Abstract
The renal proximal tubule cell line, human kidney 2 (HK-2), recapitulates many of the functional cellular and molecular characteristics of differentiated primary proximal tubule cells. These features include anchorage dependence, gluconeogenesis capability, and sodium-dependent sugar transport. In order to ascertain how well HK-2 cells can reliably reveal the toxicological profile of compounds having a potential to cause proximal tubule injury in vivo, we sought to evaluate the effects of known proximal tubule toxicants using the HK-2 cell line. We selected 20 pure nephrotoxic compounds that included chemotherapeutic drugs, antibiotics, and heavy metal-containing compounds and evaluated their ability to induce HK-2 cell injury relative to 10 innocuous pure compounds or cell culture media alone. We performed a comprehensive set of in vitro cellular toxicological assays to evaluate cell viability, oxidative stress, mitochondrial integrity, and a specific biomarker of renal injury, Kidney Injury Molecule 1. For each of our selected compounds, we were able to establish a reproducible profile of toxicological outcomes. We compared our results to those described in peer-reviewed publications to understand how well the HK-2 cellular model agrees with overall in vivo rat or human toxicological outcomes. This study begins to address the question of how well in vitro data generated with HK-2 cells can mirror in vivo animal and human outcomes.
Introduction
In vitro cellular models are an important tool for implementing the Food and Drug Administration (FDA) predictive toxicology roadmap. 1 Not only do they often allow for potential toxins to be evaluated more rapidly than in settings utilizing in vivo models, but they are also relatively less expensive, reduce animal use, and may offer mechanistic insights into organ-specific cellular processes. Like all models, however, in vitro cellular systems have limitations and thus, achieving a good understanding of their domain of applicability in predicting in vivo outcomes is critically important. Establishing the extent of such in vitro to in vivo concordance may allow data generated using in vitro models to be used more productively toward regulatory applications.
Clinical adverse events often involve the kidney, likely due to its important role in filtration, tubular reabsorption, and tubular secretion of substances from the body. 2 Within each kidney, thousands of nephrons are present to carry out the tasks of filtering the blood of waste products via the glomerulus, whereas the proximal tubules receive the filtrate as they work to maintain electrolyte and other equilibria 3 that are vital to human health. The proximal tubules are among the most common targets of renal toxicity due in part to their ability to concentrate solutes. 4 As such, having the appropriate in vitro cell lines to mimic their physiological roles is vital to support the fidelity of in vitro cellular toxicological assays using proximal tubule cell models.
One of the most widely used proximal tubule cell lines is the human kidney 2 (HK-2) cell line, which was established over 2 decades ago as a useful cellular tool for carrying out in vitro toxicology studies.5-7 These cells have been shown to capture essential characteristics of human primary proximal tubules. This cell line is similar to its primary cell counterpart in that HK-2 cells have maintained their anchorage dependence as well as their requirement for epidermal growth factor to grow. Furthermore, HK-2 cell expression of vital metabolic enzymes such as cytochrome P450 3A4 and critical cytoskeletal proteins such as cytokeratin are maintained in HK-2 cells. They have also retained their sodium-dependent glucose uptake, glycolytic metabolism, and gluconeogenesis capabilities. A study that directly compared HK-2 cells to primary human proximal tubule epithelial cells showed that under well-defined culture conditions, they were morphologically and functionally very similar. 8 The retention of these properties by HK-2 cells has made this cell line a favored tool in kidney research across many disciplines, including toxicology.
Many studies using the HK-2 cell line in research, however, have not been comprehensive in investigating its utility as a tool for yielding in vitro data that is aligned with in vivo toxicological outcomes. Thus, we asked whether testing a series of known nephrotoxins toward proximal tubules using in vitro HK-2 cellular methods would also produce data that would be concordant with published in vivo outcomes. Toward this aim, we selected a broad range of 20 compounds that are established toxins of the proximal tubule system and compared them with 10 innocuous compounds that are not known to be toxic toward the kidneys. The collection of toxic chemicals spans 6 classes of toxins: anticancer drugs, immune suppressants, heavy metal compounds, mycotoxins, antibiotics, and a surfactant. We hypothesized that diversifying our selection of toxins would afford us the opportunity to test compounds of different nephrotoxicity strengths. We collected data on the effects of these toxins on several aspects of HK-2 cell toxicity including overall cell viability, reactive oxygen species (ROS) production, mitochondrial effects, and the FDA-qualified renal-specific biomarker of injury Kidney Injury Molecule 1 (KIM-1). Using this collection of assays and proximal tubule-directed nephrotoxins, we examine the concordance between in vitro cellular toxicity data collected using HK-2 cells with that of published in vivo toxicity profiles. Our in vitro findings were concordant with in vivo reports of proximal tubule toxicity and reveal the value of using HK-2 cells as a helpful model for this purpose.
Methods
Cell Culture and Treatment With Select Compounds
Human kidney 2 cells were grown and maintained as described previously. 9 Human kidney 2 cells were exposed to select chemically pure compounds for 24 hours before end point assays were performed. Pure compound stock solutions are listed in Tables 1 and 2 and were purchased from Enzo Life Sciences. All compounds are provided at a purity of at least 95% following high-performance liquid chromatography- or thin-layer chromatography testing. All stock solutions (Tables 1 and 2) were serially diluted in HK-2 cell culture media by 102-, 103-, 104-, 105-, and 106-fold in cell culture medium and were well solubilized. Treated and nontreated HK-2 cells were incubated for 24 hours at 37 °C in a humidified incubator with 5% CO2 prior to performing end point assays. Assays were independently performed 3 times.
Toxic Test Compounds.
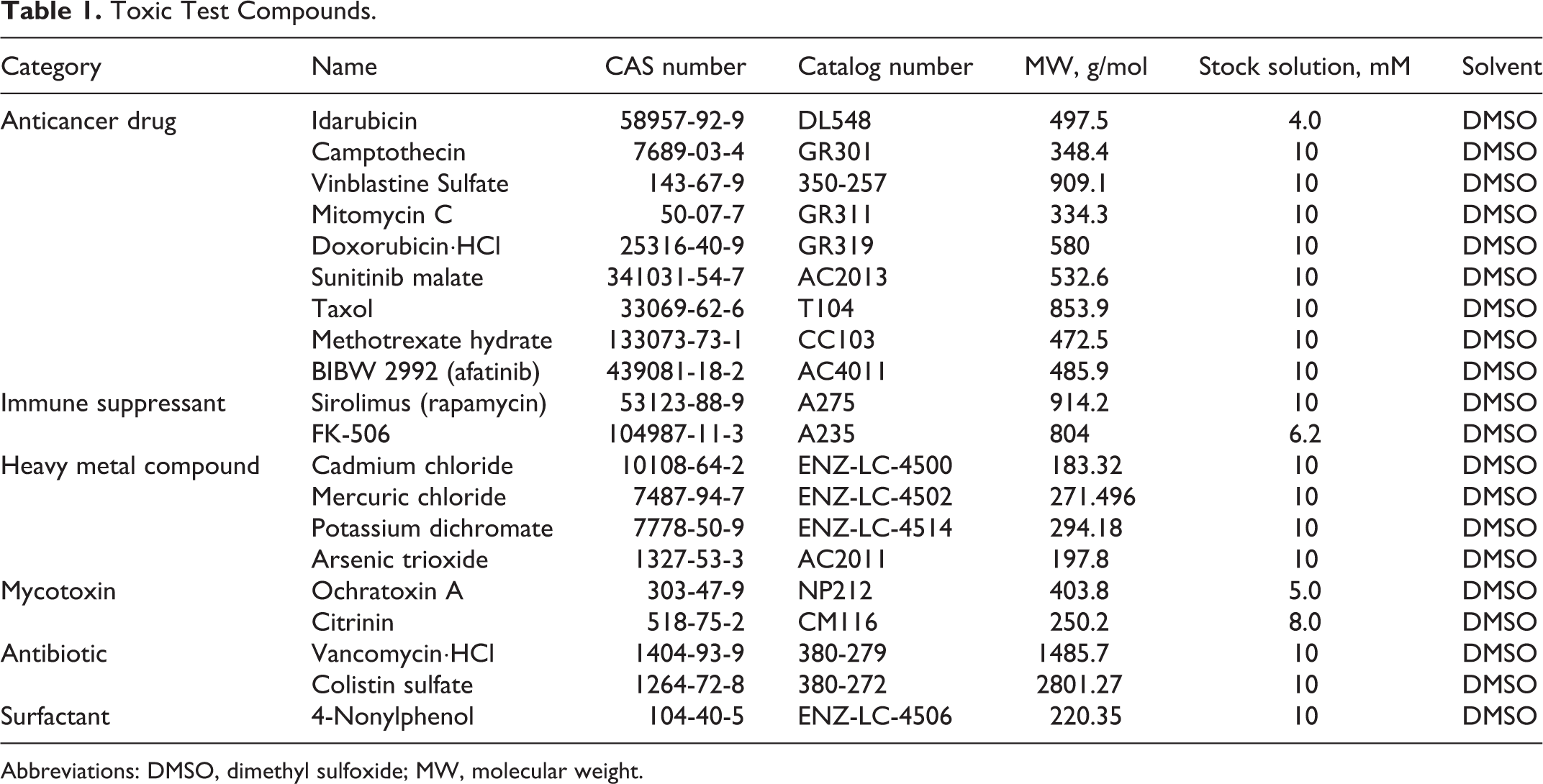
Abbreviations: DMSO, dimethyl sulfoxide; MW, molecular weight.
Innocuous Test Compounds.
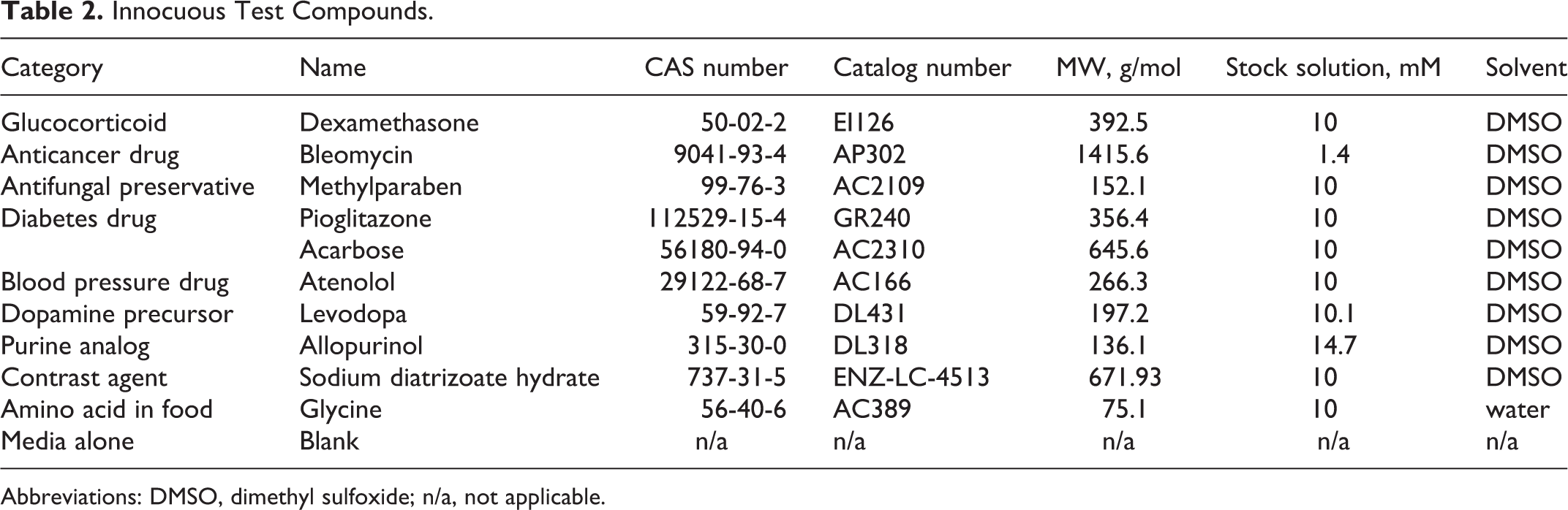
Abbreviations: DMSO, dimethyl sulfoxide; n/a, not applicable.
Cytotoxicity Assay
A luminescent assay based on the principle that adenosine triphosphate (ATP) production is a surrogate indicator of cell viability was used to evaluate compound cytotoxicity in HK-2 cells. Following the manufacturer instructions, CellTiter-Glo Cell Viability Assay (Promega) was carried out on HK-2 cells that were seeded and treated in black-wall, clear-bottom 96-well plates as described previously. 9 Plates were read using a FLUOstar plate reader (BMG Labtech).
Mitochondrial Membrane Potential Assay
Changes in the membrane potential of mitochondrial populations in control and treated HK-2 cells were evaluated using the JC-10 dye (Enzo Life Sciences). As described previously, 9 cells were assayed using fluorescence detection methods at excitation wavelength 485 nm and emission wavelengths 520 and 590 nm. Notably, we verified that the compounds themselves did not autofluorescence. Plates were read using a FLUOstar plate reader.
Reactive Oxygen Species Assay
Reactive oxygen species production from HK-2 cells treated for 24 hours was measured using Promega ROS-Glo H2O2 luminescence-based detection system, following the manufacturer instructions and data were normalized to cell viability as described previously. 9 Plates were read using a FLUOstar plate reader.
Nephrotoxicity Biomarker Assay
The concentration of the renal-specific biomarkers of KIM-1 was quantitatively determined in the cell culture supernatants of treated and nontreated HK-2 cells using the Human Kidney Toxicity kit (R&D Systems) and the Bio-plex Luminex 200 system (Bio-Rad). Biomarker measurements were normalized to cell viability, as described previously. 9
Statistics
Data were collected and analyzed using Microsoft Excel or GraphPad Prism. Statistical analyses were performed by one- or two-way analysis of variances to establish the significance of dose-matched treatment effect differences using a P value threshold of below 0.01 or 0.05 (as indicated).
Results:
Intracellular ATP Levels Reveal a Spectrum of Nephrotoxin Cytotoxicity Profiles
We selected 20 pure compounds that are established in vivo toxins of the proximal tubule system and tested their effects on HK-2 renal cell to examine their effects on cell viability. The selected nephrotoxins are listed in Table 1. We first established cytotoxicity profiles of each compound by measuring each of their effects on the intracellular ATP content of treated HK-2 cells for 24 hours relative to nontreated cells. As shown in Figure 1, ATP levels in toxin-treated HK-2 cells often declined severely as the toxin concentration was increased from a dilution factor of 106 up to 102. Within all 6 categories of toxins, the 102-fold dilution consistently produced statistically significant declines in ATP levels (P < 0.01). Within the anticancer drug category (Figure 1A), 103-fold dilutions of the majority of drugs were also associated with potent toxicity and even the 104-fold dilution (equivalent to just 0.04-1.0 µM) yielded measurable declines in ATP levels. Afatinib was by far the most toxic anticancer drug, as the highest dilution of 106 (or 0.01 µM) resulted in statistically significant lower ATP levels compared to nontreated cells (P < 0.05). By contrast, immune suppressant drugs sirolimus and FK-506 (Figure 1B) were only cytotoxic at the 102-fold dilution tested (P < 0.01), which translates to 0.01 mM and 0.05 mg/mL, respectively. Toxins composed of heavy metals (Figure 1C) had potencies similar to the anticancer drugs, with significant declines in ATP levels measured in starting from dilutions of 102- or 103-fold, which yielded concentrations of 0.1 to 0.01 mM (P < 0.01). In the case of mycotoxins, ochratoxin A was highly cytotoxic relative to citrinin, but neither compound achieved a 50% decline in ATP levels even at the most concentrated dose tested (0.05 and 0.08 mM; Figure 1D). The antibiotics vancomycin-HCl and colistin sulfate were also relatively mild in their toxicity toward HK-2 cells, achieving significant ATP reduction (P < 0.01) only at the 102-fold dilution dose (0.1 mM; Figure 1E). Surfactant 4-nonylphenol was severely toxic at the 102-fold dilution (P < 0.01) but milder in effect at higher dilutions (Figure 1F). The overall trend for this collection of proximal tubule toxins was clear reductions in cellular ATP levels in a manner that was directly proportional to the exposure dose.
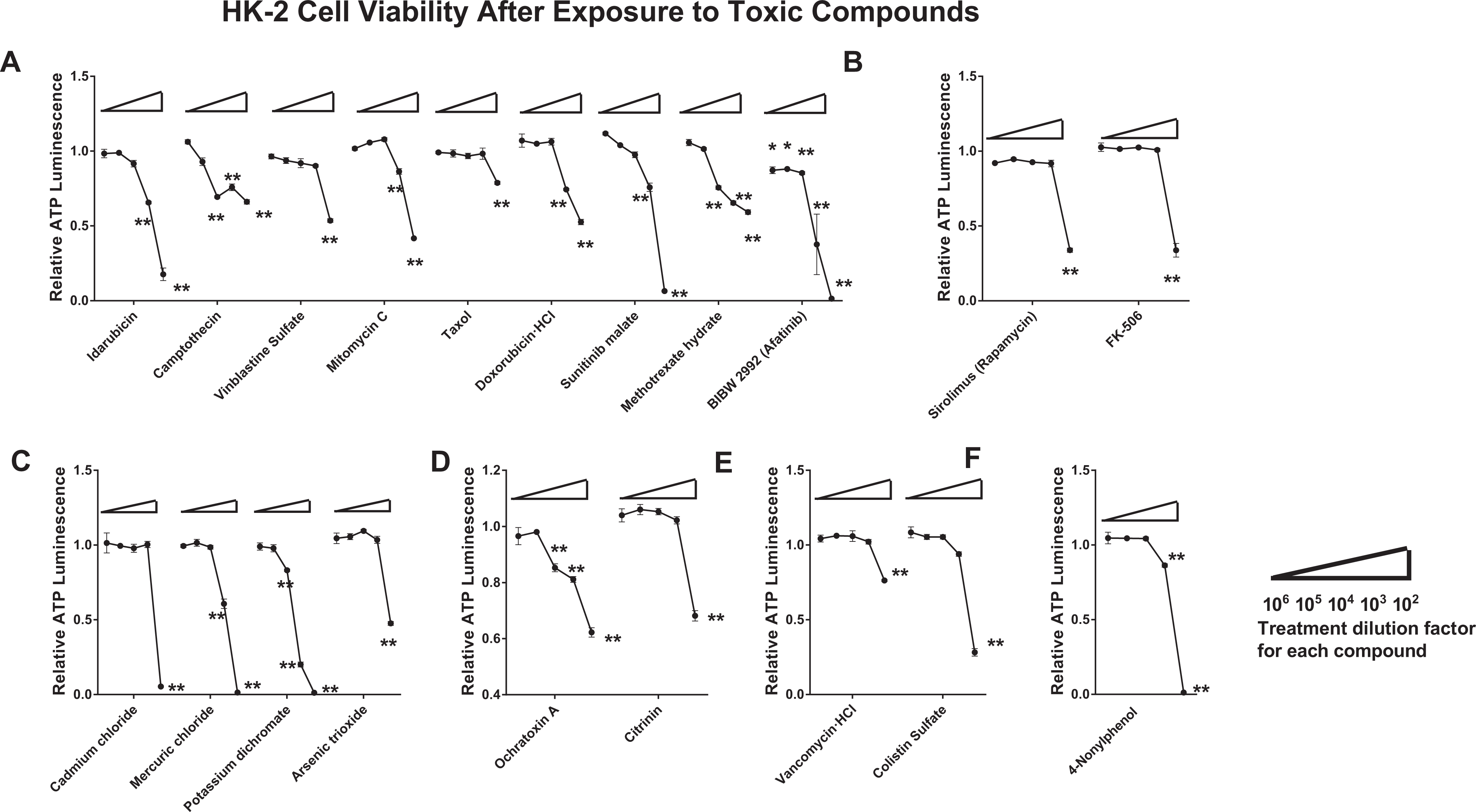
Human kidney 2 cells were treated with 1 of the 20 pure nephrotoxic compounds for 24 hours. Cell viability in treated cells was assayed by ATP luminescence and calculated relative to nontreated HK-2 cell viability. Nephrotoxin doses are increasing (left to right along x-axis for each compound) and are categorized as anticancer drugs (A), immunosuppressants (B), heavy metal-containing compounds (C), mycotoxins (D), antibiotics (E), and a surfactant (F). Data shown are representative of 3 independent experiments. Statistical significance of P < 0.01 or 0.05 for treated versus nontreated populations is indicated by ** or * symbols, respectively. ATP indicates adenosine triphosphate; HK-2, human kidney 2.
Based on the cell viability data collected from cellular ATP levels, lethal concentration 50 (LC50) values were calculated to further explore the acceptability of standard in vitro methods in accurately capturing the toxicity of established in vivo proximal tubule toxicants. As shown in Table 3, all 20 tested toxicants had LC50 values that fell between the values of 0.01 and 0.80 mM. Among the 20 toxic compounds tested, 14 were particularly potent in this cellular model, having LC50 values ranging from merely 0.01 to 0.1 mM. These included 5 of the tested anticancer drugs (idarubicin, mitomycin C, doxorubicin-HCl, sunitinib malate, and afatinib), both of the immune suppressants, all 4 of the tested heavy metal-containing compounds, 1 mycotoxin (citrinin), 1 antibiotic (colistin sulfate), and the 1 surfactant. Less potent compounds having LC50 values between 0.1 and 0.8 mM were 4 anticancer drugs (camptothecin, vinblastine sulfate, taxol, and methotrexate hydrate), 1 mycotoxin (ochratoxin A), and 1 antibiotic (vancomycin-HCl).
Values of LC50 for Each Toxic Compound.
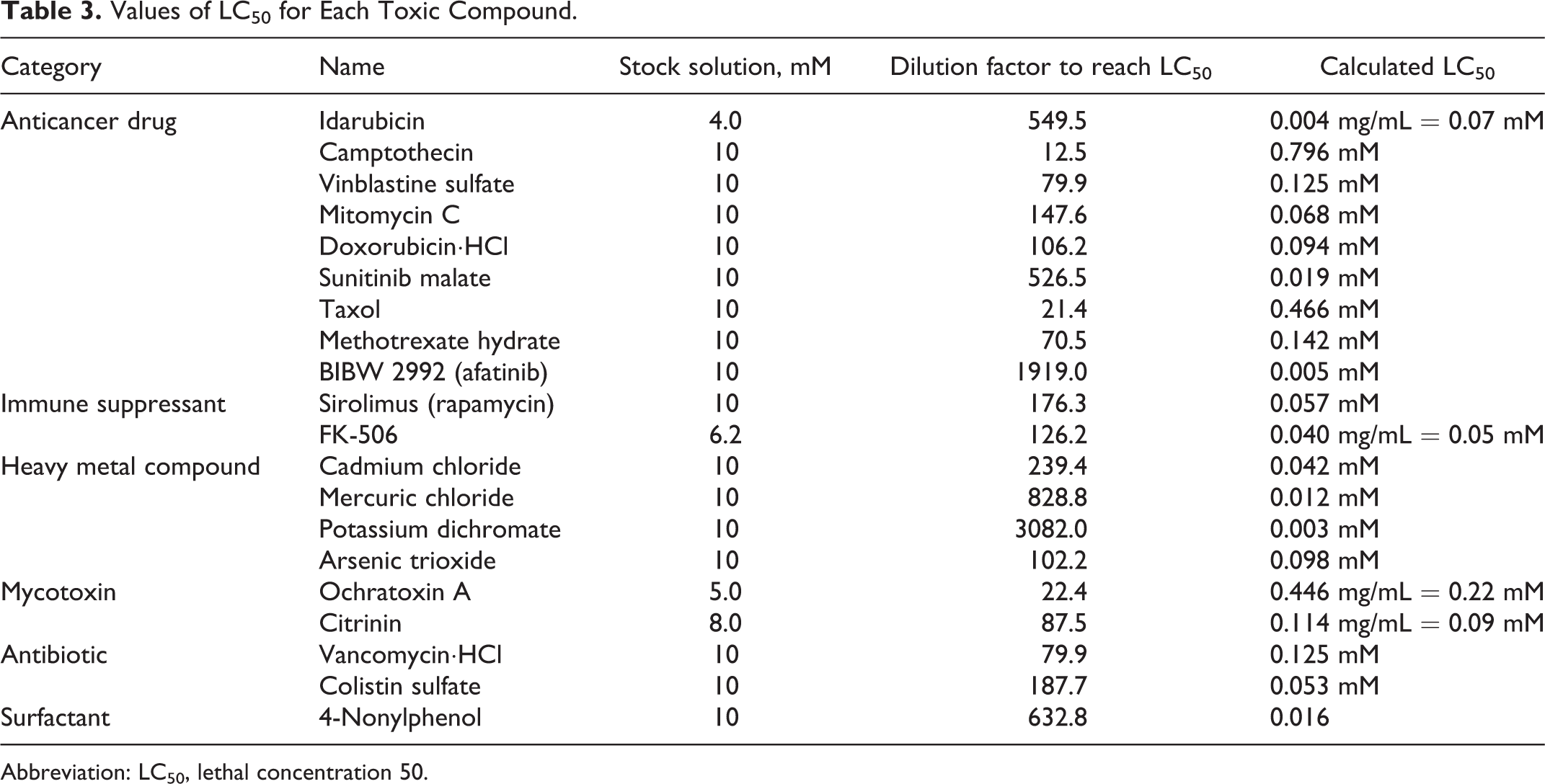
Abbreviation: LC50, lethal concentration 50.
Effects of Proximal Tubule Toxicants Include Oxidative Stress and Mitochondrial Dysfunction
Having confirmed that all tested proximal tubule cell toxicants were capable of consistently producing significant cytotoxicity in the HK-2 model, we sought to investigate how much oxidative stress was occurring in treated cells. Levels of ROS have been shown to be a strong biomarker of cellular toxicity 10 and we found that oxidative stress and cell viability following exposure to some compounds had similar trends, but large changes in ROS were generally not measured (Figure 2). Across all 6 categories of toxins, we found the trend that high dilutions of toxins did produce slight elevations in ROS levels until the highest dose was applied. These high toxin doses yielded significantly decreased ROS production relative to nontreated cells, as the treated cells were nonviable under such potent cytotoxicity.
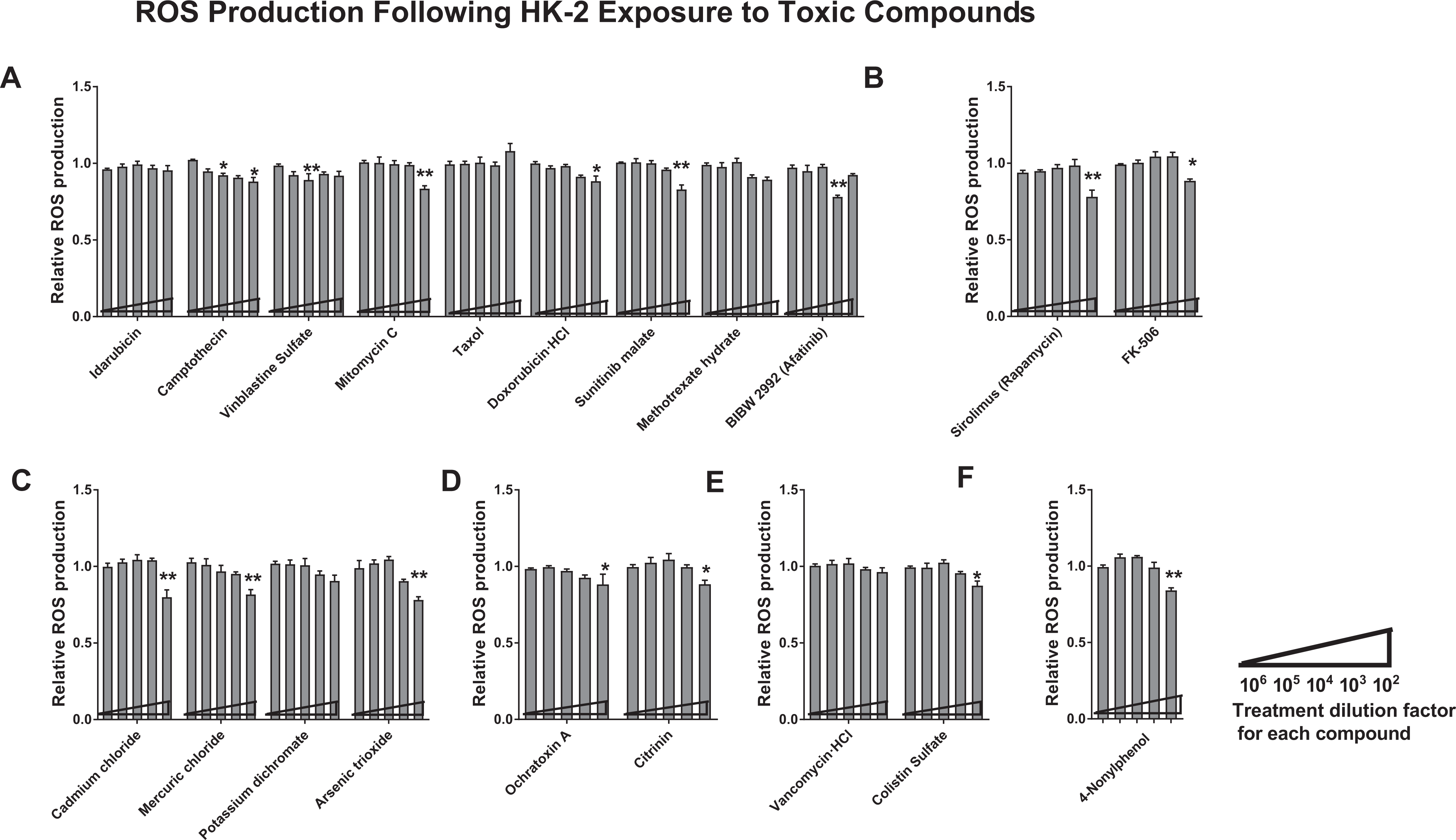
Human kidney 2 cells were exposed to each of 20 nephrotoxins and quantitatively assayed for ROS levels 24 hours posttreatment. Nephrotoxins are categorized as anticancer drugs (A), immunosuppressants (B), heavy metal-containing compounds (C), mycotoxins (D), antibiotics (E), and a surfactant (F). For each nephrotoxin, dosing strength increases along the x-axis. Data shown are representative of 3 independent experiments. Statistical significance of treated versus nontreated populations is indicated by the ** (P < 0.01) or * (P < 0.05) symbols. HK-2 indicates human kidney 2; ROS, reactive oxygen species.
As oxidative stress is a manifestation of dysfunctional mitochondria, we next investigated effects on mitochondrial health of exposing HK-2 cells to each of the 20 toxins. Mitochondria use their inner and outer membranes to maintain a voltage potential that is critical for the ATP-producing electron transport chain to function. We measured the disruption of this membrane potential using the well-established dye JC-10, which fluoresces at one wavelength (590 nm) when it is present inside the cell, but outside the mitochondria it fluoresces at a different wavelength (520 nm) when it is able to penetrate into mitochondria with damaged membranes. As shown in Figure 3, loss in mitochondrial membrane potential (MMP), as measured by increases in the 520:590 nm ratio of wavelengths, was only detectable at low toxin doses. These increases did not reach statistical significance, except in the case of the heavy metal compound cadmium chloride (Figure 3C). High doses of toxins typically resulted in the overall loss of signal, as in the case of ROS production levels, which is aligned with a severe cytotoxic state.
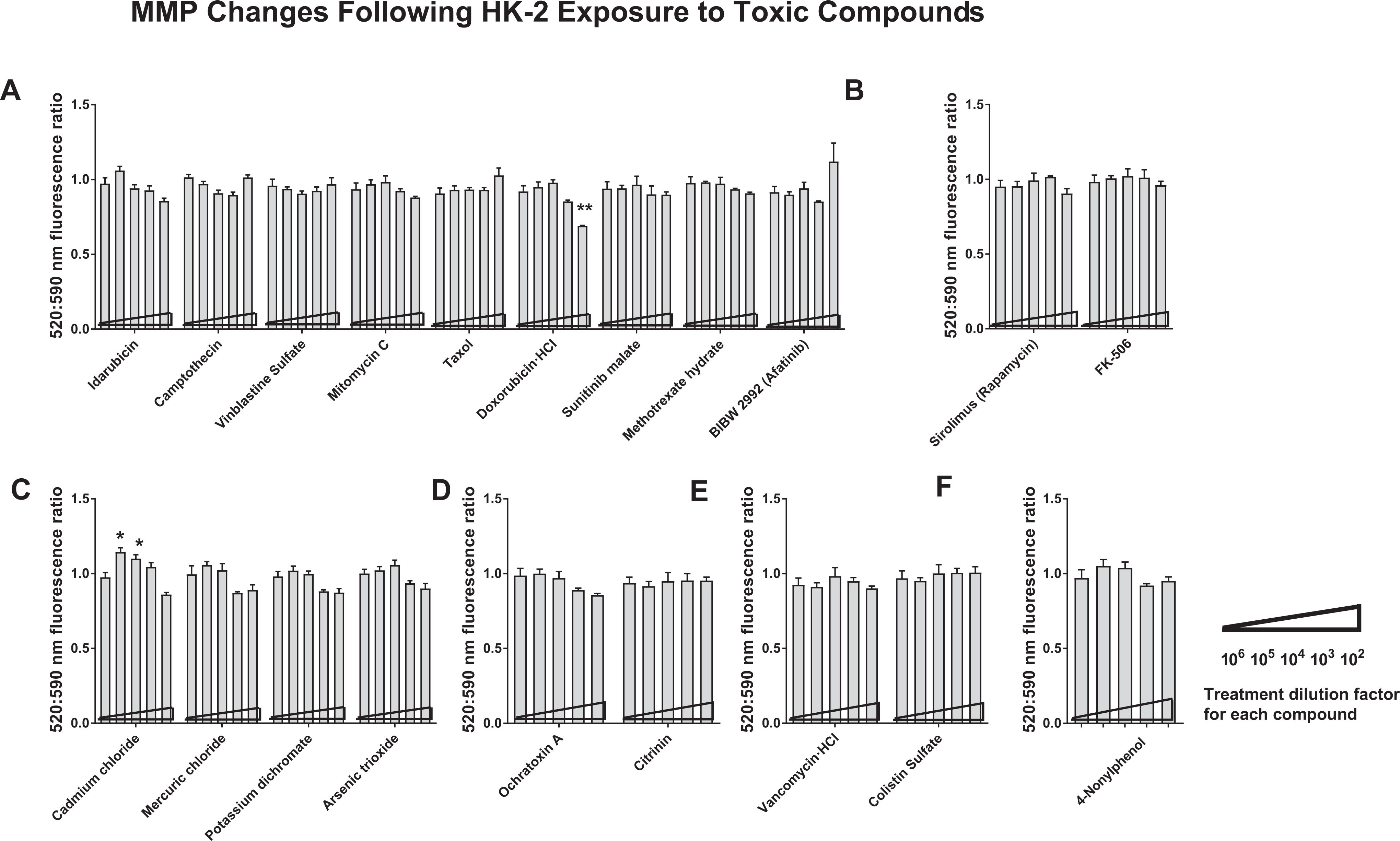
Shifts toward MMP loss were measured in treated HK-2 cells treated for 24 hours with cell toxins by determining changes in the relative amounts of mitochondria that undergo loss versus maintenance of membrane potential, calculated as a ratio of fluorescence emission at 520 versus 590 nm. Nephrotoxins are categorized as anticancer drugs (A), immunosuppressants (B), heavy metal-containing compounds (C), mycotoxins (D), antibiotics (E), and a surfactant (F). For each nephrotoxin, dosing strength increases along the x-axis. The graphs shown are representative of 3 independent experiments. Statistical significance of treated versus nontreated populations is indicated by individual ** or * symbols (P < 0.01 or 0.05, respectively). HK-2 indicates human kidney 2; MMP, mitochondrial membrane potential.
Renal-Specific Injury Marker, KIM-1, Is Elevated Following Exposure to Renal Toxins
We next investigated how effectively changes in KIM-1, an FDA-qualified biomarker of renal injury, could be reliably captured in HK-2 cells following 24-hour exposure to each of the 20 proximal tubule toxins. After collecting the culture supernatants of cells treated with 103-fold diluted compounds, we analyzed the concentrations of KIM-1 by Luminex. As shown in Figure 4, elevations in KIM-1 levels were not consistently measured across all toxin categories. Anticancer drugs idarubicin and afatinib produced about two times as much as KIM-1 in nontreated cell supernatants (P < 0.01; Figure 4A) but only mild changes were found in immune suppressant-treated HK-2 cells (Figure 4B). As shown in Figure 4C, exposure to heavy metal compound mercuric chloride was associated with high KIM-1 levels (P < 0.05), which reached 5-fold higher KIM-1 levels following exposure to potassium dichromate (P < 0.01). Similar to the tested immune suppressants, the mycotoxins, antibiotics, and surfactant produced mild to no elevations of KIM-1 in treated HK-2 cells.
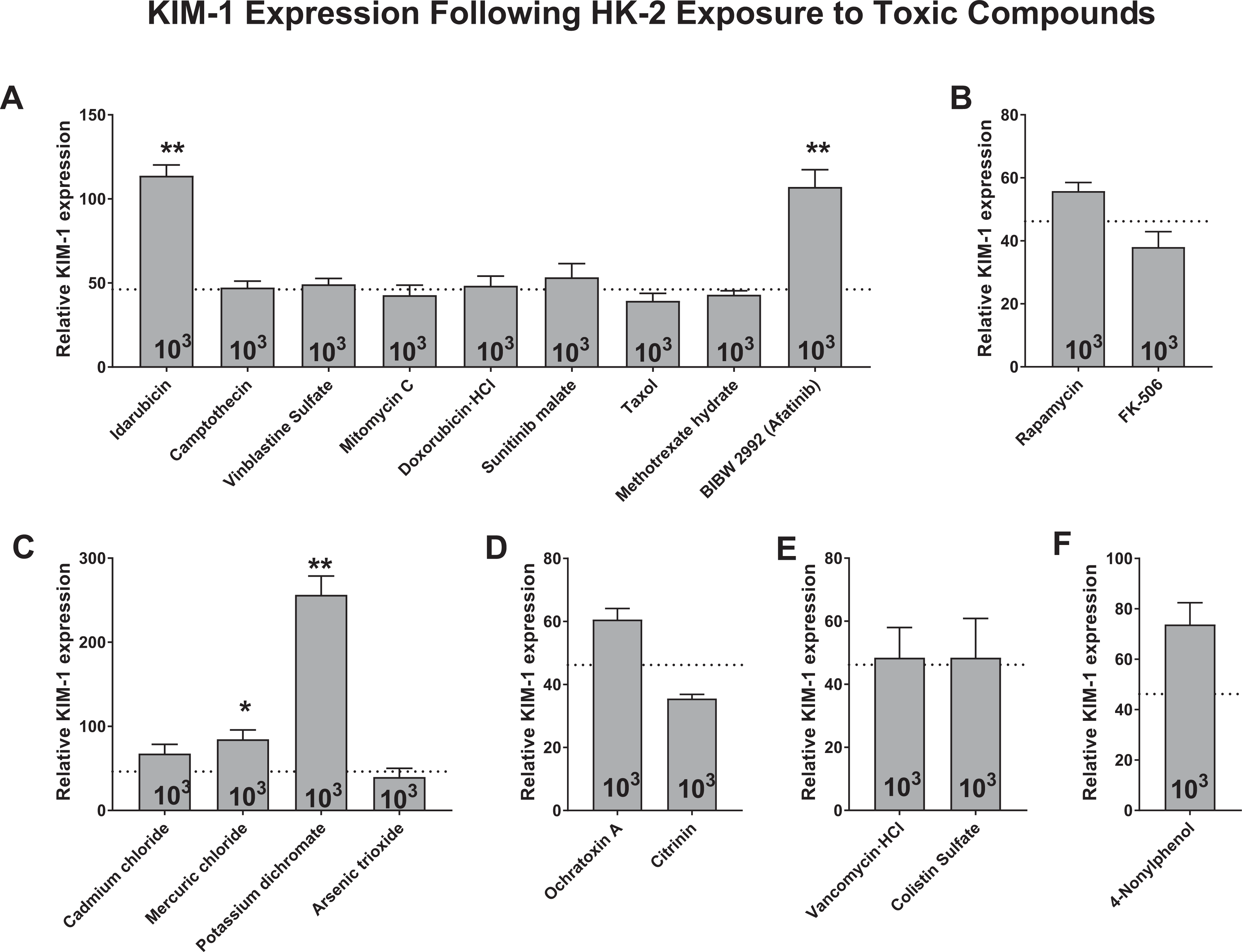
Biomarker signature of relative KIM-1 expression following 24 hours of exposing HK-2 cells to each nephrotoxin at a single dose determined by diluting stock solutions by 103-fold. Human kidney 2 cell culture supernatants from cells treated at this single dose were assayed by luminex technology. Nephrotoxins are categorized as anticancer drugs (A), immunosuppressants (B), heavy metal-containing compounds (C), mycotoxins (D), antibiotics (E), and a surfactant (F). Horizonal line indicates KIM-1 levels when HK-2 cells are left untreated. Data are representative of 3 independent experiments. Statistical significance of treated versus nontreated populations is indicated by the symbol ** for P < 0.01 or * for P < 0.05. HK-2 indicates human kidney 2; KIM-1, Kidney Injury Molecule 1.
Innocuous Compounds Yield Only Minor Effects on HK-2 Cells
To better understand how the assortment of proximal tubule toxins compared to innocuous compounds, we selected 10 pure chemicals that have been shown not to be nephrotoxic toward the proximal tubules (Table 2). We performed the same collection of assays using these innocuous compounds to be able to establish whether false-positive effects would be obtained using the HK-2 model and to be able to compare the results from the 2 lists of compounds. As shown in Figure 5A, cytotoxic effects were typically measured only at the highest dose tested, but these declines in ATP levels were not statistically significant. Similarly, changes in ROS production and MMP were not detected, except in the case of pioglitazone drug, which mildly compromised mitochondrial integrity in a dose-dependent manner (Figure 5B and C). Additionally, when the 103-fold dilution of compounds was tested for their effects on KIM-1 levels, only sodium diatrizoate hydrate yielded mild elevation that reached statistical significance (P < 0.05).
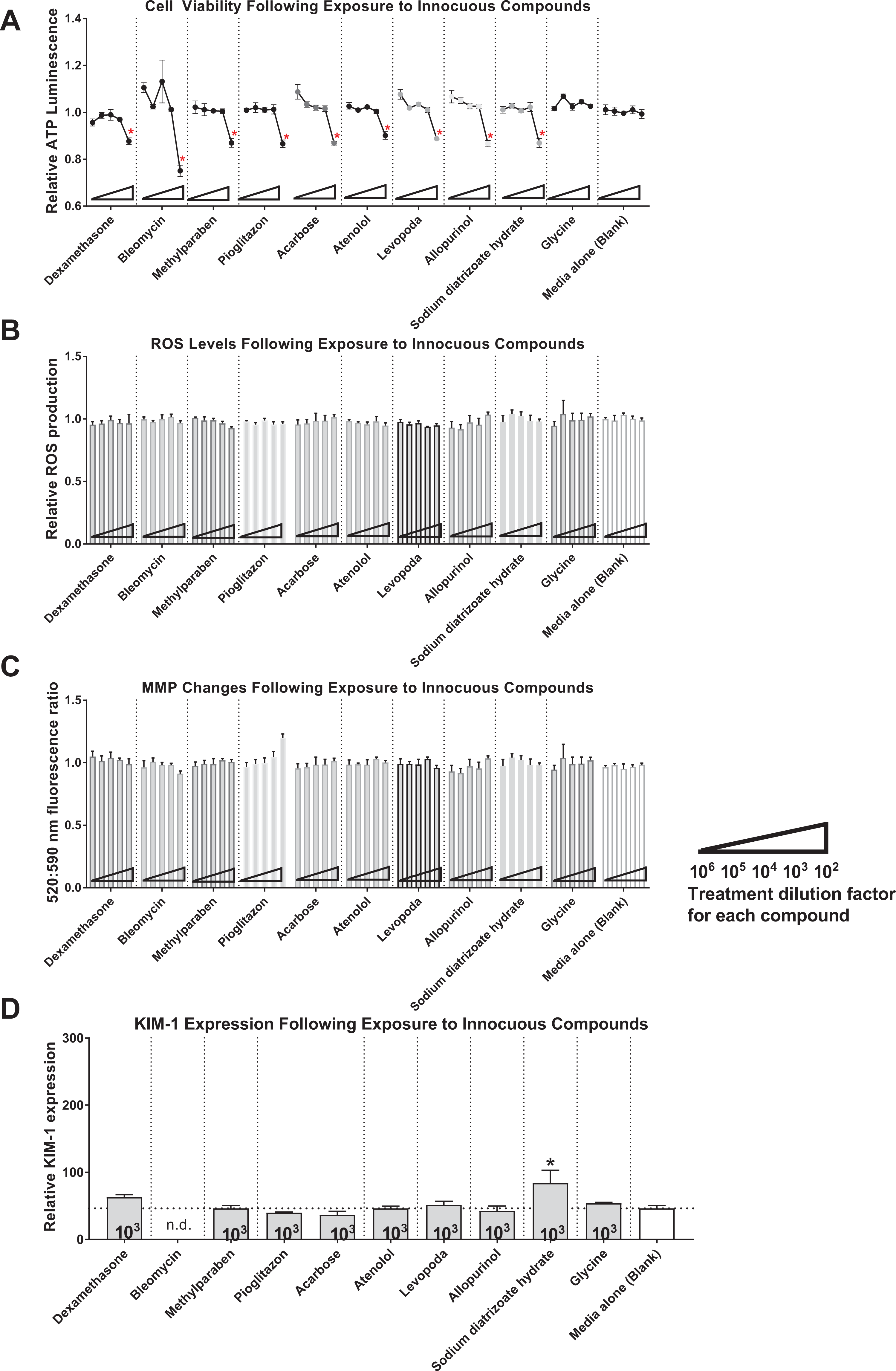
Human kidney 2 cells were treated with 1 of 10 pure innocuous compounds for 24 hours. Cell viability in treated cells was assayed by ATP luminescence and calculated relative to nontreated HK-2 cell viability (A). The ROS production was measured relative to cell viability in treated versus nontreated cells (B). Mitochondrial changes in membrane potential were evaluated by JC-10 assay (C). Relative KIM-1 expression was measured at the treatment concentration of 10 µM (D). The graphs shown are representative of 3 independent experiments. Statistical significance of treated versus nontreated populations is indicated by individual ** or * symbols (P < 0.01 or 0.05, respectively). ATP indicates adenosine triphosphate; HK-2, human kidney 2; KIM-1, Kidney Injury Molecule 1; nd, not determined; ROS, reactive oxygen species.
Discussion
Although the human proximal tubule cell line, HK-2, has been used in a plethora of studies to help establish the nephrotoxicity potential of select toxins, 11 -16 there is a lack of comprehensive research focused on the use of the HK-2 cell line as a tool to effectively predict and verify in vivo toxicity on proximal tubule injury. Understanding the concordance between data generated from in vitro cell models, in vivo animal models, and human clinical studies lends credence to in vitro studies in settings where risk assessments of potential toxins are being pursued. Thus, our goal in this study was to perform an initial test to begin answering the question of how well in vitro data generated with HK-2 cells can mirror in vivo animal and human outcomes. We expected that all selected proximal tubule toxicants would be toxic in some capacity in our assays, as we specifically chose them based on their published toxicity toward proximal tubules. We also expected that since these toxicants spanned multiple classes of compounds with varying chemical structures and modes of action, we would see considerable variation in the results of our general assays of toxicity (cytotoxicity, MMP, and ROS assays) as well as the renal-specific KIM-1 assay. Toward our goal, we selected a total of 20 proximal tubule toxicants and 10 compounds that are not known to be toxic toward the proximal tubules to test their effects on HK-2 cells using 4 different in vitro toxicity assays. We found that the established toxins of the proximal tubules can show their toxic effects most prominently on cell viability, as measured by cellular ATP levels, but that their effects on mechanisms of toxicity relating to oxidative stress, mitochondrial dysfunction, and KIM-1 induction were not variable. We also found that the collection of tested innocuous compounds produced mild or undetectable effects.
The design of this study entailed serially diluting purchased stock solutions of select compounds to reduce the hazards of extensively handling so many noxious chemicals. All stock solutions were usually provided at the concentration of 1 mM or 2 mg/mL and their dilution by 102-fold reliably yielded potent cytotoxicity when ATP levels in HK-2 cells were measured. Further dilutions by factors of 10 resulted in a straight-forward, dose-dependent lessening of cytotoxicity, as expected. Additionally, the collection of 10 innocuous chemicals that we tested consistently yielded relatively mild effects that were not significant on HK-2 cell viability. Their highest tested doses did produce detectable and significant (P < 0.05) declines in ATP levels, most notably with bleomycin; however, it is well known that harmful osmotic pressures from high concentrations of most solutes will eventually result in untoward effects on cells. Nevertheless, it appeared that HK-2 cells performed reliably well in discriminating toxins from nontoxins in terms of cell viability. Since there are no official standards for innocuous compounds toward human proximal tubule cells, it would be helpful to expand this list to better explore the best way to define a non-nephrotoxic effect. Expanding this list of innocuous compounds and including them in future testing efforts would help to standardize our testing platform.
Upon testing the usefulness of HK-2 cells to yield additional mechanistic information about the levels of oxidative stress and mitochondrial membrane integrity, striking differences between the list of toxic and innocuous chemicals were lacking. In many cases, changes induced by the toxic chemicals were detectable, but not significant. In other cases, such as camptothecin, vinblastine sulfate, and afatinib, statistically significant ROS levels were measured, but dose-dependent responses were lacking. It is possible that very high doses of these toxins induced intracellular changes that were upstream of ROS changes that would have been induced at lower toxin doses. Alternatively, such unexpected findings could simply reflect the possibility that nonmitochondrial-related mechanisms of action were being induced by these toxins. In other cases, however, attempts to glean useful information by performing ROS and JC-10 assays were undermined by the steep toxicity curves induced by higher dose exposures; dead cells are not suitable for ROS and other mitochondria-related tests. Adjusting the range of testing doses may mitigate this issue, but it is also possible that chemical interference occurred between the selected compounds and the proprietary chemicals used in the assay kits. All chemicals evaluated in our study were checked for autoluminescence and autofluorescence (data not shown) to ensure that erroneous signals would not confound our data sets. Thus, when evaluating the utility of HK-2 cells as a suitable model for in vitro cellular toxicity studies, the selection of assays is just as important as the appropriateness of the cell model itself.
Since KIM-1 is an officially qualified biomarker of nephrotoxicity in vivo and has been used in several published reports to serve as a biomarker of in vitro proximal tubule cell injury as well, we were surprised that it did not yield a robust pattern of elevation that mirrored the cytotoxicity data trends collected for each toxin. Since the highest concentrations tested resulted in such potent cytotoxicity, we avoided using those doses and instead evaluated KIM-1 levels in the 103-fold dilution dose. Perhaps additional intermediate doses of toxins would be needed in order to observe consistent KIM-1 changes, but the lack of upregulation may also reflect a limitation of the HK-2 cell line in this aspect of its use. In vivo, KIM-1 expression by the proximal tubules is associated with their regeneration process following injury. 17 Although studies have demonstrated that the HK-2 cell line is capable of increasing its KIM-1 expression following its exposure to toxins 18 -21 it is possible that certain toxins may actually inhibit the regenerative potential of these cells. Thus, despite its utility as a supportive assay, KIM-1 expression analyses in HK-2 cells cannot reliably be used as a stand-alone assay. This notion is further underscored by the unexpected finding that sodium diatrizoate hydrate yielded significantly high KIM-1 expression for reasons that are not clear. It has been shown by Isamail et al. 22 that KIM-1 upregulation can help protect against kidney damage by blocking guanosine triphosphate loading, but further experiments would be needed to determine if this hypothesis is pertinent in this setting.
Other than KIM-1 levels, commonalities between end point measurements from in vitro cellular assays and in vivo outcomes are not established. In vitro to in vivo extrapolation is an active field in toxicology but suffers from difficulties in finding common ground between the 2 systems. Thus, our attempt to understand how the HK-2 cell line can be used as an in vitro cellular model that yields data which are concordant with in vivo outcomes relies on using a collection of in vitro assays to establish if any given chemical is toxic to HK-2 cells and then comparing this conclusion to published determinations on whether the same toxin is harmful to proximal tubules in vivo.
Within each of the 6 categories of selected toxins, variation was observed in the extent of their potency against HK-2 cells, but overall, anticancer drugs and heavy metal-containing compounds were the most toxic. Within the anticancer drug category, in vivo animal and clinical studies have demonstrated potentially profound proximal tubular nephrotoxicity of idarubicin, 23,24 camptothecin, 25,26 vinblastine sulfate, 27 mitomycin C, 28,29 taxol, 30 doxorubicin-HCl, 24,31 sunitinib malate, 32,33 methotrexate hydrate, 28,34 and afatinib 35,36 These pharmaceutical drugs which possess vastly different mechanisms of action for their intended purposes of affecting the tubular and other parts of the kidney are not well studied. However, what they have in common is that these drugs and their metabolites are commonly eliminated through the renal system. 37 Immunosuppressants sirolimus and FK-506 also work differently from one another, through the inhibition of mammalian target of rapamycin and calcineurin, respectively, to inhibit T cell activation. At least in the case of rapamycin, its harmful effects on the proximal tubules of humans and rodent models are thought to involve their antiproliferative action against tubular cells. 38,39 It seems that HK-2 cells were able to effectively capture the cytotoxicity associated with these drugs because their action against proximal tubule cells is not dependent on the tubular structure of the cell. Research on heavy metal-containing compounds as well as fungal mycotoxins reveals extensive toxicity profiles to multiple organs. Their nephrotoxicity within the proximal tubules, however, is at least due in part to their ability to settle within the tubules as well as interfere with mitochondrial function. 40 -43 If their toxicity was entirely reliant on the tubular structure of the proximal tubule cells, then HK-2 cells (cultured as 2-dimensional monolayers) would not have demonstrated concordance with the in vivo outcomes of these compounds. Antibiotics are also long considered to be nephrotoxic in part due to their ability to accumulate in the cortex, including the proximal tubules of cortical nephrons 29,30 and through direct cellular damage. 44,45 Our in vitro findings of toxicity match up with data collected from animal model studies, since both vancomycin-HCl and colistin sulfate were cytotoxic. The last compound among the 20 toxins we tested, 4-nonylphenol, is a known surfactant used in many detergents and can induce a variety of tubular-related effects including cyst formation and tubular dilation, 46,47 However, because surfactants also disrupt the lipid composition of cell membranes, 48 it is likely that the HK-2 cytotoxicity we measured was due to this effect, since HK-2 cells were not cultured to model a 3-dimensional (3D) tubular structure. Although we selected the toxins in this study for their known toxic effects on proximal tubules, it was interesting that their reported mechanisms of action varied, even for compounds within a single category. An exploration into how their effects impact other parts of the nephron (Figure 6) would provide a more profound understanding of their nephrotoxic nature in vivo, which could in turn be modeled in vitro with additional studies that ideally use 3D culture systems.
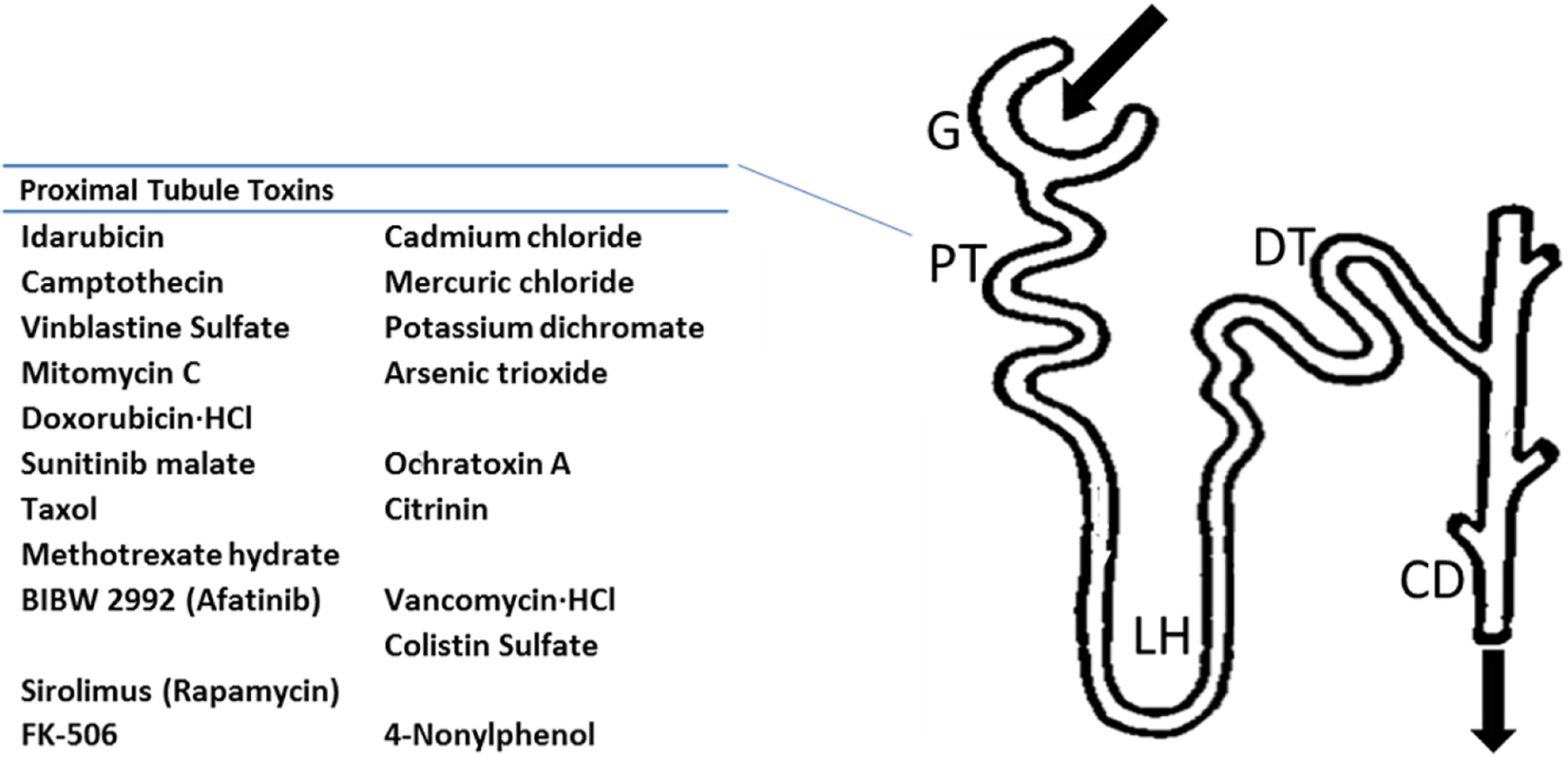
Toxic compounds used in this study affect the proximal tubules of the nephron in vivo and show toxic effects on cultured human proximal tubule HK-2 cells in vitro. CD indicates collecting duct; G, glomerulus; DT, distal convoluted tubule; HL, loop of Henle; HK-2, human kidney 2; PT, proximal convoluted tubule.
In addition to evaluating the concordance of HK-2 cell readouts and in vivo data, we also checked for false-positive results using 10 innocuous compounds. In the case of dexamethasone, pioglitazone, and glycine, published research by others has shown that these innocuous compounds can be nephroprotective in vivo. 49 -53 In other cases, such as methylparaben, levododa, sodium diatrizoate hydrate, and bleomycin, the selected innocuous chemicals were simply shown to have no significant renal effects in vivo. 54 -57 Interestingly, the diabetes drug acarbose was shown to be nephrotoxic to the glomerulus in vivo using rodent models, but we appropriately found that it was not toxic to proximal tubule HK-2 cells. 58 Similarly, atenolol is not nephrotoxic on its own but is instead reported to be nephrotoxic in combination with other medications due to its effects on the ureter. 59 Thus, the lack of in vivo toxicity reported for these compounds was similarly not mimicked in our in vitro cellular assays, showing that HK-2 cell outcomes can also effectively model nontoxins as well as toxins.
We recognize that although investigating a series of proximal tubule-damaging nephrotoxins and nonnephrotoxins has helped us center our study on proximal tubule-specific in vitro to in vivo concordance, future studies would benefit from modifications to this design. First, an expanded list of different types of compounds of clinical significance will be evaluated. This list will include expanded lists of non-nephrotoxic as well as nephrotoxic compounds that target different parts of the nephron, not just the proximal tubule cells. Taking this approach will help us to tease out differences in mechanistic vulnerabilities between individual parts of the nephron. Second, additional in vitro control model cells from other parts of the nephron and other organs (eg, liver) will also be added to better control for proximal tubule specificity of any observed cytotoxic responses. Third, KIM-1 assay measurements will be done on the full range of treatment concentrations, including toxin LC50 values. Finally, a wider variety of end point assays that investigate varied aspects of cell injury, such as toxin entry into cells and potential toxin transport within organelles of the cell, will be included to complement our current collection of evaluation assays.
In conclusion, the HK-2 cell line can serve as an appropriate cellular model for in vitro toxicity research, as its cytotoxic responses to known toxins and innocuous chemicals are concordant with published in vivo outcomes. Our study is the first of its kind to directly compare the concordance of in vitro to in vivo data and we ascertain that HK-2 cells merit further, more extensive investigations as a tool to (1) predict whether a more exhaustive list of compounds can cause injury to the epithelial cells that make up the proximal tubules and (2) establish the limits of the predictability of this cell line. It is clear from our work that one limitation is the evaluation of toxins that need a full tubule to induce injury. Thus, in addition to having an expanded multiparameter study design, future studies addressing the value of 3D cultured HK-2 cells arranged as tubules would be especially useful to properly evaluate chemicals with mechanisms involving tubular structures. Such advances would not only allow better concordance to be attained but allow this important topic to be explored from other angles, such as the potential reversibility of toxicity.
Footnotes
Author Contributions
Mossoba contributed to conception and design, contributed to acquisition, drafted the manuscript, and critically revised the manuscript; Sprando contributed to conception and design and critically revised the manuscript. Both authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no external financial support for the research, authorship, and/or publication of this article.
