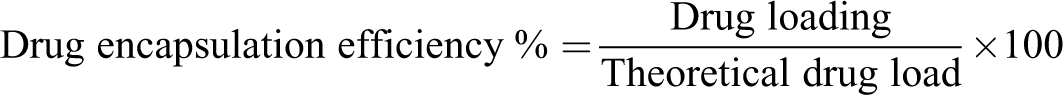Abstract
Clinical use of the chemotherapeutic agent vincristine (VCR) is limited by chemotherapy-induced peripheral neuropathy (CiPN). A new formulation of VCR encapsulated by nanoparticles has been proposed and developed to alleviate CiPN. We hypothesized in nonclinical animals that the nanoparticle drug would be less neurotoxic due to different absorption and distribution properties to the peripheral nerve from the unencapsulated free drug. Here, we assessed whether VCR encapsulation in nanoparticles alleviates CiPN using behavioral gait analysis (CatWalk), histopathologic and molecular biological (RT-qPCR) approaches. Adult male C57BL/6 mice were assigned to 3 groups (empty nanoparticle, nano-VCR, solution-based VCR, each n = 8). After 15 days of dosing, animals were euthanized for tissue collection. It was shown that intraperitoneal administration of nano-VCR (0.15 mg/kg, every other day) and the empty nanoparticle resulted in no changes in gait parameters; whereas, injection of solution-based VCR resulted in decreased run speed and increased step cycle and stance (P < 0.05). There were no differences in incidence and severity of degeneration in the sciatic nerves between the nano-VCR-dosed and solution-based VCR-dosed animals. Likewise, decreased levels of a nervous tissue-enriched microRNA-183 in circulating blood did not show a significant difference between the nano- and solution-based VCR groups (P > 0.05). Empty nanoparticle administration did not cause any behavioral, microRNA, or structural changes. In conclusion, this study suggests that the nano-VCR formulation may alleviate behavioral changes in CiPN, but it does not improve the structural changes of CiPN in peripheral nerve. Nanoparticle properties may need to be optimized to improve biological observations.
Introduction
Chemotherapy-induced peripheral neuropathy (CiPN) is a common adverse event, which inconsistently results in dose-limiting adverse effects due to severe disturbance of functional or ultrastructural changes in the peripheral nerves. 1,2 The prevalence of CiPN in patients with cancer varies considerably from 10% to 100%, depending upon the particular chemotherapeutic drug or drug combination administered, the dosing regimen, the methods of pain assessment, and the particular patient situation. 3 Furthermore, these trends are rising, as many forms of cancer are becoming chronic conditions. 4 CiPN may lead to dose reduction of chemotherapeutic drugs or the possible cessation of therapy, which may impact success of cancer treatment and negative disease outcomes. 5 To date, it remains difficult to elucidate the exact mechanisms or pathophysiology of CiPN, including vincristine (VCR) induced peripheral neuropathy. 6 It is generally believed that axonopathy through axonal damage is involved. Neuroprotective agents for VCR-related neurotoxicity, including folinic acid; vitamins B1, B6, and B12; isaxonine; glutamic acid; gangliosides; adrenocorticotropin hormone analogues; insulin-like growth factor 1; and nerve growth factor, have been tested in experimental setting but have not been deemed sufficiently efficacious for clinical use. 7 Vitamin-E 8,9 and selenium 10 have also been proposed and tested in several clinical trials to protect from CiPN, but none has been tested in VCR-induced CiPN patients. Therefore, prevention or treatment of VCR-induced peripheral neuropathy remains a challenge. 11,12
Vincristine has been one of the most common anticancer drugs widely used since 1962 in the treatment of many types of cancers such as leukemias, lymphomas, and other types of neoplastic processes. A common side effect is a dose- and duration-dependent peripheral neuropathy, which frequently results in an autonomic and peripheral sensory-motor polyneuropathy. 13 This effect is caused by its antimitotic property, and, in part, explains the neurotoxic effects. 14 -16 The antitumor action of VCR is due to its binding to β-tubulin 17 which also leads to disorganization of the microtubule cytoskeleton in the axons of the peripheral nerves. The most common clinical consequences of neurotoxicity include loss of deep tendon reflexes which may be associated with foot drop and gait disturbance, jaw pain, constipation, and ileus. Currently, novel tubulin inhibitors with more effective antitumor activity and less neurotoxicity have been explored, 18 but these have not been tested in the clinic. Currently, there are no effective preventive or treatment options for CiPN, forcing many patients to either reduce or discontinue use of these potentially highly effective and curative antitumor, neurotoxic drugs. 19,20 Even symptomatic treatment of pain in CiPN may be difficult.
Given the absence of effective therapies for CiPN, the modification in formulations of VCR aimed at preventing or delaying the onset of neuropathy has potential to enhance the quality of life for patients with cancer. Equally important, if tolerability were improved by lessening the frequency or occurrence of neuropathy, physicians would be more inclined to increase dose level or duration and enhance tumor cell killing. Indeed, modified formulations of VCR have already been tested. For instance, VCR has been encapsulated in sphingomyelin/cholesterol, distearoylphosphatidylcholine/cholesterol (DSPC/Chol) liposomes, 21 and sulfate liposome. 22 The most investigated formation is liposomal VCR, which is a 3-part formulation. However, it needs to be prepared by the pharmacist prior to administration. Once prepared, the drug is stable for less than 12 hours at room temperature. 23 The safety profile of this investigative formulation of VCR has not been thoroughly evaluated in animal nonclinical studies nor clinically in humans. 23 A new polymer-based nanoparticle formulation of docetaxel has been proposed and developed based on encapsulation of this drug in nanoparticles. 24 We hypothesized that nanoparticle VCR might be less neurotoxic by differentiating absorption and distribution to the peripheral nerve compared with the unencapsulated (solution-based) drug.
In drug discovery and development, histopathology has been the most commonly used method in assessing axonal degeneration or demyelination of the peripheral nerves, and sometimes, neuronal degeneration in the dorsal root ganglia (DRG) in neurotoxicity studies. 25 The behavioral changes in CiPN animals have often been observed, 26 and, in recent years, gait analysis has been used in evaluating the behavioral changes in the rodent model of CiPN. 27,28 The expression of microRNA (miR)-124, miR-338, and miR-183 has been reported highly expressed in neurons and peripheral nerves compared with that of other organs in mice. 29,30 MicroRNAs (miRNAs) appear in extracellular fluid once cellular membrane integrity is compromised. Global changes in miRNA expression in DRG have been reported in a variety of peripheral nerve injury models. 31,32 Nerve injury has also been shown to cause changes in miRNA expression in the sciatic nerves 33 and circulating blood. 34 Furthermore, in an in vitro study, miR-338 was shown to enhance axonal regeneration and myelination in neurons. 35 In this study, we assessed whether VCR encapsulation in nanoparticle alleviates CiPN using behavioral gait analysis, histopathologic and molecular biological biomarker approaches.
Methods
Preparation of VCR-Encapsulated Nanoparticles
Vincristine-encapsulated nanoparticles (NP-639) were created via a nano-emulsion process using the modified oil in water (o/w) emulsification solvent extraction method as described by Song et al 36 and Ashton et al. 37 Briefly, an organic phase composed of VCR sulfate, decanoic acid (hydrophobic counterions to form ion-pairing with VCR), and 16–5 PLA–PEG in an EA/BA solvent mixture was rapidly mixed and dispersed with an immiscible aqueous phase containing the optimal concentration of surfactant (BrijS 100) for particle size control. First, a coarse emulsion was formed by a handheld rotor/stator homogenizer. This coarse emulsion was subsequently processed by a high-pressure microfluidizer (Microfluidics, Inc) to form a fine nano-emulsion. The emulsions were quenched by addition of 0.17M phosphate buffer pH 6.5 quench solution, resulting in extraction of solvent from the organic phase and hardening of nanoparticles. Subsequent processing included tangential flow filtration, which removes unencapsulated active pharmaceutical ingredient and processing aids, and addition of sucrose for stabilization and cryopreservation of the nanoparticle suspension. The nano-emulsion process of ACCURINS is shown schematically in Figure 1. Empty nanoparticles were essentially polymer only (without VCR sulfate nor decanoic acid) with no additional excipients, and empty nanoparticles were created via a same nano-emulsion process described above.
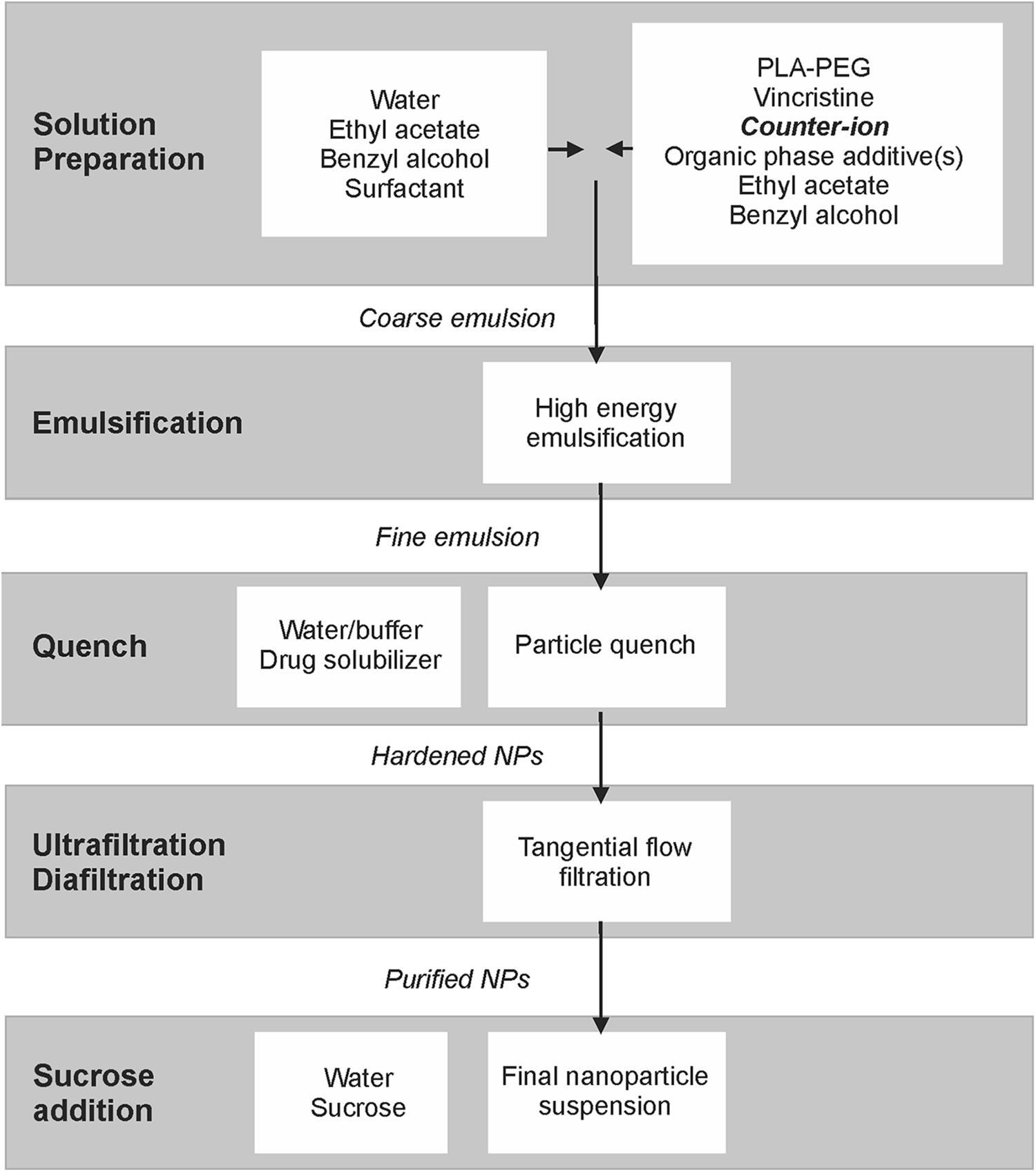
Flowchart of VCR nanoparticle preparation. NP = nanoparticle.
Nanoparticle Characterization
Vincristine nanoparticle formulations were characterized with respect to particle size, drug load, and in vitro release kinetics as described in Song et al. 36 Briefly, particle size was determined by dynamic light scattering of a dilute aqueous suspension at 25 °C on a Brookhaven ZetaPALS instrument with a 660 nm laser scattered at 90°. Dynamic light scattering data were analyzed using the cumulants method. Three measurements are performed on each sample and the mean and standard deviation were reported as well as the polydispersity index. Zeta potential was also measured on this instrument for select samples. The VCR load was calculated from the ratio of VCR to the gravimetric dry weight of the sample prior to adding sucrose to the suspension.
Vincristine release kinetics were determined in vitro under physiological sink conditions. Nanoparticles were suspended in 10% urea in 0.01M phosphate-buffered saline (PBS, pH 7.4) and incubated with mild agitation in a 37 °C water bath. Periodically, an aliquot of the suspension was removed and ultracentrifuged at 264,000 g for 30 minutes. Samples of the supernatant and the suspension prior to ultracentrifugation were analyzed by ultra-performance liquid chromatography, and the percent release was calculated by comparing the released VCR concentration in the supernatant with the total concentration in the uncentrifuged sample.
Experimental Animals
Twenty-four adult male C57BL/6 mice (Charles River Laboratories) weighing 25 to 32 g (6-7 weeks old) at the start of the study were used in this study. Mice were pair-housed in individually ventilated disposable plastic cages (Innovive) on paper bedding (ALPHA-dri, Shepherd Specialty Papers Inc) with ad libitum access to sterile feed (LabDiet 5053) and water (Innovive) in animal rooms with controlled temperature (20 °C-26 °C) and humidity (30%-70%) and a 12/12 h light/dark cycle. All activities involving animals were carried out in accordance with federal, state, local, and institutional guidelines governing the use of laboratory animals in research in an American Association for Accreditation of Laboratory Animal Care (AAALAC)-accredited facility following the Guide for the Care and Use of Laboratory Animals (Institute for Laboratory Animal Research, 2011) and were reviewed and approved by Pfizer’s Institutional Animal Care and Use Committee.
Animal Study Design
Three groups, each comprising 8 animals, were employed in this study. Group 1 served as empty nanoparticle control group in which animals received NP-684 (235 µL NP-684 + 14.765 mL PBS) administration for 7 doses at 10 mL/kg via intraperitoneal injection (IP) every other day. Group 2 served as nanoparticle VCR treatment group in which animals were administered NP-639 every other day at dose of 0.15 mg/kg for totally 7 doses at dose volume of 10 mL/kg via IP. Animals in group 3 were dosed via IP route with solution-based VCR sulfate (Sigma) in the same dose regimen as group 2, which was adopted from our 27 and other’s 38 -40 previous VCR studies. The solutions were prepared at a volume of 10 mL/kg with PBS being used as the vehicle. In order to assess general toxicity and potential mortality, animals in all groups were closely monitored daily, while body weight and food consumption were measured every other day (except on weekends to mimic clinical dose regimen). On day 16, all animals were euthanized with an overdose of inhaled CO2. Nervous tissues and blood were collected for histopathologic evaluation and miRNA quantification, respectively.
CatWalk Gait Analysis
The method was adapted from previously reported literature. 27,41 -43 The animals were trained in 3 sessions prior to the initiation of dosing to capture consecutive uninterrupted runs on the 1.5-m long walkway of a CatWalk unit (Noldus Information Technology). Baseline values were obtained from the last training session. Following the first dose, the animal’s gait behavior was tested on days 4, 10, and 15. The cutoff time for running was 5 seconds. On testing days, a total of 2 or 3 uninterrupted runs constituted an acceptable run for each mouse. Data were collected and analyzed using CatWalk XT 10.6 software (Noldus Information Technology). Automated analyses of the recorded videos yielded many parameters, of which the following were of most interest for this study: swing and stance, stride length, print area, and print intensity. 44
Anatomic Pathology
Following euthanasia on day 16, left lumbar 4 (L4) and L5 DRG and sciatic nerve samples were collected from all animals for pathologic evaluation. Samples of sciatic nerve were carefully dissected from the proximal aspects of the thigh to the knee joint just proximal to the point of division into the common peroneal, tibial, and sural nerves; the middle segment of this sample was collected and fixed in 4% methanol-free formaldehyde. DRGs were also fixed in this fixative. Tissues were processed, embedded in paraffin, sectioned only longitudinally at 5-µm thickness and stained with hematoxylin and eosin as described by previous investigators 45 for microscopic examination. The nerve and DRG sections were evaluated under light microscopy and images were scanned using a Leica Aperio AT2 whole slide digital scanner and acquired using ImageScope viewing software (Leica Biosystems). Microscopic lesions were graded on a scale of 1 to 5 as minimal, mild, moderate, marked, or severe first by a board-certified pathologist. Subsequently, a blinded, board-certified pathologist performed peer-review.
Plasma and Tissue MicroRNA Measurement
Right L4 and L5 DRGs and whole blood samples were collected from all animals for miRNA quantification. Plasma RNA was extracted as previously described 43,46 and tissue RNA extraction was conducted on DRG. 43 Briefly, right L4 and L5 DRGs were dissected under a dissection microscope, placed in Ambion RNAlater solution (Cat #: AM70201, Thermo Fisher Scientific) at 4 °C overnight, and then transferred to −20 °C freezer until further tissue processing. Frozen tissues were transferred into tubes containing Qiazol and one stainless steel bead (5 mm, cat #: 69984, Qiagen) and exposed to frequency of 25 Hz Qiagen TissueLyser II for 10 minutes. Total RNA was extracted from the tissue lysate or 100 µL of plasma using Qiagen’s miRNeasy kit according to the manufacturer’s protocol. One hundred ng of the purified total RNA from the tissues or 5 µL of total RNA purified from plasma was then subjected to quantitative real-time polymerase chain reaction (RT-qPCR). Quantification of miRNAs was performed in duplicate in the final PCR reactions. All reactions were run on a ViiA 7 Real Time PCR System (Thermo Fisher Scientific) using the following conditions: 95 °C for 10 minutes, followed by 40 cycles at 95 °C for 5 seconds, and then 60 °C for 30 seconds. Data are presented as relative copy number. Standard curves were generated using synthetic RNA oligos. Reference RNAs such as endogenous miRNAs of miR-92, miR-181, and miR-192, and exogenous spiked-in Cel-miR-39 were detected in plasma samples, while endogenous U6 snRNA was quantified with tissue samples for normalization purposes. 47 miR-124, miR-338, and miR-183 were quantified.
Statistical Analysis
All the results were reported as mean ± standard error of the mean (SEM). The data were statistically analyzed by Student t-test and 1- or 2-way analysis of variance (ANOVA) followed by Bonferroni post hoc test using Graphpad Prism 8 software (GraphPad). P values of less than 0.05 (*) were considered statistically significant.
Results
In Vitro Nanoparticle Characterization
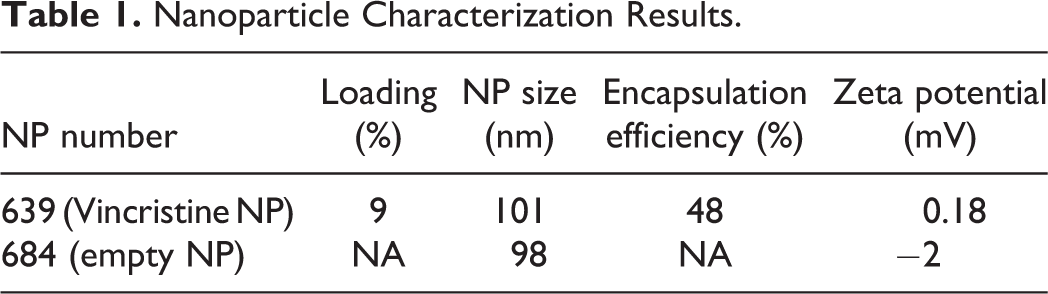
The results of encapsulated VCR and empty NP are summarized in Table 1. In vitro release profiles of the batch prepared with VCR and decanoic acid by hydrophobic ion pairing are displayed in Figure 2.
Nanoparticle Characterization Results.
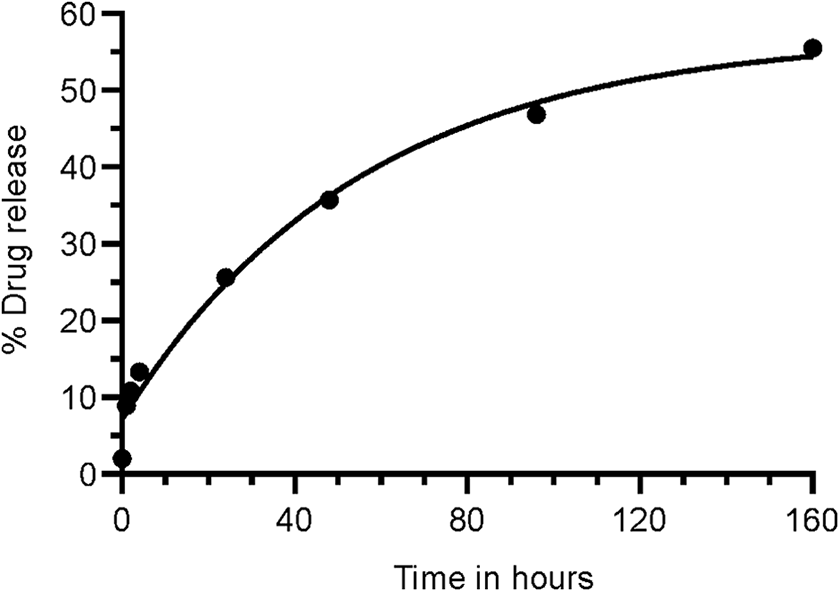
In vitro cumulative release of vincristine (VCR) over time up to 160 hours (7 days). Y-axis shows VCR nanoparticle (NP-630) in vitro release (IVR) at 37 °C in 10% urea in PBS. X-axis shows time in hours. The curve was fitted with Boltzmann sigmoidal equation (GraphPad Prism 8).
General Toxicity
As reported in our previous study on C57BL/6 mice, no body weight gain (Figure 3A) or daily food consumption (Figure 3B) changes were observed following injection of solution-based VCR at 0.15 mg/kg. 27 Likewise, in the animals dosed with nano-VCR or empty nanoparticle, body weight gain or food intake was not altered either following injections (all P > 0.05). Furthermore, no noticeable test article-related clinical signs or symptoms were reported in any animal dosed.
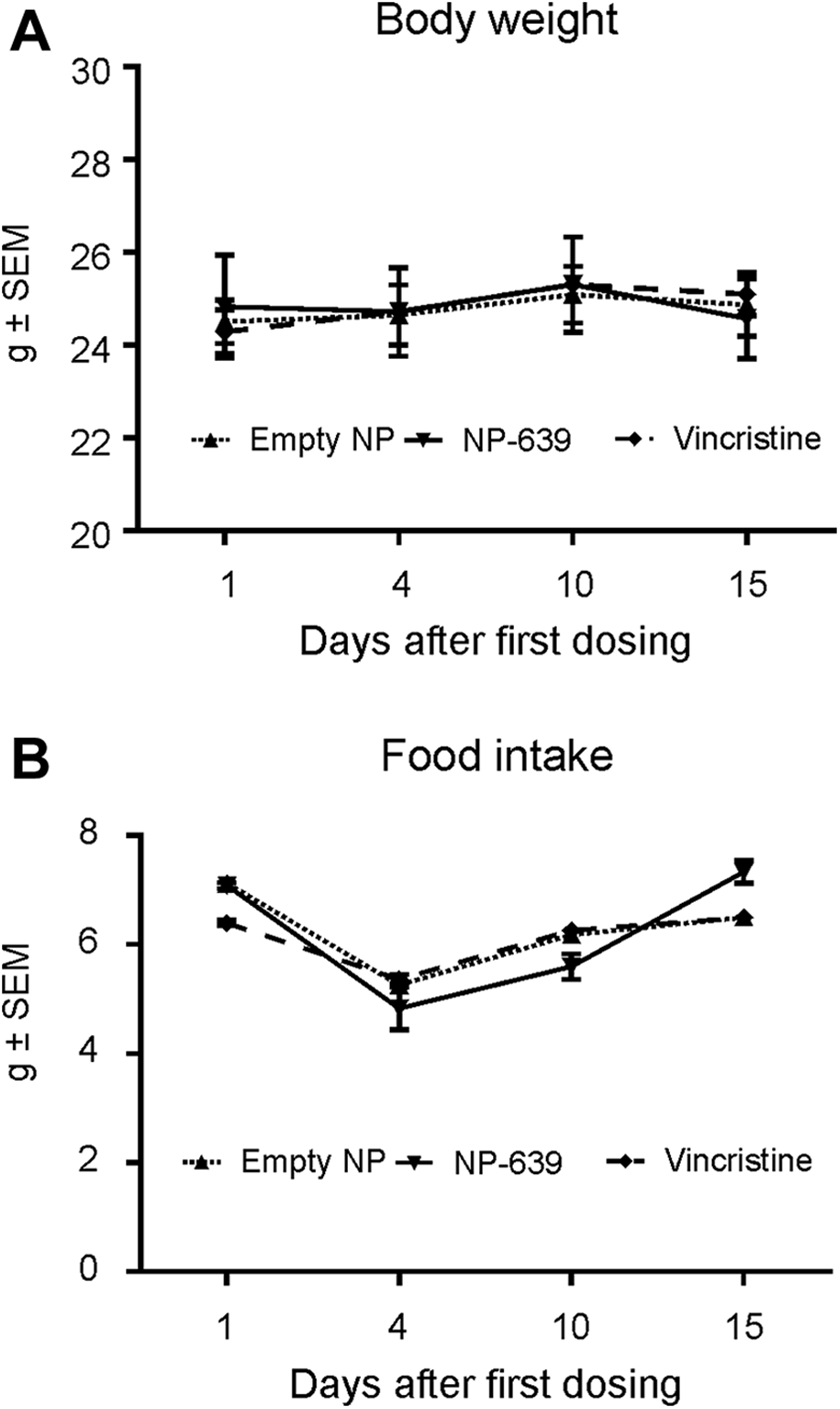
General toxicity of adult C57BL/6 mice following solution-based-, nano-vincristine (VCR), and empty nanoparticle. No changes in body weight (A) or daily food consumption (B) were observed following injection of empty nanoparticle, nano-VCR, or solution-based VCR (all P > 0.05).
In Vivo Gait Analysis
Only the mice dosed with solution-based VCR (group 3) showed a statistically significant decrease in walk/run speed on days 10 and 15 (P < 0.05 and P < 0.01, respectively, compared with the baseline on day 1, Figure 4). The ambulation effects observed were considered to be resulting from treatment with the solution-based VCR, since body weight changes were not observed. These effects were likely not the result of general poor health due to the VCR treatment as reported earlier in other studies with similar dose regimen. 27,48,49 Mice dosed with solution-based VCR also showed a statistically significant increase in duration of step cycle in all limbs on day 10 (Figure 5A-D) and in the hind limbs on day 15 (Figure 5C and D). In addition, mice in this group showed a statistically significant increase in stance time in the front limbs on day 10 (Figure 5E and F). There were no changes in any gait timing parameters evaluated in all animals dosed with empty NP or nano-VCR (Figure 5A-H). We did not detect changes in other gait parameters, that is, swing time, stride length, print area, and print intensity in any group.
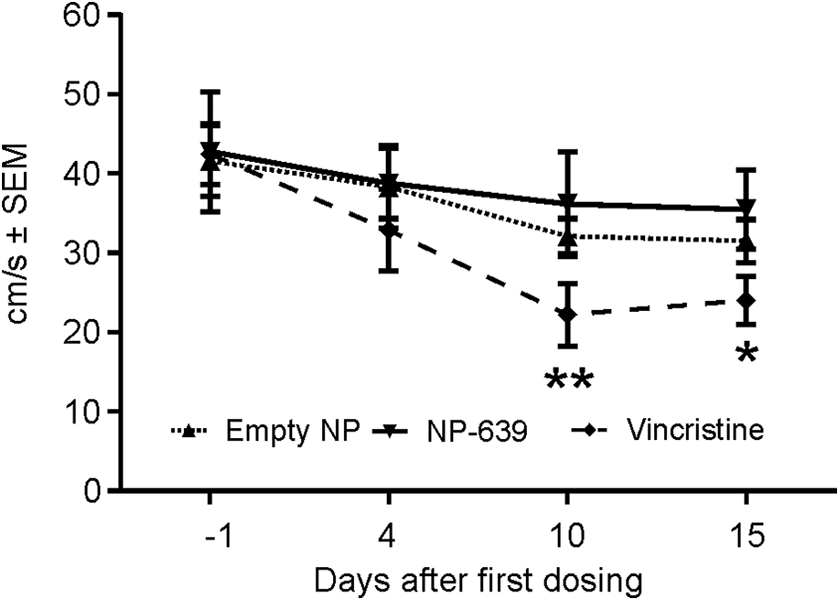
Running speed. Only the mice dosed with solution-based vincristine (VCR; dotted dash line with filled rhombuses) showed a statistically significant decrease in walk/run speed on days 10 and 15. *P < 0.05; **P < 0.01, compared with the baseline values on day 1. There was no statistically significant decrease in running speed in animals dosed with nano-vincristine (solid line with filled upside-down triangles) or empty nanoparticles (dash line with filled triangles).
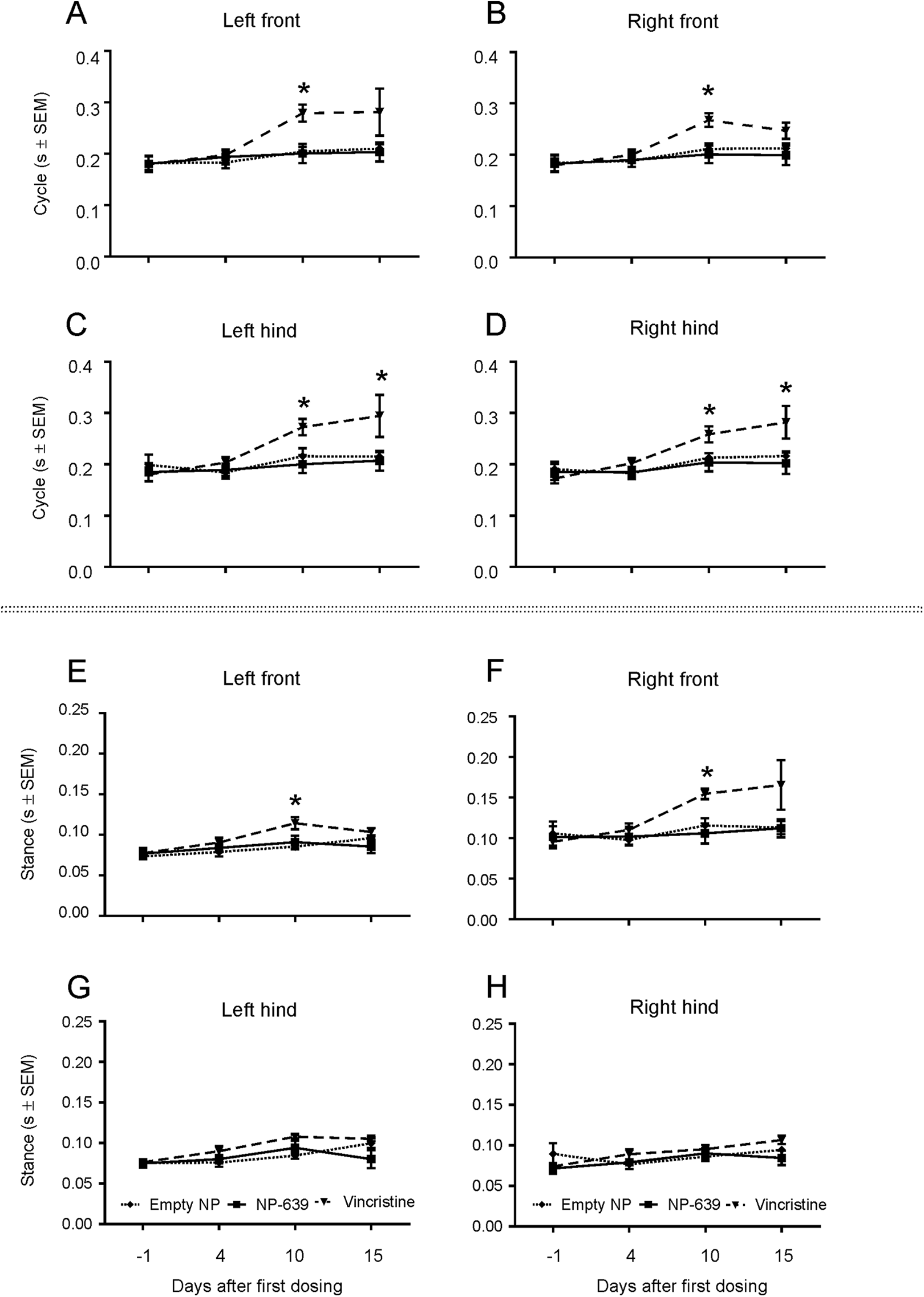
Gait analysis. Mice dosed with solution-based vincristine (VCR; dotted dash line with filled upside-down triangles) showed a statistically significant increase in duration of step cycle on days 10 (front and hind limbs) and 15 (hind limbs, A-D) and in stance time in the front limbs on day 10 (E and F), but not in the hind limbs (G and F). *P < 0.05. There were no statistically significant changes in step cycle or stance time in animals dosed with nano-vincristine (solid line with filled squares) or empty nanoparticles (dash line with filled rhombuses).
Histopathologic Evaluation of DRG and Sciatic Nerve
There were no macroscopic findings related to administration of NP-639 or solution-based VCR. No microscopic findings were noted in DRG in all dosed animals. Additionally, there were no microscopic findings in the sciatic nerves from animals dosed NP-684, that is, empty nanoparticle controls (Figure 6A). Nerve fiber (axonal) degeneration was present in the animals administrated NP-639 (minimal to mild in severity) or solution-based VCR (minimal to moderate) in the sciatic nerve (group 2 and 3, Figure 6B and C, Table 2) with increased severity and/or incidences compared with controls. Axonal degeneration was characterized by focal to multipleaxon fragmentation and myelin disruption or vacuolation (digestion chambers as shown in the insets of Figure 6B and C).
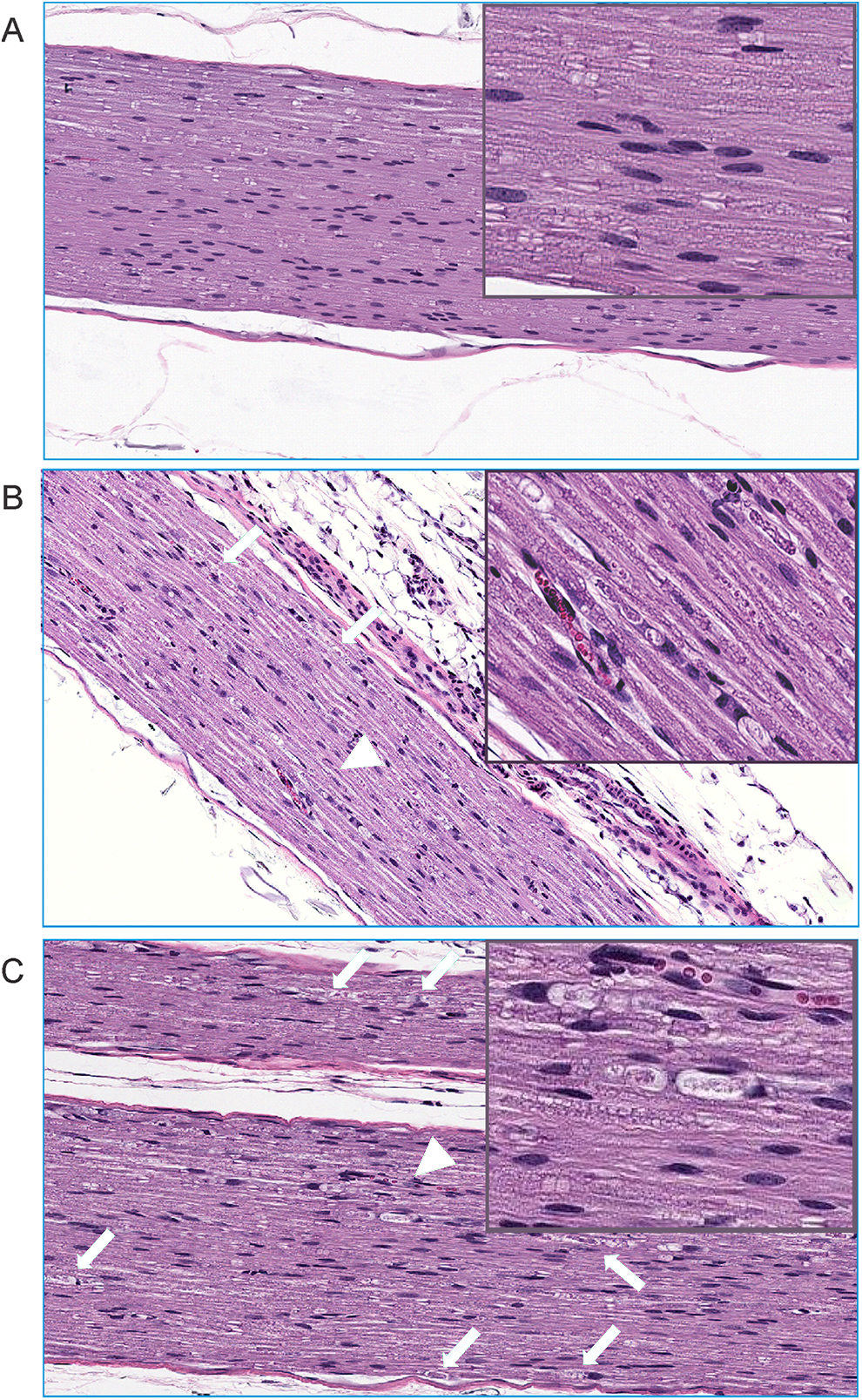
Illustrative micrographs of microscopic findings of nerve degeneration in the sciatic nerve in mice dosed with empty nanoparticle (A), nano-vincristine (VCR; mild degeneration, B) or solution-based VCR (moderate, C) at ×10 magnification. Green arrows and arrow heads in B and C show nerve fiber degeneration characterized by presence of digestion chamber containing fragments of degenerate axon and/or myelin and adjacent Schwann cells with enlarged nuclei. The insets in B and C show the arrowhead-pointed digestion chambers at higher magnification.
Group Incidences (With Severities) of Microscopic Findings in Sciatic Nerve.
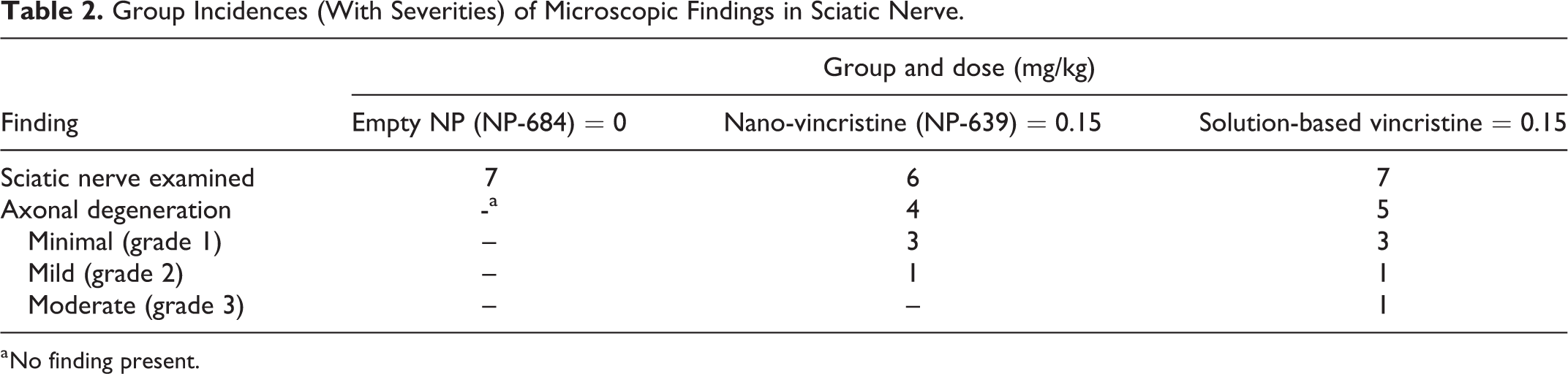
a No finding present.
MicroRNA Changes in the Circulating Blood and DRG
MiR-183 levels in plasma collected on day 16, but not in DRG, of animals dosed with nano-VCR and solution-based VCR showed statistically significant decrease (P < 0.05) compared with that of animals dosed with empty-NP (Figure 7). There was no difference in other miRNAs tested (miR-338 and miR-128) levels in plasma or DRGs.
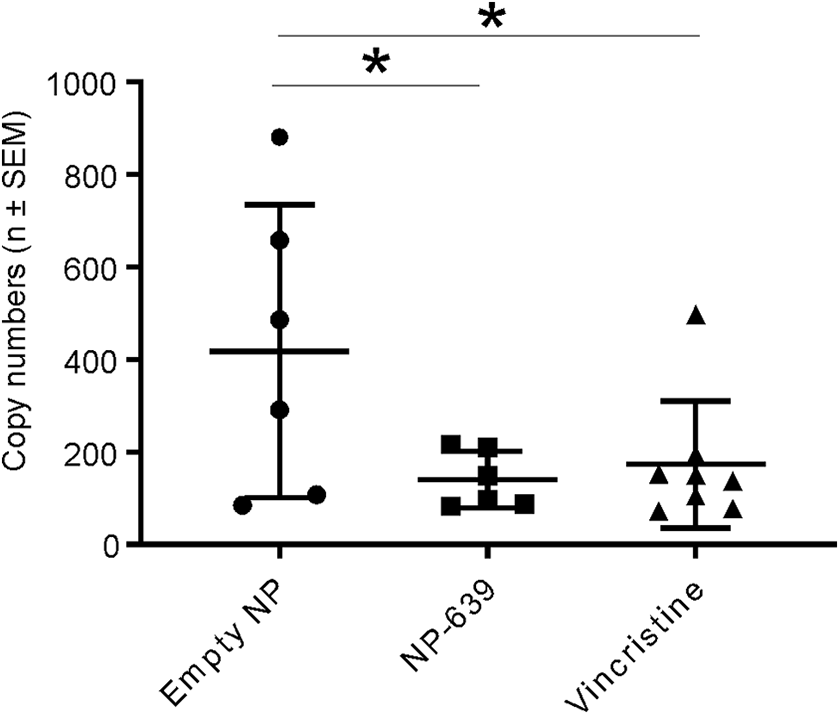
MiR-183 quantification in plasma. MiR-183 levels in the plasma collected on day 16 of animals dosed with nano-vincristine (VCR) and solution-based-VCR showed statistically significant decrease (*P < 0.05, 1-way analysis of variance [ANOVA]) compared with those in empty-NP doses animal.
Discussion
The present study demonstrated that administration of nano-VCR was associated with reduced behavioral evidence of allodynic pain with unexpectedly decreased miR-183 and unexpected histologic evidence of nerve fiber injury in the sciatic nerve of mice. This suggests that polymer-based nanoparticle formulation of VCR may functionally, but not structurally, alleviate CiPN toxicity of VCR treatment in mice.
One functional hallmark of murine CiPN is mechanical allodynia, 50 and automated gait analysis with the CatWalk technique has been used previously to detect gait alterations induced by VCR in C57BL/6 mice. 38 In this historical study, an increase in the swing phase was observed as well as a decrease in the stance phase (6%) and duty cycle (16%) after 10 days of VCR administration. In our current study, we also detected gait alternations after day 10, but we observed increases in cycle and stance phases, and observed decreases only in walking speed. In our analysis, we measured the gait parameters in each individual limb and the observed changes occurred symmetrically in both fore and hindlimbs, which is similar to the polyneuropathy characterized by numbness and tingling in both hands and feet reported by most CiPN patients. However, the differences in the gait parameters between the front limb and hind limbs is intriguing. It might reflect anatomic or physiologic differences between the muscles or motor nerves innervating the limbs. This discrepancy might be due to the dose regimens. In the previous study, the researchers tested the animals after single dose at 200 µg/kg, but in this current study we administered 7 doses at 150 µg/kg/d, which is more similar to the dose regimen often reported by the majority of citations. 48,51,52 Technically, comparing the results across laboratories and investigators is difficult since the inconsistencies can be attributed to the lack of standardization for animal age, weight, sex, and drug dosing regimen. 53 However, like this study and our previous study, 27 we did not observe changes in behavior or body weight gain even at higher concentration (0.15 vs 0.10 mg/kg we used previously. 43 ). This implies that solution-based VCR-induced gait changes were due to peripheral sensory or motor peripheral neuropathy, in particular, allodynia or pain as reported previously, 50,51 rather than general health decline.
Less translatable than the behavioral end points discussed above, microscopic lesions in the peripheral nerves, for example, sciatic nerve, have been reported in VCR-induced neuropathy rodent models 38,50 and in vitro. 54 It has also been reported that VCR administration damaged Schwann cells 55 and DRG neurons 56 in mice. Surprisingly, we did not notice any pathological changes in the soma of the primary sensory neurons in DRGs. There were typical microscopic changes indicative of nerve fiber degeneration (insets in Figure 6B and C) in animals administrated NP-639 or solution-based VCR. This suggests the animals with these pathological findings may have mechanical allodynia or pain on their paws. 57 MiR-183 has been reported to be a potential biomarker of inflammatory and neuropathic pain condition. This potential biomarker has been found to decrease in the spinal cord of a rat osteoarthritic pain model, 58 in the DRGs of spinal nerve lesion mouse model 59 and in the trigeminal ganglion neurons in rat inflammatory muscle pain model, 60 and intrathecal miR-183 administration suppressed mechanical allodynia in mononeuropathy rats. 61 On the contrary, loss of function of the miR-183 cluster results in mechanical hypersensitivity and nocifensive-like hyperalgesia in mice. 62 In our current study, miR-183 level decreases were observed in the circulating blood of the animals dosed with solution-based VCR or nano-VCR in a similar magnitude. However, unlike solution-based VCR, nano-VCR did not induce gait changes in our study. It is possible that other nonmorphological factors also play a role in animal’s pain behavioral manifestation, including gait behaviors. It has been documented that VCR treatment induces macrophage 63 and monocyte 64 infiltration in the peripheral nervous system and macrophage-derived IL-6 elicited mechanical allodynia. The characteristic early finding (acute lesions) of sciatic nerve degeneration including the characteristic swollen, eosinophilic axons (spheroids), and occasional late-stage lesions such as axonal fragmentation and/or myelin disruption or vacuolation (digestion chambers) noted in this study highly suggest Schwann cell activation and axonal degradation. 6 It is likely that solution-based VCR, but not nano-VCR, caused activation or injury of Schwann cells, which produce and release the inflammatory cytokines IL-1β, IL-6, and/or TNF-α 65,66 or directly induced infiltration of immune cells in the peripheral nervous tissues. 67 It was unlikely that the nanoparticle shell (eg, PEG) inhibited pain sensation, since PEG component did not increase the pain thresholds in a rat injury-induced neuropathic pain model. 68 The potential alternation in the VCR release kinetic might affect the innate immune cells besides nerve injury in this model. Although the nano-VCR pharmacokinetics and biodistribution properties were not evaluated in vivo, our in vitro data show that up to 60% VCR released by 6.5 day if single dosed. Here, we were unable to determine whether the observed neuropathy was due to maximal drug level or accumulated drug over the duration of the exposure. Indeed, in numerous clinical studies, continuous infusion (which may similar to the pharmaceutics of our nanoparticle formulation in the current study) instead of bolus injection has been studied in an attempt to decrease neurotoxicity, but results are not conclusive. 69 -72 Furthermore, perhaps other areas (other peripheral nerves, spinal cord tracts, brain), besides those examined, were affected (functionally or structurally) by the altered delivery system. For example, the intraepidermal nerve fibers in the paws of the animals might be less affected by nano-CVR than solution-based VCR, as reported earlier in literature. 73 In a mouse study, solution-based VCR administration caused activation of microglia and astrocyte and TNF-α mRNA upregulation in the spinal cord, and anti-TNF-α mAb attenuated VCR-induced mechanical allodynia. 49 Increased expression of α2δ1 Ca channel subunit was also found in the dorsal horn of spinal cord in rats dosed with VCR. 74 In a 5-HT transporter KO mice study, 51 VCR-induced neuropathy was attenuated, suggesting 5-HT system in CNS may also be involved.
Conclusions
Administration of nano-VCR was associated with reduced behavioral evidence of allodynic pain with unexpectedly decreased miR-183 and unexpected histologic evidence of nerve fiber injury in the sciatic nerve of mice. Nanoparticle properties, including drug load, release rate, or other factors, may need to be optimized to improve biological observations. The mechanisms underlying the behavior improvement but not peripheral nerve structural improvement following nano-VCR administration may require further investigation.
Footnotes
Acknowledgments
The authors would like to thank Jessica Kelly for test article preparation and shipment and Casey Ritenour, Hayley Pryski, and Timothy Coskran for excellent technical support of tissue processing and staining. The authors also thank the Pfizer Kendall Square research support staff for their technical expertise and Cheryl Tyszkiewicz for manuscript preparation. Thanks to Dr. Ingrid Pardo for reviewing this manuscript.
Author Contributions
Bluette, C. contributed to design, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Shoieb, A. contributed to design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Peng, Q. contributed to design, contributed to analysis, drafted manuscript, and critically revised manuscript; Manickam, B. contributed to design, contributed to interpretation, drafted manuscript, and critically revised manuscript; Huang, W. contributed to conception and design, contributed to interpretation, drafted manuscript, and critically revised manuscript; Shin, E. contributed to design, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Zhang, W. contributed to design, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Song, Y. contributed to conception and design, contributed to interpretation, drafted manuscript, and critically revised manuscript; Liu, C. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors were employees of Pfizer Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.