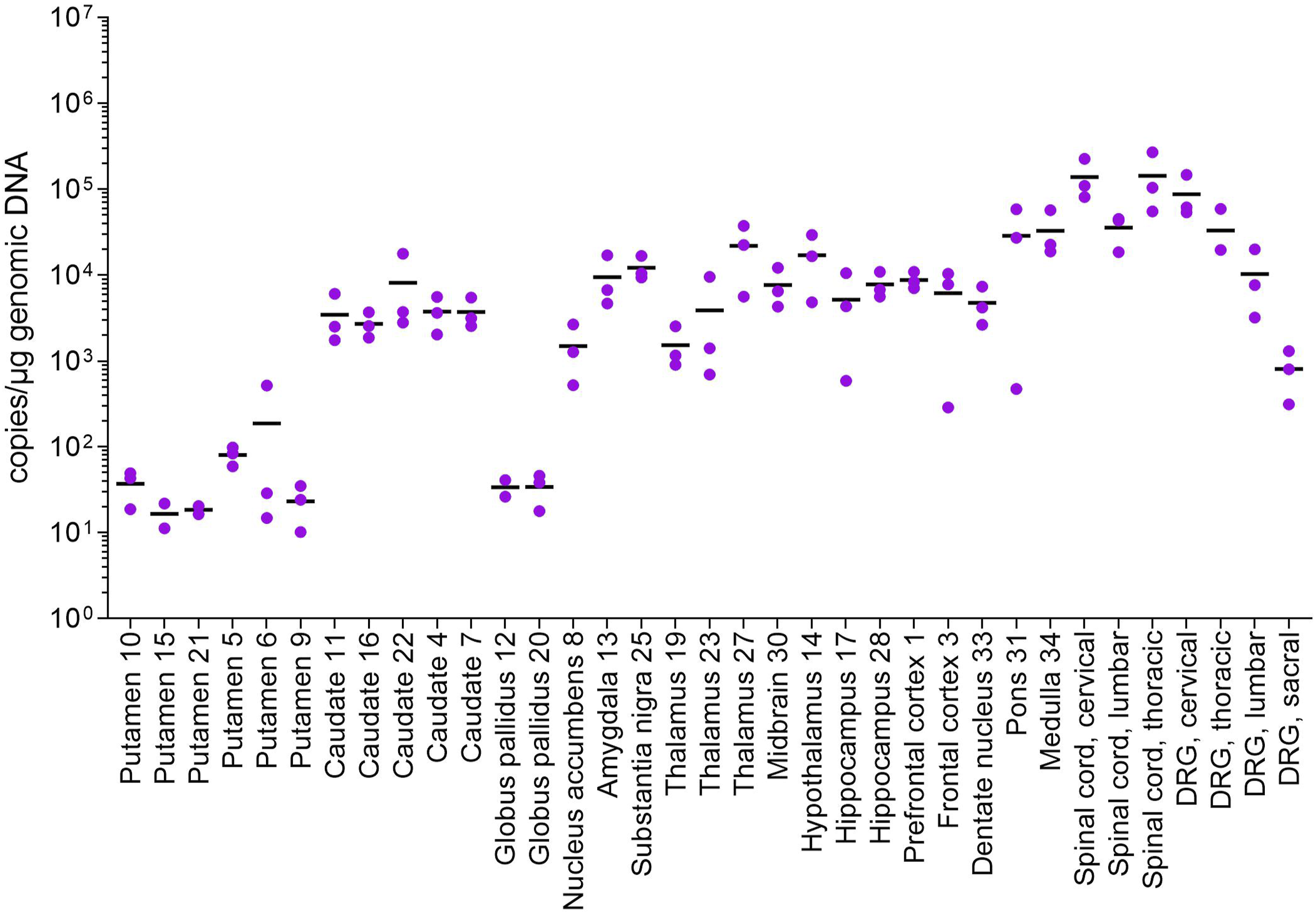
Among samples from the IT dose group, concentrations of transgene DNA were highest in the spinal cord and DRG. Transgene DNA concentration across all 3 levels of spinal cord samples ranged from >4000 to ∼230 000 copies/μg gDNA, while all levels of DRG samples ranged from 2000 to approximately 200 000 copies/μg gDNA. In contrast, concentrations of transgene DNA were lowest in the putamen, caudate, and globus pallidus as shown in Figure 4. At the highest for all samples, mean distribution in the putamen was <60 copies/μg gDNA, <5 copies/μg gDNA in the globus pallidus, and <50 copies/μg gDNA in the caudate. Biodistribution of Transgene DNA (Copies/μg genomic DNA) in Brain, Spinal Cord, and DRG Samples Obtained from Monkeys in IT (Lumbar) Dose Group. Transgene DNA concentration in tissue samples from animals in the IT dose group are shown. Transgene DNA concentrations were highest in the spinal cord and DRG. Values for individual monkeys are represented by red triangles; group means for each sample are indicated by black horizontal bars. DRG, dorsal root ganglion; IT, lumbar intrathecal.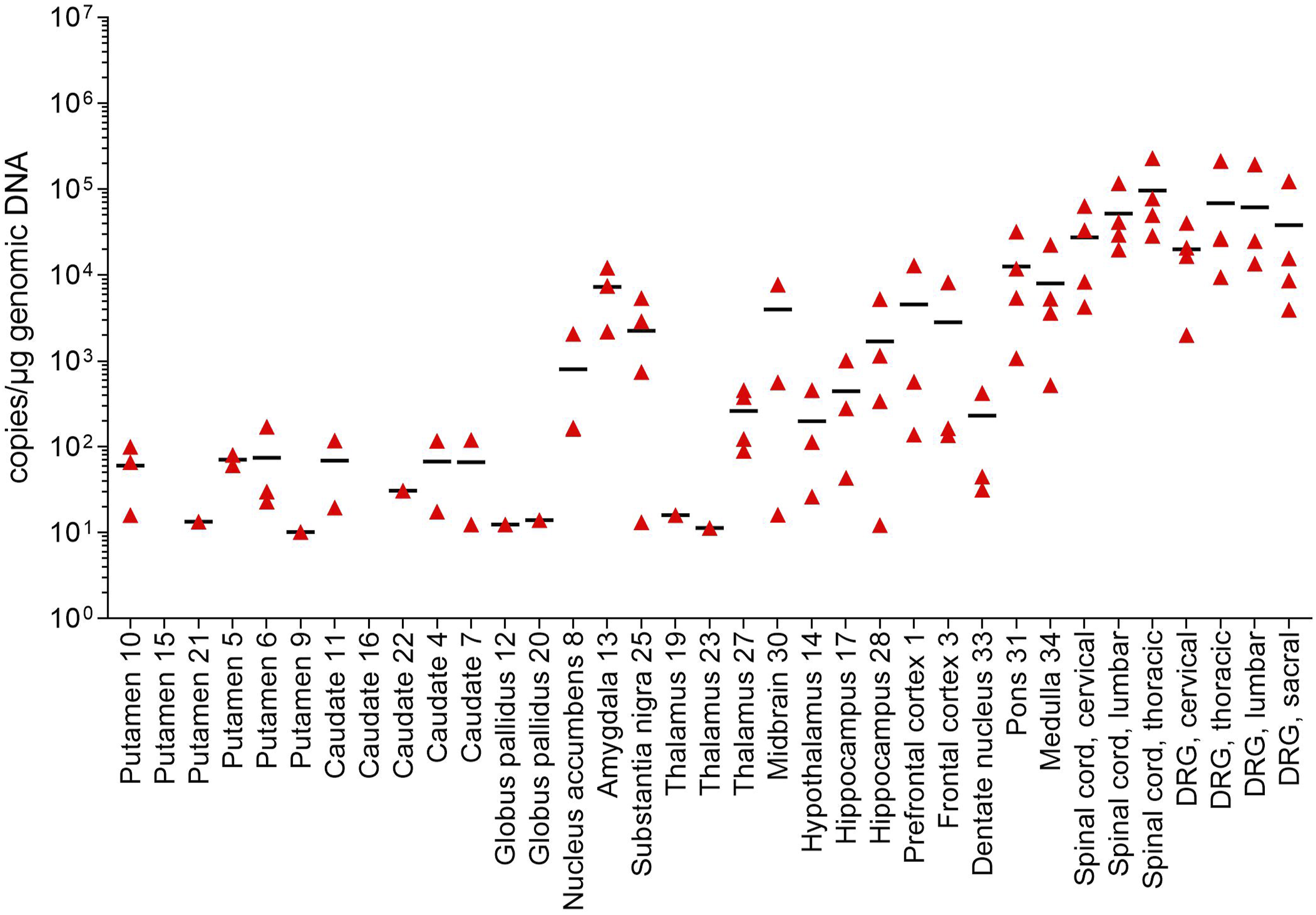
Transgene Expression Analysis
Tissues of right brain hemisphere, spinal cord, and DRGs were collected at necropsy to analyze vector-derived mRNA gene expression by reverse transcriptase quantitative polymerase chain reaction (RT-qPCR). A monkey housekeeper gene, Hprt1, was also measured to monitor overall RNA integrity.
As shown in Figure 5, animals in the intraputaminal dose group yielded the highest levels of vector-derived transgene expression in the putamen (1000 to 6000 copies/10 ng total RNA) and caudate (40 to 500 copies/10 ng total RNA), as well as globus pallidus (4000 copies/10 ng total RNA). Sporadic transgene expression was observed in the hypothalamus and amygdala, while transgene expression was BLOD in all other tissues, including the substantia nigra, spinal cord, and DRG. Transgene RNA Expression Results (Copies/10 ng total RNA) in Brain, Spinal Cord, and DRG Samples Obtained from Monkey in Intraputaminal, ICV, and IT Dose Groups. Transgene RNA expression in tissue samples are shown from animals across the 3 dose groups. Animals in the intraputaminal dose group (blue) showed the highest expression in the target therapeutic areas of the putamen and caudate, as well as the globus pallidus, with gene expression BLOD in the spinal cord and DRG. In contrast, animals in the ICV (purple) and IT (red) dose groups showed expression primarily in the midbrain, spinal cord, and DRG. Squares represent values from individual monkeys; group means for each sample are indicated by black horizontal bars. BLOD, below limit of detection; DRG, dorsal root ganglion; ICV, intracerebroventricular; IT, intrathecal.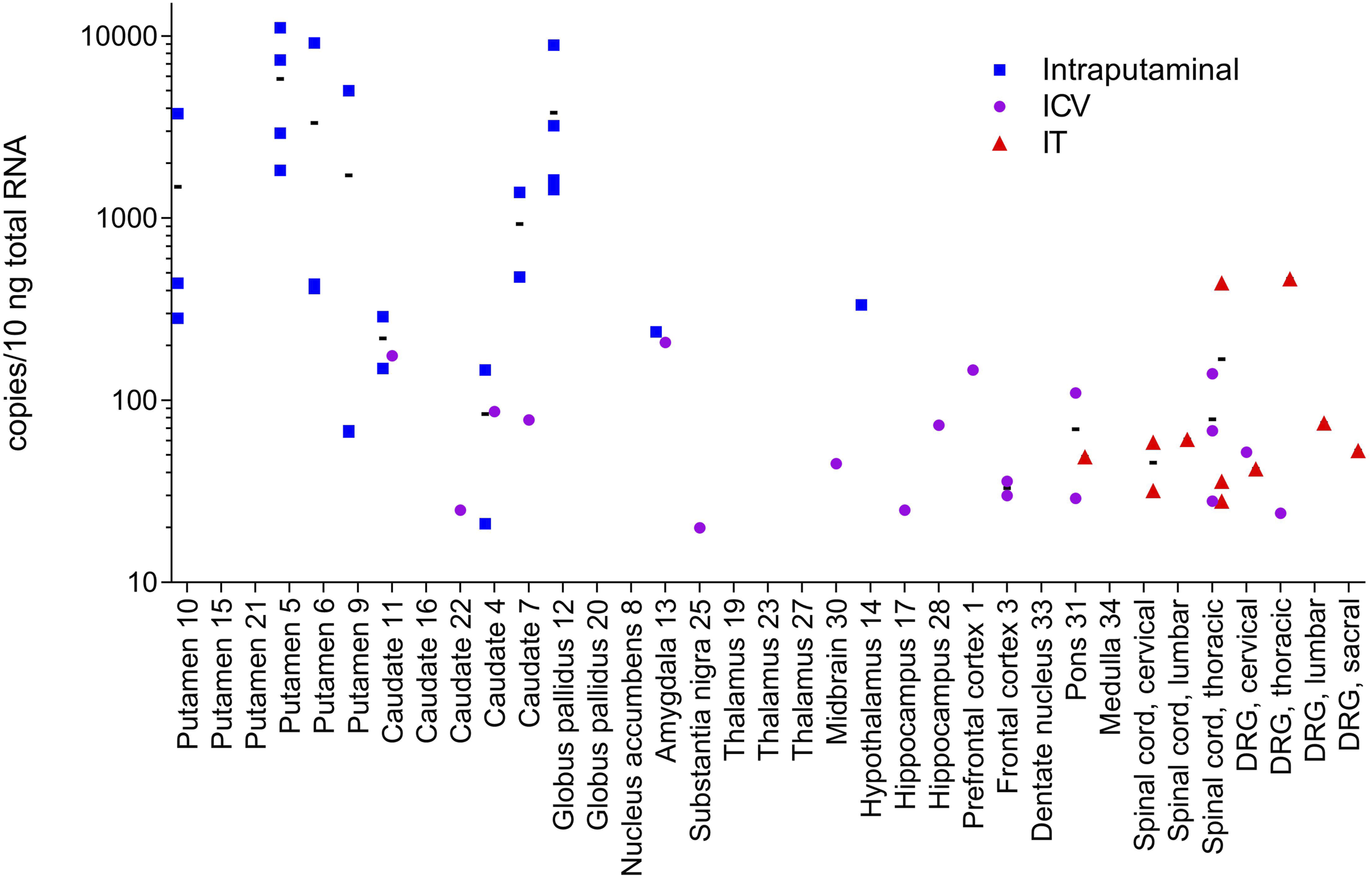
The ICV dose group showed minimal levels of transgene expression (<60 copies/10 ng total RNA), with only the thoracic spinal cord, pons, and frontal cortex yielding multiple samples with gene expression (Figure 5). Only a single sample taken from the amygdala, cervical and thoracic DRG, caudate, hippocampus, midbrain, prefrontal cortex, and substantia nigra showed positive levels of transgene expression. Samples taken from the putamen showed no gene expression, while the caudate showed no to only slight gene expression.
The IT dose group exhibited minimal transgene expression in all tissues and/or individual sample sites (<130 copies/10 ng total RNA) (Figure 5). Gene expression was observed in multiple animals in the thoracic and cervical spinal cord, while only single samples of the lumbar spinal cord, DRG, and pons demonstrated gene expression. Samples taken from the putamen and caudate, as well as the globus pallidus, were negative for gene expression.
Vector contamination was initially detected in several samples (N = 10) but following subsequent reanalysis, levels of vector DNA contamination were only confirmed in the globus pallidus and pons of two separate individual animals. Reanalysis showed that levels of vector DNA detected would account for approximately 2% of the total gene expression observed in the globus pallidus of one animal from the intraputaminal dose group, which was considered to be of no impact therefore the original result was retained. Reanalysis of the pons from the second animal, which was from the ICV dose group, yielded increased levels of vector DNA, accounting for approximately 72% of the total (110 copies) expression observed in the pons. The overall expression levels of Hprt1 were relatively consistent between tissues and between the different dose route groups. The contaminated pons sample result for the ICV dosed animal was excluded from calculated means and not represented graphically in Figure 3.
Dose-Response Evaluation
Visual inspection of results in monkeys from this study also suggested some samples, despite a high level of transgene DNA present, resulted in no expression of RNA. Thus, a graphic representation of all qPCR and RT-qPCR data, regardless of monkey or route of administration, was plotted to evaluate the relationship between vector DNA (rather than dose per se) and RNA expression (Figure 6). These data suggest that there is a vector DNA copy threshold which is necessary, but not sufficient, to result in expression. Regardless of route of administration or tissue sample analyzed, there was no RNA expression when vector DNA copies were less than 105 copies/μg gDNA. Above this ‘threshold’ there was ample evidence of robust expression. Despite this, there were numerous tissue samples for which vector DNA was >4 × 10
6
copies/μg gDNA for which there was no RNA expression. Relationship Between Level of Transgene DNA and Subsequent RNA Expression. Levels of transgene DNA (qPCR, copies/μg genomic DNA) plotted against RNA expression levels (RT-PCR, copies/10 ng total RNA) reveal a vector DNA threshold which is necessary, but not sufficient, for RNA expression (dotted line). Blue dots represent tissue samples with robust RNA expression; black dots represent tissue samples with little to no RNA expression.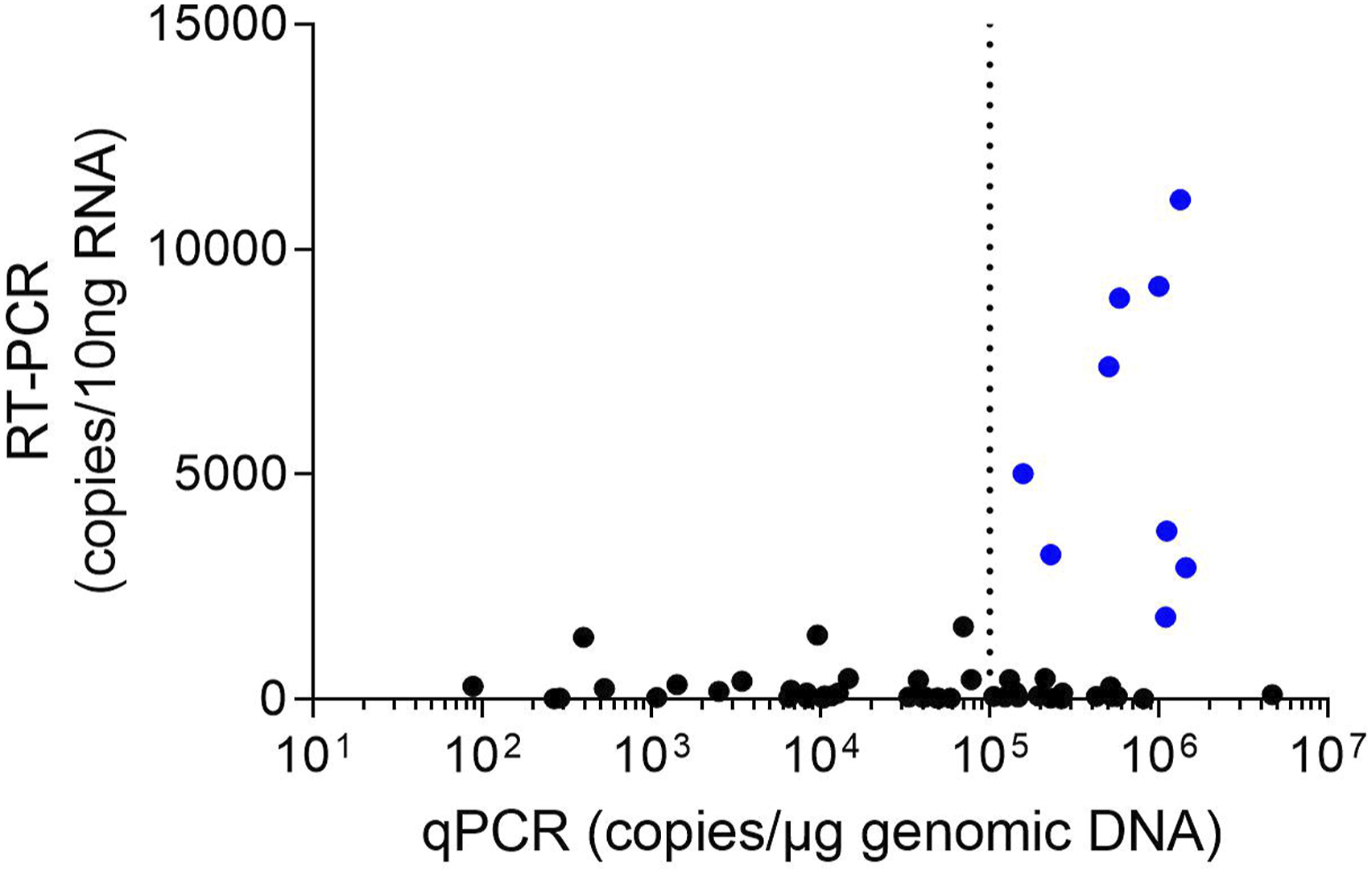
Clinical Pathology Parameters
Fluctuations among individual and mean hematological, coagulation, and clinical chemistry values were sporadic, consistent with biologic and procedure-related variation, negligible in magnitude, and were not related to administration of AAV2-hAADC. One female monkey in the ICV dose group had a moderate decrease in individual platelet count, but this change was not considered related to AAV2-hAADC administration due to a lack of correlative findings with other clinical pathology endpoints. One female in the IT dose group had minimal but progressive increases in individual alanine aminotransferase on Day −1 and Day 28, relative to value on Day −11. Due to the increase prior to dosing and the unique occurrence in this individual, this change was not considered related to AAV2-hAADC administration.
Safety and Body Weight
All animals survived until the pre-scheduled necropsy. No unexpected AAV2-hAADC–related observations were noted. Sporadic observations that were noted were deemed incidental to AAV2-hAADC and/or within normal limits for the species. The most common observations were soft or discolored feces, skin discoloration, and scabbing.
The average weight at baseline ranged from 1.93 to 2.03 kg for male and 1.75 to 2.05 kg for female monkeys. Administration of AAV2-hAADC had no effect on individual weights or group weight averages. All animals either maintained or gained weight over the course of the study.
Gross Necropsy Findings
No visible lesions were observed for any monkeys.
Discussion
In this study, it was demonstrated that administration of AAV2-hAADC via intraputaminal, ICV, or IT (lumbar) routes was well tolerated as it was not associated with any mortality, adverse clinical signs, change in clinical pathology parameters (hematology, coagulation, and clinical chemistry measurements), changes in body weight, or macroscopic observations. It is noteworthy that the doses used for ICV/IT routes were 10-times that of the intraputaminal route. This was to potentially compensate for the volume effect of administration into the CSF and allow tissue levels of transgene in target regions to attain that obtained with direct parenchymal administration. The success of this approach was substantiated based on the findings of comparable levels of transgene DNA in the CSF following all three routes of administration. However, this approach did not result in comparable levels of transgene in the brain target regions due to the differences in vector route of administration.
Intraputaminal bilateral infusions of AAV2-hAADC produced detectable levels of transgene DNA in CSF, but not in blood samples, and yielded the highest transgene DNA concentrations in the putamen and caudate, as well as the associated globus pallidus. Transgene RNA expression was also highest in these brain regions, with expression below levels of detection in DRGs and spinal tissues. Finally, intraputaminal administration yielded the lowest ADA titers at the conclusion of the study.
In contrast, AAV2-hAADC administered via the ICV route in this study yielded detectable levels of transgene DNA in both CSF and blood and produced the highest transgene DNA concentrations in spinal cord and DRG tissues. Samples with the lowest concentration of transgene DNA included the putamen and caudate. Transgene RNA expression was minimal or below levels of detection in all tissues, including potential target therapeutic areas. At the end of the study, ADA titers in the ICV dose group were 5 times higher compared to titers observed in the intraputaminal dose group. Similar to the ICV route, the IT route produced detectable levels of transgene DNA with minimal transgene RNA expression in CSF, blood, spinal cord, and DRG. There were generally no to minimal transgene DNA and transgene RNA expression in the putamen and caudate in the ICV dose group. End-of-study ADA titers following ICV dosing were 11 times higher when compared with intraputaminal dosing.
An investigation of intraputaminal administration of AAV2-hAADC in patients with AADC deficiency demonstrated substantial increases in AADC tracer uptake in the putamen, globus pallidus, and substantia nigra at 6 months and 2 years following administration. Examinations of the structural connectivity of the highly transduced putamen to cortical areas suggested that the motor improvements observed following intraputaminal AAV2-hAADC are associated with the dopaminergic restoration of putaminal areas in the prefrontal corticoputaminal network, further emphasizing the clinical importance of achieving AAV2-hAADC expression in the putamen. 10
Previous work has demonstrated a 2-log linear dose-response curve of AAV vector log dose (vg/animal) vs log expression response curve. 18 Though expression would be expected to be “low” on the initial ascending curve, once it is clearly established, further limitations to expression in a given tissue would not be expected. However, a “threshold” level has been described for an AAV2 vector with respect to dose, transduction, and RNA expression. Interestingly, this research showed that despite a sufficiently high dose and degree of transduction (sufficient to produce robust expression) in multiple samples, there were other tissue samples at the same dose and level of transduction for which there was no expression of RNA. 19 Our results demonstrate that only the intraputaminal route of administration produced detectable levels of transgene expression in the putamen. This is in agreement with the findings that vector DNA copies >105 copies/μg gDNA are necessary, but not sufficient, for detectable RNA expression, as only intraputaminal administration produced vector DNA levels above that threshold in the putamen. In addition to superior transgene expression in the putamen, the results suggest that the intraputaminal route may pose a lower safety risk compared to ICV and IT routes. CSF-based delivery of therapeutic genes is associated with vector leakage into the bloodstream, off-target tissue transduction, and subsequent DRG and liver toxicity. 6 Indeed, both ICV and IT administration resulted in detectable concentrations of transgene DNA in blood, while intraputaminal administration resulted in no detectable transgene DNA in blood. Similarly, while all three routes of administration resulted in slight to moderate levels of transgene DNA in the CSF, intraputaminal administration produced the lowest levels within that range. Additionally, ICV and IT administration both resulted in high concentrations of transgene DNA in the DRG, a target of toxicological concern. Finally, while administration of AAV2-hAADC resulted in positive immunogenicity in many (11 of 24) test samples, anti-AAV2 antibody titers following intraputaminal dosing were 9 and 11 times lower compared to ICV and IT dosing, respectively.
In summary, these results suggest that intraparenchymal delivery directly to the putamen via stereotactic infusion is the most appropriate method for administering gene therapy for the treatment of AADC deficiency. Only the intraputaminal route effectively delivers AAV2-hAADC vector to the intended therapeutic areas of the brain, demonstrates meaningful transgene expression in those areas, results in a very low ADA response, and does not distribute to DRG or blood and thereby minimizes concerns for vector leakage or shedding and off-target toxicity. 6 Given the challenges associated with the stereotactic neurosurgery required for intraparenchymal delivery in some patient populations, our results emphasize the need for advancements in stereotactic technology to facilitate timely treatment and improve clinical outcomes in patients with AADC deficiency.
Supplemental Material
Supplemental Material - AAV2-hAADC (Eladocagene Exuparvovec) Biodistribution and Expression: Superiority of Intraputaminal versus Intracerebroventricular and Intrathecal (Lumbar) Routes of Administration
Supplemental Material for AAV2-hAADC (Eladocagene Exuparvovec) Biodistribution and Expression: Superiority of Intraputaminal versus Intracerebroventricular and Intrathecal (Lumbar) Routes of Administration by David R Compton, Steven J DeMarco, Padmaja Yalamanchili in International Journal of Toxicology
Footnotes
Acknowledgments
The authors wish to thank and acknowledge Barak Gunter, PhD, for his expertise as the Study Director during the conduct of this study at Charles River Laboratories in 2020. This study was sponsored by PTC Therapeutics, Inc. Medical writing assistance and manuscript revision under the direction of the authors was provided by PRECISIONscientia, funded by PTC Therapeutics, Inc.
Author Contributions
Compton, D. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; DeMarco, S. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Yalamanchili, P. contributed to interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors DRC, SJD, and PY are employees of PTC Therapeutics, Inc. and hold shares in the company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the PTC Therapeutics.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
