Abstract
Ototoxicity and ocular toxicity screening are but two examples of specialty product lines that are often employed as Tier II or III nonclinical safety/hazard screening assessments. Compared to the regulatory guidelines that govern over standard toxicology or neurotoxicology programs, there is a paucity of regulatory strategies to address these specialized product lines. With respect to ototoxicity testing, we argue for the inclusion of the “least burdensome principles” adopted by the US FDA in providing the most pragmatic, efficient, and directed identification of potential harm to auditory function in the nonclinical safety arena. We argue for the exclusive use of the auditory brainstem response and the exclusion of the distortion product otoacoustic emissions (DPOAEs) in these Tiered II safety assessment programs. The inclusion of both are a burden on operational staff and, due to the extended episodes of anesthesia required to conduct both assays, this strategy poses a health and welfare concern for the selected animal species to be used. The DPOAE does not provide any sufficiently valid or reliable data above and beyond the gold standard ABR data, followed by complete oto-histopathology and cytocochleogram combination designs.
Introduction
There has been a concerted effort by drug and chemical control agencies of the U.S., and internationally (OECD, and the World Health Organization) to harmonize the general methods and processes involved in the development, manufacturing, and distribution of all new chemical entities to their intended targets. Based on expected human use or exposure, there may be a need to conduct specialized testing in some new chemical entities that are formulated for use through the dermal, inhalation, or intra-auricular routes-of-administration, or by other routes of exposure. These other routes-of-administration can impose scheduling or other constraints on the practical conduct of the study which may affect, for example, the timing and frequency of observations. These need to be carefully considered in the design and conduct of the study. 1
FDA COMIS Reference Table for Routes-of-Administration for Drugs Harmonized With E2B of ICH Guidelines.

A more recent route-of-administration in auditory pathway research is the direct drug administration into the inner ear by intracochlear delivery.2,3 Sampling methods have been used in the past to measure drug concentrations in the cochlear fluids, but in this novel route-of-administration the drug is injected directly into the cochlea’s scala tympani compartment. Under the current FDA coding system, this would be either an “unassigned” or “other” route of dosing the ear.
The contemporary definition for ototoxicity was generated by the U.S. and international federal, private, and academic subject matter experts of the Ototoxicity Committee of the Department of Defense Hearing Center of Excellence Pharmaceutical Interventions for Hearing Loss (PIHL) Group.4–6 Ototoxicity refers to the damage to structures and function of the auditory-vestibular system caused by exogenous agents such as pharmaceuticals, chemicals, and ionizing radiation. There are many potentially ototoxic substances. For example, depending on how ototoxicity is defined, there are 200 to 600 medications that can cause damage to hearing and/or balance. Ototoxicity encompasses cochleotoxicity, vestibulotoxicity, and neurotoxicity.6,7
Ototoxicity is primarily considered a peripheral, inner ear phenomenon; however, ototoxic agents can also affect central neural (or auditory) pathways and are then considered to be neurotoxic.8,9 Ototoxins can also cause kidney damage and associated renal dysfunction (i.e., nephrotoxicity).10–12 For regulatory review for risk/hazard assessments by the FDA, it is the ABR data that are needed for submission, along with the cytocochleogram, and a formal histopathology report by a board certified anatomic pathologist.
Contemporary industry- and governmental-standards have established a tiered safety testing pathway to marketing approval.13–15 Tier 1 testing strategies are applicable to all new chemical entities that require regulatory review and approval for marketing. 16 Tier 1 tests of the central nervous system (CNS) function employ the functional observational battery (FOB) or Irwin screen to evaluate broad neurological functions whereas in Tier 2, motor, sensory, and memory functions are assessed only if needed. Tier II testing is best described as “fit-for-purpose” testing and is conducted based on Tier I findings of conditional or systemic chemical availability. 15 Tier I findings direct subsequent Tier II testing of specific systems or processes of identified toxicity that demand further investigation. Finally, Tier III testing refers to specialized testing that is triggered by Tier II protocols and directed on a case-by-case basis. 15
In the US, the FDA has issued federal guidance documents on the background and objectives of conducting obligatory studies with all new molecular entities that target novel routes-of-administration (New Drug Application Type 1) or in the examination of toxicity when there is a change in the route-of-administration with a new formulation.
16
Under this 2015 guidance, the FDA’s thinking on studies required to be submitted for all new or modified otic dose administration formulations that may affect the auditory system, even in young children, are: 1. The dermal irritation and potential for delayed contact hypersensitivity of the new formulation should be evaluated. 2. The ability of the drug product to penetrate an intact tympanic membrane should be determined and the exposure to the middle and inner ears in an animal model should be estimated when this barrier is or is not intact. 3. If the drug product is expected to reach the middle or inner ear during clinical use or is introduced directly to those regions, evaluation of the auditory brainstem response (ABR), as well as microscopy of relevant otic tissues, including a cytocochleogram, should be included in acute and/or repeat-dose studies conducted by intratympanic administration.
16
Even reformulations that do not change the route-of-administration might have local toxic effects not previously observed (New Drug Application Type 2) because new combinations of active and inactive ingredients can produce additive or new effects. In the past, neurotoxicity was equated with neuropathy involving neuropathological lesions or neurological dysfunctions, such as seizure, paralysis or tremor. Although neuropathy is an important manifestation of neurotoxicity testing, it is now clear that there are many other signs of nervous system toxicity (e.g., loss of motor co-ordination, sensory deficits, learning and memory dysfunctions) that may not be reflected in neuropathy or other types of studies conducted using more common routes-of-administration (p. 7/15). 17 Similar to the detailed clinical observations, functional tests should also be conducted once prior to exposure and frequently thereafter in all animals selected for this purpose. The frequency of functional testing is also dependent on the study duration. In addition to the observation periods, functional observations on satellite recovery groups should also be made as close as possible to the scheduled euthanasia on the study. Functional tests should include sensory reactivity to stimuli of different modalities.
Core Battery for Ototoxicity
Neurotoxic effects in animals and humans can be assessed with a wide array of methods, including neurochemical, anatomic, physiologic, and behavioral. For example, neurochemical effects of an agent can include selective effects on synthesis, reuptake, release, or metabolism of specific neurotransmitters. Anatomic changes can include alterations of the cell body, the axon, or the myelin sheath of neurons or of the thickness of cell layers in specific brain regions. At the physiologic level, a chemical might reduce the speed of neurotransmission or change the thresholds for neural activation. Behavioral alterations can include changes in sensations of sight, hearing, or touch; alterations in simple or complex reflexes and motor functions; alterations in cognitive functions, such as learning, memory, and attention; and changes in a wide array of psychologic and social behaviors.18,19 This fact underscores the importance of using the professional judgement of the legally competent authority and other interested drug regulatory agencies (e.g., National Institutes of Health, National Institute on Drug Abuse, Center for Disease Control, etc.) together with the contractor/laboratory to determine the design of tests 1 (#20, Item 25). 1 Neurotoxicity evaluations are also required by the US EPA and the OECD guidelines for standard acute, sub-chronic, and chronic toxicity testing of all new molecular entities prior to marketing approval by these agencies. The tests must, at a minimum, include detailed clinical observations, functional testing of system, and post mortem evaluations (page 39) 1 similar to those described in standard toxicity studies (EPA, FDA; OPPTS 870.6200) but require perfusion-fixed CNS and non-CNS tissues, more frequent measurement of functional tests of the CNS, and behavioral observations to be conducted without the knowledge of treatment levels administered in the study (page 39). 1 Consistent with the basic strategy advocated by US drug and chemical evaluators for standard toxicological testing, the assessment of neurotoxic potential is most effectively carried out through a structured process of tiered testing in which chemicals are initially screened for signs of neurotoxicity as part of those toxicity studies recommended for entrance-level testing of proposed food ingredients. Chemicals identified as possible neurotoxicants become candidates for subsequent special neurotoxicity testing designed to confirm and characterize the scope of nervous system involvement and to determine dose-response characteristics, including a quantitative determination of the no-observed-adverse-effect level. 19
In contemporary toxicology screening of new drugs and chemicals, “CNS adversity” is defined as “a diminished ability to perform a function.”
20
As shown in Figure 1, above, ototoxicity of chemicals, drugs, and food additives must be identified and reported to the applicable federal agency, such as EPA or FDA. One aspect of the criteria used to identify a substance as a neurotoxicant that must be considered is the purpose of the identification. If identification is made on an interim basis to obtain further test data (Tier II or III testing) that will either confirm or refute the duration, magnitude or recovery of CNS substrates, and a broad definition of neurotoxicity seems appropriate. If the classification is intended to “kill a compound” in early development, or restrict manufacturing, a more narrow definition might be reasonable. There is a third level of testing in which previous data suggest a specific neurotoxic effect (i.e., sensory defect, cognitive loss) and subsequent experiments are performed to characterize or study the mechanism of action associated with a neurotoxicant.
21
Finally, as shown in Figure 1, additional insights into cochleotoxicity (outer hair cell (OHC), inner hair cell (IHC), stria cell deaths) and vestibulotoxicity will come from widespread ototoxicity monitoring; improved objective, data-driven measures of hearing loss (rather than blunt categories of relative hearing deficits), and widespread adoption of extended higher frequency audiometry, potentially via use of rat operant-based frequency discrimination tasks similar to those reported by Sederholm and Swedberg.
22
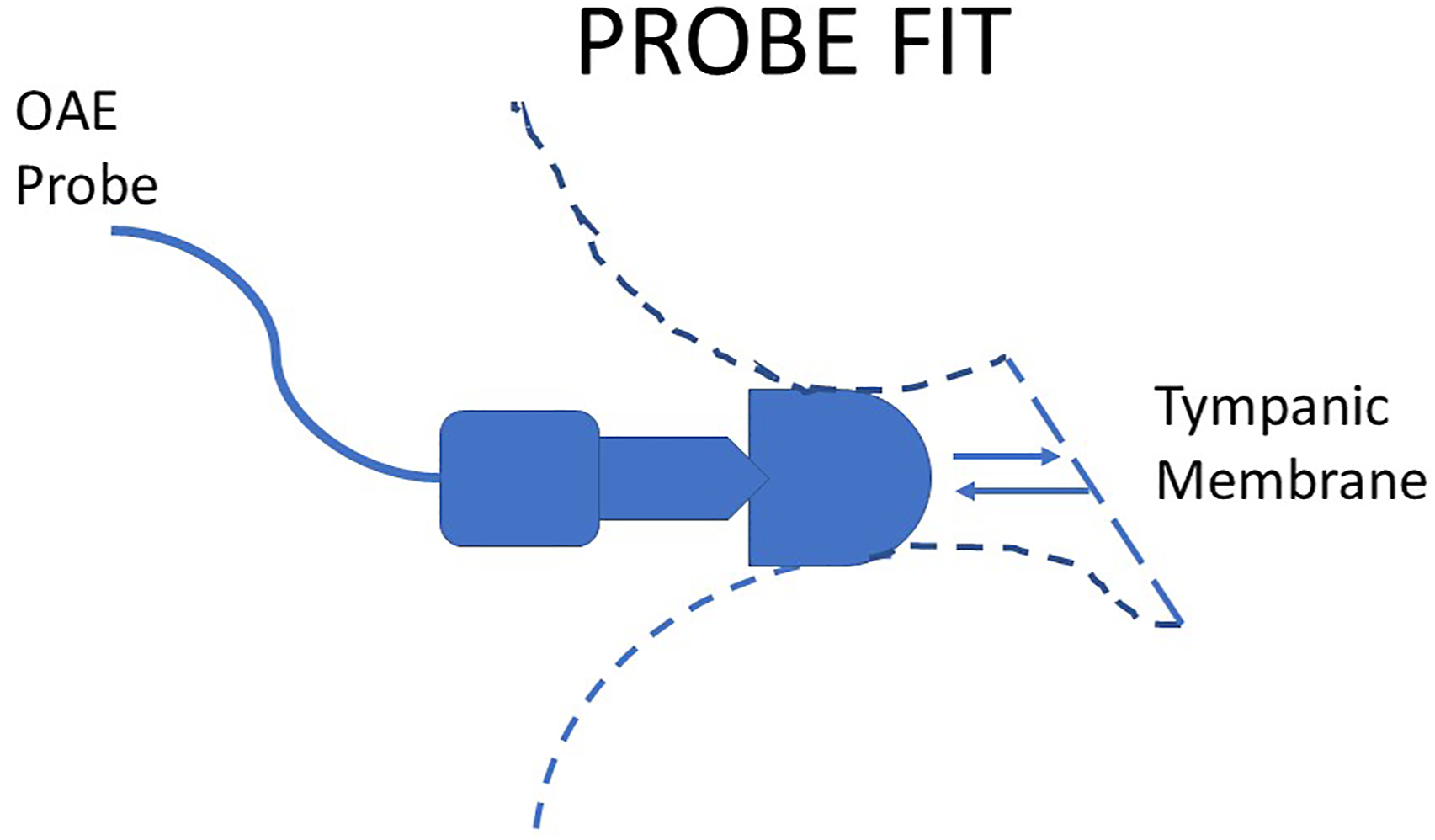
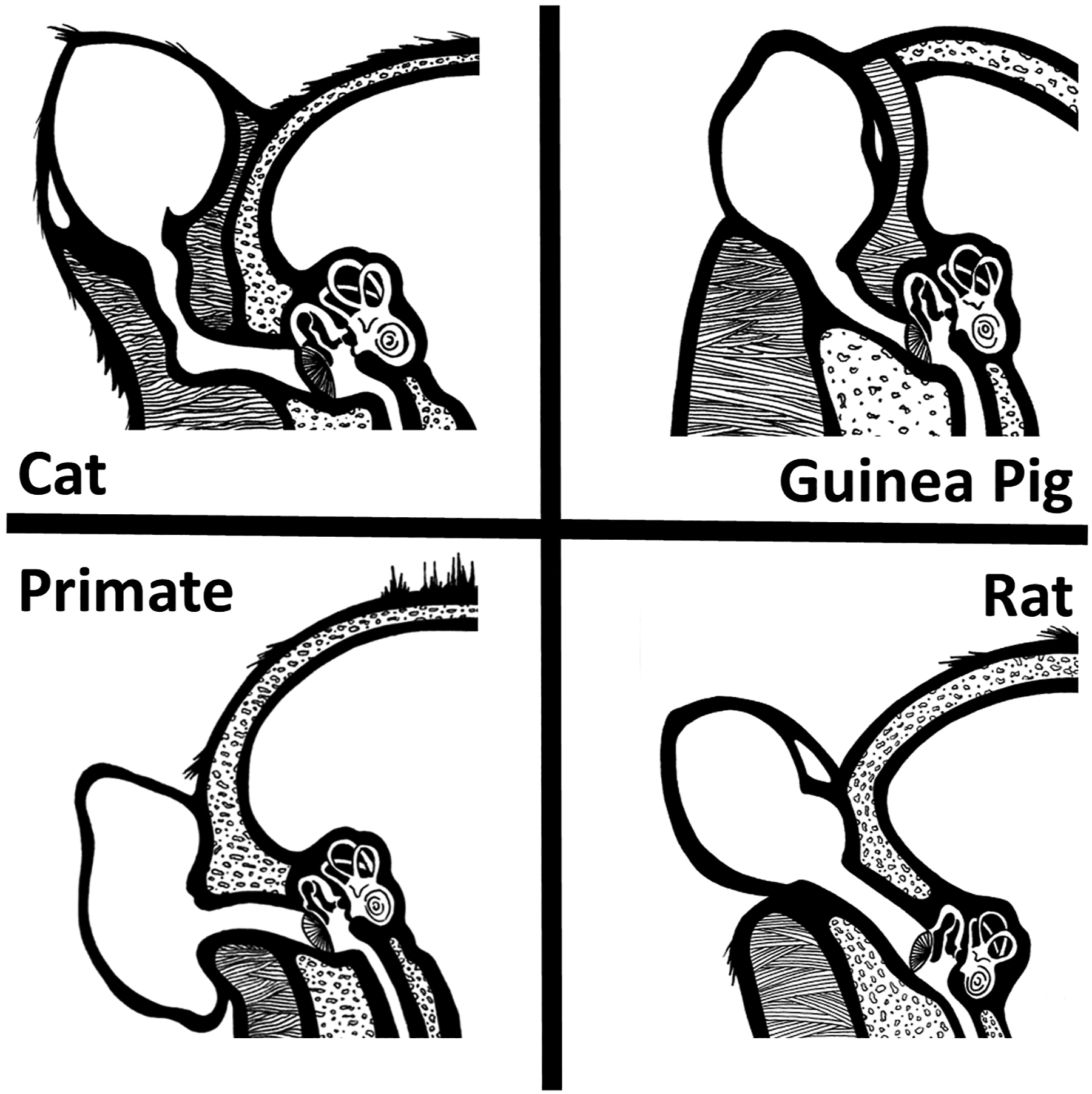
(Figures 2 and 3). Illustration of the probe fit required for a reliable (test–retest) recording of the DPOAE within the ear canal. The sides of the probe must block any external noise from interfering in the microphone recording as it is transmitted from the ear drug (tympanum). The canal must be free of debris, and the signals must be transmitted in direct line with the eardrum. Any deviation from this “ideal” recording environment are experimental confounds. Illustrations of the ear canals of the cat (upper left), guinea pig (upper right), nonhuman primate, lower left and rat (lower right) designed to show the lack of direct sound pathways from an inserted ear bud fitted into the auditory meatus (opening at the base of the pinna) and the tympanum (eardrum) which propagates the otoacoustic emissions from the inner ear to the ear canal. Only the non-human primates (NHP) and rat have a distinctive “straight shot” at the eardrum from the external ear placement of the OAE ear bud (ear plug).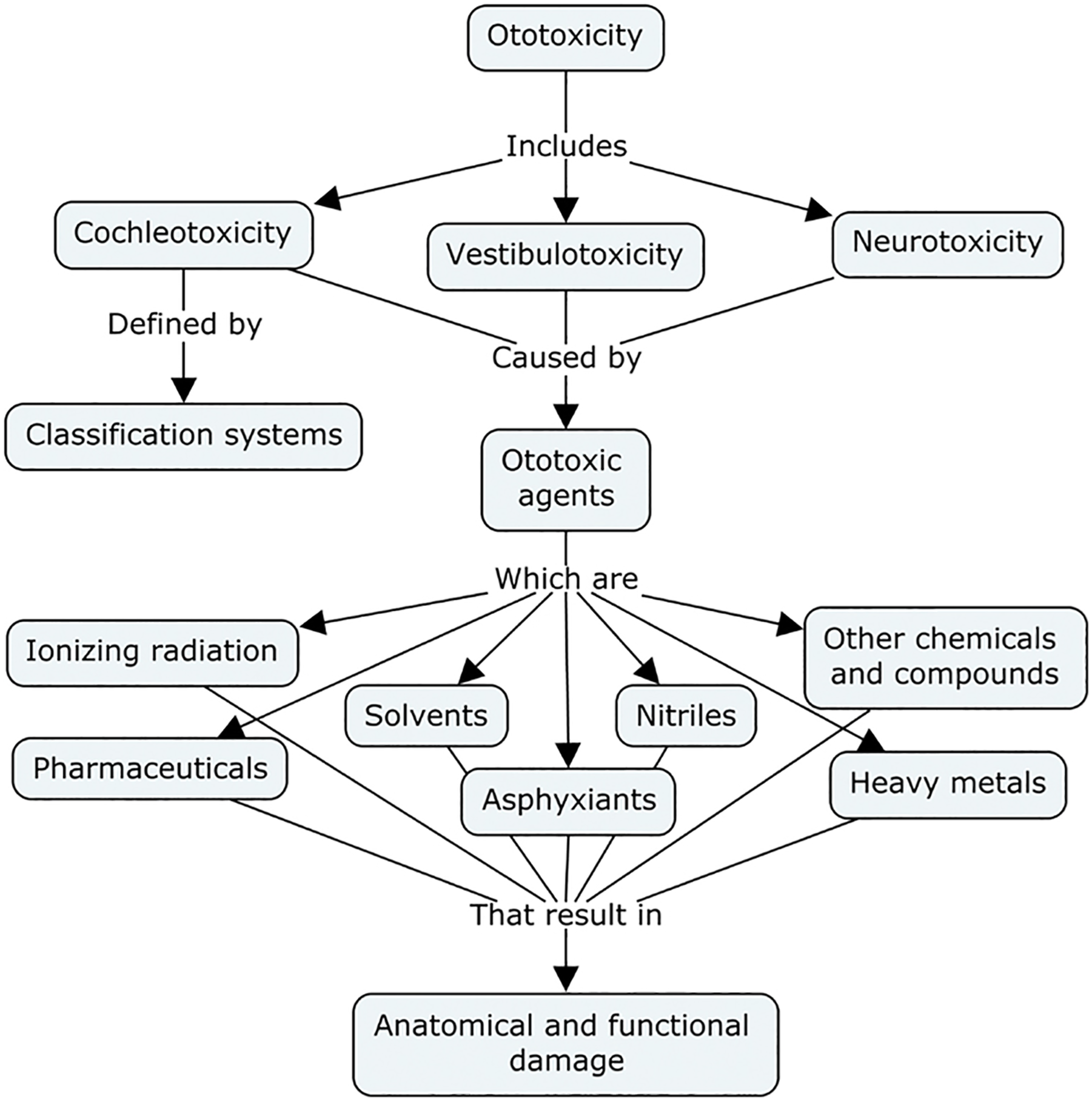
In 2018, the consensus of the Ototoxicity Working Group of Pharmaceutical Interventions for Hearing Loss 23 defined ototoxicity as damage to the inner ear, targeting cochlear and vestibular structures and sensory function, due to exposure to certain pharmaceuticals, chemicals, and/or ionizing radiation (i.e., iatrogenic hearing loss).
Electrophysiological techniques are commonly used by neurophysiologists and clinical neurologists. Thus, the experimental and clinical literature provides a large body of background information to assist interpretation of test data (for example, Thompson and Patterson 24 ). The neural basis of most electrophysiological tests is known, and many of them can be readily applied across species, including man. The latter points, facilitate their interpretation and extrapolation to man. Nevertheless, a multidisciplinary approach (e.g., including behavioral and neuropathological endpoints in the study) is recommended to facilitate a better understanding of the effects of chemicals on the nervous system.18,25
Auditory Brainstem Response (ABR)
The 2015 FDA guidance document specifically suggests the use of the ABR as the preferred electrophysiological assay to monitor the qualitative and quantitative assessments of auditory function testing. Functional testing of sensory systems include those for vision, audition, taste, olfaction, thermoregulation, somatosensation (e.g., pressure, light touch, limb position) and nociception (painful stimuli). Simple tests such as the tail flick or hot plate are used routinely to measure chemical-induced changes in nociception. More complex operant behavioral procedures have been used to determine changes in thresholds to aversive stimuli or changes in somatosensory thresholds. Some chemicals produce ototoxicity, so that procedures such as reactivity to an acoustic stimulus have been developed to assess sensory function. More complex tests such as pre-pulse modification of the acoustic startle response and operant procedures to assess auditory thresholds have also been used. Tests of olfaction and taste are available, but have not been commonly used in routine neurotoxicological testing (Item 43, page 21). 1 The auditory system transforms sound pressure waves into distinct patterns of neural activity, which are then integrated with information from other sensory systems to guide behavior, including orienting movements to acoustical stimuli and intraspecies communication.26,27 The earliest stage of central processing occurs at on the transduced electrochemical message of the cochlear hair cells that terminate in the cochlear nucleus, where the peripheral auditory information diverges into a number of parallel central pathways. Accordingly, the output of the cochlear nucleus has several targets. One of these is the superior olivary complex (SOC), the first place that information from the two ears interacts and the site of the initial parallel processing of the cues that allow us to localize sound in space. The cochlear nucleus also projects to the inferior colliculus of the midbrain (IC), a major integrative center and the first place where binaural information can interact with the motor system. The inferior colliculus is an obligatory relay for information traveling to the thalamus and cortex, where additional integrative aspects of sound that are especially germane to speech (such as sound combinations that vary over time) are processed.26,27
The 2015 guidance document specifically calls for the use of the ABR in monitoring and quantifying changes in auditory function over the course of the study plan. In the case of sensory functions of the auditory pathway, the operating status is most often assessed by the measurement of the auditory brainstem response (ABR). The ABR is an auditory evoked potential extracted from ongoing electrical activity in the brain and recorded via electrodes placed on the scalp. The measured recording is a series of six to seven vertex positive waves of which I through V are evaluated. The ABR audiometry is a neurologic test of auditory brainstem function in response to a controlled presentation of a repeated series of auditory stimuli (click, or peep). First described by Jewett and Williston in 1971, ABR audiometry is the industry standard application of auditory evoked responses. 28 ABRs do not represent hearing thresholds, but rather it measures the health and efficiency of the auditory pathways from the transduction of the sound pressure waves to electrical neuronal current by the cochlea, and to transmission through the spinal cord to the auditory cortex. The ABR is the standard test to measure changes in auditory function in standard ototoxicity programs.
The electrophysiological tracings are similar to an electroencephalogram (EEG) in that the test is shown as a series of vertex-positive waves, with waves I through V being evaluated by the examiner. These waves, labeled with roman numerals, occur in the first 10 msec after the onset of an auditory stimulus. Considering the fact that the ABR is dependent on external factors, the ABR is considered an exogenous response. To interpret the ABR, the examiner considers the wave amplitude, which indicates the grand sum response of a larger number of neurons firing; latency of the wave, which indicates the speed of transmission; interpeak latency, which shows the time between peaks; and finally the interaural latency, which shows the difference in wave latency between the two ears. The ABR is, by its nature, a test of auditory function. Other bioassays of sound signal processing were available well before the 2015 publication of the FDA guidance. As early as 1978, Kemp 29 first reported the phenomenon of the otoacoustic emissions (OAEs) recorded in the external ear canal meant to identify common causes of hearing loss in young children, namely middle ear disorders and outer hair cell dysfunction of the cochlear.29,30 In the 1980s, some authors considered the OAE responses as equivalent to the then-established ABR screening technique, however any advantages of OAEs were not always supported by actual evidence from formal peer-reviewed investigations with head-to-head comparisons between the ABR and OAE. 31
The Distortion Product Otoacoustic Emission
Recently, there has been noted interest in using an electrophysiological “snap shot” of an active cochlear process referred to as the cochlear microphonic or the summating potential to substitute for, or to combine with, the ABR to document changes in auditory function by dose administrations in drug-induced ototoxicity studies. When recorded with optimal parameters and interpreted appropriately, DPOAEs may provide important information about the integrity of the cochlea. 32 Like any hearing assessment tool, it must be used correctly to ensure appropriate evaluation and interpretation of test outcome. Otoacoustic emissions (OAEs) have become a commonly used clinical pediatric tool for assessing cochlear health status, in particular, the integrity of the cochlear amplifier or motor component of cochlear function of neonates. Predicting hearing thresholds from OAEs, however, remains a research challenge. 33
In mammals, hearing sensitivity depends on the normal function of two-distinct cell types in the cochlea. 34 Inner hair cells (IHCs) serve as the primary output of acoustic energy to the central auditory nervous system through connections with type-I afferents (>90%). In contrast, outer hair cells (OHCs) are innervated by the remaining 10% and there is no convincing evidence that OHCs transmit acoustic information to the central auditory nervous system.35,36 Some laboratories have attempted to use short-term decrements in DPOAEs to anticipate eventual permanent hearing threshold shifts. According to Ohlemiller, 37 neither DPOAEs or transient OAEs, at present, appear to indicate which subjects are most vulnerable to damage nor do they reliably identify those who are in the initial stages of permanent hearing threshold shifts.
Bramhall, McMillan and Mashburn 38 (2021) reviewed some of the most comprehensive animal data on the relationship between DPOAE levels and OHC function, including those conducted by Harding et al. 39 and Harding and Bohne 40 with respect to DPOAE-level shifts and histopathology findings in noise-exposed chinchillas. Harding et al.39,40 reported that permanent DPOAE-level shifts were associated with moderate to substantial OHC loss; however, they also found that small focal lesions with 100% loss of OHCs did not result in DPOAE-level shifts at the corresponding frequency on the tonotopic representation of the basilar membrane. Bramhall, McMillan and Mashburn 38 concluded that these data suggest that DPOAEs can be generated or supplemented by the activity of OHCs in other cochlear regions, particularly those basal to the damaged region. These data also showed that DPOAE levels are sensitive not only to OHC loss but also to the condition of the supporting cells. DPOAE levels are useful for detecting broad OHC losses of greater than 10% and large focal OHC lesions greater than 0.6 mm. Although these findings illustrate that DPOAEs generally reflect OHC integrity, they also show that there are limitations in terms of the accuracy with which DPOAEs can represent OHC damage. 38
As a valid and reliable assay for federal and international regulatory review to assess the safety and hazards of new chemicals on auditory function, the DPOAE remains controversial. Models and experimental data suggest that there are two mechanisms involved in the generation of OAEs. When two tones are presented simultaneously to an ear, a normal functioning cochlea processes the two signals nonlinearly, resulting in the creation of intermodulation distortion products. The concept of “intermodulation distortion products (IDP)” comes from the field of acoustic engineering. IDP comes from signal combinations being processed in a nonlinear information processing system. This nonlinear characteristic along the intensity continuum (loudness) is not a straight-line function but is curved, which generally implies a power law. Stevens41,42 attempted to make “sense” of stimulus processing by the CNS and in the 1950–60s generated “power laws” for visual and auditory information processing by the CNS. In simple terms, a power law is a relationship which does relate one set of values (the input voltage) to another (the output voltage) by some mathematical constant, which would yield a straight-line relationship—and incidentally a distortionless amplifier. Stevens understood stimulus response functions to be made up of both constant and a multiplication factor which is related to itself. Some of these distortion products escapes from the cochlea and can be recorded in the ear canal as a “distortion product otoacoustic emission” (DPOAEs). 43 Not all combined stimulus signals escape, just some. For a distortion product, transient, and high-level stimulus frequency emissions, the interaction of multiple sources of emissions in the cochlea leads to amplitude variation in the composite ear canal signal (refer to Lasky et al.,44,45). The middle ear has to be working efficiently to allow the vibration from the cochlea to be transmitted to the backside of the tympanic membrane. Given a healthy middle ear, the most important aspect recording of the DPOAE is the probe fitting used to create a “closed system” within that ear canal when testing. A good fitting probe ensures closure of the ear canal, excluding unwanted external sounds and increasing the sound pressure in the ear canal itself. 20 Multiple sources of emissions complicate simple correlations between audiometric test frequencies and otoacoustic emission frequencies.46,47 A clear sound-tunnel is expected but in some cases such as nonhuman primates or other animals species auditory canals provide in imperfect acoustic sound chamber.
When conducted in the optimal manner, with a clear understanding of interpretation, DPOAEs can add significantly to a human infant’s auditory assessment battery and may provide a means of detecting hearing loss in this pediatric population that cannot be tested using standard behavioral techniques. 45 An equally beneficial application of the DPOAE is in its use as a scientific tool to explore the cochlea process of transduction.32,46 However, it is questionable as to the relevance of this biometric technique in older human patients and in nonclinical animal safety assessment programs44,45 where the testing environment (the ear canal) is less than adequate.
Ear canal acoustics critically influence the stimuli used to record the OAEs. The reliability and validity of the stimulus signal in nonhuman subjects as a “risk assessment tool” for the basis of federal regulatory decisions is tenuous at best. The typical OAE probe houses one or two signal delivery tubes that are connected to an ultra-miniature speaker as well as one or more ultra-miniature microphones that actually record the collective acoustic emissions signal within that small ear canal, itself. The stimulus probe must fit into an appropriately sized ear tip (ear buds) to create an isolated acoustic chamber between the probe tip (auditory meatus) and the eardrum (tympanum). Both insertion depth and canal acoustics are critical in establishing a valid and reliable recording. In the human ear the ear canal runs straight and perpendicular to the eardrum—signal and microphone are located in the same plane of axis relative to the eardrum.
Improper orientation of the probe could lead to a distortion of the signal delivery system if one or more of the sound delivery tubes is blocked or angled toward the wall of the ear canal instead of directly focused onto the eardrum. On the other hand, a “leaky fit” ear bud could contaminate the OAE recording allowing environmental sounds into the canal or biologically produced sounds of the cochlea to leak out of the canal. In addition to the normal variations in auditory canal structures that will affect externa ear canal acoustics, such as cerumen or other debris in the ear canal will impeded accurate recording of the OAEs. In the case of the NHP, cerumen is a natural immunological response of the normally functioning ear. Simply inserting an ear bud into the ear canal can impede accurate recordings of OAEs projecting from the eardrum down the canal. In the extreme case, the cerumen can block one or more of the apertures of the sound delivery channels, thereby confounding critical delivery of the sound stimulus itself or block the microphone channels which will impede accurate data collection of microvolt signals of the OAEs. These small differences may not be important in screening deafness in a human neonate, but they become significant in applications of the DPOAEs in ototoxicity monitoring where the agreement between repeated measures is the metric of concern. 31
Factors that may influence the validity and reliability of otoacoustic emissions in purpose bred laboratory animals. 31

Ding, Allman, and Salvi 53 suggest that the major differences in the toxicity of these three platinum compounds arise from several different metal transporters that selectively regulate the influx, efflux, and sequestration of these drugs in the stria vascularis and/or hair cells (also refer to54,55). Cell death is not the only mechanism by which emissions can be distorted or modified since DPOAEs are significantly affected by middle ear pathology such as otitis media, etc.56,57
Clearly, the DPOAE is not a measure of auditory function, as required by the guidelines. Any interest in specific outer hair cell changes are already addressed by the inclusion of both histopathology of the ear structure and cytocochleogram. The complex study design described in the 2015 FDA guidance document regarding special routes-of-administration is considered a Tier III level of testing under contemporary iterative approach to safety testing. 21 This Tier III test is to be conducted during nonclinical safety assessment of new drugs that are intended for dose administrations into the external ear canal, or any other FDA categorized routes of intra-auricular administration.
Nonclinical Research vs Clinical Practice
The clinical use of DPOAEs in neonatology departments has a firm position based on their utility and safety in this “fragile” population. The data are clear that there is a correlation between repeated testing of DPOAEs and hearing loss in infants and aged human patients, but this correlation does not reflect a cause and effect relationship with auditory threshold shifts or the status of OHCs along the tonotopic map of the basilar membrane of the cochlea in these patients. As stated above, there is no single mechanism or specific target tissue characteristic of ototoxicity. The use of DPOAEs in infants is not an issue here. However, Dhar and Hall 31 have concluded that with careful recording and analysis of DPOAE functions in human patients, it might be possible to more accurately predict tone thresholds (i.e.,59,60) but problems remain in the low-frequency region when hearing loss exceeds 30 dB HL and even in cases of “rather complex analysis techniques” applied to human patients, accurate hearing status was not possible for normal and hearing-impaired persons. 61 Dhar and Hall 31 are in agreement with Shaffer et al. 33 in concluding that the complexity of the DPOAEs, specifically multiple generator sources, makes it difficult to accurately predict auditory thresholds from DPOAE data in clinical practice. With these clinical conclusions, the use of DPOAEs in nonclinical safety/hazard assessments by federal regulatory agencies is, for us, the major concern.
Otoacoustic emission and ABR recordings are attractive laboratory measurement techniques because they are reliable, do not require the cooperation of the subject (i.e., they can be recorded while sleeping or under anesthetic), and can be recorded efficiently. 45 These attributes make them especially desirable for applications in which extensive training and testing is prohibitive, as in nonclinical safety assessment programs. These two techniques provide quantitative assessments at different stages of auditory stimulus processing. DPOAE assesses the ongoing process of stimulus transduction and provides information on the quality and integrity of conversion of sound pressure waves to their electrochemical attributes within the Organ of Corti. In contrast, the ABR provides for a measure of change in the speed and quality of auditory information processing of the transduced sound stimulus signal. Therefore, DPOAE and ABR thresholds should not be expected to equate directly with behavioral auditory thresholds. In addition to methodological differences, it is not surprising that DPOAE, ABR, and behavioral thresholds differ because the three biometric tools assess different levels of information processing within the auditory system. That is, DPOAEs and ABRs provide information concerning the processing of sound pressure waves at different levels of the auditory pathway, which cannot be inferred from behavioral data alone44,45
According to Barker, Lesperance and Kileny 61 there are no universally accepted standards for DPOAE pass criteria in hearing screening. In the majority of publications dealing with this subject, a “pass” for DPOAE is defined by the DPOAE/noise floor amplitude ratio, with a range of SNRs varying from 13 dB 62 to 15 dB. 63 The ABR has the advantage of being the “gold standard” by which comparative analyses of auditory function is conducted in the industry. The ABR reflects quantitative and qualitative changes on both the auditory periphery and a segment of the central auditory pathway. Adding DPOAE and ABR assays to a FDA-regulatory standard nonclinical ototoxicity safety assessment protocol requires additional anesthesia exposure to each animal on study. If possible, the standard ABR recording session must be extended to add DPOAE measurements an additional DPOAE anesthesia schedule must be added in tandem to each scheduled ABR anesthesia schedules over the full duration of the study schedule. Either way this has the potential to pose a critical concern of unwarranted stress to the animal and a major concern for the health and welfare of each animal on study. Supplemental anesthesia is always a risk and depending on the complexity of the ABR program separate DPOAE and ABR recording sessions may be required.
Barker, Lesperance, and Kileny 61 recommend the most logical manner of inclusion of DPOAE into hearing assessment protocols would be to use it in a diagnostic capacity, in conjunction with a known hearing deficit defined by the ABR. As measure of cochleotoxicity, the DPOAE testing should supplement the ABR in a secondary protocol if, and only if, the Sponsor is interested in providing further diagnostic information about the integrity of the cochlear hair cells and the status of the middle ear using specialized histopathological staining techniques.4,5 Both OHC and IHC losses are part of the general findings that would already be available by the ancillary immunohistochemical cytocochleogram and detailed otichistopathology conducted in the stand-alone CNS neurotoxicity protocol as described in the FDA guidance document. 16
Harding et al., 39 and Harding and Bohne 40 observed that permanent DPOAE-level shifts were associated with moderate to substantial OHC loss in chinchillas. They also found that small focal lesions with 100% loss of OHCs did not result in DPOAE-level shifts at the corresponding tonotopic mapped regions on the basilar membrane. Bramhall, McMillan, and Mashburn 57 recently suggested that these data indicate that DPOAEs can be generated or supplemented by the activity of OHCs in other cochlear regions, particularly those basal to the damaged region. Harding et al. also reported that DPOAE levels are sensitive not only to OHC loss but also to the condition of the supporting cells. These authors convincingly conclude that DPOAE levels are useful for detecting broad OHC losses of greater than 10% and large focal OHC lesions greater than 0.6 mm along the basilar membrane. Although these findings illustrate that DPOAEs generally reflect OHC integrity, Bramhall, McMillan, and Mashburn 57 highlight the fact that these data also show that there are severe limitations in terms of the accuracy with which DPOAEs can represent OHC damage. According to Hopkins 64 when all data are taken into consideration, the amplitude of otoacoustic emissions is only weakly correlated with degree of outer hair cell dysfunction. 65
As currently defined in the existing US and international guidances, the most cost-effective Tier III safety assessment protocol would use a single-stage screening consisting of (1) an ABR, (2) standard otic histopathology, and (3) cytocochleograms as the “industry standard” nonclinical ototoxicity study plan. The ABR has the advantage of reflecting the functioning of both the auditory periphery and a segment of the central auditory pathway required for NDA submissions. Any interest in OHC or IHC loss is captured in the otic histopathology report required under the 2015 guidance document protocol.
Footnotes
Author Contributions
David V. Gauvin contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Joshua Yoder contributed to conception and design, contributed to acquisition and analysis and drafted manuscript; Margaret McComb contributed to design, contributed to interpretation, and critically revised manuscript; Rachel Tapp contributed to conception, contributed to analysis, and critically revised manuscript; Zachary Zimmermann contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
This manuscript was considered “work product” as a function of employment at MPI Research.
