Abstract
Respiratory monitoring, using impedance with implanted telemetry in socially housed animals, was not possible until the recent development of digital signal transmission. The objective of this study was to evaluate digital telemetry monitoring of cardiopulmonary parameters (respiratory rate, tidal volume, minute volume, electrocardiography (DII), systemic arterial blood pressure, physical activity, and body temperature) in conscious, single-housed, non-rodent species commonly used in toxicology studies following administration of positive/negative controls (saline, dexmedetomidine, morphine, amphetamine, and doxapram), and also, the effects of various social housing arrangements in untreated female and/or male cynomolgus monkeys, Beagle dogs, and Göttingen minipigs (n = 4 per species). Aggressions were observed in socially housed male minipigs, however, which prevented pair-housed assessments in this species. All tested pharmacological agents significantly altered more than one organ system, highlighting important inter-organ dependencies when analyzing functional endpoints. Stress-related physiological changes were observed with single-housing or pair-housing with a new cage mate in cynomolgus monkeys and Beagle dogs, suggesting that stable social structures are preferable to limit variability, especially around dosing. Concomitant monitoring of cardiovascular and respiratory parameters from the same animals may help reduce the number of animals (3 Rs) needed to fulfill the S7A guidelines and allows for identification of organ system functional correlations. Globally, the data support the use of social housing in non-rodents for safety pharmacology multi-organ system (heart and lungs) monitoring investigations.
Introduction
Cardiopulmonary monitoring can be used in non-rodent species such as cynomolgus monkeys (Macaca fascicularis), Beagle dogs (Canis familiaris), and Göttingen minipigs (Sus scrofa domesticus) enabling an assessment of organ system inter-dependencies.1,2 While legacy analog telemetry transmitters were restricted by cross-talk during signal transmission from various animals, digital telemeters allow for the recording of cardiovascular (electrocardiography [ECG] and hemodynamics such as arterial blood pressure [BP] and heart rate [HR]) and respiratory parameters (minute volume [MV] and its components, respiratory rate [RR], and tidal volume [TV]) as well as body temperature and physical activity data from the same animal while housed in a social group. These measures may be considered to fulfill the requirements established in the S7A guidelines 3 on safety pharmacology assessment in animals for cardiovascular and respiratory safety pharmacology. Social housing is associated with improved animal welfare and ultimately with higher quality results.4,5 While safety pharmacology monitoring is typically for 24 hours after each dose, total study duration will often exceed 6 weeks when the pre-study acclimation and inter-treatment washout are considered. In this context, enabling the conduct of safety pharmacology studies in socially housed animals is likely to have a major beneficial impact on animal welfare. The objective of this study was to evaluate MV, RR, TV, ECG, HR, and hemodynamics recorded with digital telemetry including respiratory impedance from conscious socially housed non-human primates (NHPs), Beagle dogs, and Göttingen minipigs. To that effect, animals were exposed to positive control drugs (dexmedetomidine, morphine, amphetamine, and doxapram) and controls, as well as different social structures.
Materials and Methods
Statement on Use and Care of Animals
All the procedures performed in this study were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) prior to initiation. The research facility where the study was conducted is accredited by the Canadian Council on Animal Care—Guide for the Care and Use of Laboratory Animals endorsed by the Institute of Laboratory Animal Resources and Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). 6 All procedures were conducted as per Standard Operating Procedures in place.
Animal Housing
The animal room environment was controlled (temperature 21 ± 3 C, humidity 30-70%, 12 hours light, 12 hours dark, and 10-15 air changes per h) and temperature and relative humidity were monitored continuously. A standard certified commercial primate chow (Certified Primate Diet 2055C™, Harlan Teklad, Madison, WI, USA) was available to each NHP twice daily. Beagle dogs were fed daily with 400 g of Envigo Teklad (Certified 25% Lab Dog Diet 8727C™). Minipigs were given a standard certified commercial chow (approximately 125 g of Envigo Teklad Certified Mini-swine Diet 7037C™ per day). On days of dose administration, animals were fed prior to dosing and approximately 4 hours after the last animal was dosed. Feeding was done after the end of telemetry data acquisition on the day following dosing.
Animal Preparation: Telemetry Implantation for Cardiopulmonary Monitoring
Four cynomolgus (Macaca fascicularis) monkeys (2 males and 2 females, age: 3–4 years, 3.1–5.1 kg), four (4) Beagle (canis familiaris) dogs (4 males, age: 1 –2 years, 9.3–12.4 kg), and four (4) Göttingen minipigs (Sus scrofa) (2 males and 2 females, age: 7–9 months, 9.3–24.4 kg) were surgically prepared with digital telemetry transmitters (PhysioTel™ Digital L11R, Data Science International Ltd., St-Paul, MN, USA) for cardiovascular and respiratory monitoring.
Surface ECG traces were obtained from all animals prior to surgery to ensure normal morphologies were present and the absence of arrhythmias or conduction abnormalities. For the surgical procedures, prophylactic antibiotic enrofloxacin (5 mg/kg, Baytril 50 mg/mL, Bayer, Mississauga, ON, Canada) was administered intramuscularly (IM) to animals once before surgery (at least 15 minutes before) and daily for 2 days following surgery. Prophylactic antibiotics (30 000 UI/kg, Penicillin G procaine 300 000 UI/mL, Dominion Veterinary Laboratories Ltd., Winnipeg, MB, Canada) was given IM twice daily starting at least 30 minutes prior to surgery and for at least 2 days post-surgery. Opioid buprenorphine (0.03 mg/kg, Vetergesic 0.3 mg/mL, Ceva Animal Health Inc., N. Cambridge, ON, Canada) was administered IM, bis in die (BID) for 3 days starting at least 30 minutes prior to surgery. Intravenous fluid therapy was given at least during surgery using sterile lactated Ringer’s and 5% dextrose solution at a rate of 10 mL/kg/h, or as needed. A local anesthetic (50:50, Marcaine 0.25% [Marcaine, Hospira Health care Corp., Saint-Laurent, QC, Canada] and lidocaine 2% [Zoetis Canada Inc., Kirkland, QC, Canada] was injected subcutaneously (SC) in 6-10 sites (maximum of 0.1 mL/site, maximum total volume of 0.8 mL) distributed over the surgical site. Meloxicam was administered SC (0.2 mg/kg, Metacam 5 mg/mL, Boehringer Ingelheim Ltd., Burlington, ON, Canada) just after the surgery and 0.1 mg/kg SC 1 day after the surgery.
Prior to the surgery, animals were fasted overnight and anesthetized by IV injection of propofol (6 mg/kg,propoflo-28 10 mg/mL, Zoetis Canada Inc.) for Beagle dogs; for NHPs and minipigs, ketamine hydrochloride (9.09 mg/kg, Narketan 100 mg/mL, Vétoquinol N.-A. Inc., Lavaltrie, QC, Canada) and acepromazine (0.09 mg/kg, Atravet 10 mg/mL, Boehringer Ingelheim Ltd.) IM injection was used, followed by endotracheal intubation. Ophthalmic ointment was applied to both eyes to prevent drying of the cornea shortly after initiation of anesthesia and after the end of anesthesia. The animals were placed on a heating pad with warming water system to maintain adequate body temperature. Isoflurane in oxygen was administered at a flow rate of approximately 200 mL/kg/min (approximately 2% on vaporizer). A ventilator was used to maintain the RR between 8 and 20 breaths/min with a positive ventilatory pressure never exceeding 25 cm H2O. Anesthesia monitoring included pulse oxygen saturation (SpO2) and HR.
The implantation of the transmitter and intravascular lead was performed as follows. A longitudinal incision was performed lateral but close to the linea alba. The abdominal internal oblique muscle was separated from the aponeurosis of the transverse abdominal by blunt dissection. A sterile cardiovascular transmitter PhysioTel™ Digital L11R was inserted between the abdominal internal oblique muscle and the aponeurosis of the transverse abdominal. Hemostasis was maintained using appropriate non-absorbable suture material. Before placement, the L11R transmitter was placed in sterile saline. Catheterization of the artery was performed as follows. An arteriotomy was performed, and a catheter inserted in the femoral artery. The catheter was then secured in place using polybutester or other appropriate non-absorbable suturing material. The ECG lead was placed near the jugular and the other lead was placed on the thorax near the sternum to have an ECG electrode in DII lead. To measure thoracic impedance, 4 different leads were implanted SC on the thorax, 2 on each side on the midplane between the sternum and the spine at the mid thorax level, with a 3 intercostal space interval between leads on each side. The impedance leads were identical to the standard ECG leads and were attached in a common fashion to the intercostal tissue by creating a loop from approximately 2-3 cm of exposed lead coils and with approximately 2-3 cm of distance between each electrode in a pair.
Monitoring of each signal (ECG, systemic arterial BP and impedance signal for respiration) using Ponemah v5.32 telemetry system (DSI Ltd.) confirmed the proper placement upon completion of the suturing. All sites were flushed with warm sterile saline and the incisions were closed with absorbable suture material using simple continuous sutures while the skin was closed with discontinuous buried sutures using absorbable suture material.
Rectal body temperature was monitored in the post-operative period. Once the body temperature was considered within an acceptable range and the animal was alert, the animal was returned to its cage. Nutritional support was provided to the animals (NHPs and minipigs: fruit and vegetable buffet; Beagle dogs: 156 g of canned food mixed with 244 g of regular food) for at least 3 days post-surgery. The correlation between impedance values and tracheal airflow was calibrated for each animal once after implantation using the telemetry system (DSI Ltd.) and the same calibration values were applied for each animal throughout the experiments.
Experimental Methods: Cardiopulmonary Monitoring
Following surgery, at least 3-weeks of recovery were allowed prior to positive/negative control administration and the initiation of cardiovascular and respiratory monitoring in single-housed animals. Three to six weeks after the dosing period, the effects of different social structures on cardiopulmonary parameters were investigated using 8 animals (Beagle dogs and NHPs) which were first pair-housed with a known cage mate for 1 day, then single-housed for 2 consecutive days, and finally, pair-housed with a new cage mate for 2 consecutive days:
Day 1: pair-housed with a known cage mate (at least 1 month with the known cage mate).
Days 2 and 3: single-housed.
Days 4 and 5: pair-housed with a novel cage mate.
Respiratory and cardiovascular parameters were evaluated to assess the impact of different social structures on cardiopulmonary parameters.
The impedance signal was calibrated to respiratory flow by simultaneously monitoring signal from the telemetry implant and a pneumotachometer (Pneumotach Amplifier, Hans Rudolph Inc., Shawnee, KS, USA) with a bias flow unit (Buxco large animal, DSI Ltd.). Each spontaneously breathing animal was restrained using a sling (Beagle dog and minipig) or chair (non-human primate) to acquire at least 50 high quality breaths within a 5-minute segment for the calibration. Data were acquired for a 24-hour period with 1-hour baseline minimum. Respiratory flow signals were modified using ACQ7700 Carrier signal conditioner amplifiers (DSI Ltd.). Respiratory and cardiovascular function monitoring included lung volume (via impedance), ECG, systemic arterial BP, body temperature, and physical activity. The following parameters were evaluated: MV (mL/min), RR (breaths per minute), and TV (mL) derived from the impedance signal, HR (beats per minute (bpm)) derived from the ECG, and HR (bpm) and mean systemic arterial BP (mmHg) derived from the BP signal.
Test Drugs
Drugs Tested in Conscious Telemetered NHP, Beagle Dogs, and Minipigs for Effects on Minute Volume, Respiratory Rate, Tidal Volume, and Blood Pressure.

NHP, non-human primate; PO, orally; SC, subcutaneous; Drugs doses calculated via Nair method. 7
Cardiovascular and Respiratory Data Analysis
Respiratory signal analysis was conducted using Ponemah v5.32 (DSI Ltd.), and analysis attributes were adjusted following impedance signal calibration to provide automated breath detection. Adjustments were made between the different species based on signal characteristics (e.g., TV and RR ranges) to improve detection of each breathing cycle. ECG and impedance analysis were conducted using semi-automated methods by a single reader to minimize variability using Ponemah v5.32 (DSI Ltd.). Arterial BP was analyzed using a fully automated method with Ponemah v5.32 (DSI Ltd.).
Statistical analysis was undertaken using SAS version 9.2 (SAS Institute, Cary, NC, USA). For pharmacological effects, an analysis of variance (ANOVA) for repeated measures on 15 minutes means was used as described below. Parameters were reported as 15-minute periods over 1 hour prior to dosing, every 15 minutes post dosing (up to a maximum of 24 hours). No bioanalysis of drug exposure levels was conducted for this analysis. Mean ± SEM data are presented, and a two-sided threshold alpha of 5% was applied for statistical significance with correction for multiple comparisons. For the cardiopulmonary parameter, the individual post-dose measurements were adjusted for possible differences in baseline (pre-dose) values by expressing the values as changes from pre-dose (percentage changes).
Inferential analysis was performed for the following parameters: MV, RR, TV, BP, and HR. The differences between post-treatment and baseline were statistically assessed using a repeated measures linear mixed model. 8 Significant results were reported as P ≤ 0.05, where p represents the considered probability.
To express the correlations across measurements, the Compound Symmetry and the Heterogeneous Compound Symmetry were considered as possible covariance structures in the repeated measures ANOVA. 9 Each repeated measure analysis was defined by using the treatment as repeated factor, each one of the two-covariance structures and by using the Kenward and Roger’s method when computing the denominator degrees of freedom for the test involving the fixed effects. 10
If the Hessian matrix of the model was not positive definite or computational limitation/convergence problems were encountered when fitting a covariance structure, the related model and its results were discarded. If both covariance candidate models differing by their structures converged and their final Hessian were positive definite, the one having the lowest small-sample-corrected Akaike’s Information Criterion was selected. 11
For the different drugs, if there was a significant treatment effect, each test item dose level results were compared to the control using Dunnett tests on least-squares means.
For the social structures (difference between each day), if there was a significant housing effect, the single-housed and pair-housed results were compared using a t-test or an F-test. When the fixed effect includes only 2 levels of classification, the F-test is equivalent to a t-test and could be reported without distinction.
Results
Respiratory Monitoring: Pharmacological Effects
Dexmedetomidine
Dexmedetomidine decreased MV in NHPs from 0.50 to 3.25 hours reaching statistical significance at 0.75 hours after treatment compared to control (P < 0.05, Figure 1A). No statistically significant change in RR was observed compared to the control (Figure 1B) but a non-statistically significant trend toward moderately lower values was observed from approximately 0.50 to 3.5 hours post-dose. A similar effect on MV was observed in Beagle dogs, with a significant reduction at 0.50 to 0.75 hours post-dose compared to control (P < 0.05, Figure 2A). Furthermore, a significant decrease in RR was observed in Beagle dogs following dexmedetomidine treatment at 0.25 to 0.50 hours compared to control (P < 0.01, Figure 2B). Dexmedetomidine administration to Göttingen minipigs was not associated with significant changes to MV and RR but a trend toward lower MV values was observed between 1.75 and 2.50 hours post-dose (Figures 3A and 3B). Time course for the effect of different drugs on the (A) minute volume (MV) and (B) respiratory rate (RR), being measured with PhysioTel Digital L11R telemetry implants, over 6 hours in telemetered non-human primates (n = 4). ⍰ = control, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ =amphetamine (0.5 mg/kg PO), ♦ = doxapram (3.79 mg/kg SC). SC, subcutaneously; PO, orally. Time course for the effect of different drugs on the (A) minute volume (MV) and (B) respiratory rate (RR), being measured with PhysioTel Digital L11R telemetry implants, over 6 hours in telemetered Beagle dogs (n = 4). ⍰ = control, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ = amphetamine (2.0 mg/kg PO), ♦ = doxapram (10.0 mg/kg SC). SC, subcutaneously; PO, orally. Time course for the effect of different drugs on the (A) minute volume (MV) and (B) respiratory rate (RR), being measured with PhysioTel Digital L11R telemetry implants, over 6 hours in telemetered Göttingen minipigs (n = 4). ⍰ = control□, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ = amphetamine (3.0 mg/kg PO), ♦ = doxapram (10.0 mg/kg SC). SC, subcutaneously; PO, orally.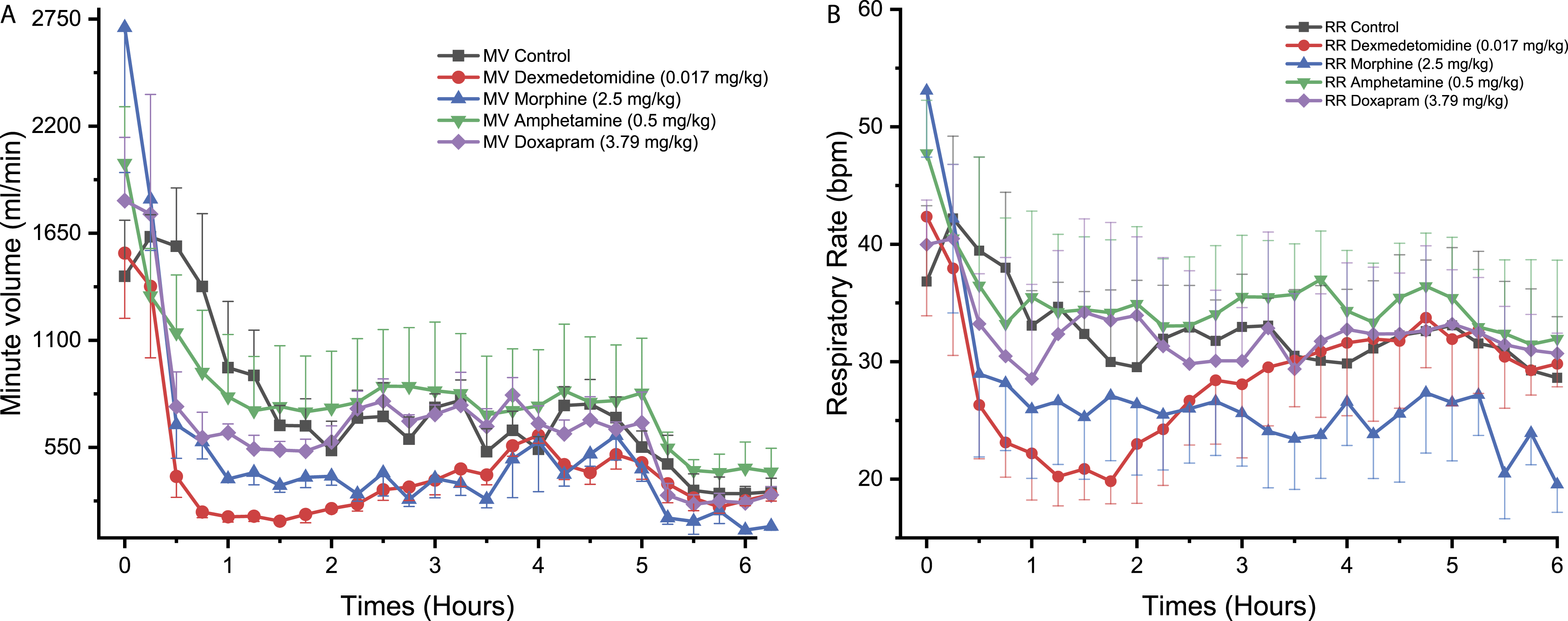
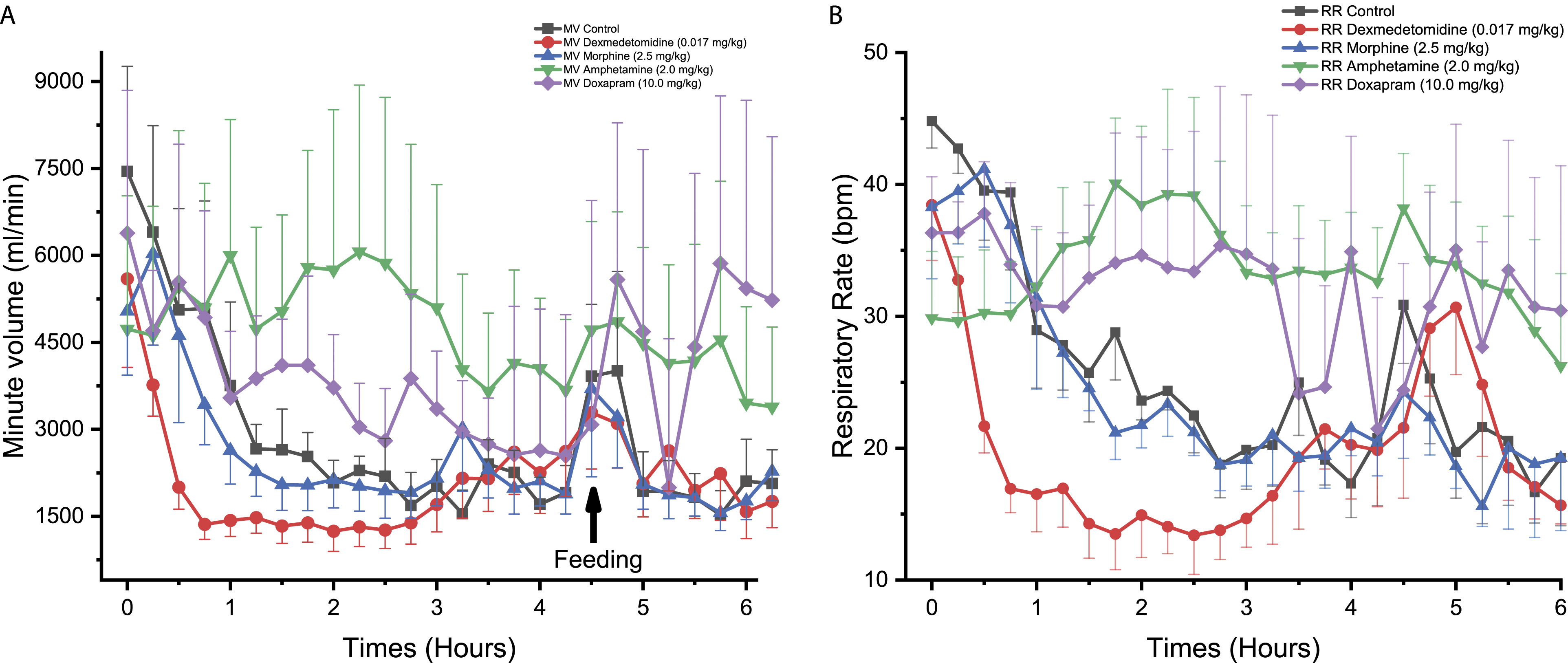
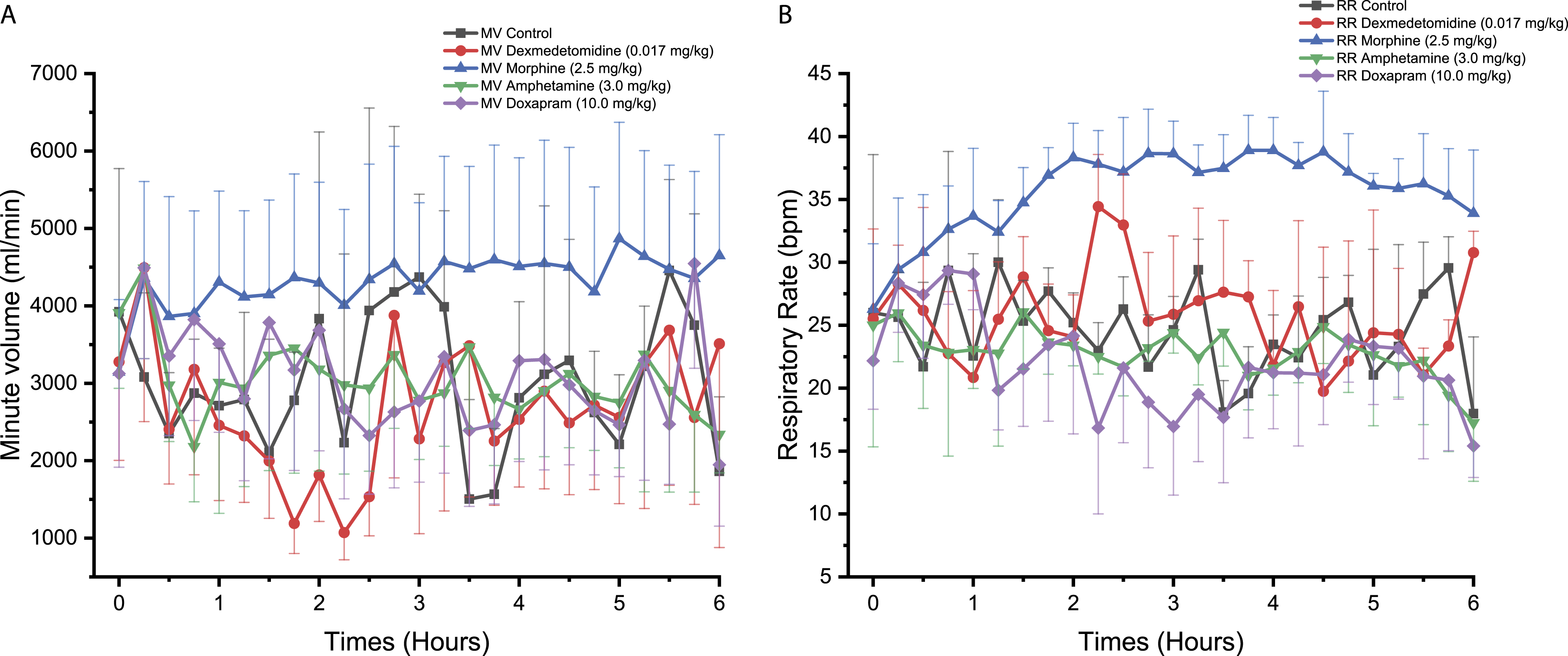
Morphine
Morphine administration to NHPs significantly reduced MV reaching statistical significance at one time-point, that is, 5.50 hours (P < 0.05, Figure 1A). No statistically significant effect in RR was observed (Figure 1B). Morphine was not associated with significant effects on MV and RR in Beagle dogs (Figures 2A and 2B). In Göttingen minipigs, morphine significantly increased MV at 3.75 hours (P < 0.05, Figure 3A). Morphine also increased RR with changes reaching statistical significance at 1.00 hour (P < 0.05), 3.00 hours (P < 0.05), 3.75 hours (P < 0.01), 4.00 hours (P < 0.001), 4.50 to 4.75 hours (P < 0.05), 5.00 hours (P < 0.01), and 5.50 to 5.75 hours (P < 0.05) compared to control (Figure 3B).
Amphetamine
In NHPs, amphetamine was not associated with any significant effect on MV and RR (Figures 1A and 1B). In Beagle dogs, MV was significantly increased following amphetamine administration reaching statistical significance at 2.00 hours (P < 0.05), 2.50 to 3.25 hours (P < 0.01), and 4.00 hours (P < 0.05) compared to control (Figure 2A). In the same species, RR was also significantly increased at 2.75 to 3.00 hours (P < 0.05, Figure 2B). In Göttingen minipigs, no significant change in MV and RR was observed (Figures 3A and 3B).
Doxapram
No significant effect on MV or RR was observed in NHPs following doxapram administration (Figures 1A and 1B). In Beagle dogs, doxapram caused an increase in MV reaching statistical significance at 3.25 hours (P < 0.01, Figure 2A). An increase in RR was also noticed but the effects did not attain statistical significance which was likely due to the individual variability that was observed (Figure 2B). No significant difference in MV or RR was observed for Göttingen minipigs following administration of doxapram (Figures 3A and 3B).
Cardiovascular Monitoring: Pharmacological Effects
Dexmedetomidine
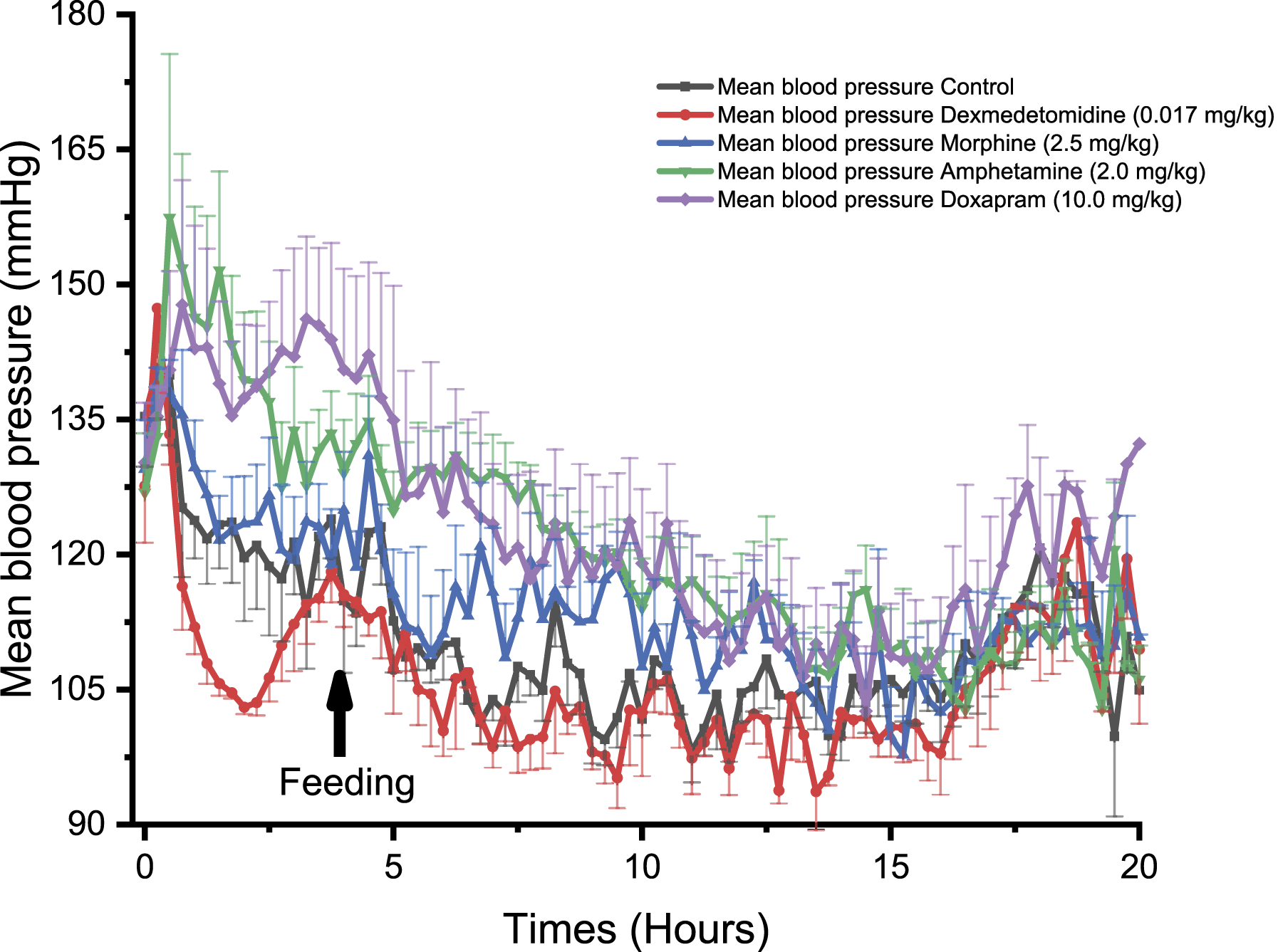
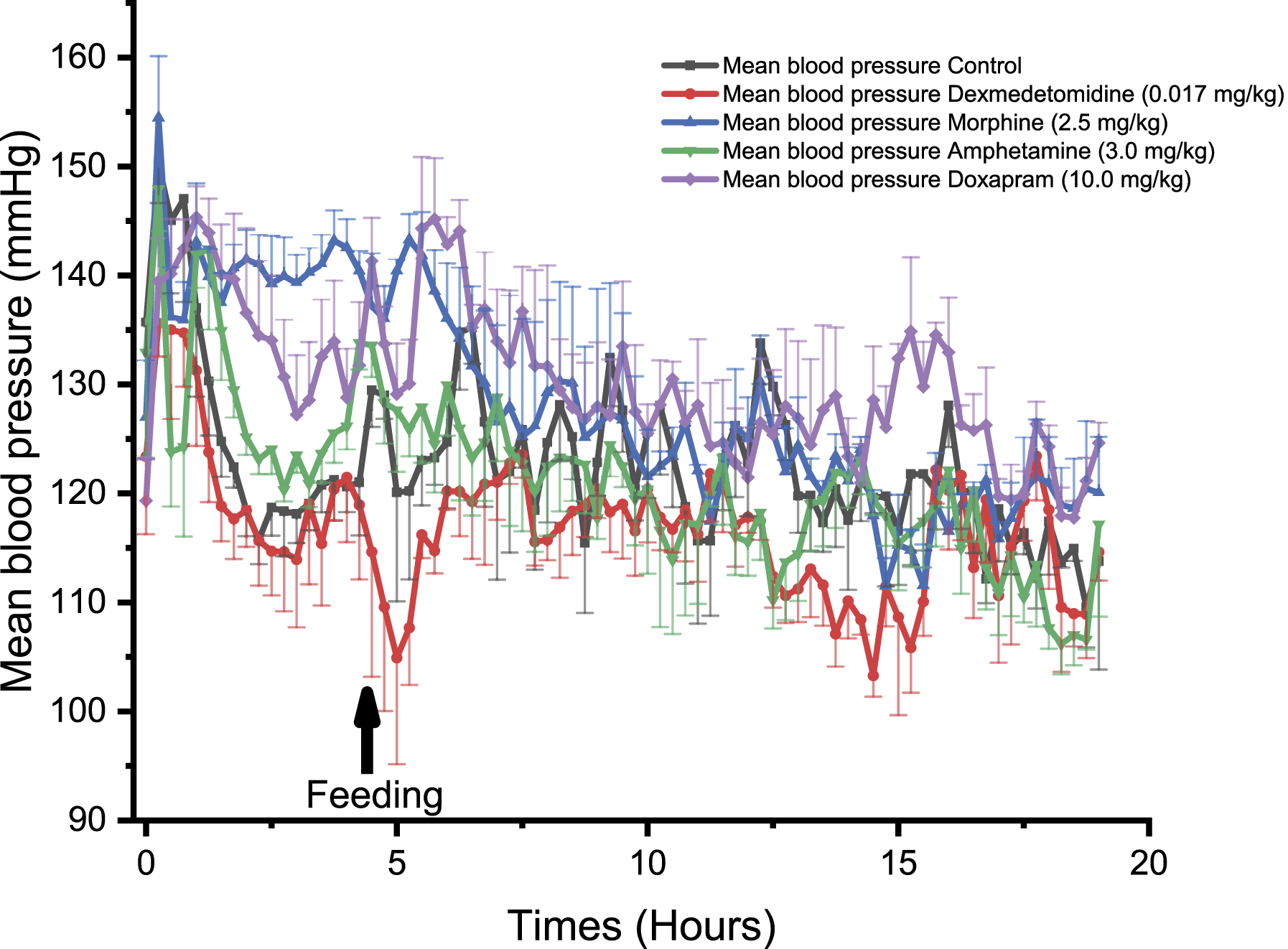
In NHPs, dexmedetomidine induced a significant decrease in BP at 0.50, 1.00, and 1.25 hours compared to control (P < 0.05, Figure 4). Similar effects were observed in Beagle dogs with a significant decrease in BP at 1.75 hours following treatment (P < 0.05, Figure 5). Dexmedetomidine treatment induced a significant decrease in BP in Göttingen minipigs at 5.25 hours (P < 0.05), 6.50 hours (P < 0.05) and 14.25 to 14.50 hours, compared to control (P < 0.05, Figure 6). Comparison of the effect of different drugs on the systemic mean arterial blood pressure (BP), being measured with PhysioTel Digital L11R telemetry implants, over 20 hours in telemetered non-human primates (n = 4). ⍰ = control, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ = amphetamine (0.5 mg/kg PO), ♦ = doxapram (3.79 mg/kg SC). SC, subcutaneously; PO, orally. Comparison of the effect of different drugs on the systemic mean arterial blood pressure, being measured with PhysioTel Digital L11R telemetry implants, over 20 hours in telemetered Beagle dogs (n = 4). ⍰ = control, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ = amphetamine (2.0 mg/kg PO), ♦ = doxapram (10.0 mg/kg SC). SC, subcutaneously; PO, orally. Comparison of the effect of different drugs on the systemic mean arterial blood pressure, being measured with PhysioTel Digital L11R telemetry implant, over 20 hours in telemetered minipigs (n = 4). ⍰ = control, ● = dexmedetomidine (0.017 mg/kg SC), ▲ = morphine (2.5 mg/kg SC), ▼ = amphetamine (3.0 mg/kg PO), ♦ = doxapram (10.0 mg/kg SC). SC, subcutaneously; PO, orally.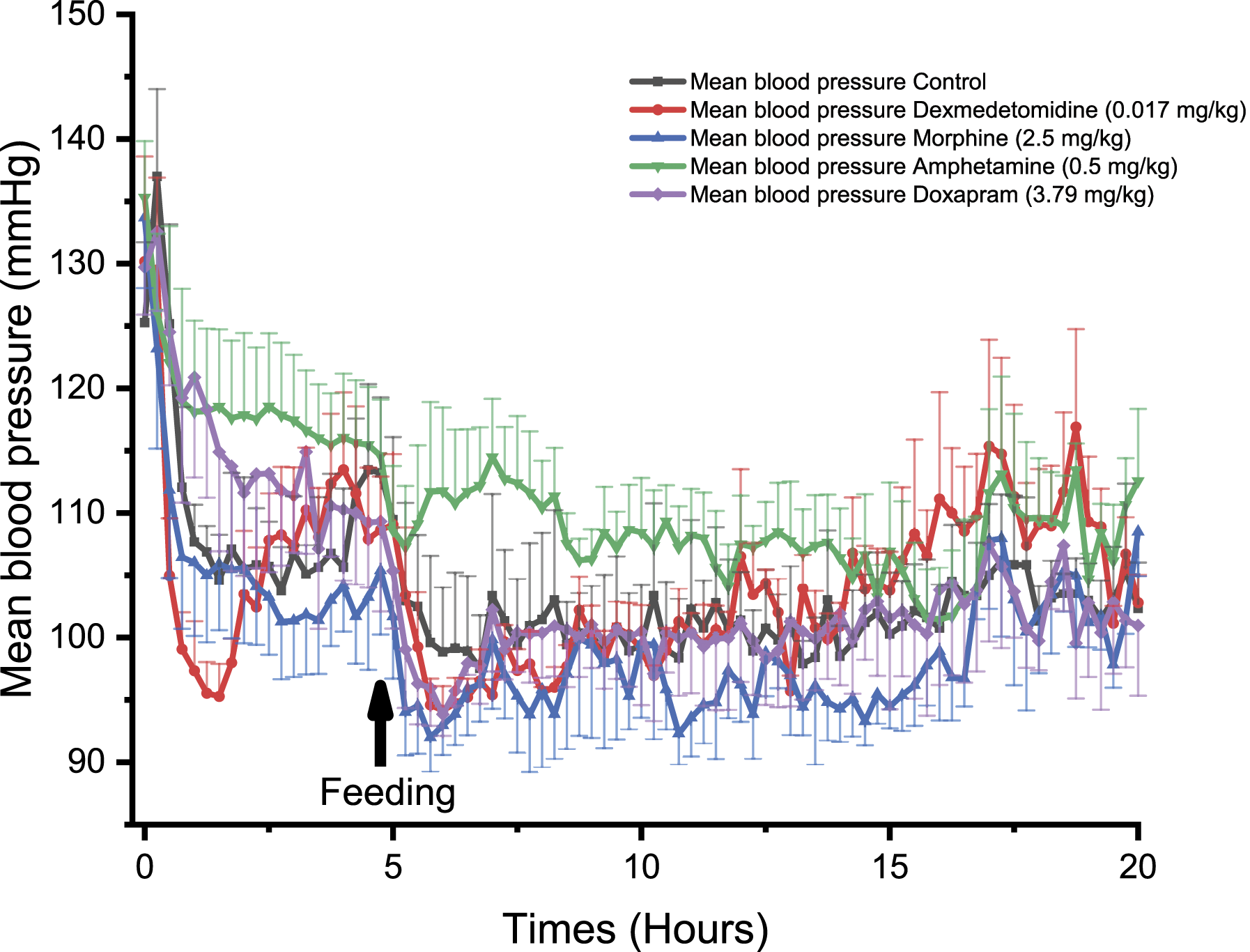
Morphine
In NHPs, morphine significantly decreased BP reaching statistical significance at 4.25 hours and at 14.50 hours (P < 0.05, Figure 4). In Beagle dogs, morphine was not associated with significant effects on BP (Figure 5) but a trend toward higher BP values was observed. In Göttingen minipigs, morphine induced a significant increase in BP at 2.00 to 2.25 hours (P < 0.05, Figure 6).
Amphetamine
In NHPs, amphetamine significantly increased BP at 2.75 to 3.00 hours and at 6.75 hours compared to control (P < 0.05, Figure 4). Similarly, amphetamine increased BP in Beagle dogs at 1.00 to 1.25 hours (P < 0.05), 1.50 to 2.00 hours (P < 0.01), 2.25 to 2.50 hours (P < 0.05), 5.50 hours (P < 0.05), 6.25 hours (P < 0.05), 6.50 to 7.50 hours (P < 0.01), 7.75 to 8.00 hours (P < 0.05), 9.00 to 9.25 hours (P < 0.05), and 11.25 hours (P < 0.05, Figure 5). In Göttingen minipigs, amphetamine did not significantly alter BP (Figure 6).
Doxapram
Doxapram significantly increased BP in NHPs at 1.00 hour post-dose (P < 0.05, Figure 4). In Beagle dogs, doxapram significantly increased BP at 1.25 hours (P < 0.05), 2.00 to 2.75 hours (P < 0.05), 3.25 to 3.50 hours (P < 0.05), 5.50 hours (P < 0.05), 6.25 to 6.50 hours (P < 0.05), 7.00 hours (P < 0.05), and 9.25 hours (P < 0.05, Figure 5). In Göttingen minipigs, doxapram administration was associated with an increase in BP but the long lasting effect did not reach statistical significance with the group size that was used (Figure 6).
Comparison Between the Heart Rate Derived from the Blood Pressure and the ECG in Non-Human Primates
The NHP was selected for telemetry signal comparisons as this species generally present a higher level of movement artifacts when compared to dogs and minipigs. Comparison of HR derived from the BP signal and HR derived from the ECG signal in NHPs (n = 4) over a 24-hour period revealed that both HR signals were highly correlated. The maximum difference in HR between the 2 signals was 1.5 bpm, and overall, the average difference when considering timepoints was 0.17 bpm (Figure 7). No significant differences were identified by a one-way ANOVA test. Comparison between the heart rate (HR) derived from either the blood pressure (BP) or the ECG signal, recorded over 24.50 hours in telemetered non-human primates (n = 4). ⍰ = HR from the control BP channel, ● = HR from the control ECG channel. One-way ANOVA Test F (1, 196) = 0.03; P > 0.05. ECG, electrocardiography; ANOVA F Test, analysis of variance.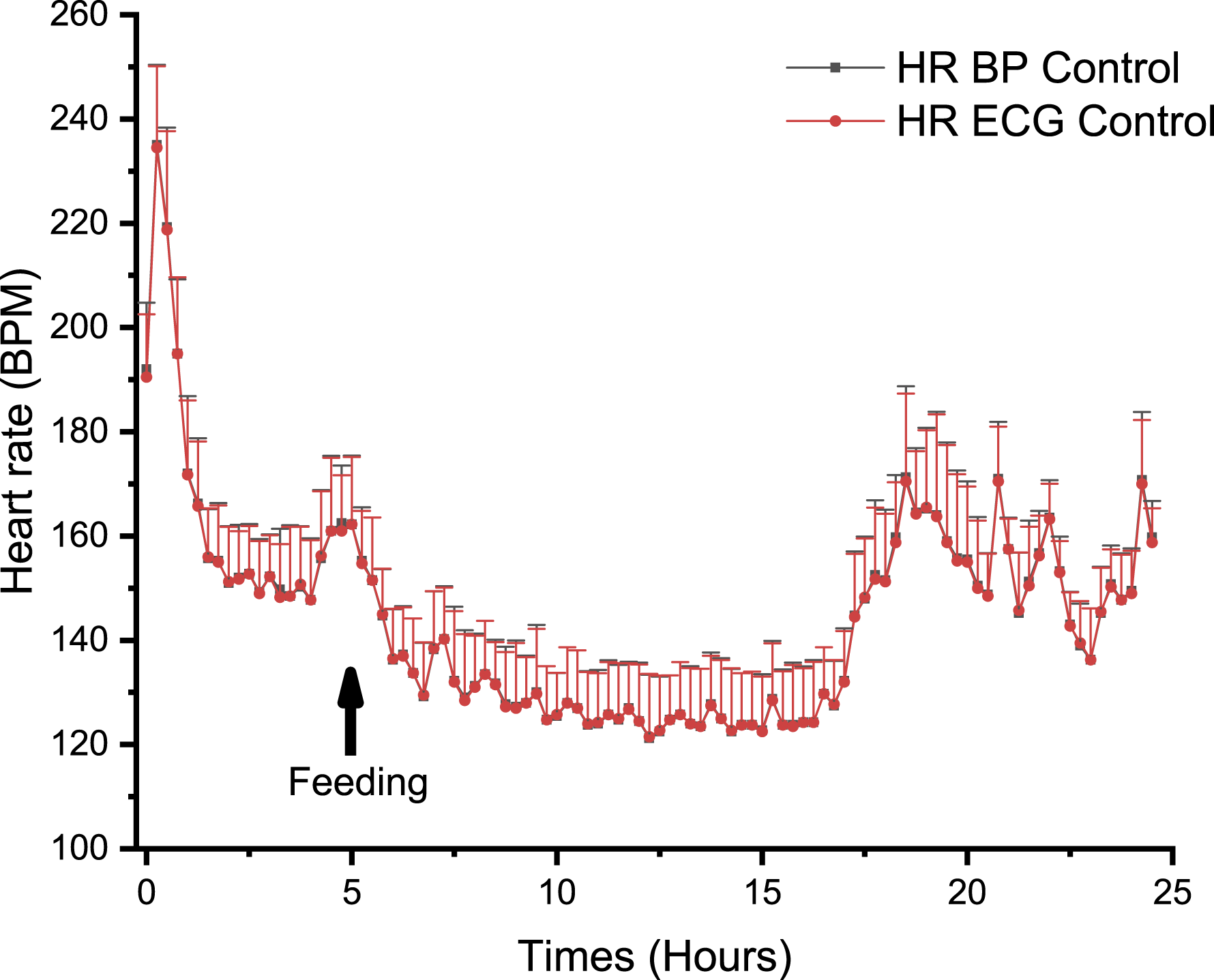
Correlation between Cardiovascular and Respiratory Parameters in Non-Human Primates
Changes in HR for NHPs (n = 4) were positively correlated with MV changes: Figure 8 illustrates the association between the cardiac and pulmonary systems; HR and impedance-based MV had a R2 of 0.87. Correlation between minute volume (MV) and heart rate over a 24-hour period (15 minutes average) in a control group composed of telemetered non-human primates (n = 4). The figure demonstrates cardiopulmonary relationship as an increase in heart rate is expected to be associated with an increase in MV. The coefficient of correlation was 0.869 and the slope 13.97. ⍰ = MV control.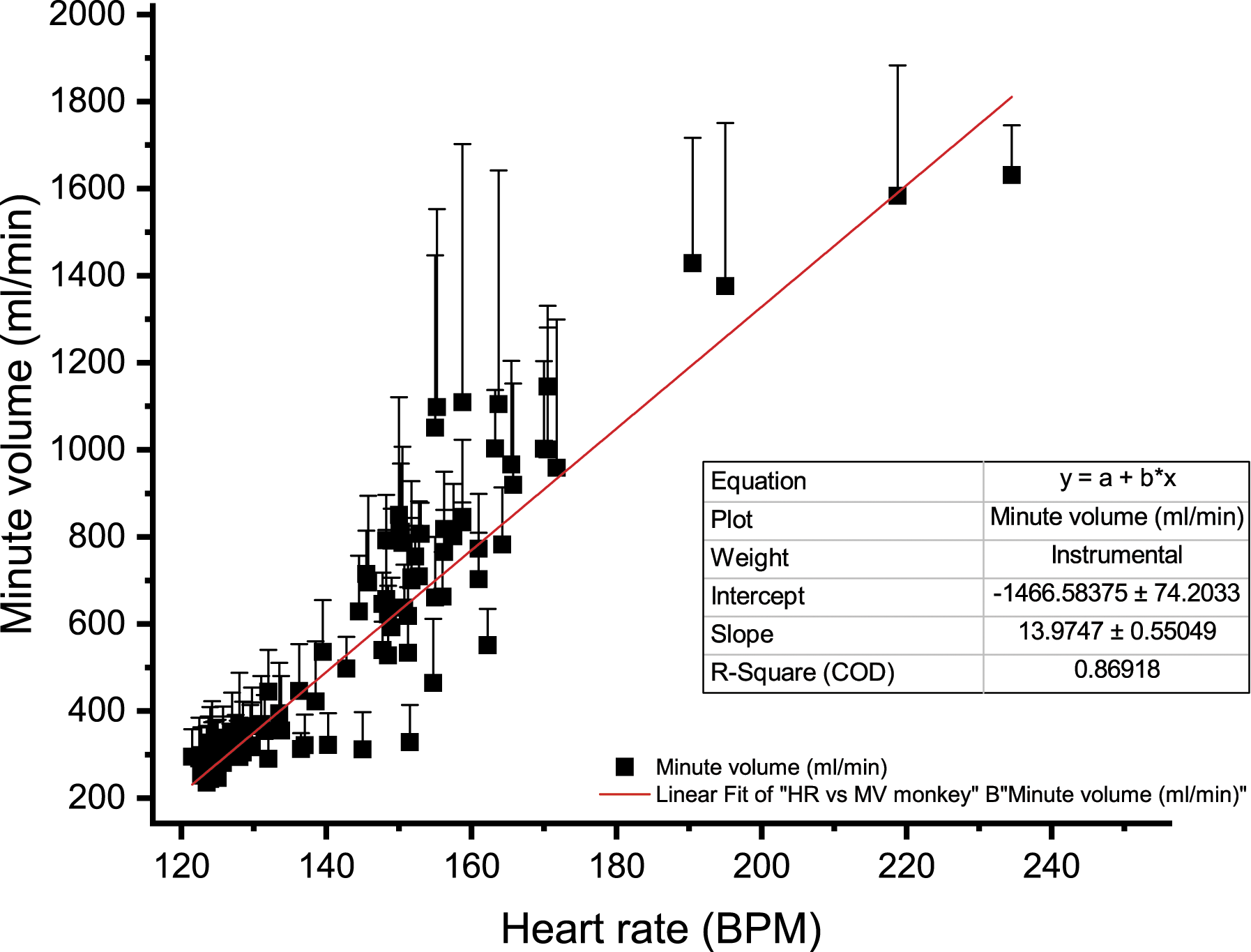
Effects of Social Structure: Single-Housed Versus Pair-Housed
The digital telemetry implant used in the current study allowed for recording of telemetry data when animals were pair-housed, a feature that previous telemetry implant technology could not achieve. MV was monitored and results are presented (Figure 9). Time course for the effects of social structure (single-house vs pair-house) on the (A) minute volume (MV), (B) respiratory rate (RR), (C) tidal volume (TV), being measured with PhysioTel Digital L11R telemetry implants, represented over 5 days (6 hours of continuous data are presented for each day, right after the manipulation of saline injection which was time 0) in telemetered non-human primates (n = 4). ⍰ = day 1 pair-housed, ● = day 2 single-housed, ▲ = day 3 single-housed, ▼ = day 4 pair-housed with a new cage mate, ♦ = day 5 pair-housed with a new cage mate.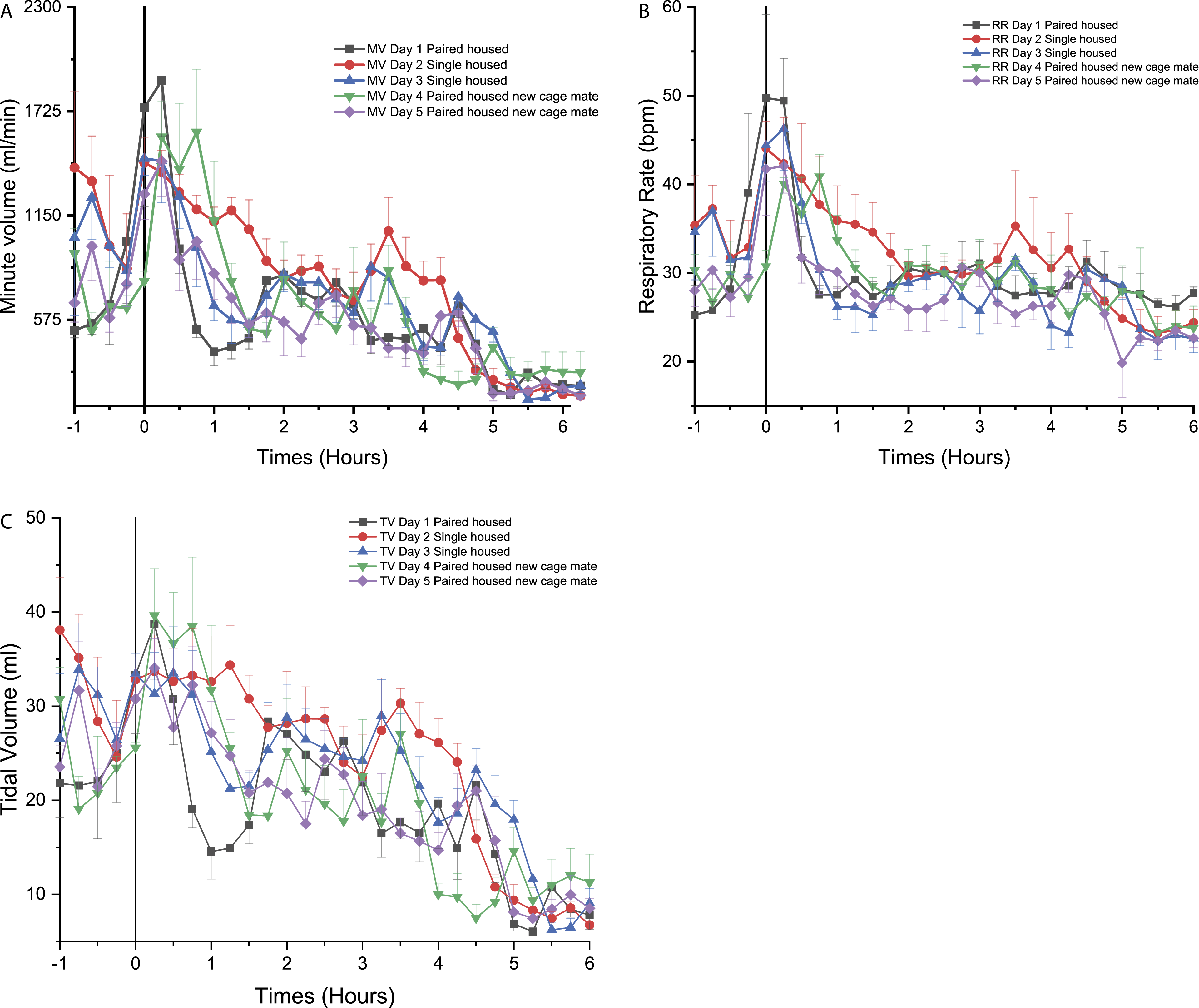
Effects of social structure for the non-human primates
On day 1, pair housing was associated with lower MV values which ranged from 1900 to 400 mL/min in NHPs after animal handling (Figure 9A). The MV remained at a generally steady level for the remainder of day 1 (Figure 9A). On day 2, the first day of single-housing caused an increase in MV, from 800 to 1200 mL/min from 0.50 to 4.25 hours (with a more pronounced difference when compared to day 1 around 3.75 to 4.00 hours). On day 3, the second consecutive single-housing day, data were similar to day 1 (Figure 9A). On day 4, the addition of a new cage mate (different from the cage mate from the first day) led to an increase in MV higher than the second day, but only for the first hour (significantly different from 0.75 to 1.00 hour, P < 0.05) (Figure 9A). From 4.00 to 6.00 hours after the social change, MV decreased to 290 mL/min (significant at 4.5 to 5.0 hours (P < 0.05)). MV values on day 5 (i.e., second day with a novel cage mate) were similar to day 1. The MV remained low from 1.25 to 4.75 hours (Figure 9A).
Respiratory rate on days 1, 3, and 5 followed a similar trend as MV with values ranging from 25 to 31 breaths per minute (Figure 9B). However, on day 2 and 4 (introduction to single-housing, and new cage mate, respectively), RR increased transiently: On day 2, an initial increase was observed (44.5 bpm at 0.00 hours, not different from day 1), followed by a progressive return to day 1 equivalent values (30 bpm at 2.00 hours) (Figure 9B). On day 4, a statistically significant increase in RR was noticed at 0.00 and 3.00 hours.
On day 2, while being single-housed, a greater and a longer increase in TV was observed (24 to 34 mL) from 0.00 to 4.25 hours with a significant increase in TV at 0.25 hours (P < 0.05) followed by a decrease in TV at 4.5 hours (P < 0.05). TV was higher than on day 1 for days 3, 4, and 5 (21 to 40 mL from 0.25 to 1.5 hours) (Figure 9C).
Effect of the social structure for the beagle dogs
On day 2, when the dogs were single-housed, a rapid decrease in MV was noticed, when compared to day 1 with a known cage mate, followed by a recovery to day 1 comparable levels after 0.25 hours (Figure 10A). On day 3 (second day of single-housing), the animals presented MV values that were lower than the previous 2 days (Figure 10A) with a significant decrease at 3.5 hours and 4.25 to 4.5 hours when compared to day 1 (P < 0.05). On the fourth day, the addition of a novel cage mate was associated to a rapid increase in MV that persisted until 3.00 hours after the social change and attaining statistical significance from 0.5 to 2.25 hours (P < 0.05) followed by sporadic increases at 3.25, 3.75, 4.25 to 4.5 hours and at 5.75 hours (P < 0.05). On day 5, MV remained comparable to day 1 (Figure 10A). Time course for the effects of social structure (single-house vs pair house) on the (A) minute volume (MV), (B) respiratory rate (RR), (C) tidal volume (TV), being measured with PhysioTel Digital L11R telemetry implants, represented over 5 days (6 hours of continuous data are presented for each day, right after the manipulation of saline injection which was time 0) in telemetered Beagle dogs (n = 4). ⍰ = day 1 pair-housed, ● = day 2 single-housed, ▲ = day 3 single-housed, ▼ = day 4 pair-housed with a new cage mate, ♦ = day 5 pair-housed with a new cage mate.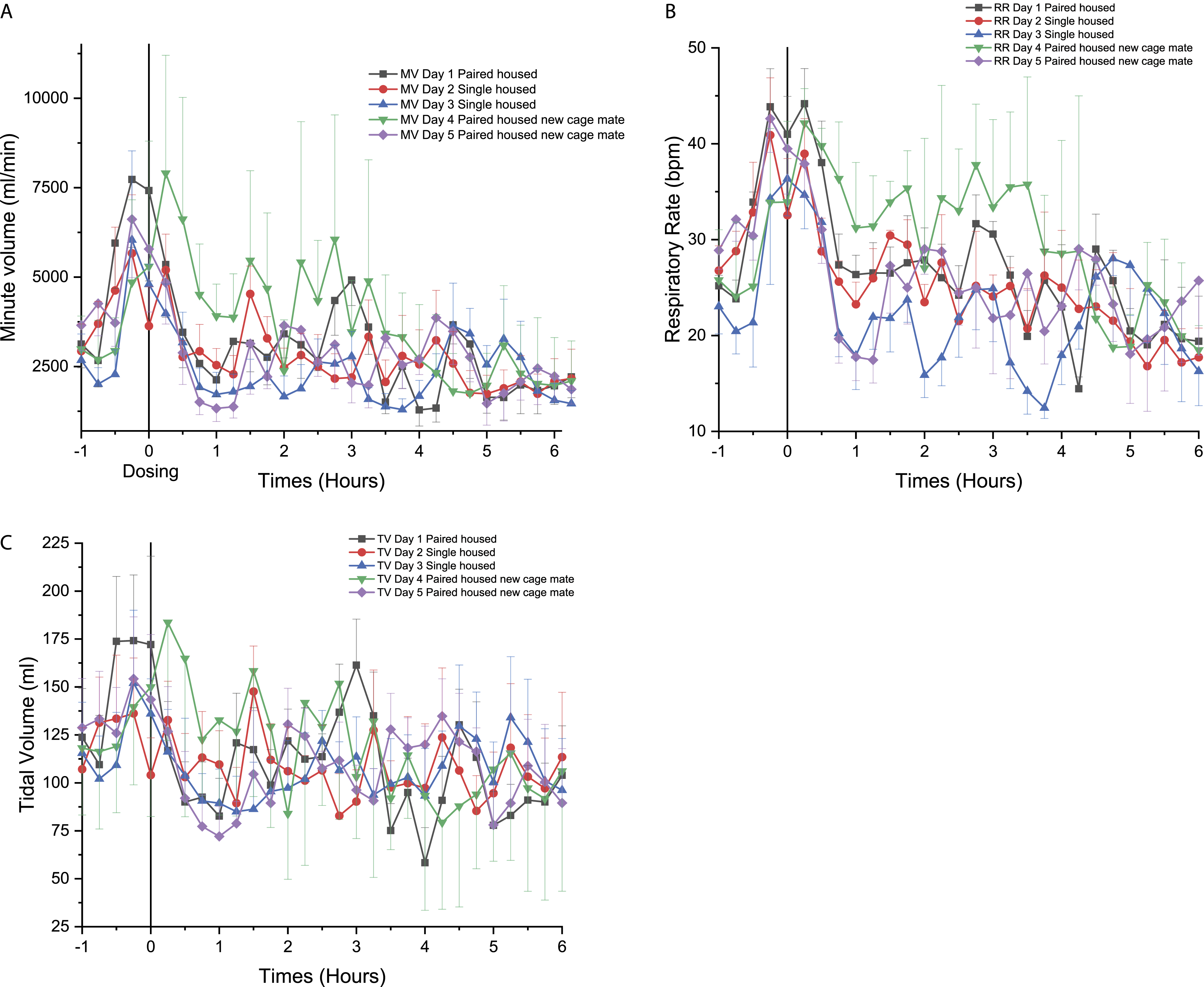
RR on days 1 and 2 followed a similar trend as MV with a range of 20–44 breaths per minute (Figure 10B). However, on day 3, RR was generally lower than day 1 (36 to 12 bpm from 0.00 to 3.75 hours) (Figure 10B). On day 4, when dogs were housed with a new cage mate, an increase in RR was observed from 0.25 to 4.25 hours (P < 0.05) (Figure 10B). For day 5, there was a rapid decrease in RR at around 0.5 hour and RR returned to values similar to day 1, 1.50 hours following handling (Figure 10B) and then a significant decrease of the RR was observed from 0.5 to 4.25 hours (P < 0.05).
Finally, TV was similar for day 1, 2, 3, and 5 with minor increase and decrease throughout the initial 6 hours of the day (Figure 10C). Moreover, on day 4, pair housing with a new cage mate led to an increase in TV (83 to 183 mL from 0.00 to 2.75 hours) (Figure 10C) reaching statistical significance at 0.5 to 1.75 hours (P < 0.05).
Discussion
The current study suggests that cardiopulmonary monitoring using digital implants can be used for drug safety investigations and importantly, enables continuous functional monitoring in socially housed animals which significantly contributes to implementation of the 3Rs by reducing the number of animals needed to fulfill the requirements from ICH S7A.
Monitoring of Physiological Parameters after Pharmacological Treatments
Dexmedetomidine is an α2-adrenoreceptor agonist that is recognized for its sedative and analgesic properties and is associated with cardiac and respiratory (dose-dependent) depression. Therefore, a reduction in RR leading to a reduction in MV, as well as a decrease in HR, was expected. 12 In this study, we observed a significant but transient reduction in MV for NHPs and Beagle dogs, with a corresponding decrease in RR only in Beagle dogs. In NHPs and dogs, respiratory depression induced by dexmedetomidine was observed only for approximately 30 minutes. No significant effect of dexmedetomidine was observed for respiratory parameters in minipigs, although a trend toward lower MV values was noticeable between 1.75 and 2.5 hours. A short duration of effect is expected as the dexmedetomidine half-life is about 2 hours (in humans) after single administration. 13 Dexmedetomidine was also associated with a transient reduction in BP for all the animal species tested. For NHPs and Beagle dogs, the reduction in BP occurred within the first 2 hours post-dose, quite concomitant with the respiratory depression previously described, while the effect of dexmedetomidine on BP in minipigs occurred later (at 5.25, 6.50 and up to 14.50 hours post-treatment), although this species presented higher variability when monitored with this technology. These results illustrate the relevance of functional monitoring for a longer period of time following drug administration as toxicopharmacological profiles may vary between species. Drug exposure was not quantified in the current study and would be relevant to assess potential inter-species differences.
Morphine is an opioid used in the management of moderate to severe pain. Its effects are mediated by activation of the mu opioid receptors in the central and peripheral nervous system. Morphine is known to have different side effects, the one most recognized being the dose-dependent respiratory depression, whereas it is usually associated with relatively limited cardiovascular effects. 14 In NHPs, a long-lasting reduction in MV was observed reaching statistical significance at one time-point, that is, 5.50 hours following treatment. Morphine also decreased BP in NHPs, reaching statistical significance at 4.25 and 14.50 hours post-injection.14,15 In Beagle dogs, morphine had no statistically significant effect on respiratory and cardiovascular parameters, although a trend toward lower RR values was observed. This observation is somewhat unexpected as morphine administration in dogs has been shown to induce respiratory depression in this species. 16 It remains that milder pharmacological effects, especially when higher variability is present, maybe difficult to detect with an n = 4 study designs as was used in the current study. In contrast, morphine treatment in minipigs increased RR and MV combined with an increase in BP, highlighting species differences even when using the same pharmacological agent. This corroborates observations from pigs in other studies where morphine impaired blood gas exchange in the lungs leading to an adaptative increase in RR and BP.17,18
Amphetamine is a centrally active nervous system stimulant with the potential to increase BP, MV, and RR. 19 As expected, a severe and sustained increase in MV, RR, and BP was observed in Beagle dogs after amphetamine. No significant change in BP, MV, and RR was observed in Göttingen minipigs but a sustained increase in BP was noted in NHPs. The dose of amphetamine for NHPs that results in a clinical effect (acute toxic effect) is approximately 1.0 mg/kg IV. 20 A lower amphetamine dose was administered PO to NHPs in the current study (i.e., 0.5 mg/kg) leading to a moderate long-lasting increase in BP. These results highlight the diverse functional profiles that may be observed when the same drug is administered to different species.
Doxapram is a respiratory stimulant with analeptic activity acting on the peripheral carotid chemoreceptor by inhibiting potassium channels, thereby increasing central inspiratory and expiratory neuronal activity.21,22 In rats, doxapram increases BP even when the carotid body is denervated, indicating that at least one other mechanism is involved. 23 In our study, doxapram administration resulted in no significant changes for MV and RR in NHPs and Göttingen minipigs. In contrast, an important long-lasting increase in MV and RR was observed in dogs. BP increased over a period of approximately 4 hours in NHPs, and sustained BP elevations were observed in Beagle dogs after the injection of doxapram again revealing important inter-species differences in the response to the same positive control drug.
In conclusion, relevant respiratory and cardiovascular effects were observed with simultaneous multi-organ system monitoring and significantly different inter-species responses to drugs were identified. Functional changes were identified in the 3 tested species evaluated and with a signal performance similar to previous publications but with the major advantage of enabling social housing. Different functional responses to the same pharmacological agent are documented across species (animal species but also different responses when compared to humans) which could be due to differences in exposure, target expression/sensitivity, or functional system responsiveness. These potential differences are commonly discussed when considering the translational value of animal models to predict the human response.
Heart Rate Signal Correlation (Blood Pressure, or ECG-Derived) in Non-Human Primates
Heart rate derived from BP or from ECG traces yielded highly concordant measures for NHPs. These data suggest both methodologies are equivalent in assessing chronotropic effects in NHPs, which are amongst the most prevalent findings in drug safety testing. A clear linear correlation between HR and MV changes also illustrates the cardiovascular and respiratory dependencies. 24 A relatively high correlation was achieved, despite the inherent variations related to the individual animals tested. 25
The Effects of Social Housing Structure
Single-housing of a social species is one of the main factors that leads to chronic stress. 26 Stress can interfere with either behavioral (anxiety, depression, and memory tests) or physiological endpoints.4,5 The level of cortisol increases when NHPs or dogs are singly housed for an extended period and decreases when pair-housed.27,28 Typical signs of stress such as increased respiratory parameters MV, RR, and TV were observed in NHPs and Beagle dogs that were single-housed or placed in a cage with the new cage mate. The acute stress developed in these social animals was transient, leading to minor increase (in intensity and duration) in MV, RR, and TV. NHPs showed more severe signs of stress on day 2, while single-housed, and for a longer period, as opposed to Beagle dogs that rapidly returned to a quiet state during the same day. Dogs were noticeably more active, for a longer period, when paired with a new cage companion on day 4. This suggests that NHPs critically need a social housing structure to minimize stress and this configuration is allowed by the digital technology studied in the current article. As for Beagle dogs, physiological parameters remained in the normal range with a known cage mate, but single-housing or a new cage mate both altered the functional endpoint in this species. Adult male Göttingen minipigs showed signs of aggressiveness that precluded from having them socially housed. Social incompatibility of adult male minipigs had been anecdotally reported (personal communication), and the data obtained in this study suggest that single-housing may be preferable when using adult male minipigs. Changes to the social structure (i.e., separation of a pair to single house and animal or introduction of a new cage mate) altered physiological parameters that were monitored in this study with increases in HR and/or respiratory parameters such as RR and MV observed with separation and with new pairs. Acclimation and habituation are important to ensure animal welfare and are expected to translate into more stable function parameters in telemetry studies. Results from the current study highlight the importance of social stability and acclimation when evaluating functional endpoints as stress from social changes may become a significant confounding factor.
Conclusion
This is one of the first studies to use a digital telemetry implant for cardiopulmonary monitoring in socially housed non-rodent species in the context of drug safety testing. The study highlights important inter-species differences when monitoring functional endpoints with known pharmacological agents. This study also illustrates the impacts of changes to the social structure with noticeable inter-species differences. Maintaining stable social groups can avoid confounding effects on telemetry parameters that are related to changes in social structure. Ultimately, multi-organ system monitoring offers an opportunity to improve designs in drug safety studies with the application of the 3Rs principles by reducing the number of animals needed to fulfill the ICH S7A guidelines.
Footnotes
Acknowledgments
The authors would like to thank the technical staff who worked on this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
