Abstract
Per- and polyfluorinated alkyl substances (PFAS) are ubiquitous, persistent, and toxic chemicals that pose public health risks. Recent carcinogenicity concerns have arisen based on epidemiological studies, animal tumor findings, and mechanistic data. Thousands of PFAS exist; however, current understanding of their toxicity is informed by studies of a select few, namely, perfluorooctanoic acid and perfluorooctanesulfonic acid. Hence, the computational, high-throughput screening tool, the US EPA CompTox Chemical Dashboard’s ToxCast, was utilized to explore the carcinogenicity potential of PFAS. Twenty-three major PFAS that had sufficient in vitro ToxCast data and covered a range of structural subclasses were analyzed with the visual analytics software ToxPi, yielding a qualitative and quantitative assessment of PFAS activity in realms closely linked with carcinogenicity. A comprehensive literature search was also conducted to check the consistency of analyses with other mechanistic data streams. The PFAS were found to induce a vast range of biological perturbations, in line with several of the International Agency for Research on Cancer–defined key carcinogen characteristics. Patterns observed varied by length of fluorine-bonded chains and/or functional group within and between each key characteristic, suggesting some structure-based variability in activity. In general, the major conclusions drawn from the analysis, that is, the most notable activities being modulation of receptor-mediated effects and induction of oxidative stress, were supported by literature findings. The study helps enhance understanding of the mechanistic pathways that underlie the potential carcinogenicity of various PFAS and hence could assist in hazard identification and risk assessment for this emerging and relevant class of environmental toxicants.
Keywords
Introduction
Per- and polyfluorinated alkyl substances (PFAS) are a series of synthetic compounds, utilized for several commercial and industrial purposes. They are found in a variety of consumer products, ranging from food packaging materials (e.g., microwave popcorn bags) and nonstick cookware to carpets and furniture to cosmetics and personal care products. 1 Per- and polyfluorinated alkyl substances are derived by 2 major processes: (i) electrochemical fluorination and (2) telomerization. 2 Typically, they consist of a partially or fully fluorinated carbon skeleton and a hydrophilic head group. Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) are the 2 most common and well-studied PFAS. However, this class of compounds includes a huge number of structural variants, based on the presence of different functional groups and varying chain lengths. The strength of the C–F bond, 3 resulting from its partial ionic character, lends extraordinary stability to these compounds and, hence, they are resistant to the major environmental, chemical, and biological degradation pathways. 4
Per- and polyfluorinated alkyl substances are distributed globally and have been detected in every trophic level and environmental phase. 5 Additionally, human exposure can occur through multiple routes, including ingestion via drinking water or food, inhalation, and contact with residual levels in packaging materials. 6 Furthermore, several health concerns regarding these compounds have arisen. 7 The PFAS tend to bioaccumulate in humans and wildlife due to their metabolic stability and long half-lives. 8 Their unique hydrophobic and lipophobic properties 9 lead them to bind to plasma proteins and concentrate in organs (e.g., liver, kidney), and levels have been detected in human serum and tissue samples from around the world. 10 For instance, a 2018 regional exposure study in women (n = 425) from the Los Angeles county indicated that the mean serum concentrations of PFAS range from 0.0681 ng/mL for lesser distributed PFAS to as much as 2.13 ng/mL for PFOS (https://biomonitoring.ca.gov). Several of the more well studied PFAS have been found to be toxic. Exposure to PFAS has been associated with developmental and reproductive toxicity, immunological effects, metabolic disorders, and organ-specific (e.g., hepatic, renal) toxicity.7,11
One major health concern regarding PFAS is their potential to act as carcinogens. 12 Epidemiological links between exposure to PFAS and incidences of cancer, 13 mechanistic studies in line with the International Agency for Research on Cancer (IARC) key carcinogen characteristics, and in vivo tumor data 14 suggest that carcinogenic activity is associated with exposure to PFAS.
One limitation that scientists including toxicologists face today is the lack of data available to evaluate the health concerns posed by a vast number of environmental chemicals. To mitigate this issue, the US Environmental Protection Agency (EPA) has developed a tool known as Computational Toxicology (CompTox) Chemicals Dashboard (https://comptox.epa.gov/dashboard). It integrates information for thousands of chemicals into a comprehensive database and, utilizing high-throughput screening models such as Toxicity Forecaster (ToxCast), generates in vitro toxicity data for chemicals by measuring the perturbations they induce in biological systems. This allows quick and efficient assessments concerning the toxicity of chemical(s) of interest and reduces the need for expensive and time-consuming animal testing.
Several of the ToxCast in vitro bioassay end points can be mapped directly to one of the 10 IARC key characteristics of carcinogens and thus data can be applied toward gauging toxicity in areas related to carcinogenicity.15,16 The wide range of health effects currently associated with PFAS exposure suggests they might possess multiple IARC-defined carcinogen characteristics. Moreover, structural variability such as different number of C–F bonds in the molecular backbone and/or the presence of unique functional groups could further influence the possible carcinogenic action of PFAS. Hence, the purpose of this study is to utilize data from a high-throughput toxicological tool to examine the potential biochemical modes by which a highly persistent, diverse, and widespread class of environmental chemicals could behave as carcinogens. The results were compared with literature-based findings to validate the conclusions.
Methods
Chemicals Dashboard and PFAS Selection
The PFAS of interest were identified from EPA’s CompTox Chemicals Dashboard (https://comptox.epa.gov/dashboard/chemical_lists). Utilizing EPA’s master list of potential PFAS, thousands of chemicals were initially examined (as of May 25, 2020), of which a set of 23 PFAS was selected for analysis (Figure 1). The chemicals were selected based on 3 main factors: close structural relation to the most relevant PFAS (i.e., PFOA and PFOS), distinct functional groups to cover a range of structural subclasses, and availability of sufficient high-throughput toxicity data. Alongside PFOA, the carboxylic acid–based PFAS series was selected and included both short- (6-7 carbons) and long-chain (9-11 carbons) variants as well as both free acid and salt forms. Together with PFOS, other sulfonic acids (short chains and salts) and sulfonic derivatives (ie, sulfonamides) were also chosen. Additionally, a variety of alternative functional group (eg, alcohols, methacrylate) based PFAS were selected to study moiety-specific differences. Finally, sufficient toxicity data necessary for inclusion were defined as an active or inactive hit in at least 1 ToxCast assay of interest. Hence, chemicals that were not tested in any of the ToxCast assays were excluded from analysis.
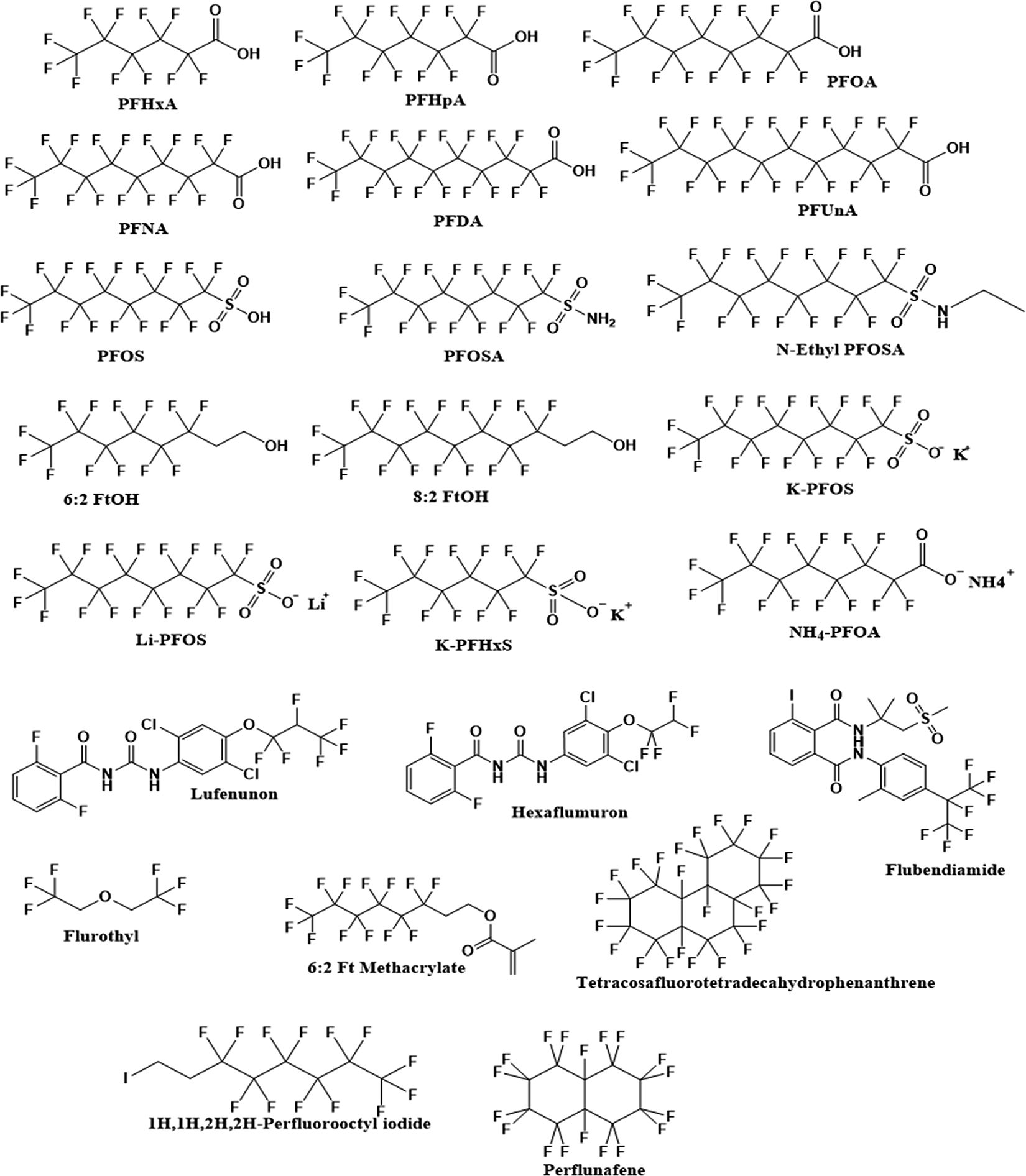
Chemical structures of the 23 per- and polyfluorinated alkyl substances selected from CompTox chemicals dashboard.
ToxCast Assays
Data from EPA’s ToxCast, a high-throughput screening program, were utilized to assess activity of the selected PFAS in areas related to carcinogenicity. Each chemical of interest had toxicity data for a series of in vitro assays that measured changes in either (1) cellular responses or (2) biochemical pathways, upon exposure to the chemical. The toxicity readout per assay indicated 1 of 3 possible outcomes: active, inactive, or not tested yet. Each active hit corresponded to a particular half-maximal activity concentration (AC50) to indicate potency with respect to that end point. The detailed experimental procedures employed to acquire the in vitro ToxCast data are readily available online (https://comptox.epa.gov/dashboard/assay_endpoints/), as well as previously documented.17-20 The ToxCast assays of interest were ones that could be mapped to 1 of 10 IARC described key characteristics of carcinogens. 15 They were primarily selected using a previously described 2018 study (Chiu et al) which matched the descriptions and end points of assays to the key carcinogen characteristics based on identified mechanistic similarities. 16 The exceptions include inclusion of a new group of nuclear receptor targeting assays (mapped to key characteristic #8) and exclusion of key characteristic #1 mapped assays since this class of chemicals is not metabolically activated. Names and end point details of the 242 ToxCast assays are described in Tables S1-6 (in Supplementary material).
Toxicological Prioritization Index
Toxicological Prioritization Index (ToxPi, http://toxpi.org) is a visual analytics tool that integrates bioassay data from various sources and outputs the results in a graphical manner (i.e., a pie chart), allowing more efficient interpretation of findings. 21 Additionally, it provides a quantitative score that assists in ranking and prioritization of chemicals. A numerical chemical-assay matrix was generated, which accounted for active (AC50), inactive (1000000), or untested (NaN) assay results and inputted into ToxPi for holistic visualization of activity. For a chemical, each pie slice represents a key characteristic of carcinogens, with the slice width and length referring to the number of assays tested and ratio of positive to total hits, respectively. To calculate scores (y), a negative log scale (i.e., y = −log10(x) + 6) was applied to each slice, as previously described. 22 The cumulative score is an aggregation of slice-wise scores and is proportional to the overall predicted activity in areas related to carcinogenicity.
Results
ToxPi Output
Figure 2 depicts the ToxPi graphical output, overall scores, and legend following integration and analysis of ToxCast data of the 23 selected PFAS. Chemicals that displayed the greatest overall activity in assays linked to carcinogenicity were perfluorooctanesulfonamide (PFOSA), PFOS, N-ethyl PFOSA, and the potassium (K) salt of PFOS (scores = 0.7032-0.8235). Perfluorononanoic acid (PFNA), perfluorodecanoic acid (PFDA), and perfluoroundecanoic acid (PFUnA), carboxylic acids with a fluorine-bonded carbon skeleton longer than PFOA, were more active than PFOA. However, there was no significant difference (scores = 0.4975-0.5353) between the overall activity of the 3 chemicals. Nevertheless, PFOA (score = 0.3502) was more active than perfluoroheptanoic acid (PFHpA; score = 0.2438), which was far more active than perfluorohexanoic acid (PFHxA; score = 0.0428). Similarly, the 8:2 fluorotelomer alcohol (8:2 FtOH; score = 0.4320) ranked higher than the 6:2 fluorotelomer alcohol (6:2 FtOH (score = 0.2121) as did K-PFOS (score = 0.7032) compared to K-perfluorohexanesulfonic acid (K-PFHxS; score = 0.2299). Miscellaneous functional group (eg, methacrylate, urea, amide) based PFAS tended to be generally less active (scores <0.2100), though they had considerably more untested assays. Thus, for certain chemicals, particularly those with aromatic rings or an alkyl iodide, the smaller scores might be more indicative of lesser data availability than lower toxicity and so should be interpreted carefully.

ToxPi output: Plots and scores after integration of ToxCast data for the 23 per- and polyfluorinated alkyl substances. For a chemical, each pie slice represents a key characteristic (as per the legend), with the slice width and length referring to the number of assays tested and ratio of positive to total hits, respectively. A y = −log10(x) + 6 scale was applied to calculate scores (y) for each slice, and the displayed, cumulative score is an aggregation of slice-wise scores and is proportional to the overall predicted activity in areas related to carcinogenicity.
Genotoxicity
Key characteristic #2 (i.e., Is genotoxic) was generally the most minor carcinogenic activity component for most chemicals, with the notable exception being PFOSA (Figure 2). However, all the ToxCast assays corresponding to genotoxicity measured p53-regulated genotoxic stress, an indirect and constrained end point. Hence, the scope for assessing genotoxic activity was limited in this analysis.
Epigenetic Alterations
With regard to key characteristic #4 (i.e., Induces epigenetic alterations), the fluorotelomer alcohols tended to be the most consistently active when it came to enhancing expression of certain transcription factors (Table 1) that are indicative of changes to the epigenome.23,24 In a previous study, carboxylic and sulfonic acid–based PFAS did not influence expression of FoxA2. 25 This finding supports the ToxCast result that only 8:2 FtOH affected FoxA2. On the other hand, sulfonates (free acid and salt) and short-chain carboxylic acids inhibited activity of key epigenome-linked histone and sirtuin deacetylase enzymes (Table 1).
PFAS Active in ToxCast Assays Correlating With IARC Key Characteristic #4 End Points.

Abbreviations: IARC, International Agency for Research on Cancer; K-PFHxS, K-perfluorohexanesulfonic acid; NA, not applicable; PFAS, per- and polyfluorinated alkyl substances; PFDA, perfluorodecanoic acid; PFHxA, perfluorohexanoic acid; PFHxS, perfluorohexanesulfonic acid; PFNA, perfluorononanoic acid, PFOA, perfluorooctanoic acid; PFOS, perfluorooctanesulfonic acid; PFOSA, perfluorooctanesulfonamide; PFUnA, perfluoroundecanoic acid.
Oxidative Stress
Key characteristic #5 (i.e., Induces oxidative stress) is assessed in ToxCast by examining the upregulation of several oxidative stress markers and antioxidative responses (Table 2). Phosphorylation of histone protein H2A and mitogen-activated protein kinase JNK acts as a marker for reactive oxygen species (ROS) triggered DNA and cellular damage26,27 and hence higher levels of phosphorylated variants following exposure to various PFAS suggested induction of oxidative stress. Relevant PFAS included PFHpA, which has been previously shown to mediate hepatic and cardiac toxicity via activation of the phosphorylated JNK pathway, 28 thus supporting the ToxCast finding. Upregulation of the collagenase MMP-1 is another oxidative stress marker 29 and was observed to be increased following PFOA and PFOSA exposure. Hypoxic conditions can accumulate ROS, 30 and, accordingly, PFOSA, PFUnA, and a methacrylate fluorotelomer increased expression of H1F1α, a hypoxia-inducible factor. NRF2-ARE signaling is the central antioxidative pathway that attempts to restore homeostatic balance. It was triggered in response to exposure to a variety of PFAS, including PFOA, with enhanced NRF2 gene expression. Increased NRF2-ARE signaling in rat testes and liver of frogs exposed to PFOA, with greater expression of downstream NRF2 antioxidative targets (e.g., superoxide dismutase, catalase, heme oxygenase-1), has been documented.31,32 Other oxidative stress protective pathways include the upregulation of metallothionein proteins (MTs) as well as recruitment of heat shock proteins (HSPs).33,34 Activation of the metal response element promotes transcription of MTs and was observed in response to several PFAS. Heat shock factor 1 (HSF1) transcribes HSPs, and PFOSA and flubendiamide enhanced expression of the HSF1 gene.
PFAS Active in ToxCast Assays Correlating With IARC Key Characteristic #5 End Points.

Abbreviations: IARC, International Agency for Research on Cancer;K-PFHxS, K-perfluorohexanesulfonic acid; NA, not applicable; PFAS, per- and polyfluorinated alkyl substances; PFDA, perfluorodecanoic acid; PFHpA, perfluoroheptanoic acid; PFNA, perfluorononanoic acid, PFOA, perfluorooctanoic acid; PFOS, perfluorooctanesulfonic acid; PFOSA, perfluorooctanesulfonamide; PFUnA, perfluoroundecanoic acid.
Inflammation
In line with key characteristic #6 (i.e., Induces chronic inflammation), the increase in cell adhesion molecules (CAMs) and pro-inflammatory signaling factors (Table 3) is used to evaluate upregulation of inflammation pathways in ToxCast. Both PFOA and the potassium salt of PFOS were predicted to elevate levels of CAMs, specifically E-selectin and P-selectin. Prolonged exposure to K-PFOS has previously increased P-selectin in human umbilical vascular endothelial cells. 35 Nuclear factor (NF)-κB is a protein complex that acts as the master transcription factor in the activation of a systemic pro-inflammatory response. Macrophage generated cytokine tumor necrosis factor α (TNFα) activates NF-κB and results in the downstream production of pro-inflammatory cytokine interleukin-6. 36 Increased expression of the 3 proteins in this axis was only observed with exposure to alcohol fluorotelomers and sulfonamides in ToxCast. This observation was reinforced by a study, 37 which indicated PFAS fluorotelomers and sulfonamides did not interfere with the degradation of I-κB, inhibitory proteins that block NF-κB transcriptional activity. This contrasted with PFOS, which inhibited the degradation of I-κB, facilitating the sequestration of NF-κB in the cytosol, and possibly even decreasing downstream pro-inflammatory cytokine activity.
PFAS Active in ToxCast Assays Correlating With IARC Key Characteristic #6 End Points.
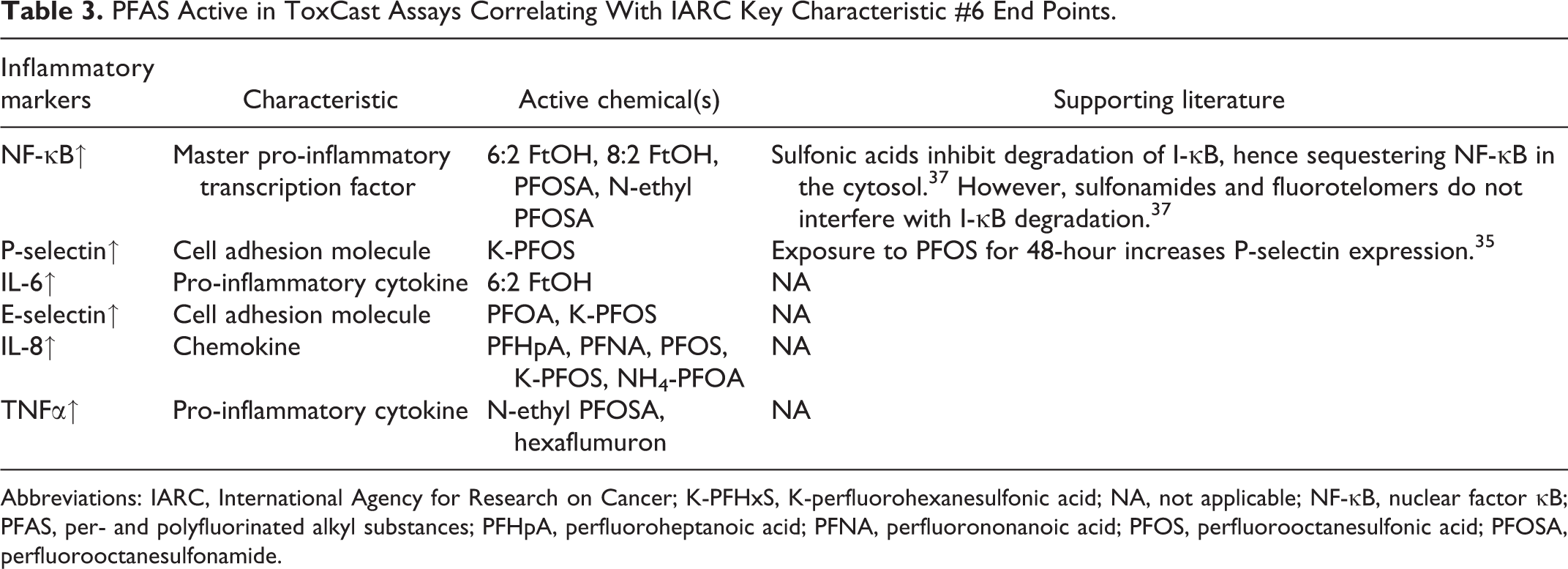
Abbreviations: IARC, International Agency for Research on Cancer; K-PFHxS, K-perfluorohexanesulfonic acid; NA, not applicable; NF-κB, nuclear factor κB; PFAS, per- and polyfluorinated alkyl substances; PFHpA, perfluoroheptanoic acid; PFNA, perfluorononanoic acid; PFOS, perfluorooctanesulfonic acid; PFOSA, perfluorooctanesulfonamide.
Receptor-Mediated Effects
Key characteristic #8 (i.e., Modulates receptor-mediated effects) is studied in ToxCast as per changes in the activity of a variety of receptor targets (Table 4 and 5). PFOA, PFOS, and certain studied PFAS are known endocrine disruptors as well as peroxisome proliferators.38-41 Major targets include the estrogen receptor α (ERα) and peroxisome proliferator–activated receptor γ (PPARγ), respectively. They are also considered mixed ERα agonists/antagonists due to their ability to bind as both. Predictably, PFAS-mediated ERα agonistic and antagonistic effects as well as PPARγ agonism in ToxCast were observed and, in general, the potency of action correlated with the length of the fluorine-bonded carbon chain (Table 4). Additionally, general differences in functional group activity were also seen, including stronger ERα antagonistic action by carboxylic acids relative to sulfonic acids, weaker ERα agonism by carboxylic acids compared to sulfonamides, and stronger PPARγ agonism by carboxylic acids relative to sulfonamides (Table 4).
Potency (1/μM) of PFAS-Modulated ERα and PPARγ Effects in ToxCast Assays.
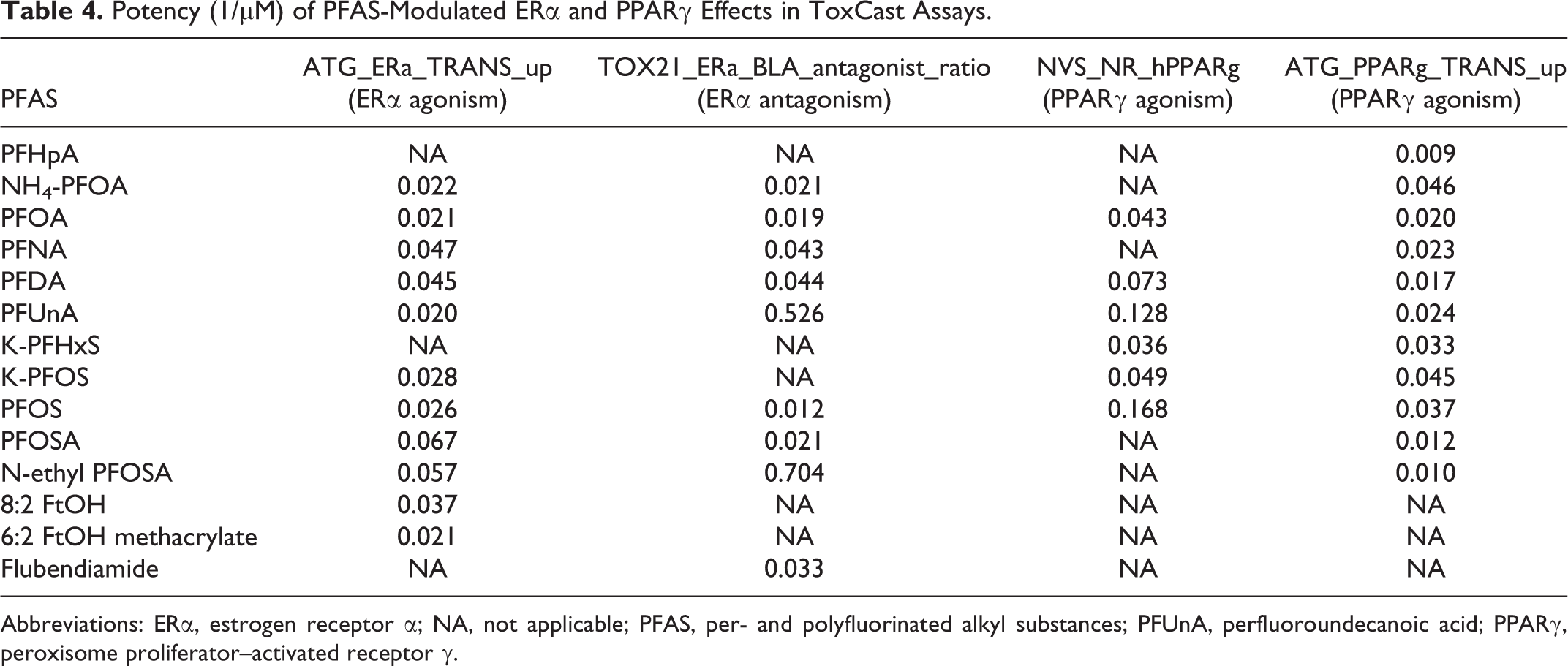
Abbreviations: ERα, estrogen receptor α; NA, not applicable; PFAS, per- and polyfluorinated alkyl substances; PFUnA, perfluoroundecanoic acid; PPARγ, peroxisome proliferator–activated receptor γ.
PFAS Active in ToxCast Assays Correlating With IARC Key Characteristic #8 Endpoints.
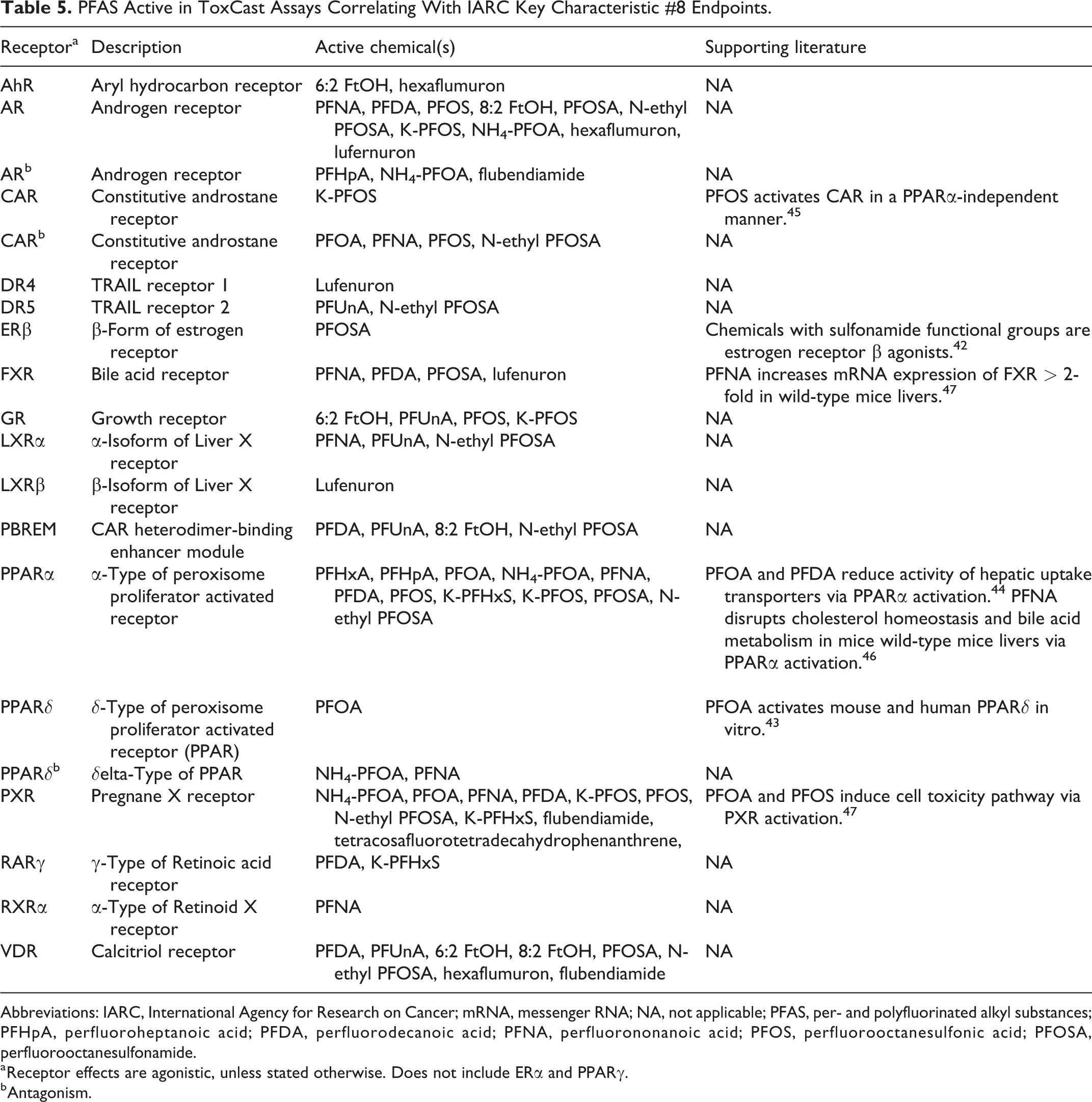
Abbreviations: IARC, International Agency for Research on Cancer; mRNA, messenger RNA; NA, not applicable; PFAS, per- and polyfluorinated alkyl substances; PFHpA, perfluoroheptanoic acid; PFDA, perfluorodecanoic acid; PFNA, perfluorononanoic acid; PFOS, perfluorooctanesulfonic acid; PFOSA, perfluorooctanesulfonamide.
a Receptor effects are agonistic, unless stated otherwise. Does not include ERα and PPARγ.
b Antagonism.
ToxCast data also demonstrated PFAS to be ligands of other estrogen and PPAR receptor types, as well as of the androgen receptor (AR; Table 5). The ERβ was activated only by PFOSA, an observation in line with the literature finding that sulfonamide-based chemicals are selective ERβ agonists. 42 A number of carboxylic and sulfonic PFAS were PPARα and PPARδ agonists, a finding consistent with the activation of human and mouse α and δ PPAR isoforms by PFOA and PFOS. 43 Moreover, PPARα activation is considered to drive the hepatotoxicity of PFOA and other long-chain carboxylic PFAS. 44 The activity toward the AR was mixed. The shorter chain PFHpA was an antagonist, longer chain PFNA and PFDA were agonists while the ammonium salt of PFOA was both.
In addition, several miscellaneous nuclear receptors were considered targets for PFAS binding (Table 5). Examples include activation of CAR by the potassium salt of PFOS, a notable observation since a study implicated CAR agonism by PFOS as a mode of PPARα-independent toxicity. 45 Both PFNA and PFDA activated FXR, while increased expression of FXR genes by PFNA exposure in mouse livers has been found in literature. 46 The PXR was also activated by a number of PFAS, and a study considers agonism of PXR by PFOA and PFOS as yet another mode of toxicity. 47 Non-nuclear receptor targets included the cell-surface TRAIL receptors (DR4 and DR5), but they were activated by only a few PFAS.
Alterations in Cell Proliferation
With regard to key characteristic #10 (i.e., Alters cell proliferation, cell death, or nutrient supply), many mapped ToxCast assays measured changes in cell growth, viability, and cytotoxicity by utilizing tools such as the SRB assay and mitochondrial toxicity end points. Certain cell-cycle markers were also assessed (Table 6), and these were consistently altered by long chain (≥8) carboxylic acids, alongside various functional group–based PFAS. Higher levels of transforming growth factor β1 (TGFβ1) messenger RNA were expressed, and this observation is bolstered by the findings that PFOA and PFOS stimulate epithelial–mesenchymal transition and subsequent renal fibrosis through upregulation of TGFβ1.48,49 Increased expression of proto-oncogenes transcribed by Myb and Myc was also seen. HFPO-TA, an ether-based PFAS recently developed as an PFOA alternative, has demonstrated the ability to elevate Myc protein levels, 50 further validating the involvement of this pathway. Finally, greater expression of Xbp1-transcribed genes by PFAS was supported by higher levels of the active, spliced version of Xbp1 (sXbp1), being observed following PFOA exposure in liver cells. 51 This would suggest PFAS mediate induction of prosurvival early-stage endoplasmic reticulum stress. 52
PFAS Active in ToxCast Assays Correlating With IARC Key Characteristic #10 End Points.

Abbreviations: IARC, International Agency for Research on Cancer; mRNA, messenger RNA; NA, not applicable; PFAS, per- and polyfluorinated alkyl substances; PFHpA, perfluoroheptanoic acid; PFDA, perfluorodecanoic acid; PFNA, perfluorononanoic acid; PFOS, perfluorooctanesulfonic acid; PFOSA, perfluorooctanesulfonamide; PFUnA, perfluoroundecanoic acid.
Discussion
In recent years, there have been rising health concerns regarding the carcinogenic potential of PFAS. 12 As a result, studying the mechanistic modes of PFAS toxicity, in relation to key carcinogen characteristics, could play a key role in understanding how they might act as carcinogens.
Analysis of in vitro ToxCast data revealed that PFAS induce a huge number of diverse biological perturbations in several carcinogenicity-implicated molecular pathways (Table 7), many of which were validated by literature findings. The relatively higher overall scores for longer chain (≥8) sulfonamide and sulfonate PFAS (Figure 2) indicate they might be particularly toxic chemicals in mechanistic domains associated with carcinogenicity. This result would be in line with certain toxicity studies that found PFAS-containing sulfonic acid derivative moieties to be more potent than their carboxylic equivalents.53,54
Summary of Major Biological Perturbations Induced by PFAS in ToxCast Assays.

Abbreviations: AR, androgen receptor; ER, estrogen receptor; IL-6, interleukin-6; NF-κB, nuclear factor κB; PFAS, per- and polyfluorinated alkyl substances; TNFα, tumor necrosis factor α.
While the potential of PFAS to act as genotoxins (key characteristic #2) was relatively minor (Figure 2), their influence on gene expression through other modes of action was found to be of considerable interest from a standpoint of epigenetic and nuclear receptor-mediated toxicity. In accordance with key characteristic #4, epigenetic alterations involve nonsequence-based changes to the DNA-regulatory protein structure, often leading to different heritable phenotypic traits. For instance, blocking the deacetylation of histone proteins loosens the chromatin complex and is typically linked with transcriptional activation. 55 Changes in epigenomic markers and inhibition of deacetylase activity (Table 1) in response to PFAS exposure signifies a possible pathway for certain PFAS to influence gene expression. Key characteristic #8 covers the ability of chemicals to mediate effects by acting on receptors and was the most significant area of activity for PFAS. The vast majority of active ToxCast receptor-based assay hits were for nuclear receptors (Tables 4 and 5). This, in conjunction with the recognized endocrine disrupting and peroxisome proliferating action of PFAS, highlighted the capacity of PFAS to mimic endogenous hydrophobic signaling molecules and influence transcription of genes 56 as a major concern. The PFAS induce generation of ROS in both in vitro and in vivo toxicity models.57-59 The upregulation of multiple oxidative stress-involved pathways and protein markers as well as elevated antioxidative responses was observed following PFAS exposure in ToxCast assays (Table 2). This implies an exogenously triggered systemic imbalance between ROS and antioxidants and hence further solidifies key characteristic #5 as another major component of PFAS toxicity. Persistent and unregulated inflammatory factor activity results in conditions of chronic inflammation. Correspondingly, the enhancement of CAMs or cytokines by particular PFAS (Table 3) suggests an ability of some of these chemicals to promote cell recruitment, aggregation, and sustained pro-inflammatory signaling (i.e., key characteristic #6). Chemicals that induce changes in cellular proliferation or death can be considered carcinogens as per key characteristic #10. The generally observed increase in cell growth, prosurvival, and transformation markers (Table 6) as well as a decrease in apoptosis suggest PFAS could also potentially exert pro-proliferative effects and inhibit cell death.
Generally, within each functional group class, the activity of PFAS was found to be directly proportional to the length of their fluorine-bonded carbon skeleton. This trend is consistent with literature findings, which deem long-chain PFAS more potent due to the presence of additional C–F bonds.60-62 Hence, the analysis provides more evidence to support the phasing out of longer chain PFAS, in favor of short-chain alternatives. Some functional group-selective activities were also observed, including fluorotelomer alcohol-predominated epigenetic effects (Table 1) and discrepancy in inflammatory effects exerted by PFAS (Table 3), the latter due to variable influence on the nuclear translocation of NF-κB. The mixed activity toward the AR (Table 5) also indicates that chain length might influence the orientation of interaction with receptor-binding sites. Though, despite the prediction of some specific ligand–receptor interactions (e.g., sulfonamides and ERβ), the sheer variety of receptor targets (Table 5) points to the generally nonselective and promiscuous nature of PFAS as ligands.
Limitations
Certain limitations of the ToxCast data sets have been identified and need to be considered. First, the ToxCast assays do not account for all IARC-defined key characteristics, namely, #3 (Alters DNA repair or causes genomic instability), #7 (Is immunosuppressive), and #9 (Causes immortalization). In particular, the scarcity of assays that can be mapped to key characteristic #7 restricts the extent of the analysis since there is strong epidemiological and toxicological evidence connecting immunosuppression with exposure to PFOA and PFOS. 63 The chemical purity of the test substance PFAS utilized in assays has also not been determined and quality control analyses are still underway. Recently, a few assay-associated curve fitting flags that raise concerns of false positives have been identified 64 and subsequently addressed. 65 Finally, the surfactant-like properties of PFAS introduce some uncertainty about chemical disposition and partitioning, factors that can influence results of in vitro testing systems. Thus, in general, caution should be exercised before any conclusions or generalizations are made based on the findings of the study.
Conclusion
Overall, the inferences drawn here could act as initial steps in helping uncover the potential carcinogenic nature of PFAS. They might be applied toward further studying the hazards posed by PFAS by examining biomarkers of effect following PFAS exposure, thus providing potential tools to identify possible cancer risk.
Supplemental Material
Supplemental Material, sj-docx-1-ijt-10.1177_10915818211010490 - Exploring Potential Carcinogenic Activity of Per- and Polyfluorinated Alkyl Substances Utilizing High-Throughput Toxicity Screening Data
Supplemental Material, sj-docx-1-ijt-10.1177_10915818211010490 for Exploring Potential Carcinogenic Activity of Per- and Polyfluorinated Alkyl Substances Utilizing High-Throughput Toxicity Screening Data by Nalin Singh and Ching Yi Jennifer Hsieh in International Journal of Toxicology
Footnotes
Authors’ Contribution
Singh, N. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Hsieh, C. J. contributed to conception and design; acquisition, analysis, and interpretation; and critically revised manuscript. Both authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. The content of this manuscript is solely the responsibility of the authors and does not represent the official views of the Office of Environmental Health Hazard Assessment, California Environmental Protection Agency, or the State of California.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
