Abstract
Increasing evidence indicates that environmental pollutants can change human gut microbiota. Microcystin–leucine arginine (MC-LR), considered a major hazard to mammals, is one of the important contaminants. However, little is known about the long-term influence of MC-LR on gut microbial communities. We aimed to investigate the effect of MC-LR on gut microbiota composition and functions by conducting a chronic exposure of male mice to MC-LR via the oral route. Using 16S rRNA gene sequencing analysis on cecum samples of mice, our results showed that significant changes of species diversity were observed in the gut microbiota of MC-LR-exposed mice. In addition, comparative analysis of the microbial communities showed that the reduction of the
Introduction
Microcystins (MCs) released from dead cyanobacteria are considered to be a harmful contamination to mammalian health. 1 Microcystin–leucine arginine (MC-LR) is the most widely and frequently studied isoform, accounting for 46% to 99.8% of the total MCs in freshwater. 2 The World Health Organization established a provisional guideline value of 1 µg/L for drinking water and a tolerable daily intake of 0.04 µg/kg body weight per day for MC-LR in contaminated seafood due to its toxicity and based on the results of liver toxicity studies in mice according to the California Office of Environmental Health Hazard Assessment. World Health Organization also categorized swimming risk levels as mild, moderate, high, or very high based on the water concentration of MCs. In 2010, the International Agency for Research on Cancer classified MC-LR as a possible human carcinogen. 3 Recent studies demonstrate that MC-LR has toxic effect on the intestine. 4 However, the mechanism of this phenomenon is still unclear.
Commensal microbiota is present in different body parts, including skin, oral cavity, and gut. 5 Dysregulation in the composition of the gut microbiome may lead to the occurrence of many diseases, such as obesity and diabetes, due to the complex involvement of microbial populations in many biological processes, including immune regulation and energy metabolism. 6,7 The profile and abundance of gut microbes are highly sensitive to dietary habits and environmental pollutants. 8 Previous studies have shown that some pollutants, such as persistent organic pollutants, lead, and bisphenol A, can change the composition of the gut microbiomes. 8 Thus, we speculate that MC-LR induces intestinal injury due to the alteration of gut microbes. In addition, intraperitoneal injection of high concentrations of MC-LR is the main treatment to establish acute mouse injury model. 3 However, this method does not represent a typical method for animals to be exposed to this toxic substance. The animals and humans are more likely to accumulate MC-LR in the body by ingesting food or water contaminated with this toxin.
Previous studies demonstrated that MC-LR can affect the microbiota profile from the jejunum, ileum, and colon, and it specially affects the microbial diversity in the cecum. 9 Several studies have revealed that microbiota patterns in cecal contents are different from those in feces because of the specific ecological conditions. 10 Therefore, cecal microbiota was chosen to investigate in our study.
Taken together, the purpose of this article is to investigate the mechanism of MC-LR-induced intestinal toxicity on intestinal flora based on the susceptibility and importance of gut microbiota using the chronic exposure method. Adult male mice were exposed to MC-LR (0, 10, and 100 μg/L) for 6 months. Then, the composition of intestinal flora was evaluated by high-throughput 16S rRNA amplicon sequencing to clarify the influence of MC-LR on gut microorganisms.
Method and Materials
Animal Maintenance and Sample Collection
A total of 15 eight-week-old male, specific pathogens free (SPF) BALB/c mice weighting range from 20 to 25 g were obtained from the Chinese Experimental Animal Center of the Academy of Military Medical Science Institute. The mice were raised in an SPF environment at the animal facilities on a 12-hour light/12-hour dark cycle in a controlled temperature (20 °C-22 °C) and relative humidity (45%-55%). All experiments were applied under protocols approved by the Animal Care and Use Committee of the Zhejiang University. Mice were housed in static polycarbonate cages and fed ad libitum. The SPF diet (Vital River Laboratory Animal Company) was refreshed every 2 days. The wood-chip bedding (Vital River Laboratory Animal Company) and static polycarbonate duplex cages were refreshed every week during the experimental period. Two weeks after adaption to the environment, mice were randomly divided into 3 groups of 5 mice each. The MC-LR (purity >96%) was purchased from Enzo Life Sciences (Dieter Schapfel). The MC-LR was dissolved in a minimum amount of methanol and diluted to required concentration using sterilized reverse osmosis water. The density of methanol is 0.792 g/mL and 20 µL were added to dilute the MC-LR. A 500 mg/L of MC-LR was then diluted to 10 and 100 μg/L. Thus, the highest concentration of methanol is approximately equal to 48.9 μmol/L, which is far lower than the toxic concentration. 11 Mice were exposed orally at 10 and 100 μg/L MC-LR in the drinking water. One literature reference suggests mouse mean daily water intakes 1.5 mL/10 g body weight. 3 The body weights were evaluated once 2 weeks. Thus, the average concentration of MC-LR was intake at 1.5 μg/kg body weight and 15 μg/kg body weight per day according to the previous references. 1 The control animals received the same water containing the same amount of methanol. Right before the mice were euthanized with carbon dioxide, fresh fecal samples were collected in sterile tubes and immediately placed on dry ice. Cecal contents were collected from the cecum upon sacrifice and dissection of the mice. All the samples were stored at −80 °C until use.
DNA Isolation, Polymerase Chain Reaction Amplification, and 16s rRNA Sequencing
Total DNA was extracted using the Qiagen Fast DNA Stool Mini Kit (Qiagen) following the manufacture’s instruction. The DNA concentrations were evaluated through a Nanodrop (Thermo Scientific), and the quality was assessed by agarose gel electrophoresis. The bacteria 16S rRNA gene sequences spanning the hypervariable regions V3-V4 were amplified with primers 338F-806R. 12 Polymerase chain reaction (PCR) reactions were performed in triplicate. The purified amplicons were pooled in equimolar amounts and paired-end sequenced on MiSeq platform according to the standard protocols by Majorbio Bio-Pharm Technology Co Ltd.
Processing of Sequencing Data
Raw FASTQ files were demultiplexed and quality-filtered by Trimmomatic. Operational taxonomic units (OTUs) were clustered with 97% similarity cutoff using UPARSE (version 7.1) with a novel “greedy” algorithm that performs chimera filtering and OTU clustering simultaneously. The taxonomy of each 16S rRNA gene sequence was analyzed by Ribosomal Database Project Classifier algorithm (http://rdp.cme.msu.edu/) against the Silva (SSU123) 16S rRNA database with a confidence threshold of 70%. Alpha diversity indices (Shannon index) and richness estimator (Chao index) calculations were performed.
Total RNA Extraction and Quantitative-PCR
Total RNA was extracted using TRIzol reagent (Invitrogen). The purity of RNA was determined with a spectrophotometer (NanoDrop). Then the RNA was reverse transcribed by a first strand cDNA synthesis kit (Roche). Quantitative PCR (q-PCR) was performed using the SYBR Green qRT-PCR kit (Invitrogen) using a Roche Real-Time PCR System (Roche). The relative quantification values for each mRNA were calculated by the 2−ΔΔCT method using GAPDH as an internal reference. All reactions, including no-template controls, were run in triplicate. The sequences of the primer pairs used in this assay were claudin5, forward primer GCAAGGTGTATGAATCTGTGCT, reverse primer GTCAAGGTAACAAAGAGTGCCA; occludin, forward primer TGAAAGTCCACCTCCTTACAGA, reverse primer CCGGATAAAAAGAGTACGCTGG; ZO-1, forward primer CTCCAGAGCACCGAGAGCTA, reverse primer GGCGTTTGCGAAGTTCTTCAT; and GAPDH, forward primer TGGATTTGGACGCATTGGTC, reverse primer TTTGCACTGGTACGTGTTGAT.
Results
Sequencing Summary
To detect the changes in microbial communities associated with MC-LR exposure, 16S rRNA gene sequencing analysis of gut microbes was conducted in the mice. A total of 525,005 clean sequence reads were obtained, made up of 171,540 sequences from the control group, 174,960 sequences from the 1.5 μg/kg body weight group, and 178,505 from the 15 μg/kg body weight group (Figure 1A). The OTU was used to classify microbial diversity in terms of bacterial strains, based on rRNA sequence similarity. Of the OTUs recognized in control (386), 1.5 μg/kg body weight (447), and 15 μg/kg body weight (451) groups, 353 were found to be common among the 3 groups of mice (Figure 1B).
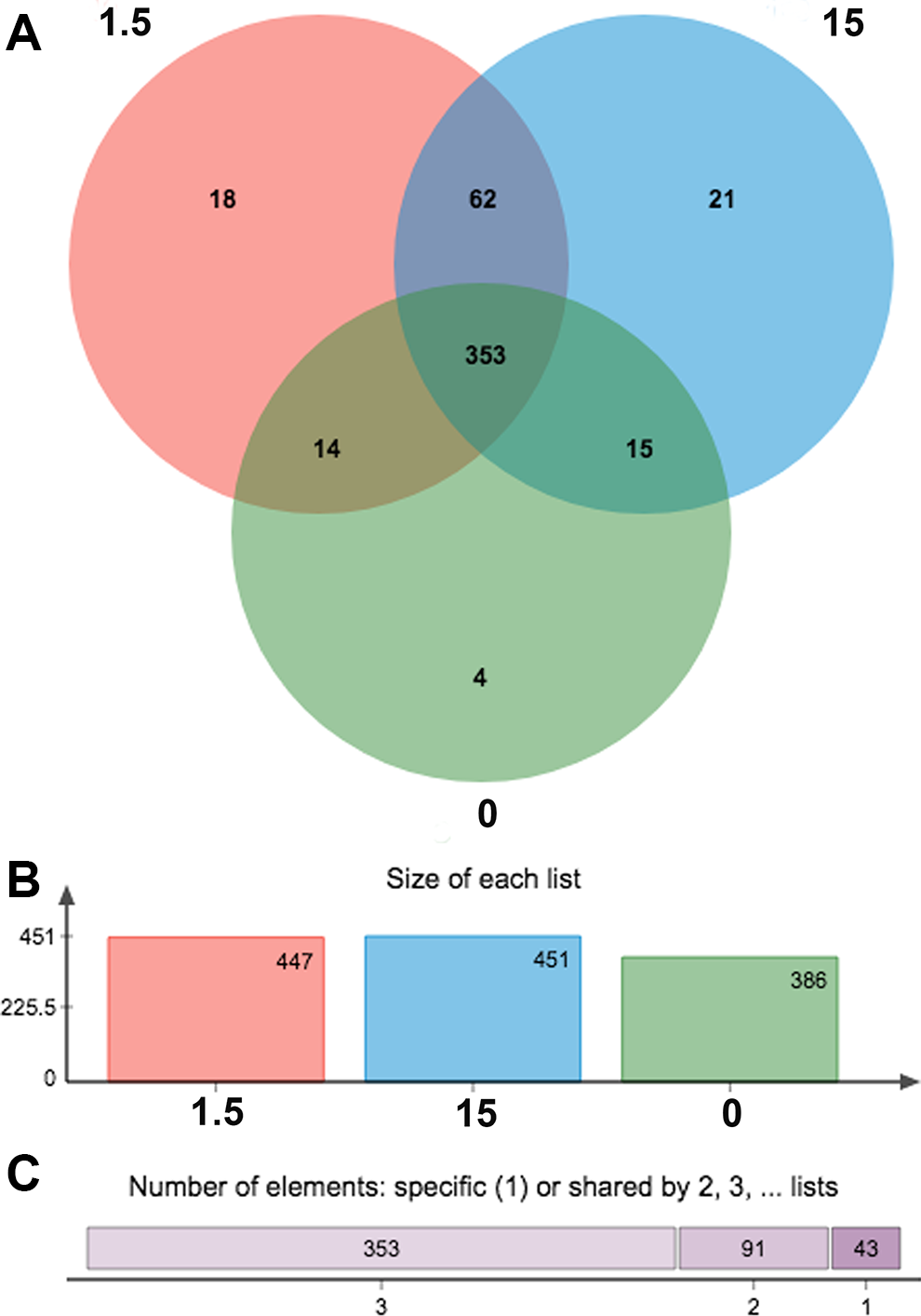
Comparison of operational taxonomic unit (OTU). A, Different color represents different groups (control, 1.5 and 15 μg/kg body weight), the intersection represents the set of OTU commonly present in the counterpart groups. Likewise, the single-layer zone represents the number of OTUs uniquely found in the certain group. B, A bar chart of the total number of species in each group at the selected taxonomy level. C, The abscissa is the number of common or unique groups and the length of upper bar indicates the number of corresponding species.
Effects of the MC-LR on the Number and Diversity of Gut Microbial Species
Alpha diversity was applied for analyzing the complexity of the species diversity. The flattening of rarefaction curve based on the values of observed species demonstrated that our data volume covered all species of the community in the samples (Figure 2A). The species diversity and richness of gut microbial increased both in 1.5 and 15 μg/kg body weight groups according to the Shannon and Chao index (Figure 2B and C). Our data suggested that MC-LR affected the species diversity and richness of the microbial community in the animal gut.
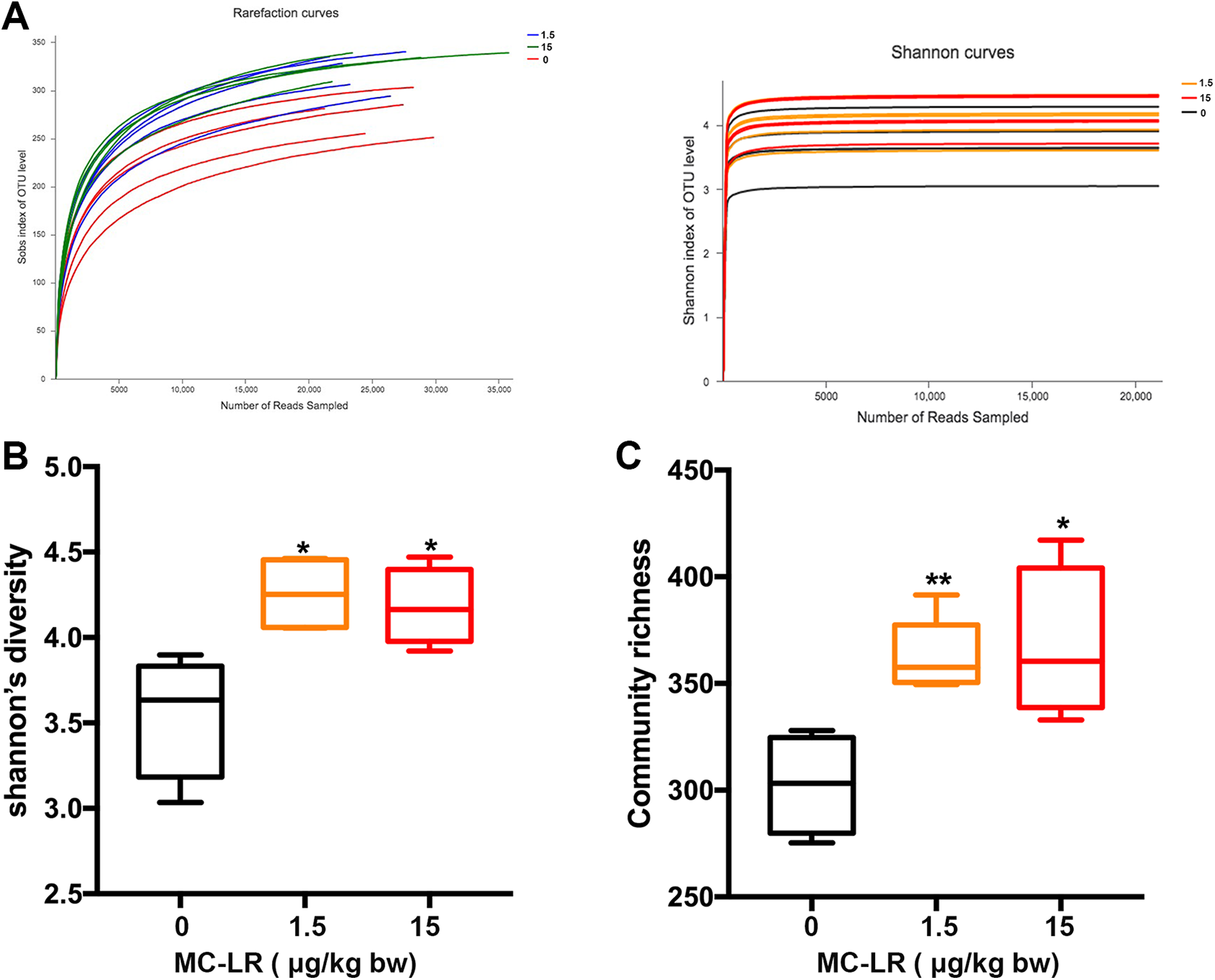
Changes in number and diversity of gut microbial species by various MC-LR exposure. Rarefaction curves based on the (A) observed species values and Shannon indices are used to show that the data volume cover all species in the gut microbial community. The changes in (B) Shannon’s diversity and (C) Chao indices demonstrate that MC-LR increase the species diversity of the gut microbial community. MC-LR indicates microcystin–leucine arginine.
Effects of the MC-LR on Gut Microbial Communities
A comparative analysis of the microbial communities was conducted. Distribution histograms of the taxonomic composition of each sample from the respective groups were constructed from the Phylum and Class levels (Figure 3). After MC-LR treatment, we calculated the abundance ratio of the
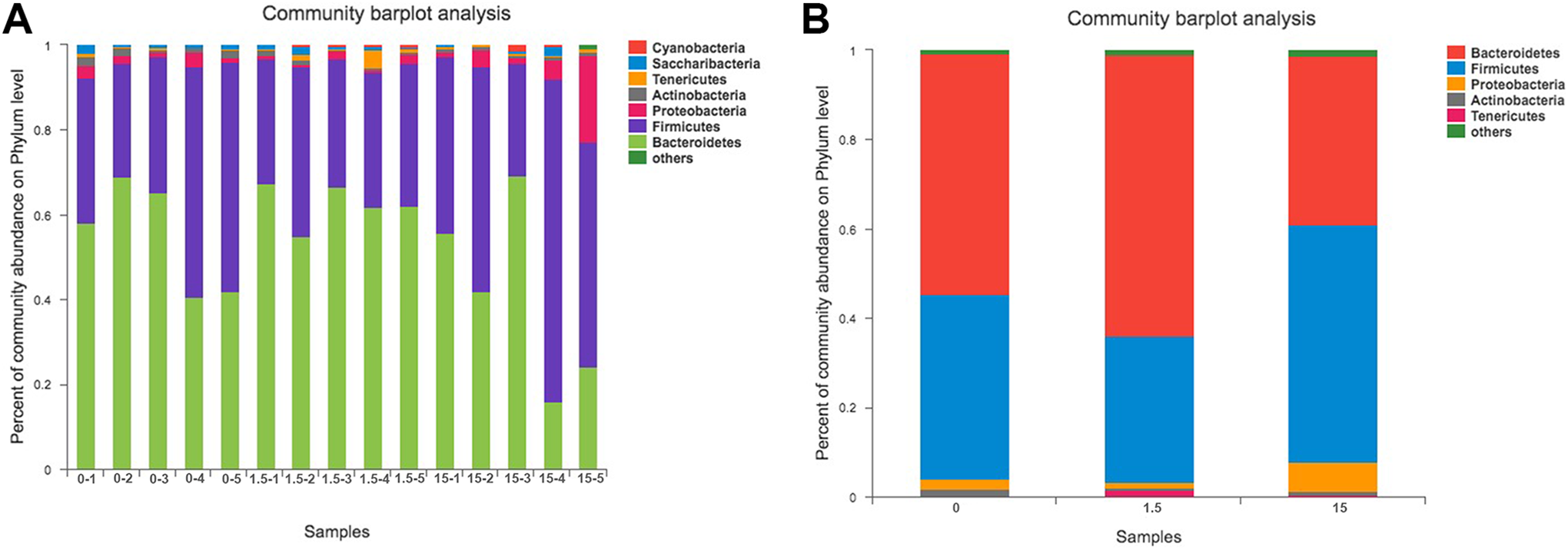
Analysis of the composition of bacteria at the phylum level. A and B,
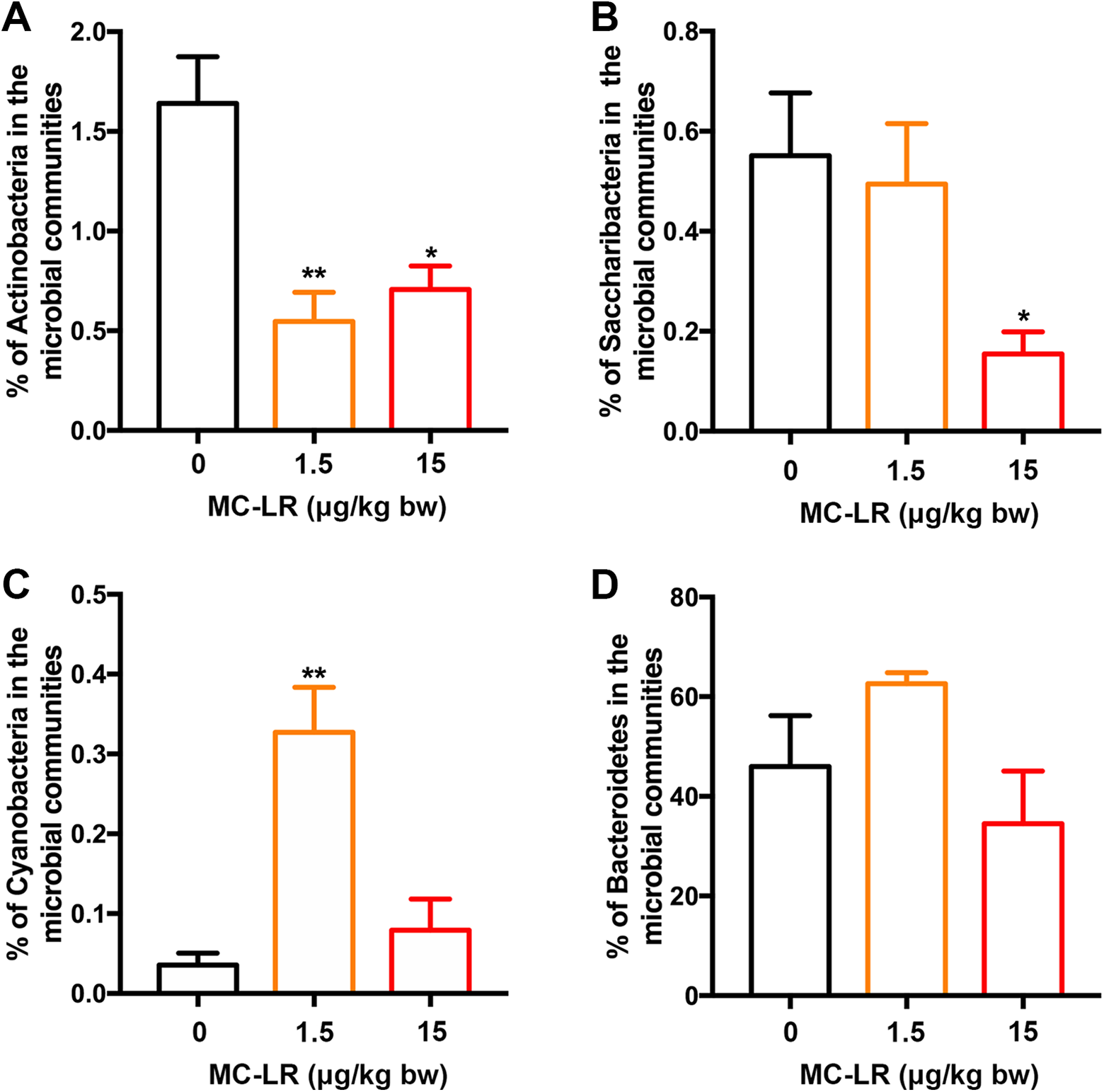
Changes in the percentage of gut microbial communities. A,
Effects of the MC-LR on the Bodyweight and Gut Tight Junction
The bodyweights of mice were significantly altered at 1.5 and 15 μg/kg body weight MC-LR compared with the control after exposed for 6 months (Figure 5A). We analyzed the gene expression of claudin, occlusion, and ZO-1 by q-PCR (Figure 5B-D). After a 6-month treatment, the results showed that the mRNA expression levels of claudin-5, occludin, and ZO-1 were significantly reduced in the gut following exposure to 1.5 μg/kg body weight group.
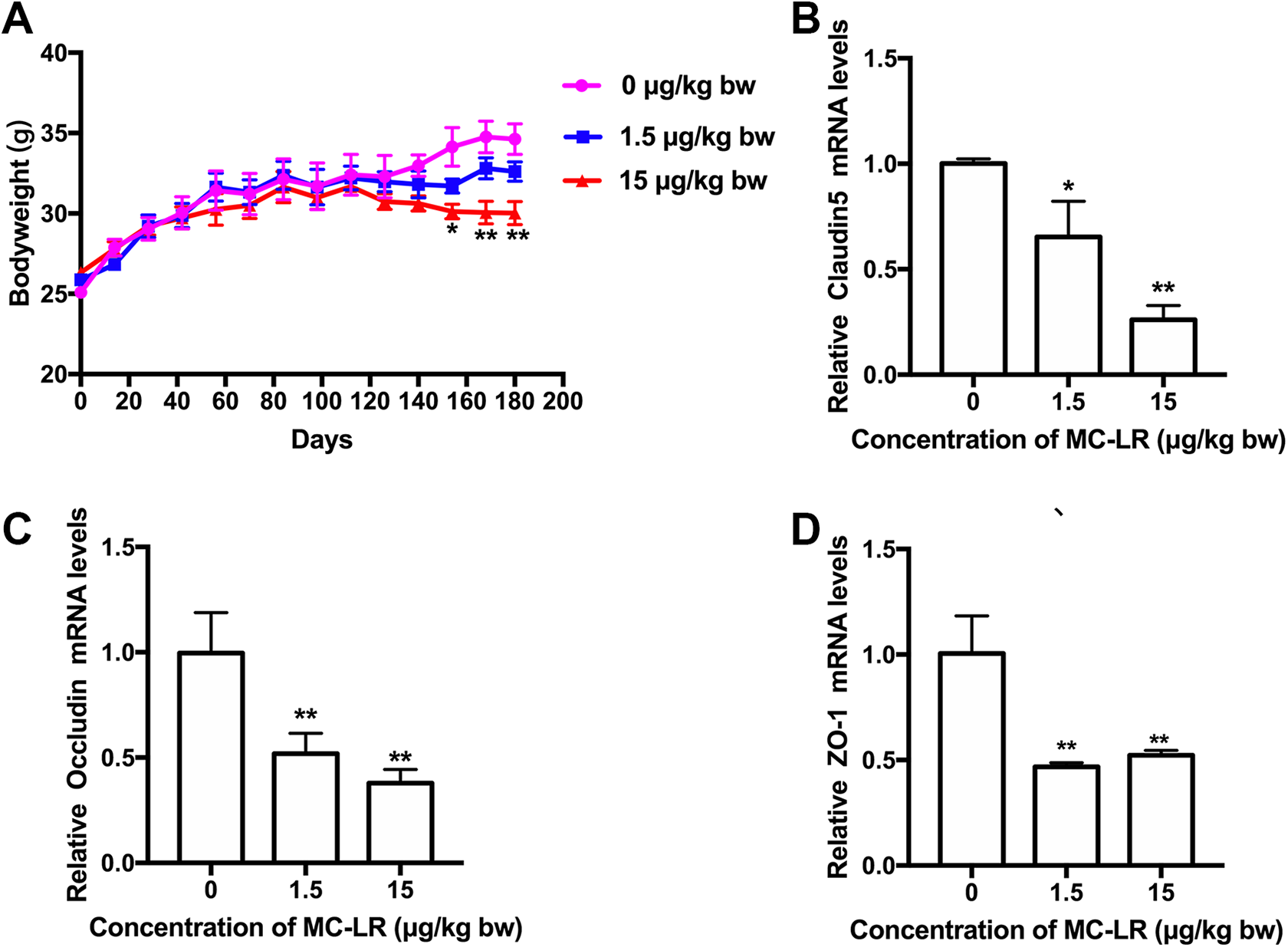
The bodyweight (A) and the mRNA levels of cluadin5 (B), occludin (C), and ZO-1(D) are measured with q-PCR in gut exposed to MC-LR. The data are expressed as the mean ± SD. (
Discussion
Recently, studies of the toxicity of MCs have mostly focused on the acute and subchronic treatment, while the effect of chronic MCs exposure is largely unknown. In this study, we established a chronic MC-LR exposure animal model by treating mice with MC-LR at 1.5 or 15 μg/kg body weight in the drinking water for 6 months, which mimics the natural exposure to MC-LR, and evaluated the effects of MC-LR on intestinal microbes. Our results demonstrated that chronic MC-LR exposure in mice significantly changes the number of bacterial species in the gut. Consistent with previous studies, we observed an increased species diversity and abundance of gut flora, and significantly decreased body weight in mice with chronic MC-LR treatment. 1 Given that the gut microbe diversity is strongly associated with individual body weight, 13,14 our findings suggest that chronic MC-LR exposure induces body weight loss in mice through changing the microbe diversity.
Magnitudes of toxic effects, induced by MCs, depend on route and magnitude of exposure to the toxin.
1
In the study by Chen et al, intragastric administration was used.
9
Animals and humans are more likely to consume low doses of MC-LR contaminated food or water.
1
Thus, we conducted a chronic exposure of mice to MC-LR by oral exposure in the drinking water. Using 16S rRNA gene sequencing analysis on cecum samples of mice, our results showed that MC-LR significantly increase species diversity, which was similar to the previous study. However, the kinds of altered microbial communities in our study is different from the previous one. In the present study, the
The gut barrier is a functional unit that is exposed to the external environment daily.
16
It is mainly composed with the tight junctions of epithelia cells.
16
Intestinal TJs barrier dysfunction or increased permeability plays an important role in the pathogenesis of various intestinal diseases, including the celiac disease and Crohn disease.
4
In the present study, we found that the expression of the tight junction genes was significantly reduced in the gut after 6-month MC-LR treatment, which was similar to a previous in vitro study.
4
However, the underlying mechanism of MC-LR-caused tight junction dysfunction is still unclear.
In summary, our present study demonstrated that low-dose, chronic MC-LR exposure significantly changed the composition of the intestinal flora in mice, drawing attention to the impact of environmental pollutants on the pathogenesis of gut microbiota-associated intestinal diseases.
Footnotes
Author Contribution
Zhuang, Lilei contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Jin, Zhonghai contributed to interpretation and critically revised manuscript. Li, Hongguang contributed to analysis and drafted manuscript. Wu, Shenbao contributed to analysis and critically revised manuscript. Tong, Xiuping and Wang, Haiying contributed to analysis and interpretation and drafted manuscript. Li, Miaomiao contributed to conception and design; acquisition, analysis, and interpretation; and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
